Abstract
Double-unit umbilical cord blood (DU-UCB) may extend the use of UCB transplantation and improve clinical outcomes. Data in the literature show that single-unit dominance happened in a vast majority of recipients, and the mechanism is unknown. We examined the clinical relevance and engraftment kinetics of DU-UCB transplant in 65 consecutive children who underwent unrelated single-unit (n = 25) and double-unit (n = 40) UCB transplantation for various hematological malignancies (n = 45) and nonmalignant disorders (n = 20). Our result showed no discernible benefit to children receiving double-unit transplant over those receiving single-unit transplant when the total nucleated cell (TNC) doses are ≥2.5 × 107/kg, in terms of the hastening of the engraftment of neutrophils and platelets, reduction of nonengraftment, disease recurrence, early mortality, and graft-versus-host disease, despite significantly higher numbers of TNCs in double units. Further analyses demonstrated that the phenomena were not associated with underlying disease, duration of UCB storage, postthaw viability, HLA disparity, ABO incompatibility, gender, or doses of TNCs, CD34+ cells, CD3+ cells, or colony-forming units. Engrafting units in DU-UCB transplants were notably associated with higher CD34+ cell dose. Chimerism studies demonstrated that single-unit dominance started before neutrophil engraftment in DU-UCB transplants. Data from the study suggested no advantage of infusing double-unit UCB, if an adequately dosed single-unit UCB is available. Successful prediction of the dominant graft would optimize algorithms of UCB selection and maximize the long-term engraftment of chosen units.
Introduction
Umbilical cord blood (UCB) transplantation has been increasingly used for patients with hematological malignancies and nonmalignant disorders after the first report on successful application in a child with Fanconi anemia in 1988 1 . The advantages include availability in a short time period, an unlikelihood of donor attrition compared to bone marrow donation, higher tolerance of HLA disparity between recipient–donor pair, and a reduced risk of graft-versus-host disease (GvHD) 2 . UCB transplantation has been limited to children and adults of low body weight because of a finite number of cells in a single unit. Various strategies to overcome the hurdle have been explored in clinical settings, including ex vivo expansion of hematopoietic stem cells, intraosseous administration, and modulation of the homing signal to bone marrow3–5. So far, the most commonly used approach appeared to be the use of double-unit UCB (DU-UCB) transplantation6–10.
In view of reports of promising results, we conducted an open-label study of DU-UCB transplantation at the Lady Pao Children's Cancer Centre at the Prince of Wales Hospital in Hong Kong. The objectives were to compare double-unit and single-unit UCB transplantation in terms of engraftment kinetics, chimerism, incidence of acute and chronic GvHD, relapse, disease progression, primary graft failure, and mortality on day ≤100 posttransplant.
Materials and Methods
Patients
Sixty–five children and adolescents (21 girls and 44 boys, female/male = 1:2.2) of a median (range) age of 5.3 (0.7–21.2) years and body weight of 18.9 kg (5.3–50.5 kg) were studied. They received first unrelated UCB transplantation. Patients receiving second or third UCB transplantation were excluded. The underlying diseases included hematologic malignancies (n = 45): high-risk acute leukemia, juvenile myelomonocytic leukemia, therapy-related myelodysplastic syndrome, or T-cell lymphoma and nonmalignant diseases (n = 20): β-thalassemia major, Fanconi's anemia, severe aplastic anemia, adrenoleukodystrophy, mucopolysaccharidoses, Langerhans’ cell histiocytosis, hemophagocytic lymphohistiocytosis, or dyskeratosis congenita. Based on the availability of UCB (see the following paragraph), patients were transplanted with either a single unit (n = 25) or a double unit (n = 40) of UCB (Table 1). The inclusion criteria were patients with diseases and clinical conditions suitable for transplant and without an HLA-identical sibling or unrelated donor. Written informed consents from the parents or legal guardians of minors were obtained. Assents were also obtained from patients at age of ≥8 years. The protocol was approved by Clinical Research Ethics Committee of The Chinese University of Hong Kong and New Territories East Cluster of Hong Kong Hospital Authority in 2006.
Demographics of 65 Pediatric Patients Who Underwent Unrelated UCB Transplant

Acute biphenotypic leukemia, therapy-related myelodysplastic syndrome, myelodysplastic syndrome, juvenile myelomonocytic leukemia, and T-cell lymphoma.
