Abstract
Joint injury dramatically enhances the onset of osteoarthritis (OA) and is responsible for an estimated 12% of OA. Posttraumatic arthritis (PTA) is especially common after intra-articular fracture, and no disease-modifying therapies are currently available. We hypothesized that the delivery of mesenchymal stem cells (MSCs) would prevent PTA by altering the balance of inflammation and regeneration after fracture of the mouse knee. Additionally, we examined the hypothesis that MSCs from the MRL/MpJ (MRL) “superhealer” mouse strain would show increased multilineage and therapeutic potentials as compared to those from C57BL/6 (B6) mice, as MRL mice have shown exceptional in vivo regenerative abilities. A highly purified population of MSCs was prospectively isolated from bone marrow using cell surface markers (CD45-/TER119-/PDGFRa+/Sca-1+). B6 MSCs expanded greater than 100,000-fold in 3 weeks when cultured at 2% oxygen and displayed greater adipogenic, osteogenic, and chondrogenic differentiation as compared to MRL MSCs. Mice receiving only a control saline injection after fracture demonstrated PTA after 8 weeks, but the delivery of 10,000 B6 or MRL MSCs to the joint prevented the development of PTA. Cytokine levels in serum and synovial fluid were affected by treatment with stem cells, including elevated systemic interleukin-10 at several time points. The delivery of MSCs did not reduce the degree of synovial inflammation but did show increased bone volume during repair. This study provides evidence that intra-articular stem cell therapy can prevent the development of PTA after fracture and has implications for possible clinical interventions after joint injury before evidence of significant OA.
Keywords
Introduction
An estimated 27 million Americans have clinical osteoarthritis (OA) (45), and the risk of OA increases 10-to 20-fold following joint trauma such as ligament injury, meniscal tear, or intra-articular fracture (2). Posttraumatic arthritis (PTA) represents 12% of lower extremity OA cases and causes a large economic burden due to the young age of the PTA population (8). The presence of inflammatory cytokines such as interleukin-1 (IL-1) and tumor necrosis factor-α (TNF-α) in the joint fluid, synovium, and other joint tissues has emerged as an important contributor to the pathogenesis of both idiopathic and secondary OA (5,15,23,27,32,33). Furthermore, the rapid development of OA after closed-joint intra-articular fracture of the mouse knee has confirmed the central role of inflammation and provides a model system for examining the effects of different therapeutic approaches to prevent the onset or progression of PTA (22,24,50).
The delivery of mesenchymal stem cells (MSCs) has been proposed as a regenerative therapy for a wide range of disease states. An emerging paradigm suggests that long-term engraftment and differentiation may not be the primary regenerative mechanisms of exogenously delivered MSCs. Instead, MSCs modulate inflammation and provide a regenerative environment either by direct secretion of bioactive factors or by altering the cytokine and growth factor production of endogenous cells (9,39,63,66). In several models of disease, MSCs exert protective effects by producing anti-inflammatory molecules such as IL-1 receptor antagonist (IL-1ra) and interleukin-10 (IL-10) (59,60). While stem cell-based solutions have been studied for musculoskeletal repair and regeneration (3,46,58,73), MSC therapy for the prevention of PTA after closed intra-articular fracture has not been investigated.
Different mouse strains possess significantly different regenerative phenotypes, suggesting that their MSCs may have different therapeutic effectiveness. For example, the MRL/MpJ (MRL) “superhealer” inbred mouse strain has shown enhanced regeneration after injury in a variety of tissues such as the ear, cornea, heart, digit tips, and articular cartilage (11,12,20,48,76). Of particular interest was the observation that MRL mice were protected from PTA after intra-articular fracture (80). Regeneration in MRL mice is correlated with a reduced inflammatory signature after injury (22,28,36,52), suggesting an altered transition from the acute inflammatory phase to a resolution phase that allows for regeneration. The contribution of MSCs to this transition is unknown, but bone marrow-derived MSCs from MRL mice exhibited enhanced engraftment, deposition of granulation tissue, and functional improvement in a model of myocardial injury (1).
The evaluation of MSC therapy in preclinical models is hampered by the technical challenges associated with in vitro culture of murine MSCs, such as slow expansion and the persistence of contaminating cell populations (62). Strategies to overcome these limitations have included extended culture periods at low density (56,61), removing undesired cell types by modified plating techniques or immunodepletion (4,72,74), expanding cells in hypoxic conditions (71), or utilizing compact bone as an enriched MSC source as compared to bone marrow (71,81,84). In recent studies, Morikawa et al. reported a method for prospectively isolating a pure population of bone marrow-derived MSCs utilizing collagenase digestion and flow cytometry cell sorting for cells coexpressing platelet-derived growth factor receptor a (PDGFRa, also known as CD140a) and stem cell antigen-1 (Sca-1) (57). In this study, we found that culturing PDGFRa+/Sca-1+ MSCs at low oxygen tension greatly increased the rate of cell proliferation, thus providing a pure and extensive source of murine MSCs with multilineage potential.
