Abstract
Previous studies using genetic-deficient murine models suggest that different T-helper subsets may contribute to different types of tissue damages in graft-versus-host disease (GvHD). However, there is limited information available on the distribution of T-helper cytokines in the various GvHD target tissues. In the current study, an acute GvHD murine model was set up to directly assess the in situ cytokine profiles in various GvHD tissue lesions; in addition, we also studied GvHD tissues from patients who had undergone bone marrow transplantation procedures. We observed that interferon-γ (IFN-γ was dominant in murine liver and gastrointestinal tissue lesions, whereas IFN-γ and interleukin 17 (IL-17) were abundant in murine skin lesions. Furthermore, in human GvHD tissues, interleukin 4 (IL-4) and IFN-γ were predominant in liver lesions and colon lesions, respectively, while no specific cytokine was prevalent in human GvHD skin lesions. In addition, a low ratio of CD4+ T helper (Th) versus CD8+ T cytotoxic (Tc) cells in human GvHD tissue lesions, especially in the liver, was detected, and this contrasts with the situation in murine GvHD tissues where CD4+ Th cells were predominant. Dual staining for CD markers and cytokine expression showed that IFN-γ-secreting T cells were enriched in all murine GvHD target tissue lesions, and Tc1 and Tc2 cells were predominant in human GvHD colon and liver sections, respectively. However, IFN-γ+ Th1, IL-17+ Th17, IFN-γ+ Tc1, and IL-17+ Tc17 cells were slightly more frequent in human skin lesions compared to IL-4+ Th2 and IL-4+ Tc2 cells. To sum up, these results suggest that differences in cytokine imbalances may significantly contribute to tissue-specific pathogenesis in GvHD target organs, and CD8+ Tc cells may play an important role in human GvHD induction.
Keywords
Introduction
Hematopoietic stem cell transplantation (HSCT) is the major therapy for malignant blood diseases (16). At present, around 25,000 HSCT procedures are carried out annually. Graft-versus-host disease (GvHD) is a critical clinical condition and the major complication of allogeneic HSCT (9). Until now, no definitive treatment is able to ensure a cure for this disease.
Acute GvHD usually affects the skin, the gastrointestinal (GI) system, and the liver. Less often, idiopathic pneumonia, an acute noninfectious lung injury, may develop around 1 month after HSCT (5). It has been demonstrated that donor T cells play a pivotal role in the induction of GvHD, but which functional subset of T cells are responsible for GvHD and how they mediate tissue inflammation remain elusive (4). Interferon-γ (IFN-γ) is the hallmark cytokine of T helper 1 (Th1) cells, whereas interleukin 4 (IL-4) has the same role in Th2 cells, while that of Th17 cells is interleukin 17 (IL-17). For many years, it has been widely acknowledged that acute GvHD is mediated by Th1 cells, which mainly secrete IFN-γ; however, it has been found that IFN-γ knockout donor T cells induce even more severe GvHD than wild-type donor T cells, which contradicts this assumption (25). Yi et al. have suggested that IFN-γ-secreting Th1 cells preferentially induce GvHD tissue damage in the gut and liver, while Th2 and Th17 cells tend to cause injury to the lungs and skin, respectively (37). In addition, blocking Th2 cell responses has been found to lead to increased GI symptoms and reduced levels of hepatic and skin damage (26). Moreover, in vitro-differentiated Th17 cells mediate lethally acute GvHD with severe cutaneous and pulmonary pathological manifestations (2). These studies raised the possibility that the various GvHD tissue lesions may be caused by different T-cell subsets and that the damage to different GvHD target organs may be caused by a specific T-helper cytokine imbalance. In this context, the question of which cytokine is predominant in various in situ GvHD tissue lesions needs to be clarified. Based on related gene-deficient murine GvHD model studies published previously, it is hypothesized that Th1 dominates in the GI and liver, while Th2 and Th17 predominate in the lungs and skin, respectively. Therefore, the purpose of this study is to explore the differences in dominant types of immune cells that are enriched in various GvHD target organs; these findings may have major implications for the role of various types of immune cells in the tissue-specific pathogenesis of GvHD.
Materials and Methods
Mice
Male C57BL/6JNarl (B6) and Balb/cByJNarl (Balb/c) mice were purchased from the National Laboratory Animal Center, Taiwan, and were bred and maintained under specific pathogen-free conditions in the Laboratory Animal Center of Taipei Veterans General Hospital, Taiwan. The age of the mice used as donors and recipients was between 10 and 13 weeks. They were housed in sterilized microisolator cages and, after bone marrow transplantation (BMT), received autoclaved water supplemented with antibiotics, 25 mg/ml neomycin (Sigma-Aldrich, USA) and 0.3 U/ml polymyxin B (Sigma-Aldrich, USA), for 2 weeks. All animal experiments were performed in accordance with protocols approved by the animal care committee of Taipei Veterans General Hospital.