Adrenoleukodystrophy, mucopolysaccharidosis, and I-cell disease.
Langerhans’ cell histiocytosis, hemophagocytic lymphohistiocytosis, and dyskeratosis congenital.
Umbilical Cord Blood
The selection of UCB units was based on doses of total nucleated cells (TNCs) and CD34+ cells, and HLA matching to the recipient and HLA matching between the two units of UCB in DU-UCB transplants. The minimum dose of TNCs for single-unit UCB transplants was 2.5 × 107/kg recipient body weight. In DU-UCB transplants, a required dose for TNCs and CD34+ cells was 4 × 107/kg and 1.5 × 105/kg, respectively. At least four of six loci of HLA were matched at the antigen level for HLA-A and HLA-B and at the allelic level for HLA-DRB1 between UCB (single or double) and recipient.
A total of 105 UCB units were transplanted into 65 patients between June 1998 and September 2014. Seven (6.7%) were obtained from overseas public UCB banks (Australia, China, Germany, Italy, Taiwan, and US), and 98 (93.3%) were supplied by the local public UCB bank of Hong Kong Red Cross Blood Transfusion Service.
Transplantation
Patients were conditioned with disease-dependent institutional protocols of myeloablative regimens by using different combinations and doses of multiple cytotoxic agents including busulphan (Otsuka Pharmaceutical Co., Ltd., Tokyo, Japan), cyclophosphamide (Baxter Healthcare Ltd., Norfolk, UK), etoposide (Sandoz Inc., Princeton, NJ, USA), fludarabine (Sandoz), melphalan (Aspen Pharma, NSW, Australia), thiotepa (RIEMSER Pharma GmbH, Insel Riems, Germany), and anti-thymocyte globulin (Sanofi-Aventis, Quebec, Canada) with and without total body irradiation. Cyclosporine (Novartis AG, Basel, Switzerland) and methylprednisolone (Teva Pharmaceutical Industries, Petah Tikva, Israel) were used for GvHD prophylaxis. In 2007 onward, cyclosporine and mycophenolate mofetil (Roche Pharmaceuticals, Basel, Switzerland) were administered. On the day of transplantation, a red cell-depleted UCB unit was processed in the cell processing laboratory next to the Bone Marrow Transplant (BMT) ward. Details of the processing procedure were reported elsewhere 11 . In brief, UCB units of >25 ml were diluted and washed twice in cold dextran 40 (Hospira, Lake Forest, IL, USA) and 5% normal human albumin (Hong Kong Red Cross Blood Transfusion Service). Cells were resuspended in an appropriate volume of dextran–albumin solution and infused immediately at a rate of 5–10 ml/min. UCB units frozen in 25 ml were not washed to retain the maximal number of TNCs. Infusions were completed within an hour of thawing. In DU-UCB transplantation, the second unit was processed and infused without a hiatus. Successful neutrophil engraftment was defined as the recovery of absolute neutrophil count ≥0.5 × 109/L for at least 3 consecutive days, and the first of 3 consecutive days of achieving such counts was regarded as the day of neutrophil engraftment. Similarly, the day of platelet engraftment was the first of 3 consecutive days with platelet counts of ≥20 × 109/L without transfusion for at least 7 days.
Assessments
Assessments of TNCs, CD34+ cells, CD3+ cells, colony-forming units (CFUs), and viability were performed on an aliquot from thawed UCB. Viability was determined by a trypan blue (Invitrogen, Carlsbad, CA, USA) dye exclusion test. Flow cytometry was used to enumerate CD34+ cells and CD3+ cells. A clonogenic assay was employed to enumerate CFU. FISH analysis for X and Y chromosomes was performed to assess chimerism in sex-mismatched single-unit UCB transplants. Chimerism studies were performed on mononuclear cells of UCB, bone marrow, or peripheral blood samples from recipients pretransplant and at weekly intervals for the first 3 months posttransplant and then at 6, 8, 12, and 24 months posttransplant. Details of methodologies including the specificity and sensitivity of FISH were reported elsewhere 12 . Both false positivity and false negativity were ≤0.4%.