We hypothesized that the delivery of MSCs directly to the joint space after fracture would prevent the development of PTA by altering the inflammatory environment. Additionally, we used this model system to compare the regenerative capabilities of MSCs isolated from control C57BL/6 mice and MRL/MpJ “superhealer” mice.
Materials and Methods
MSC Isolation
All procedures were performed in accordance with a protocol approved by the Duke University Institutional Animal Care and Use Committee. For stem cell isolation, male MRL/MpJ (The Jackson Laboratory, Bar Harbor, ME, USA) or C57BL/6 (Charles River Laboratories, Wilmington, MA, USA) mice were sacrificed at 8–10 weeks of age with CO2. Excised femurs and tibias were crushed into fragments and the exposed bone marrow was gently removed by washing. Digestion with 5 ml of 0.2% collagenase type 1 (Worthington Biochemical, Lakewood, NJ, USA) per mouse for 60 min at 37°C released cells that were filtered through a 70-μm strainer (BD Biosciences, San Jose, CA, USA) and treated with ammonium chloride potassium (ACK) buffer (0.15 M NH4Cl, 10 mM KHCO3, and 0.1 mM Na2EDTA pH 7.3; Sigma-Aldrich, St. Louis, MO, USA) to lyse erythrocytes. Cells were treated with Fc block and labeled with antibodies to mouse CD45 [fluorescein isothiocyanate (FITC)], TER-119 [lymphocyte antigen-76 (Ly76); FITC], Sca-1 (Alexa 647), and PDGFRa [phycoerythrin (PE)] or isotype controls for 30 min at 4°C (all from Biolegend, San Diego, CA, USA). A Cytomation MoFlo® sorter (Beckman Coulter, Bray, CA, USA) captured cells negative for CD45/TER-119 and positive for both Sca-1 and PDGFRα (57).
MSC Expansion
After sorting, MSCs were cultured at 100 cells/cm2 in expansion medium consisting of α-modified Eagle's medium (aMEM; Invitrogen, Carlsbad, CA, USA), 20% lot-selected fetal bovine serum (FBS; Sigma-Aldrich), and 1% penicillin/streptomycin/fungizone (P/S/F; Invitrogen) in a hypoxic incubator (37°C, 2% O2, 5% CO2, remaining gas N2; NAPCO, Winchester, VA, USA). After 8 days with media changes every 3 days, cells were passaged and plated at 3,000 cells/cm2 with subsequent passages carried out every 3–4 days upon 90% confluence. Expansion rates were calculated from three independent isolations of three to six mice of each strain.
Colony-Forming Unit (CFU-F) Assay
Two-hundred fifty freshly sorted MSCs were plated in wells of a six-well plate (9.5 cm2; Costar, Corning, NY, USA) in expansion medium. After 7 days with no medium changes, wells were stained with 3% crystal violet (Harleco, EMD Chemicals, Millipore, Billerica, MA, USA) washed repeatedly with PBS and then analyzed for defined colonies of greater than 10 cells using standard microscopy, with data presented from three wells of one of two independent experiments.
Flow Cytometry
Passage 3 cells were treated with Fc block and labeled with antibodies to the following cell surface markers and appropriate isotype controls (all from Biolegend): mouse CD45 (FITC), CD49d (FITC), TER-119 (FITC), CD44 [PE-Cyanine 5 (Cy5)], CD29 (PE-Cy5), C-X-C chemokine receptor type 4 (CXCR4; Alexa 647), CD11b [allophycocyanin (APC)], PDGFRa (PE), Sca-1 (Alexa 647). A C6 benchtop flow cytometer (Accuri Cytometers, Ann Arbor, MI, USA) was used for analysis and percentages obtained by subtracting the value of isotype controls.
Adipogenic Differentiation
Ten thousand cells at passage 2 or 3 were plated in wells of 48-well plates (0.95 cm2; Costar) for 2 days in expansion medium at normoxic conditions. Medium was then switched to control medium consisting of Dulbecco's modified Eagle's medium/F12 (DMEM/F12) (Lonza, Walkersville, MD, USA) with 3% FBS and 1% P/S/F or adipogenic differentiation medium (34) consisting of control medium plus (all from Sigma-Aldrich) 33 μM biotin, 17 μM pantothenate, 1 μM bovine insulin, 1 μM dexamethasone, and for the first 3 days only 250 μM isobutylmethylxanthine (IBMX) and 2 μM rosiglitazone (Avandia”, GlaxoSmithKline, London, UK). After 14 days, cells were imaged and then fixed with 4% paraformaldehyde (Electron Microscopy Sciences, Hatfield, PA, USA). Mono-layers were stained as described (34) with 0.5% Oil Red O stain (EMD Chemicals, Gibbstown, NJ, USA). Cells were washed with 60% isopropanol (Acros Organics, Thermo Fisher Scientific, Waltham, MA, USA), and the stain was released with 250 μl of 100% isopropanol and then quantified by absorbance at 535 nm, with data presented from three wells of one of two independent experiments.