Induction of GvHD in Mice
Mice received transplantation according to the protocols previously reported in the literature (29,37). Briefly, on day −1, Balb/c mice received 775 rad total body irradiation (137Cs source). On day 0, bone marrow (BM) cells and splenocytes were collected from B6 donors, and the cell suspensions were then treated with red blood cell (RBC) lysis buffer (Invitrogen Corporation, CA, USA) to remove RBCs. The cells were then resuspended in sterile injection buffer consisting of 10 mM HEPES (Sigma-Aldrich, USA), 0.5 mM EDTA (Mallinckrodt Baker, Inc., NJ, USA), and 0.5% penicillin/streptomycin (Sigma-Aldrich, St. Louis, MO, USA). Next, BM cells (1 × 107) and different doses of splenocytes (0–2 × 107) were transfused intravenously on day 0. Each recipient was administered with ≤ 200 ml of cell suspension at the time of transplantation. Recipients were monitored for survival, food intake, weight loss, and clinical GvHD symptoms twice a week.
Clinical Assessment of GvHD
The severity of systemic GvHD developed in the mice was assessed according to a mouse clinical GvHD scoring system. The scoring system for acute GvHD had six clinical criteria, and these are described in Table 1 (maximum index=11). Weight loss of <10% was scored 0, of >10% and <25% was scored as 1, and of > 25% was scored as 2. For GI symptoms, the scoring system denoted 0 as normal and 1 as suffering from diarrhea. For posture and activity, the scoring system denoted 0 as normal, while 1 was used for hunching at rest and a mild to moderate decrease in activity, and 2 was used for severe hunching and a severe decrease in activity. For fur texture and skin integrity, the scoring system denoted 0 as normal, 1 for mild to moderate fur ruffling and scaling of the paws and tails, and 2 for severe fur ruffling and an obviously denuded mouse. Each mouse's total clinical GvHD score was measured twice a week.
Mouse GvHD Clinical Scoring System

Histology
Mice with a clinical GvHD score of 6 or higher were sacrificed between days 25 and 30 after BMT. Liver, lung, skin, and GI were removed and formalin-fixed (Sigma-Aldrich, US), paraffin-embedded, sectioned, and stained with hematoxylin and eosin (H&E) (Sigma-Aldrich, US). The histopathology of the GvHD target organs was analyzed and examined with a pathologist's assistance. Images of the GvHD tissues were acquired using an Olympus AX80 microscope (Olympus, Tokyo, Japan), UPlanApo 10x/0.4, 20x/0.7 and 40x/0.85 objective lens (Olympus), a QImaging camera (Canada), and the Wright Cell Imaging facility (WCIF) ImageJ 1.37C software (National Institute of Health, USA).
Human GvHD Specimens
After informed consent, biopsies were performed as diagnostic procedures on patients receiving HSCT. Clinical grading and pathological examinations were performed by different specialized pathologists at Taipei Veterans General Hospital. Institutional review board approval was obtained prior to the commencement of the study of human specimens (VGHIRB No. 201006022IC). Tissue blocks from GvHD patients following allogenic HSCT and from non-GvHD donors were investigated, with the former selected for resectioning after histological evidence of GvHD in liver, skin, and/or colon tissue was confirmed by pathological examination. The clinical information available for the GvHD patients is summarized in Table 2.
GvHD Patients Clinical Information

Sub, subject; M, male; F, female; MDS, myelodysplastic syndrome; ALL, acute lymphoblastic leukemia; AML, acute myeloid leukemia; CML, chronic myeloid leukemia; SAA, severe aplastic anemia; PCV, polycythemia vera; TCL, T-cell lymphoma; DLI, donor lymphocyte infusion; allo-PBSCT, allogeneic peripheral blood stem cell transplantation; allo-BMT, allogeneic bone marrow transplantation.