Quantitative polymerase chain reaction (qPCR) for short tandem repeat (STR) was used to determine chimerism in other cases. Informative STR were amplified with four sets primer pairs (D19S253, D5S818, D11S554, and DXS9902) by running 30 cycles of PCR in a thermal cycler (GeneAmp PCR 9700; Applied Biosystems, Carlsbad, CA, USA). Amplicons were separated and analyzed quantitatively with a DNA sequencer and GeneScan software (ABI PRISM 3130; Applied Biosystems). STR analysis was sensitive for detecting chimerism of 0.05%. The qPCR for STR was performed with the methods based on the recommendation of United Kingdom National External Quality Assessment Service for Leucocyte Immunophenotyping Chimerism Working Group 13 .
Acute and chronic GvHD were graded according to the established criteria and treated with steroids14,15. Mycophenolate mofetil and anti-thymocyte globulin were administered as second line treatment.
Data Analysis
Data are presented as median and range, unless stated otherwise. Categorical variables related to patients, disease, UCB transplants, incidence of nonengraftment, relapse, and mortality on days ≤100 were analyzed with Fisher's exact test, if sample sizes were small, or chi-square tests. The nonparametric Mann–Whitney U test was used to analyze continuous variables such as viability, doses of TNCs, CD34+ cells, CD3+ cells, and CFU, length of UCB storage and days of neutrophil and platelet engraftment derived from single-unit and DU-UCB transplants as well as engrafting and nonengrafting UCB. “Event” was defined as death, relapse, nonengraftment, graft rejection, or disease progression. Overall survival and event-free survival were evaluated by using the log-rank test in Kaplan–Meier analysis and the SPSS statistical platform version 21.0.0 (SPSS; IBM, Armonk, NY, USA). The time-dependent Cox proportional hazard regression model was used to estimate the cumulative incidence of disease relapse and GvHD from the time of transplantation. Competing risks of disease recurrence and death without GvHD were excluded in assessing cumulative incidence of chronic GvHD. Differences in values between tested groups were regarded as significant at a value of p ≤ 0.05.
Results
Patient Characteristics
Starting June 1998, 65 eligible patients, who received either single-unit (n = 25) or double-unit (n = 40) unrelated UCB transplants, underwent myeloablative conditioning according to the institutional protocols. Characteristics of the patient cohort are detailed in Table 1. From 2006 onward, DU-UCB transplantation was more frequently performed; however, patients having transplantation with single-unit or DU-UCB were comparable in terms of sex, age, body weight, and underlying diseases.
UCB Transplant Characteristics
Upon thawing, 94.2% (99/105) UCB units were washed to reduce the residual toxicity of the cryoprotectant dimethyl sulfoxide (DMSO) and cell lysate prior to infusion. The median (range) of cell viability was 83.8% (55–100%). While UCB in double-unit transplantation was stored significantly longer than in single-unit transplantation, there was no discernible compromise of viability upon thawing (Table 2). Full HLA matching was more readily available in single-unit transplantation than double-unit transplantation. Major and minor ABO incompatibilities were frequently encountered in DU-UCB transplantation; nevertheless, transplantation was uneventful at the time of the infusion. Sex matching was 10% (4/40) among recipients and the two units of UCB in double-unit transplants, compared to 52% (13/25) of patients in single-unit transplants (p < 0.01).
Characteristics and Outcomes of 65 Unrelated UCB Transplants
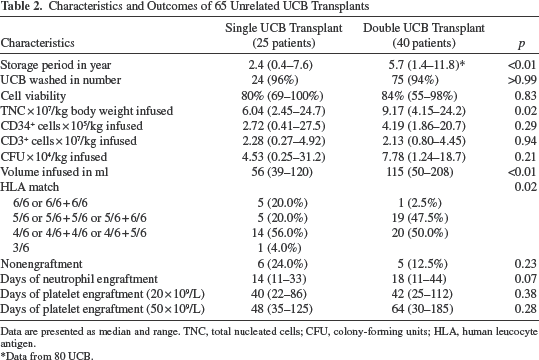
Data are presented as median and range. TNC, total nucleated cells; CFU, colony-forming units; HLA, human leucocyte antigen.
Data from 80 UCB.