Osteogenic Differentiation
Ten thousand cells at passage 2 or 3 were plated in wells of 48-well plates (0.95 cm2) for 2 days in expansion medium at normoxic conditions. Medium was then switched to control medium consisting of DMEM-high glucose (HG) with 10% FBS and 1% P/S/F or osteogenic differentiation medium (79) consisting of control medium plus 10 mM β-glycerophosphate (Sigma-Aldrich), 250 μM ascorbate (Sigma-Aldrich), 2.5 μM retinoic acid (Sigma-Aldrich), and 50 ng/ml human bone morphogenetic protein-2 (BMP-2, R&D Systems, Minneapolis, MN, USA). After 21 days, cells were fixed with 4% paraformalde hyde and stained with 2% Alizarin Red S (Electron Microscopy Sciences, Hatfield, PA, USA) for 20 min. After two washes with deionized water, wells were photographed and then Alizarin stain was released for quantification by heated acid extraction (31). Briefly, 10% v/v acetic acid (EMD Chemicals, Millipore) was added to well for 30 min at room temperature and then transferred to Eppendorfs (VWR, Radnor, PA, USA) for 10 min at 85°C. Ammonium hydroxide (10% v/v; Ricca Chemical Company, Arlington, TX, USA) was added to neutralize the acid and absorbance was measured at 405 nm, with data presented from five wells of one of two independent experiments.
Chondrogenic Differentiation
Two hundred fifty thousand cells at passage 2 or 3 in expansion medium were placed in 15-ml polypropylene tubes (Corning) and centrifuged at 300×g (Thermo Scientific Centra CL3R; Thermo Fisher Scientific) for 5 min to form rounded pellets. After 2 days, medium was switched to serum-free control medium consisting of DMEM-HG (Invitrogen), 1% insulin transferrin selenous acid + (ITS+; BD Biosciences), 50 μg/ml ascorbate (Sigma-Aldrich), 40 μg/ml proline (Sigma-Aldrich), and 1% P/S/F (Sigma-Aldrich) or chondrogenic differentiation medium (16) consisting of control medium plus 10 ng/ml human transforming growth factor-b3 (TGF-β3; R&D) and 500 ng/ml human bone morphogenetic protein-6 (BMP-6; R&D). After 28 days, pellets were processed for Safranin-O/Fast Green histological staining (Sigma-Aldrich) or biochemical analysis by measuring double-stranded DNA with the PicoGreen assay (Molecular Probes, Invitrogen) and glycosaminoglycan (GAG) content with the 1,9 dimethyl methylene blue (DMB; Sigma-Aldrich) assay as previously described (17). Data presented are from three or more pellets of one of two independent experiments.
Intra-Articular Fracture
B6 mice at the skeletally mature age of 16 weeks were used for a closed tibial plateau fracture model of PTA as previously described (24,80). Briefly, the sedated mouse was fit into a custom cradle so that the left hind limb was at neutral position under a 10 N preload, which was applied using a wedge-shaped indenter attached to a materials testing system (ElectroForce ELF3200, Bose Corp., Minnetonka, MN, USA). Compressive force was applied until −2.7 mm displacement at a rate of 20 N/s in load control. Each fracture was confirmed using high resolution digital X-ray (MX-20, Faxitron, Lincolnshire, IL, USA). Fifty-one mice received fractures of the left hind limb, while right hind limbs were not fractured and served as contralateral controls.
Stem Cell Injection
MSCs were pooled from six B6 or MRL mice and expanded three passages in monolayer culture. MSCs were delivered immediately after fracture. The sedated mice were injected with either sterile saline only (Hospira, Inc, Lake Forest, IL, USA) or 10,000 MSCs in 6 μl saline using a specialty syringe (catalog #80401, Hamilton Company, Reno, NV, USA) and 30-gauge 1/2 needle (BD Biosciences). The mouse was positioned for lateral entry with the left hind limb extended to facilitate injection of 6 μl into the joint space through the patellar tendon. This delivery technique does not result in initiation of osteoarthritis (77). To allow for cell tracking, some MSCs were first treated with 4 μM of the membrane dye chloromethylbenzamido-dialkylcarbocyanine (CM-DiI, Sigma-Aldrich) for 5 min and washed before injection.
Serum and Synovial Fluid Analysis
Retro-orbital bleeding and cardiac stick were used to collect blood at the time of sacrifice, with three mice for prefracture controls, day 1, day 3, and day 7 time points and eight mice for 8 weeks after fracture and injection. After clotting, samples were centrifuged at 2,450×g for 15 min, and serum was transferred to −80°C. At the same time points, synovial fluid from fractured and contralateral limbs was isolated from the exposed joint space as previously described (70). Briefly, the synovial fluid was absorbed onto a Melgisorb pad (Tendra, Göteborg, Sweden), and this was dissolved with 35 μl alginate lyase (Sigma-Aldrich) and 15 μl sodium citrate (Sigma-Aldrich). Serum and synovial fluid from the fractured knee were analyzed for the presence of the following cytokines by ELISA utilizing the manufacturer's instructions and a 1:5 dilution for synovial fluid samples (R&D Systems): IL-1b (cat #MLB00C), interleukin-1 receptor antagonist (IL-1ra, cat #MRA00), and interleukin-10 (IL-10, cat #M1000, serum only). Samples that were undetectable were assigned a value of one half of the lower limit of quantification for analysis.