Immunohistochemistry
Tissue sections, 3–5 mm in thickness, were prepared from formalin-fixed, paraffin-embedded specimens. Each section was first deparaffinized in xylene (Mallinckrodt Baker B.V., Deventer, Holland) and rehydrated in 100%, 95%, 70%, and 50% of ethanol (J.T. Baker, Mumbai, India) and phosphate-buffered saline (PBS) (UniRegion Bio-Tech, Taiwan) sequentially. The antigenicity of the tissue sections was restored by heat treatment in either 0.01 M citrate buffer (Sigma-Aldrich, USA), pH 6.0, or Tris-EDTA (J.T. Baker, Mumbai, India), pH 9.0, for 20 min. Next, the sections were incubated with 3% H2O2 (Shimakyu Chemical Co. Ltd., Japan) for 10 min to block endogenous peroxidase activity. Subsequent immunohistochemistry (IHC) staining was performed according to ScyTec UltraTek HRP detection system protocol (ScyTek Laboratories, Utah, USA). Briefly, sections were treated with superblock and avidin/biotin block (Sigma-Aldrich, USA) at each blocking step. Sections then were incubated with specific primary antibody at 4°C overnight. Mouse sections were treated with mouse IgG block (ScyTek Laboratories, Utah, USA) before proceeding to primary antibody incubation. After washes, the sections were incubated in UltraTek antipolyvalent solution, then UltraTek HRP solution before DAB development. Sections were washed in 0.05% PBS-Tween20 (Sigma-Aldrich, USA) between steps. If Permanent Red was used for development, UltraTek alkaline phosphotase solution was used before application of the Permanent Red development system (ScyTek Laboratories, Utah, USA). For cytokine examination, serial sections from the same tissue sample were examined.
Antibodies
To detect the expression of surface molecules on cells in the paraffin-embedded tissues by IHC, the primary antibodies used include antibodies against human/mouse CD3 (Clone LN10, Leica Biosystems Newcastle Ltd., UK), human CD4 (Clone 4B12, Leica Bio systems Newcastle Ltd., UK), human/mouse CD4 (Clone mAb51312, Abcam plc., Cambridge, UK), mouse CD4 (Clone APMAB0707, Novus Biologicals, CO, USA), human CD8 (Clone 4B11, Leica Biosystems Newcastle Ltd., UK), and mouse CD8 (Clone YTS169.4, Abcam plc., Cambridge, UK). To detect the expression of different cytokines in cells in the paraffin-embedded tissues by IHC, primary antibodies against human IFN-γ (ab9657, Abcam plc., Cambridge, UK), human/mouse IFN-γ (PAB16613, Abnova, Taiwan), mouse IFN-γ (Clone R4-6A2, Biolegend, CA, USA), human IL-4 (ab9622, Abcam plc., Cambridge, UK), mouse IL-4 (Clone 11B11, Biolegend, CA, USA), mouse IL-4 (Clone NYRmIL4, Santa Cruz Biotechnology, Inc., UK), human IL-17 (AF317NA, R&D Systems, Inc., Minneapolis, MN, USA), and human/mouse IL-17 (Clone C20, Santa Cruz Biotechnology, Inc., UK) were purchased. All tissues were stained with corresponding isotype-matched control antibodies as negative controls.
Statistics
Data in this study are expressed as mean ± standard error of the mean (SEM). Group comparisons of overall survival and pathology scores were performed using log-rank tests. Statistical analyses between groups were performed using two-way ANOVA with Bonferroni posttests. Values of p < 0.05 were considered to indicate a significant difference (GraphPad Prism).
Results
GvHD Murine Model
Lethally irradiated Balb/c mice were transfused with 1 × 107 bone marrow cells and different doses of splenocytes from major histocompatibility complex (MHC)-mismatched wild-type B6 mice. The BMT control mice received bone marrow cells only. BMT control mice had a significantly higher survival rate (100%) when compared to recipients that received both BM cells and splenocytes (Fig. 1A). The survival rate of the BMT mice decreased inversely as the dose of splenocytes transfused increased (Fig. 1A). The BMT control mice had a steady body weight change after transplantation, while the BMT mice who had received splenocytes showed markedly reduced body weights after BMT (below 50% of original body weight after 30 days) (Fig. 1B). Using the GvHD clinical scoring system (Table 1), which is modified from a previous study, the clinical status of the BMT mice was scored. In the BMT control group, the mice showed signs of few GvHD (total mean score < 1.0) throughout the period of BMT, while in the BMT mice from the splenocytes transfusion group, the clinical GvHD scores increased dramatically over time (Fig. 1C). Assessing the clinical GvHD manifestations, the BMT mice with splenocytes transfusion showed a hunching posture and a decreased activity. Furthermore, while the BMT control mice had a smooth fur texture, the BMT mice with splenocytes cotransplantation had a ruffling fur texture and obviously denuded skin (Fig. 1D). These manifestations strongly suggest the occurrence of GvHD.