Transplantation and Engraftment
Table 2 shows a larger infusion volume of a higher dose of TNCs in double–unit transplants, but comparable doses of CD34+ cells, CD3+ cells, and CFU. Engraftment was achieved in 83.1% (54/65) of the patient cohort at a median of 16 days (range: 11–44 days). Nonengraftment was observed in 16.9% (11/65) of patients and was not associated with the underlying disease [malignant vs. nonmalignant: 17.8% (8/45) vs. 15.0% (3/20), p > 0.99], duration of UCB storage, viability upon thawing, degree of HLA disparity, ABO incompatibility, gender differences (data not shown), infused doses of TNCs, CD34+ cells, CD3+ cells, and CFU per kilogram of body weight and numbers of infused UCB units (Table 3). However, there was a significantly higher nonengraftment rate in patients transplanted with UCB from overseas [overseas UCB vs. local UCB: 66.7% (4/6) vs. 11.9% (7/59), p = 0.01]. Among six children (infantile ALL, ALL, AML, MDS transformed to AML, JMML, and JMML transformed to AML) transplanted with a total of seven overseas units, only two (AML and JMML transformed to AML) were successfully engrafted. Median days for engraftment of neutrophils and platelets for patients receiving double-unit transplantation appeared longer than those receiving single-unit transplantation, though not statistically significant (Table 2).
Characteristics of Engrafting and Nonengrafting Units in 65 Unrelated UCB Transplants

Data are presented as median and range. TNC, total nucleated cells; CFU, colony-forming units; HLA, human leucocyte antigen.
Chimerism and Dominance
Complete donor chimerism was defined as >95% donor cells, irrespective of single or double donors, and <5% autologous cells. Mixed chimerism was identified when there were 10–95% donor cells and >5% autologous cells. In double-unit transplantation, predominance was regarded as one unit persistently contributing to >70% of the donor chimerism.
FISH and STR analyses were available in 98.1% (53/54) of successfully transplanted patients. Complete chimerism of donor cells was detected in 90.6% (48/53) of patients. Single unit predominance of >70% of donor cells in patients after DU-UCB was demonstrated in 88.6% (31/35) of successfully transplanted patients, and the predominance happened earlier than neutrophil engraftment occurred in 57.1% (20/35) [days: single-unit predominance vs. neutrophil engraftment: 14 (5–28) vs. 18 (11–44), p < 0.01]. Predominance reversion (decline of the proportion of cells of the predominating unit with the reciprocal replacement by the other unit) was not noted in the study. Sustained mixed chimerism was identified in 9.4% (5/53) of successfully transplanted patients (one ALL, one hemophagocytic lymphohistiocytosis, one mucopolysaccharidosis, and two thalassemia major) and prevailed in patients transplanted for nonmalignant disorders compared to patients transplanted for hematological malignancies [23.5% (4/17) vs. 2.8% (1/36), p = 0.03].
Table 4 shows the characteristics and cellularity of the engrafting and nonengrafting UCB in 30 successful double-unit transplants. Mixed chimerism (n = 5) with recipient cells >5% and absence of single-unit dominance were excluded. Apart from the infused dose of CD34+ cells, there was no association of the engrafting UCB with the duration of storage, viability, doses of TNCs, CD3+ cells, or CFU per kg body weight, HLA matching, ABO incompatibility, or gender differences.
Characteristics of Double-Unit UCB Transplants Having Achieved Complete Chimerism of ≥ 95% Cells of Donor Origin

Data are presented as median and range. TNC, total nucleated cells; CFU, colony-forming units; HLA, human leucocyte antigen.
GvHD and Mortality
Among 83% (54/65) of patients with successful engraftment, 85.1% (46/54) developed acute GvHD of which grade III/IV accounted for 19.6% (9/46). Chronic GvHD was observed in 45.8% (22/48) of successfully transplanted patients who survived more than 100 days. Among 22 patients with chronic GvHD, 17 (77.3%) limited to skin and 5 (22.7%) had extensive involvement. Table 5 displays the cumulative risks of acute/chronic GvHD and leukemia relapse, and there was no significant difference between single-unit and DU-UCB transplants. The cumulative risks of acute/chronic GvHD were similar in children transplanted for hematological malignancies or nonmalignant disorders (Table 6).
Events of GvHD, Relapse, and Mortality on Day ≤100 Among 54 Successfully Transplanted Pediatric Patients Having Undergone Unrelated UCB Transplantation

GvHD, graft versus host disease.
n = 48, excluding mortality on day ≤100.