Analysis of Mouse Joints
Eight weeks after fracture, eight mice per group were sacrificed, and both fractured and contralateral control limbs were fixed in neutral alignment using 10% neutral buffered formalin (VWR, Radnor, PA, USA) for 48 h. Joints were transferred to 70% ethanol (Koptec, VWR) and analyzed with micro-computed tomography (μCT 40, Scanco Medical, Brüttisellen, Switzerland) as previously described (24,80). Briefly, transverse slices of 16 μm were used to generate 3D reconstructions that were subjected to global threshholding to separate bone from soft tissue. A phantom was used to calibrate linear attenuation to hydroxyapatite (HA) concentration for bone density measurements. Bone density and bone volume were analyzed from eight mice per group in three regions: cancellous bone of the distal femoral condyles, tibial plateau distal to subchondral plate, and metaphyseal region of tibial plateau. After μCT analysis, the limbs were decalcified and processed for histology using an increasing ethanol series, xylenes, and paraffin steps in an automated tissue processor (ASP300S, Leica Microsystems, Buffalo Grove, IL, USA).
Coronal plane sections of 8 μm were taken for histology and stained with Safranin-O/Fast-green/hematoxylin for analysis of cartilage degradation. Modified Mankin grading of OA features including changes to cartilage structure and loss of Safranin-O staining was performed by three blinded graders and used to calculate a score with a maximum of 30 for both the medial and lateral aspects of the femur and tibia (23). Sections were stained with Harris hematoxylin and eosin (H&E; Poly Scientific, Bay Shore, NY, USA) for assessment of synovitis by three blinded graders as previously described (22). One joint was unable to be processed, leaving at least seven mice for each group for modified Mankin and synovitis grading. Three selected joints from each group were analyzed with immunohistochemistry for activated macrophages using a monoclonal antibody against F4/80 (Clone BM8, Biolegend). Antigen retrieval was aided by 0.05% Proteinase K (Sigma-Aldrich) and heated citrate buffer extraction. Chromogen detection was carried out with the Vectastain system (Vector Laboratories, Burlingame, CA, USA). For one mouse per group at each time point, sections were analyzed for CM-DiI cell tracking by clearing with xylenes (Thermo Fisher Scientific) and exciting with a 543-nm laser using confocal microscopy (LSM510, Zeiss, Peabody, MA, USA).
Statistical Analysis
Statistical analysis was carried out using a t test for comparison of two groups and analysis of variance (ANOVA) for comparison of multiple groups, with repeated measures ANOVA on control and fractured limbs for modified Mankin scoring. Fisher's LSD post hoc analysis with a = 0.05 was used. Normality was tested and data log-transformed before analysis if necessary. For nonparametric analysis of cytokine data, undetectable values were assigned the value of one half of the lower limit of detection for analysis. Kruskal–Wallis median test was used to determine the effect of time point and the main effects of treatment group at each time point. Data are presented as mean ± standard error of the mean.
Results
Rapid Expansion of Purified MSCs
Bone marrow-derived MSCs were isolated from B6 and MRL mice by utilizing collagenase digestion and the expression of specific cell surface markers. While the percentage of bone marrow cells expressing hematopoietic lineage markers varied with each isolation, a representative isolation shows 0.70% of all cells were negative for the hematopoietic markers CD45 and TER-119, and 5.09% of these cells were positive for both PDGFRa and Sca-1 (Fig. 1A). B6 and MRL mice contained a similar number of MSCs, with yields of approximately 300–600 MSCs per mouse. Within the sorted MSC population, MRL mice had a higher frequency of colony-forming cells as assessed by the CFU-F assay (19 ± 1.15 vs. 10 ± 0.58 colonies per 250 starting cells, p < 0.05).

Cell sorting and expansion. (A) Sorting strategy for representative MRL mesenchymal stem cell (MSC) isolation, with arrow indicating only cluster of differentiation 45/Ter-119-negative (CD45-,TER-119-) cells are shown in the second plot. Sca-1, stem cell antigen-1; PDGFRα, platelet derived growth factor receptor α. (B, E) MSCs plated down after sorting. (C, F) Colonies expand rapidly. (D, G) Cells retain morphology and rapid growth from B6 and MRL mice, respectively. Scale bars: 100 μm. (H) Cumulative fold increase at 2% O2; results averaged from three isolations.
MSCs were expanded in a 2% O2 environment. Individual colonies of B6 MSCs were established at day 4 (Fig. 1B), grew significantly by day 6 (Fig. 1C), and continued to proliferate at passage 3 (Fig. 1D), with similar results for MRL MSCs (Fig. 1E–G). MSCs from B6 mice showed a trend toward greater expansion as compared to MRL mice (Fig. 1H), resulting in a 13-fold higher cumulative fold increase in cell number at the end of three passages (154,141 ± 113,095 vs. 11,728 ± 5,069 cells per starting cell, p > 0.05).