GvHD mouse model. Graft versus host disease (GvHD) was induced in Balb/c mice by transfusion of both bone marrow cells (BM) and splenic T cells from B6 donors. Different doses of splenic T cells (2 × 107, n = 3; 5 × 106, n = 4; 2.5 × 106, n = 4) were transferred with 0.5–1 × 107 bone marrow cells into lethally irradiated Balb/c recipients. Animals were monitored for survival (A), body weight changes (B), and GvHD pathology score (C) twice a week. Animals receiving BM only served as controls for the GvHD (n = 5). (D) Representative photographs of recipients of BM and BM+ 2.5 × 106 splenocytes taken on day 33 after allogeneic bone marrow transplantation (BMT) are shown. Log-rank test for trend were carried out; p = 0.0013 for survival; p < 0.01 and p < 0.0004 for recipients receiving BM and splenocytes for body weight change data and pathology score data, respectively.
Characteristics of the Murine GvHD Tissues
The histopathology of liver, lung, skin, and GI tissues was assessed. In samples from the GvHD mice, we observed perivascular and peribiliary duct inflammation in the liver and interstitial/alveolar inflammation as well as perivascular cuffing and alveolar hemorrhage in the lungs. This contrasted with the BMT control mice that had a normal appearance for the central and portal areas and an intact alveolar structure with no inflammatory cell infiltration (Fig. 2A). The skin from the GvHD mice showed intense keratinization, epidermal hyperplasia with intensive collagen fibers, follicular obliteration, and dermal/subdermal inflammation with destruction of the fatty layer. The pathological GI lesions included outright crypt destruction, crypt regeneration, crypt cell apoptosis, and extensive intermucosal infiltration of the lamina propria with decreased goblet cells (Fig. 2A). These findings contrasted with the BMT control mice, where there was limited tract damage and preserved goblet cell content. Immunohistochemistry (IHC) studies also demonstrated the infiltration of CD3-positive lymphocytes into various GvHD lesions (Fig. 2B).

Histology of murine pathological GvHD tissues. GvHD was induced in Balb/c mice (n = 5) by transfusion of 1 × 107 of bone marrow cells (BM) and 2.5×106 of splenocytes from B6 donors. Animals receiving BM only served as controls for GvHD (n = 5). Histological evidence of GvHD in the liver, skin, lung, and gastrointestinal tract sections were obtained using H&E (A) or CD3 staining (B). The immunohistochemical staining sections were counterstained with hematoxylin. (C) The sections from GvHD tissues were stained with different Th cytokine antibodies. Representative pictures of immunohistochemical (IHC) staining are shown. Scale bar: 50 μm. (D) Cytokinepositive cells were counted in each of the lesions and expressed as number of cells per 100-μm2 lesion area (means ± SEM, *p < 0.05).
Cytokine Profile in the Various Different Murine GvHD Lesions
To examine the cytokine profile in the different GvHD lesions, various murine GvHD tissue sections were stained using a variety of T-helper cytokine antibodies, namely, anti-IFN-γ (Th1 cytokine), anti-IL-4 (Th2 cytokine), and anti-IL-17 (Th17 cytokine). To quantify the cytokine positivity, we measured the cytokine-positive cells in accordance with CD3-positive area in the GvHD lesions. We found that IFN-γ was the dominant cytokine in all four murine GvHD tissue lesions (Fig. 2C, D), especially in the liver. In GvHD lung and skin, IL-17 was the second dominant cytokine in the lesions, but in GvHD gut tissues, IL-4 was more abundant than IL-17 (Fig. 2C, D). To determine whether the three cytokines detected were secreted by T cells, we performed double staining of CD3 and the different cytokines in serial sections. As expected, we observed that IFN-γ-secreting T cells were predominant in all murine GvHD target tissues (Fig. 3A, B). The arrow in Fig. 3A lung photo indicates IFN-γ-positive T cells penetrating the epithelial layer lining around the alveoli. The arrow heads point to the cytokinenegative T cells in various GvHD tissues.

Distribution of T-helper and cytotoxic T cells in different murine GvHD tissues. Histological evidence of CD4+ or CD8+ T cells in different murine GvHD tissues. (A) Different T cytokine-secreting cells in murine GvHD tissues were identified by dual-color immunohistochemistry staining with CD3 and various different cytokine-specific antibodies (CD3 in pink, arrow head; cytokine in brown; doubleimmunostained cells in dark red, arrow). (B) Double-positive cells were counted, and the values are shown as mean ± SEM (*p < 0.05). (C) The images show immunohistochemical staining with CD3, CD4, and CD8 antibodies in different GvHD sections. (D) Ratio of CD4/CD8-positive cells in different murine GvHD lesions was measured and expressed as means ± SEM for each mouse. Scale bar: 25 μm.