Events of GvHD and Mortality on Day ≤100 Among 54 Successfully Transplanted Pediatric Patients Having Undergone Unrelated UCB Transplantation
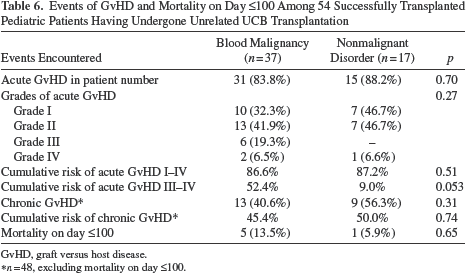
GvHD, graft versus host disease.
n = 48, excluding mortality on day ≤100.
The mortality rates on day ≤100 were comparable among patients having undergone single-unit or DU-UCB transplantation. With a median follow-up of 24 months (range: 1–192 months), the estimated 3-year overall survival probability ± standard error of the entire patient cohort was 57.5 ± 6.2%. There was no difference in the overall survival estimate between single-unit and double-unit transplants (52.3 ± 10.5% vs. 57.8 ± 7.8%, p = 0.56). The 3-year event-free survival (EFS) of the cohort was 54.3 ± 6.3%. Similarly there was no difference in the EFS estimate among patients receiving single-unit and double-unit transplant (48.1 ± 10.6% vs. 58.2 ± 7.8%, p = 0.43). Patients who underwent UCB transplantation for nonmalignant diseases had a better EFS estimate compared to that of patients transplanted for hematological malignancies (73.3 ± 10.4% vs. 46.4 ± 7.5%, p = 0.03).
Discussion
In this study, we evaluated the efficacies of DU-UCB transplantation in children with hematological malignancies and nonmalignant disorders. We found no discernible benefit to recipients of double-unit transplants for cell doses ≤2.5 × 107 TNCs/kg in terms of engraftment, early mortality, disease progression, overall survival, or event-free survival. On the contrary, longer periods of neutrophil and platelet engraftment were noted among children transplanted with DU-UCB even with significantly higher TNC and CD34+ cell doses. There was no difference in the incidence and extent of acute and chronic GvHD among children who underwent single-unit and double-unit transplantation, despite more frequent HLA disparity and ABO incompatibility among double-unit recipients.
The limited number of cells in a UCB donation hinders its application in recipients with higher body weight for fear of the likelihood of delayed engraftment, nonengraftment, and mortality. Two units of UCB containing greater numbers of cells may extend the use of UCB transplantation and improve clinical outcomes in patients for whom a single unit of UCB of >2.5 × 107 TNCs/kg body weight is not sufficient 16 . The choice of employing single-unit versus DU-UCB transplantation depended much on the available nucleated cell dose of the recipient. A widely accepted minimal threshold was initially set at 1.5 × 107 TNCs/kg and was increased to 2.5 × 107/kg in the early 21st century. The set point was further readjusted according to degree of HLA matching, 3.4 × 107/kg for HLA matching six out of six antigens and 4.0 × 107/kg for five out of six antigens.
In patients with a likely risk of graft rejection such as those with thalassemia major, a higher stem cell dose may be beneficial for successful engraftment. In this study, 87.5% (7/8) of thalassemia major patients transplanted with DU-UCB had successful engraftment. The availability of functional cells as a prerequisite for successful transplantation depends on the processing procedure, cryopreservation, storage, transportation, thawing, and infusion. Multiprocedural variations may induce substantial physical and biological losses and injuries on stem cells. If these are not properly standardized and validated, the integrity of allografts may be adversely affected for transplantation. The evaluation of cell numbers alone may not be an effective means for graft selection. It was noteworthy that UCB units of low viability were not uncommon 17 . Viability <75% were noted in 19% (20/105) of UCB allografts in the study. Nonetheless, the duration of proper storage at an ultra-low temperature would not compromise the viability and engraftment potential of the red cell-depleted UCB upon thawing and processing for immediate infusion.
Myeloengraftment has been well defined by the rebound of absolute neutrophil counts of ≥0.5 × 109/L; however, the status of donor chimerism in recipients posttransplant is also informative for engraftment kinetics and graft failure. Data from the current study demonstrated mixed chimerism at different ratios of donor cells and recipient cells shortly after transplantation. Complete chimerism and single-unit predominance in recipients having undergone double-unit transplantation were evident before neutrophil engraftment. The dominant engraftment of single-unit UCB persisted irreversibly posttransplant. Stable mixed-unit chimerism prevailed in our cohort of patients having undergone DU-UCB transplantation for nonmalignant disorders. The occurrence of long-term mixed-unit chimerism and dominance reversion after DU-UCB transplantation was scarcely available in the literature 18 . The frequency noted in this study is similar to 4% (11/280) of patients reported from a review of published literature 19 .