Characterization of MSCs From B6 and MRL Mice
Passage 3 MSCs from both strains displayed cell surface markers consistent with previous MSC isolation strategies such as being uniformly positive for CD44 and Sca-1 (>95%) and uniformly negative for CD11b (<1%). MSCs were also negative for cell surface markers related to homing capabilities (<1% for CD49d, <1% for CXCR4). MSCs retained expression of PDGFRa in culture although not uniformly (91.4% of B6 and 81.9% of MRL).
MSCs differentiated into cells with the properties of adipocytes, osteoblasts, and chondrocytes (Fig. 2). In general, B6 MSCs demonstrated enhanced differentiation as compared to MRL MSCs. After 14 days in adipogenic media, more lipid droplets formed in B6 MSC cultures (Fig. 2A, B) and quantification by release of lipid staining showed a significant increase in adipogenesis as compared to MRL MSCs (0.11 ± 0.005 vs. 0.092 ± 0.005 absorbance units, p < 0.05) (Fig. 2C). B6 MSCs also demonstrated a trend toward increased osteogenesis (Fig. 2D) as compared to MRL MSCs (Fig. 2E) when quantified by heated extraction of Alizarin S mineralization staining (0.73 ± 0.26 vs. 0.22 ± 0.064 adjusted absorbance units, p = 0.13) (Fig. 2F). Chondrogenesis was assessed by the production of GAGs after 28 days of pellet culture (Fig. 2G, H). B6 MSC pellets contained more GAGs than MRL MSCs (68.65 ± 1.23 vs. 20.25 ± 0.077 μg per pellet, p < 0.05) (Fig. 2I) despite a smaller overall pellet size.

Multilineage differentiation. (A, B) Adipogenic lipid accumulation. (D, E) Osteogenic Alizarin Red S staining. (G, H) Chondrogenic Safranin-O/Fast Green staining. (C, F, I) Quantification, results from ≥3 samples per group of one representative isolation with standard error of the mean displayed. *Significant effect by t test. Scale bar: 100 μm (A, B, G, H) or 25 mm (D, E). GAG, glycosaminoglycan.
Prevention of Posttraumatic Arthritis by Stem Cell Therapy
Fracture of the left hind limb treated with only a control saline injection resulted in a significantly higher modified Mankin osteoarthritis score as compared to the contralateral control limb, indicating the presence of PTA in this group of mice (Fig. 3A). Direct intra-articular delivery of either B6 or MRL MSCs immediately after fracture eliminated the difference in total joint modified Mankin score between the fractured limb and the control limb, showing MSC therapy was able to mitigate the development of PTA. Due to the location of the fracture, degeneration was most severe at the lateral tibia and the protective effect of the MSC therapy was most apparent on the lateral side of the joint (Fig. 3B–D). A small number of MSCs were detected in various joint structures at days 1, 3, 7 as well as 8 weeks (Fig. 4).

Evaluation of posttraumatic arthritis. (A) Total joint modified Mankin score of joint degeneration, mean of ≥7 joints per group. *Significant effect by ANOVA, Fisher's post hoc. (B–D) Safranin-O/Fast-Green/hematoxylin-stained coronal section showing the articulation of the lateral femur (top) and lateral tibia (bottom) 8 weeks after fracture. Joint of each group with the highest structural Mankin scores on lateral side shown. Scale bar: 100 μm. Arrow, fracture site.

Cell tracking. CM-DiI labeling of stem cells before injection. B6 MSCs were found at day 1 in (A) bone marrow, (B) synovium, and (C) muscle. B6 MSCs were also found at (D) day 3 in the lateral femoral synovium, (E) day 7 near lateral ligamentous tissue, and (F) 8 weeks in the tibial subchondral bone.
Serum and Synovial Fluid Analysis
The level of IL-1β in serum was increased at early time points after fracture and returned towards the prefracture values after 8 weeks in all groups with no significant effect of treatment (Fig. 5A). The treatment group did have a significant effect on synovial fluid levels of IL-1β at day 3 and at 8 weeks, with MSC treatment being associated with higher values at day 3 but reduced levels at 8 weeks (Fig. 5B). Serum levels of IL-1ra were affected by the treatment group at 8 weeks, with elevated IL-1ra in the B6 MSC treatment group (Fig. 5C), and all groups demonstrated a trend towards increased synovial fluid IL-1ra 7 days after fracture (Fig. 5D). The treatment group significantly affected the serum levels of IL-10 at days 3 and 7 after fracture, with higher values in the MSC-treated groups (Fig. 5E).