Distribution of CD4/8-Positive Cells in Murine GvHD Lesions
To pinpoint which subtype of T cells may be involved in tissue-specific GvHD pathogenesis, we examined the distribution of CD4+ T helper cells and CD8+ cytotoxic T cells in the various murine GvHD tissue lesions. Higher numbers of CD4+ T helper cells than CD8+ cytotoxic T cells were found in all four GvHD organs (Fig. 3C, D). In skin lesions, the ratio of CD4+/CD8+ T cell counts was around 4, which was higher than the ratios of 2.6 and 2.8 for lung and gut lesions, respectively (Fig. 3C, D). In GvHD liver lesions, the number of T helper cells was only slightly higher than that of cytotoxic T cells (ratio = 1.8) (Fig. 3C, D).
Characteristics of Human GvHD Tissues
The histology of human GvHD tissues was analyzed by selecting GvHD tissue blocks for resectioning with a pathologist's assistance. Clinical information on the GvHD patients following HSCT is presented in Table 2. The resectioned human GvHD sections were stained with CD3 to reconfirm the presence of GvHD damaged lesions with lymphocyte infiltration (Fig. 4A).
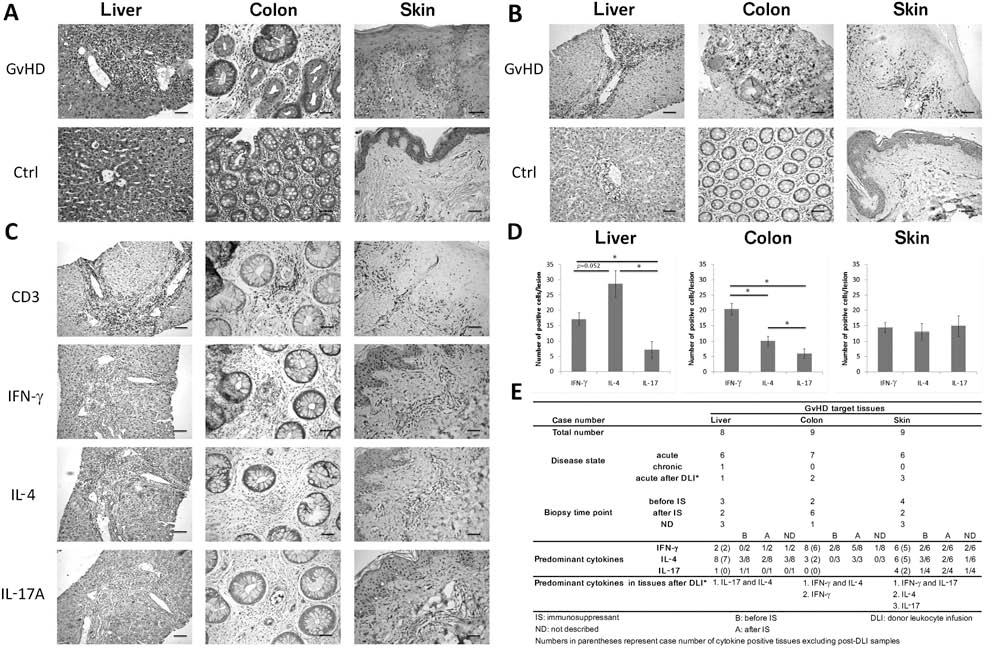
Histology of human pathological GvHD tissues. Human GvHD sections were obtained from patients after BMT transplantation. (A) H&E and (B) CD3 staining of liver, colon, and skin tissue sections of GvHD patients and non-GvHD donors (Ctrl, control). Histological evidence of GvHD in the liver, skin, and colon sections are shown. (C) The images show immunohistochemical staining with CD3, IFN-γ, IL-4, or IL-17 antibodies in different GvHD sections and mean±SEM of cytokine-positive cells in each 100-μm2 lesion area (*p < 0.05) (D). (E) Stratification of GvHD patients according to their disease state and biopsy time point. Two predominant cytokines may be present in one GvHD target tissue. Scale bar: 50 μm.
We focused on the three target tissues in this study of acute human GvHD, namely, colon, liver, and skin. In normal colon tissues, lymphocytes mainly reside in lamina propria, and the intestinal glands are intact. However, in the GvHD colon tissues, large numbers of lymphocytes have infiltrated the lamina propria area, destroying the integrated crypts and penetrating through the lining epithelium (Fig. 4A). Large numbers of disorganized glands can also be observed. In GvHD liver tissue, the sections show bile duct damage and lymphocyte infiltration within the epithelial layer in liver portal areas (Fig. 4A, B). Increased numbers of T cells in the sinusoidal portal, periductal, and centrilobular areas were also observed. These findings contrast with the situation in normal liver tissue, where the portal triads are without inflammatory cell infiltration and are surrounded by plates of hepatocytes and sinusoids (Fig. 4A). In normal skin tissue, the epidermis and dermis layers are tightly linked, while in the GvHD skin tissue, vacuoles have appeared, and adhesion between the epidermis and dermis layers has been lost (Fig. 4A, B). Meanwhile, great numbers of lymphocytes can be seen to have infiltrated into the epidermal–dermal junction (Fig. 4A, B). All the above features are compatible with the presence of GvHD.