The mechanism of predominance of a particular unit in recipients after DU-UCB transplantation is largely unknown. Many factors have been surveyed including the infused doses of TNCs, CD34+ cells, CD3+ cells, and CFU-GM, viability, HLA match, donor-directed HLA antibodies, sex, ABO compatibility, order and route of delivery, length of time between infusions, immune interaction of host–graft and graft–graft, homing properties, and proliferative potential of hematopoietic stem cells4,17,20–25. Nonetheless, no single factor was identified to be consistently related to single-unit dominance. Data from the current study suggested that engraftment was independent of UCB processing in terms of the storage duration and washing prior to infusion, viability, cell numbers, HLA disparity, ABO incompatibility and gender match between recipient–UCB pairs, with the exception of the number of infused CD34+ cells.
It was suggested that mixed chimerism was associated with disease relapse post-hematopoietic stem cell transplantation 26 . DU-UCB transplants were reported to have lower relapse rates than those derived from HLA-matched donors, HLA-matched unrelated donors, and HLA one-antigen-mismatched unrelated donors 27 . In the current study, the risks of relapse in children transplanted with either single-unit or DU-UCB were comparable and in accord with a recent randomized study on childhood leukemia 10 . Acknowledging the limitation of a few transplanted patients with stable mixed chimerism, more studies on larger cohorts are necessary to ascertain the observations of the current study.
GvHD has been attributed to HLA disparity. The impact of donor-directed HLA-specific antibodies on graft failure after UCB transplantation has been elusive20,28. Incidences of GvHD of the present study were in agreement with the findings of a recent report from the Australian and New Zealand children's hematology and oncology group 29 . There was no impact of HLA disparity on neutrophil and platelet engraftment. Minor histocompatibility antigens are not included in the standard HLA matching; however, significant differences among populations exist, and they might be associated with poor clinical outcomes and affect the overall survival posttransplant30,31. Results of a recent study suggested no effect of donor race matching on relapse, GvHD, or overall survival 32 . Nonetheless, there are few reports of ethnicity matching and graft failure in recipients having undergone UCB transplantation. In the current study, there was no engraftment of overseas non-Chinese UCB in contrast to patients engrafted with ethnic Chinese UCB collected at nonlocal banks. This observation implies an effect of ethnicity/race on engraftment. Multicenter studies with larger cohorts of patients would be beneficial to confirm this finding.
ABO incompatibility has been reported as a risk factor for GvHD in bone marrow and UCB transplantation33,34. Notwithstanding, UCB-derived lymphocytes are relatively immune naive. Results of the current study suggested that ABO mismatch exerted no impact on both acute and chronic GvHD, and favored the assertion that ABO compatibility need not be considered in UCB recipients 33 .
DU-UCB transplantation was developed to circumvent the limitation of cell dose derived from a single unit; however, the predominance of one unit emerged in a vast majority of recipients posttransplant. Successful prediction of the dominant graft would optimize the algorithms of UCB selection and maximize the long-term engraftment of chosen units. In the characterization of engrafting units and nonengrafting units in double-unit transplantation, there was no association of the dominant units with the duration of storage, viability, doses of TNCs, CD3+ cells or CFU, HLA matching, ABO incompatibility, or gender differences, except for the infused dose of CD34+ cells. There was also no clinical benefit in terms of engraftment and survival among recipients of double-unit transplants over recipients of adequately dosed single-unit transplants. A trend of longer time for myeloid and platelet engraftment was noted among recipients of double-unit transplants. Data from the current study are in agreement with a recent report of an open-label, multicenter, randomized trial 10 . In conclusion, an adequately dosed single-unit UCB transplant would be preferable to a DU-UCB transplant.
Footnotes
Acknowledgments
The study was generously supported in part by Hong Kong Pediatric Bone Marrow Transplant Fund. The authors would like to thank the provision of umbilical cord blood donations from Hong Kong Red Cross Blood Transfusion Service. Authors also express gratitude to Dr. Karen Li of Department of Pediatrics, The Chinese University of Hong Kong and Dr. Ping Law of Department of Bone Marrow Transplantation and Cellular Therapy, St. Jude Children's Research Hospital for reviewing the manuscript. The authors declare no conflicts of interest.