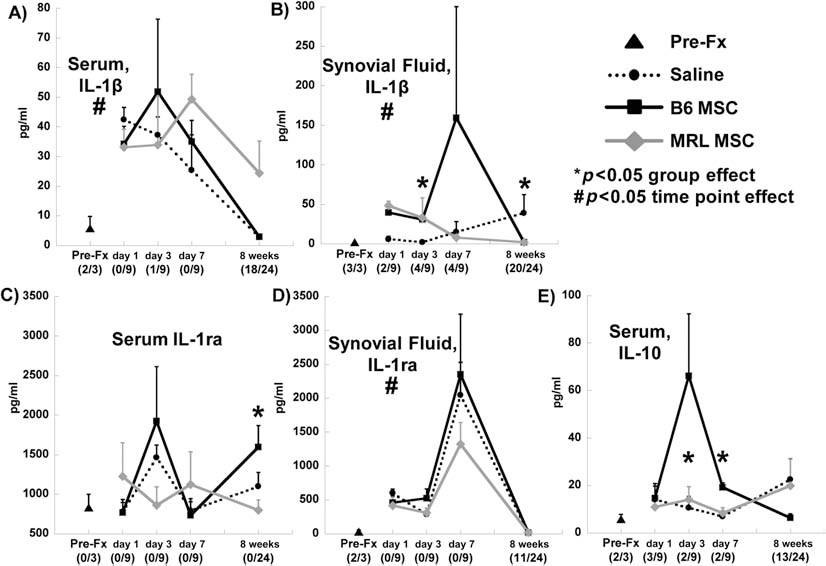
Systemic (serum) and local (synovial fluid from fractured knee) cytokine concentrations. (A, B) Interleukin-1β (IL-1β). (C, D) Interleukin-1 receptor antagonist (IL-1ra). (E) Interleukin-10 (IL-10). Number of undetectable samples is given in parentheses. #Significant effect of time point by Kruskal-Wallis test. *Significant effect of treatment group at that time point by Kruskal–Wallis test. Prefracture (Pre-Fx) and days 1, 3, 7 (n = 3) and 8 weeks (n = 8) after fracture.
Synovial Inflammation and Macrophage Immunohistochemistry
The inflammation of the synovium 8 weeks after fracture was measured by a synovitis score that assesses the thickening of the synovial lining as well as the cellularity of the surrounding stroma. Fracture clearly increased the degree of synovitis in all treatment groups as compared to the contralateral control and MRL MSCs resulted in more inflammation than B6 MSCs (Fig. 6A). The typical synovial lining of one to two cell layers (Fig. 6B) showed extensive thickening after fracture (Fig. 6C). An antibody against F4/80, used to identify activated macrophages, demonstrated that some of the cells contributing to the synovial response are macrophages (Fig. 6D). No clear differences in macrophage staining were apparent between treatment groups.

Synovial response to fracture. (A) Total joint synovitis score, mean of ≥7 joints per group. Significance to group shown (*) or all control groups (#) by ANOVA, Fisher's post hoc. (B) Hematoxylin/eosin (H&E) staining of lateral femur from a nonfractured control limb without synovial inflammation (mirror image shown). (C) Hematoxylin/eosin staining of lateral femur 8 weeks after fracture and injection of B6 MSCs. (D) F4/80 staining for macrophages in the same joint as panel C. Scale bar: 50 μm. Arrows, synovium.
Morphologic Bone Changes
Micro-CT analysis of morphologic bone changes showed increased bone volume and decreased bone density in the tibia and tibial metaphysis of the fractured limb as compared to the contralateral control limb (Fig. 7). The stem cell groups appeared to contribute to a larger bone volume, with the fractured limbs receiving MRL MSCs having a significantly larger volume than all control limbs in the tibial metaphysis (Fig. 7C). In the femur, the saline group showed significantly lower cancellous bone fraction (bone volume/total volume) in the fractured limb compared to the control limb and compared to both MSC therapy groups (Fig. 7E). With MSC treatment, there was no significant decrease in cancellous bone fraction following fracture as compared to the contralateral control limbs.

Morphological bone changes. (A) Tibial bone volume. (B) Tibial bone density. (C) Bone volume in tibial metaphysis. (D) Bone density in tibial metaphysis. (E) Femoral bone volume/total volume. (F) Femoral bone density. Significance to contralateral control (*), all control groups (#), or all groups (&) by ANOVA, Fisher's post hoc; eight joints per group. HA, hydroxyapatite.
Discussion
The goal of this study was to prevent the development of OA after intra-articular fracture using a single injection of expanded MSCs directly into the murine knee joint. Our findings show that an allogeneic stem cell based therapy can prevent the degenerative changes following joint trauma, and similar protective effects were observed using either B6 or MRL MSCs. The modified Mankin score of the fractured joint was significantly higher than the control joint when only saline was used as the treatment, indicative of PTA. However, intra-articular injection of 10,000 B6 or MRL MSCs after fracture eliminated the difference between control and fractured limbs. Establishing treatment options for PTA is particularly promising because, in contrast to idiopathic OA, the clear initiating event allows for early intervention before excessive degradation occurs (2).