Cytokine Profile in Different Human GvHD Lesions
To compare cytokine profiles in the various GvHD tissues, we quantified the expression of cytokines by microscopic observation with at least two different cytokine antibodies being used to confirm the expression of each cytokine. Significantly more IFN-γ-positive cells were present in the GvHD colon lesions than IL-4- or IL-17-positive cells. Figure 4C shows ongoing damage to a crypt that is mostly surrounded by IFN-γ-positive cells.
As for GvHD liver tissues, lymphocyte infiltration into portal areas can be seen (Fig. 4A, B). A more diffused cytokine IHC of the liver tissue was observed compared to the colon. If the H&E- and CD3-stained sections are examined, it can be seen that IL-4 and IFN-γ are predominant in the lesion areas. Especially, IL-4 positivity was greater than IFN-γ positivity in the liver lesions. However, no specific cytokine was dominant in human GvHD skin lesions (Fig. 4C).
GvHD tissues from patients were stratified according to their disease state and biopsy time points. Most tissues were obtained from patients with acute GvHD, with the exception of one liver tissue sample. This liver sample was collected a few years after the patient received HSCT (Fig. 4E). Some GvHD tissues were sampled after donor leukocyte infusion (DLI) treatment, which is commonly used to treat patients experiencing leukemia relapse; such treatment may induce GvHD. The liver tissue obtained during the chronic GvHD phase showed that IL-4 and IFN-γ were predominant (data not shown). The GvHD liver tissue obtained after DLI treatment showed IL-17 and IL-4 enrichment, while the post-DLI GvHD colon tissues were IFN-γ or IFN-γ and IL-4 as predominating (Fig. 4E). The predominant cytokines in specific tissues were further grouped, taking into account the immunosuppressant treatment (Fig. 4E). Based on our results, cytokine predominance of patients was not affected by immunosuppressant use.
To identify whether the dominant cytokine in different GvHD tissues was produced by T-helper cells, the tissues were double stained with anti-CD4 and anti-cytokine antibodies or anti-CD8 and anti-cytokine antibodies. Surprisingly, we observed that CD8+ Tc cells were predominant in all three human GvHD lesions. Tc2 cells (CD8+ IL-4+ cells) were the most abundant cells in human GvHD liver, while Tc1 cells (CD8+ IFN-γ+ cells) were most abundant in GvHD colon (Fig. 5A). In human GvHD skin, Tc1 and Tc17 cells (CD8+ IL-17+ cells) seemed equally predominant (Fig. 5B).

CD8+ cells are dominant in human GvHD tissues. (A) Different T subsets in human GvHD tissues were identified by detecting the expression of CD4 or CD8 and different cytokine using dual-color immunohistochemistry (CD4 or CD8 in pink; cytokine in brown; doubleimmunostained cells in dark red). (B) Double-positive cells were counted in at least 10 lesions of each tissue. Th1 stands for CD4+ IFN-γ+ cells; Th2, CD4+IL-4+ cells; Th17, CD4+IL-17+ cells; Tc1, CD8+IFN-γ+ cells; Tc2, CD8+IL-4+ cells; Tc17, CD8+ IL-17+ cells. The values are shown as mean±SEM. (C) The staining images showed CD3-, CD4-, and CD8-positive cells in a colon and a liver lesion. (D) Ratio of CD4/CD8-positive cells in different GvHD lesions was measured, and data are expressed as means±SEM of individual patients. Scale bar: 25 μm.
Distribution of CD4/CD8 in Human GvHD Lesions
When the CD4/CD8 ratio was examined, in liver lesions, only around 10% of CD3+ cells were CD4 positive (Th cells); this contrasted with the situation in colon and skin lesions where about 24% and 32% of CD3+ cells were CD4 positive (data not shown), respectively. CD8+ Tc cells were predominant in human GvHD lesions, especially in liver tissue, although a similar pattern for the various murine and human GvHD tissues examined was detected (Figs. 3D and 5D). The CD4/CD8 ratio was higher in GvHD skin lesions (0.47 ± 0.05) compared to the other two types of GvHD tissue lesions (Fig. 5D). In GvHD skin tissue lesions, the cells that had penetrated into epidermis were mainly CD8+ Tc cells, while CD4+ Th cells were mainly distributed between dermis and epidermis layers (data not shown).
Discussion
The pathophysiology of GvHD is known to be a process involving damage to host organs by activated donor T cells, and this is caused by an imbalance in the cytokine profile following allogeneic transplantation (8,17,35). Previous work has shown that donor T cells that are incapable of secreting IFN-γ, IL-4, and IL-17 cytokines induce different types of tissue damage in recipients (1,13,25,26,37), suggesting these proinflammatory cytokines play an important role in contributing to tissue-specific GvHD pathogenesis. However, little is known about the in situ cytokine distribution in different GvHD tissue lesions. To further elucidate this cytokine scheme, we investigated the cytokine profiles in lesional tissue biopsies from GvHD target organs; these were obtained from mice and humans following allogeneic HSCT.
Our results demonstrate that Th1 cells are enriched in both murine and human GvHD GI lesions and that low IL-4 and IL-17 are detected in these lesions, suggesting a prominent role for IFN-γ in mediating GvHD pathogenesis in the GI system. This is consistent with the findings of previous studies using genetic-deficient model systems (1,7,11). In one of these earlier studies, transplantation of IFN-γ-/- donor cells significantly reduced pathological damage to the recipient's colon compared to wild-type donor cells (1). In another of these studies, mothers against decapentaplegic homolog 3 (SMAD3) deficiency resulted in Th1 bias, and this increased the level of tissue damage to the colon (11). Finally, Das et al. demonstrated that IFN-γ-dependent IL-23 had a role in the pathogenesis of colon GvHD (7); specifically, significantly less CD4+ IFN-γ+ Th1 cells were detected in colon tissue lesions when IL23-/- donor BM cells were injected compared to wild type donor BM cells. However, this phenomenon was not observed in another GvHD tissue, namely, the liver, suggesting a tissue-specific role for IFN-γ in the pathogenesis of GI GvHD. As suggested by earlier reports, Th1 cells are enriched in murine GvHD liver lesions (Fig. 2C, D), indicating they play a prominent role in liver pathology (10,37). However, IL-4 was also found to be abundant in human GvHD liver lesions in this study. Although several studies have indicated that Th2 cytokine is not critical for inducing GvHD (18,37), blockage of signal transducer and activator of transcription 6 (STAT6) in donor T cells (Th2-deficient cells) has been shown to result limited liver or skin lesions in recipients (26), and the absence of IFN-γ has been found to result in increased levels of liver pathology after allogeneic BMT in mice (25). In an IFN-γ receptor-/- recipient murine BMT model, comparable liver pathology was observed in both IFN-γ signaling-deficient and wild-type recipients (1). Taken together, these results suggest a possible role of IL-4 in the liver-specific pathogenesis of GvHD. Previous studies have shown that Th17 cells are critical for GvHD skin pathology (2,12,37). In the present study, although Th1 was dominant in murine GvHD skin lesions, an elevated Th17 frequency was also detected. A similar pattern was also observed in the human GvHD skin lesions. However, all three Th cytokines were abundant in human skin lesions, which is consistent with the results from a previous study showing increased expression of IL-2, IL-4, and IFN-γ mRNA in skin biopsies from GvHD patients (31). A clinical report investigating the Th17 population in GvHD patients found that Th17 cells can be separated into two subtypes, namely, IL-17+ IFN-γ+ cells and IL-17+ IFN-γ- cells. Only IL-17+ IFN-γ+ cells, which express the IL-23 receptor, are able to infiltrate GvHD liver and skin lesions (6), suggesting both IFN-γ and IL-17 are important for the pathogenesis of GvHD skin. GvHD can also cause lung problems. Although there is some clinical evidence to suggest that the lungs are a critical target organ for acute GvHD (21,24), pulmonary injury is mainly a manifestation as part of chronic GvHD. Multiple factors, including infection, contribute to pulmonary complications (14), and normally, human lung tissues are not included in GvHD studies. However, in many murine acute GvHD models, noninfectious lung injury and idiopathic pneumonia are considered to be acute GvHD complications (1,20,32,37). Thus, murine pulmonary tissues were examined in this study. Previous studies have suggested that IL-4 and IL-17 are critical for GvHD lung pathology (1,2,23,34,37). Yet, in our murine GvHD model, lung tissues were found to show limited IL-4 positivity and only weak IL-17 positivity.