We confirmed that prospective isolation of PDGFRa+/Sca-1+ cells from bone marrow results in a highly purified population of MSCs, with approximately one of 20 cells undergoing clonal expansion, as compared to the one out of one million typically achieved from unsorted mouse bone marrow (62). A novel and important finding of this study was that expansion in 2% oxygen culture substantially increased the proliferation rate of this cell population. In just 3 weeks, purified MSCs from B6 mice expanded greater than 100,000-fold as compared to the reported value of 10,000-fold expansion over 3 months when cultured at normoxia (57). This finding is consistent with previous studies showing low oxygen tension facilitates human, rat, and mouse MSC expansion (6,13,30,49,71,75) by providing oxygen tension levels that are more representative of the in situ bone marrow microenvironment (38).
The use of cellular therapy or cell-based tissue engineering for the regeneration of cartilage after joint injury has generated promising preclinical and case study results. With regard to PTA, the intra-articular delivery of MSCs has been shown to lessen the degree of OA in goat knees after meniscectomy and ACL resection, potentially through partial regeneration of the resected mensicus (58). Recent studies have also shown a protective effect of infrapatellar fat pad cells injected in the rabbit knee following transection of the ACL (73). Furthermore, a series of studies in mice and rats demonstrated that delivery of BMP-2 overexpressing stem cells contributed to fracture healing and cartilage repair after open osteotomy of the hind limb (82,83). There are currently eight clinical trials investigating the injection of MSCs for osteoarthritis (55), including a phase I/II clinical trial to prevent OA after meniscal tear (http://clinicaltrials.gov, #NCT00702741). In a related approach, clinical case studies have shown the ability of MSCs to produce neotissue when combined with a collagen gel and placed in cartilage defects (54,78).
The concept that MSCs may prevent PTA after intra-articular fracture is consistent with the role of endogenous stem cells after bone and cartilage injury. After long-bone fracture, MSCs arrive at the fracture site to instigate endo-chondral ossification as part of the repair process [reviewed in Marsell and Einhorn (53)]. A similar mechanism is likely to occur after intra-articular fracture, as a multipotent MSC population was derived from hemarthrosis in joints after fracture (47). Indeed, treatment of chondral defects with microfracture or other marrow stimulating techniques relies on accessing the subchondral bone marrow to allow infiltration of progenitor cells (21,41,67).
Cytokines such as IL-1β are upregulated with joint trauma (37,64) and result in cartilage degradation by suppressing matrix synthesis and inducing catabolic matrix metalloproteinase (MMP) activity (18,27). In contrast, increased systemic or joint levels of IL-1ra may disrupt the inflammatory cascade by preventing IL-1 from binding to its cell receptor (10,44). Similarly, IL-10 has been identified as an important anti-inflammatory molecule that shows a chondroprotective role in several settings of joint disease [reviewed in Schulze-Tanzil et al. (69)]. We hypothesized that MSC therapy would preserve cartilage by altering the balance of these pro-inflammatory and anti-inflammatory cytokines in the joint after injury. In studies using other injury models, MSCs decreased the systemic level of IL-1β after long-bone fracture (29), protected the lung from injury by secreting IL-1ra (60), and reprogrammed macrophages to increase IL-10 production in a sepsis model (59). In this study, stem cell treatment altered the levels of IL-1β in the synovial fluid and the presence of IL-10 in the serum at several time points. However, serum levels of IL-1β were not affected by treatment group, indicating that the strong systemic inflammatory response at early time points after fracture is not eliminated by MSC therapy.
The synovium is a likely target for the therapeutic effects of MSCs because it exhibits significant cellular activity in response to injury. However, the relationship between stem cells and the synovium is complex, as endogenous MSCs in the mouse synovium contribute to a regenerative response through chondrogenic differentiation after cartilage injury (43), but a significant inflammatory environment can alter the differentiation of progenitors in the synovium and cause a pannus-like invasion (51). Inflammation of the synovium after injury is correlated to negative outcomes in clinical studies of meniscal injury (68). Synovitis was clearly caused by fracture in this study, but stem cell therapy improved OA scores without reducing the degree of synovial hyperplasia after fracture. Since macrophages have been implicated as producers of inflammatory cytokines and other destructive molecules such as MMPs (5), we performed immunohistochemical staining for activated macrophages. Similar to the overall synovial inflammation, delivery of MSCs did not appear to mitigate the presence of activated macrophages in the synovium. However, MSCs were capable of inhibiting the proliferation of in vitro stimulated splenocytes (data not shown), consistent with their proposed immunomodulatory function [reviewed in Ghannam et al. (26)].
Bone is a joint tissue with high turnover and the capacity to respond to MSC delivery. In a long-bone fracture model, systemically delivered MSCs produced BMP-2 at the fracture site and caused an increase in callus strength, total volume, and mineralization content (29). In this study, MSCs directly delivered to the joint increased several measures of bone volume. The finding that MSC therapy prevents the decrease in bone volume to total volume ratio in the femur after fracture of the tibia suggests an influence on PTA development as opposed to enhanced healing at the fracture site only. Exogenously delivered MSCs may contribute to the prevention of PTA by providing additional cells for earlier and more robust stabilization of the joint, thus affecting the function and loading of the limb. The differentiation of endogenous MSCs to an osteoblastic lineage is essential to bone repair and is organized by multiple waves of biochemical signals such as IL-1 and TNF-α (53). Since these inflammatory cytokines are essential to early fracture healing but may be catabolic for cartilage at later times, the timing and dose of MSCs or other agents that may affect cytokine levels will need to be considered for optimal fracture repair and protection from PTA.