The same cytokine may possess both proinflammatory and protective properties under different circumstances. From the perspective of protection, different cytokines may play roles in defending the body from tissue-specific GvHD. Burman et al. reported that IFN-γ exerted a protective role in the development of idiopathic pneumonia syndrome after stem cell transplantation (1). It has been found that a lack of Th2 cytokine in donor T cells leads to increased GvHD mortality and GI symptoms in mice, supporting a protective role for IL-4 (36). The role of IL-17A is controversial. While an absence of IL-17A has been found to lead to Th1 skewing and thus a worsening of GvHD GI pathology (27,38), in contrast, mice survived longer when IL-17A- Th donor cells were used rather than wild-type donor cells (13). It has been proposed that a shift from Th1 to Th2 cytokines indicates the development of tolerance (15,19,33). Thus, an imbalanced cytokine milieu may therefore be a critical factor in the pathophysiology of GvHD. To elucidate the detailed mechanisms behind the complex cytokine interaction that may be occurring in GvHD target tissues, further research is needed.
Prior studies have shown that Th cells are responsible for GvHD pathogenesis and that CD8+ Tc cells have a lower ability to induce GvHD (3,28,38). In the present study, it was observed that there were higher numbers of Th cells compared to Tc cells in different murine GvHD tissues. Conversely, in human GvHD lesions, the majority of T cells were CD8+ Tc cells, especially in liver lesions, which suggests a pathogenic role for CD8+ Tc cells in human GvHD tissue damage. Zheng et al. reported that highly purified central memory CD8+ T cells clearly induced GvHD and that the clinical pathology was manifest especially in liver (40). Zhao et al. also utilized CD8+ Tc cells to induce GvHD liver and intestine pathology (39). Furthermore, an early clinical study demonstrated that depletion of CD8+ Tc cells prevented GvHD after BMT (22), all of which support a prominent role of Tc cells in promoting GvHD.
In the present study, variation in Th/Tc dominance in GvHD lesions and the observed disparate cytokine patterns in murine and human GvHD liver tissue may be due to different MHC matching levels. The murine BMT model used in this study is a MHC-mismatched B6 to Balb/c model of allogeneic BMT. In conventional BMT procedures, the donor must have a human leukocyte antigen (HLA) type matching that of the recipient or show a very close degree of HLA matching. Another possible factor is the existence of malignant cells in the human recipients. All human specimens came from GvHD patients with a malignant blood disease (Table 2). In human recipients, malignant cells may modulate the immune response, and this may have led to the shift from CD4+ cell domination to CD8+ cell domination in the target organs analyzed. Furthermore, variations in the murine model strains may also cause variation in the results of the study. To verify the present results, murine models of graft-versus-leukemia (GvL) or different strain combinations of mice need to be investigated (30). In addition, during the human clinical procedures, immunosuppressant or conditioning regimens are always applied to prevent GvHD and to enhance GvL activity, which may also be the reason for the disparate results between murine GvHD model and the human disease. Thus, although the clinical manifestations in murine GvHD models seem to closely correlate with human GvHD, the above factors limit the direct translation of the murine GvHD model to human disease.
Undoubtedly, GvHD pathogenesis in regulation involves complex cytokine interactions and variations in cytokine functionality. Nonetheless, differences in cytokine imbalance seem to be the cause of tissue-specific pathogenesis in different GvHD target organs. In addition, CD8+ Tc cells may be as equally important as CD4+ Th cells in the development of human GvHD. To the best of our knowledge, this is the first study to provide information on the in situ cytokine profiles of various GvHD target lesions in mice and humans. However, further efforts are still needed if the various roles of the different cytokines in GvHD tissue-specific pathogenesis are to be elucidated.
Footnotes
Acknowledgments
The authors thank Zi-Ying Wu and Chun-Yen Hsiao from the Department of Pathology, Taipei Veterans General Hospital, for helping with human GvHD tissue sectioning. We also thank Kuan-Wen Wang from Taipei Veterans General Hospital for collecting the patient clinical information. This work was assisted in part by the Division of Experimental Surgery of the Department of Surgery, Taipei Veterans General Hospital, and supported by HealthBanks Biotech Co. and the UST-UCSD International Center of Excellence in Advanced Bioengineering sponsored by the Taiwan National Science Council I-RiCE Program under Grant No. NSC-100-2911-I-009-101. The authors also acknowledge financial support from the Taipei Veterans General Hospital (VGH101E1-012, VGH101C-015, and VN101-07), the National Science Council, Taiwan (NSC101-2120-M-010-002, NSC100-2314-B-010-030-MY3, NSC101-2321-B-010-009, NSC101-2911-I-010-503, and NSC 99-3114-B-002-005), the Ministry of Economic Affairs, Taiwan (101-EC-17-A-17-S1-503), and a grant from National Research Program for Biopharmaceuticals (DOH101-TD-PB-111-NSC014), as well as the National Yang-Ming University/Cheng Hsin General Hospital Grant (101F195CY07). This study was also supported by the Aim for the Top University Plan from the Ministry of Education. The authors declare no conflicts of interest.