There is controversy in the literature about the extent of MSC engraftment when used in injury models, with some studies showing significant engraftment and differentiation but most demonstrating functional improvements with few cells remaining at the site of cell delivery [reviewed in Prockop (65)]. With systemic delivery, MSCs get trapped in the lungs before distribution to organs such as the liver and spleen (25). However, intra-articular delivery of stem cells to the knee has resulted in some engraftment in joint structures (35,46,58). For cell tracking experiments, we labeled cells with CM-DiI, which is a lipophilic carbocyanine membrane dye that has been used effectively for tracking cells in vivo in the context of bone (19). Consistent with previous studies, we observed a relatively small fraction of fluorescent cells in various tissues of the joint throughout the time course studied. However, quantification was not possible due to the challenges associated with discriminating between infrequent positive cells and background fluorescence (7) and the possibility of membrane dyes transferring to other cells over a period of time (42). New methods for quantifying the rate of engraftment and longitudinally tracking the fate of delivered cells will be an important aspect for the future development of cellular therapies (14).
The identification of MSCs with exceptional properties can help elucidate how the cells contribute to healing and may provide a pathway to modify MSCs for enhanced function. Because MRL “superhealer” mice may derive some of their regenerative capabilities from altered stem cell function (1), MSCs from both control B6 and MRL mice were compared in this model system. Our hypothesis was that MSCs isolated from MRL mice would demonstrate distinct in vitro characteristics and improved therapeutic effectiveness in vivo when compared to B6 MSCs. We found that although MRL MSCs had a higher frequency of clonogenic cells, these cells have lower expansion rates as compared to B6 MSCs. This finding was surprising given that Alfaro et al. showed enhanced proliferation of MSCs from MRL mice (1). This discrepancy may be due to differences either in isolation or expansion procedures. We used PDGFRα+/Sca-1+ sorting of cells after collagenase digestion as opposed to immunodepletion of flushed bone marrow cells for isolation, and we expanded the cells at 2% oxygen as opposed to normoxia, resulting in a much higher rate of proliferation. Regardless, the relationship between in vitro proliferation rates and in vivo functionality is unclear. While rapid expansion of MSCs is desired for in vitro studies, MSCs are typically quiescent in vivo until activated as demonstrated by the finding that 71% of freshly isolated PDGFRa+/Sca-1+ MSCs are in the G0 phase (57).
Previous studies have shown enhanced repair of cartilage defects in MRL mice as compared to B6 mice, but only if the injury extended through the subchondral bone (20). These findings led us to hypothesize that the unique regenerative potential of MRL mice may arise from a superior differentiation potential of their MSCs. Contrary to this hypothesis, B6 MSCs displayed more robust differentiation down the adipogenic, osteogenic, and chondrogenic lineage than MRL MSCs. This difference was not caused by proliferation during differentiation, as the trends remained the same when data were normalized to cell number (data not shown).
The cellular therapy experiments demonstrated similar results using MSCs from either B6 or MRL mice. One explanation is that delivering exogenous stem cells of either strain to the B6 knee after fracture is sufficient to reduce inflammation and create a joint environment that is protected from PTA, as seen with MRL mice after fracture (22,80). This is supported by work showing that MRL mice had different serum and synovial fluid IL-1β profiles after fracture (22) and that macrophages from MRL mice have lower upregulation of inflammatory cytokines (40). Indeed, experiments investigating the ear wound closure of MRL mice demonstrated that the regenerative phenotype of MRL mice is lost when the inflammatory environment is changed due to a secondary injury (85). Interestingly, there was a nonsignificant trend towards increased cartilage degradation of the nonfractured control limb with MSC therapy as compared to saline treatment, suggesting that the influence of intra-articular MSC therapy may extend to systemic effects. However, this trend may also be related to MSC therapy protecting the fractured limb from PTA and therefore affecting activity levels, although this was not investigated in this study.
Conclusions
We demonstrated that a single intra-articular injection of MSCs from either control B6 or MRL “superhealer” mice prevented the development of posttraumatic arthritis 8 weeks after intra-articular fracture of the knee. MSCs did not reduce synovitis or the presence of activated macrophages in the synovium but did alter cytokine levels and the bone healing response. This work suggests that stem cell therapy is a promising treatment for preventing PTA and could possibly be extended to explore other biological interventions after joint injury before extensive osteoarthritis has occurred.
Footnotes
Acknowledgments
The authors thank Nancy Martin and Dr. Mike Cook of the Flow Cytometry Shared Resource of the Duke Cancer Institute, as well as Stephen Johnson, Evan Zeitler, and Elisabeth Flannery for contributions to this work. Funding from NIH AR50245, AR48852, AG15768, AR48182, NSF Graduate Research Fellowship (BOD), and the Arthritis Foundation. The authors declare no conflicts of interest.
