Abstract
Human amniotic epithelial cells (h-AECs), which have various merits as a cell source for cell therapy, are known to differentiate into cardiomyocytes in vitro. However, the ability of h-AECs to differentiate into cardiomyocytes in vivo and their cell transplantation effects on myocardial infarction are still unknown. In this study, we assessed whether h-AECs could differentiate into cardiomyocytes in vivo and whether h-AECs transplantation can decrease infarct size and improve cardiac function, in comparison to transplantation of cord blood-derived mesenchymal stem cells (MSCs) or adipose tissue-derived MSCs. For our study, we injected h-AECs, cord blood-derived MSCs, adipose tissue-derived MSCs, and saline into areas of myocardial infarction in athymic nude rats. After 4 weeks, 3% of the surviving h-AECs expressed myosin heavy chain, a marker specific to the myocardium. Compared with the saline group, all cell-implanted groups showed a higher ejection fraction, lower infarct area by positron emission tomography and histology, and more abundant myocardial gene and protein expression in the infarct area. We showed that h-AECs can differentiate into cardiomyocyte-like cells, decrease infarct size, and improve cardiac function in vivo. The beneficial effects of h-AECs were comparable to those of cord blood and adipose tissue-derived MSCs. These results support the need for further studies of h-AECs as a cell source for myocardial regeneration due to their plentiful availability, low immunity, and lack of ethical issues related to their use.
Introduction
Many patients who have failed timely reperfusion therapy after myocardial infarction (MI) suffer from congestive heart failure due to reduced living myocardium. After MI, the myocardium cannot regenerate adequately and traditional treatments are not effective for heart failure. Cell therapy using a wide range of cell types has been studied for myocardial regeneration (3,4,15,18,36).
Each cell type used for cell therapy has advantages; however, there are also many problems associated with the use of currently available cell sources, including issues with cell survival, immune rejection, tumor genesis, and ethical issues (12). Therefore, it is important to develop an ideal cell source to address such problems (19).
Recently, human amniotic epithelial cells (h-AECs) have been reported to have multipotent differentiation ability, and they have been shown to differentiate into cardiomyocytes in vitro (20). In addition, h-AECs have low immunogenicity and anti-inflammatory activity, are easily obtained in high cell numbers, and do not require the sacrifice of human embryos (24). Considering these characteristics, h-AECs are good candidates for use in cell therapy for MI. However, there have been no reports to assess the differentiation of h-AECs into cardiomyocytes in vivo or any evaluation of the effectiveness of h-AECs on myocardial regeneration after MI.
In this study, we investigated whether h-AECs could differentiate into cardiomyocytes in vivo and if transplantation of h-AECs could improve cardiac function and decrease infarct size in a rat MI model. The effect of h-AECs on MI was compared with cord blood-derived mesenchymal stem cells (MSCs) and adipose tissue-derived MSCs, which are already known to improve cardiac function and decrease infarct area (9,17,21,28,34).
Materials and Methods
Isolation and Culture of h-AECs
Human placentae were obtained after vaginal or caesarean section deliveries from healthy women, following informed consent. The amnion layer was mechanically peeled from the placenta and rinsed several times with Hank's balanced salt solution (without calcium or magnesium) to remove blood. The amnion membrane tissues were digested with 10 ml trypsin–ethylenediaminetetra-acetic acid (Gibco, USA) under gentle agitation for 45 min at 37°C. The digested tissue was then filtered through a 100-μm nylon sieve to remove cellular debris and centrifuged at 470 x g for 5 min to obtain a pellet. The pellet was resuspended in amniotic epithelial cell media (RAEM, RNLBIO, Seoul, South Korea) containing 10% fetal bovine serum (FBS), and the cell suspension was centrifuged at 470 x g for 5 min to collect the cell pellet. The cell fraction was then cultured overnight at 37°C in 5% CO2 in RAEM containing 10% FBS. The next day, cell adhesion was checked using an inverted microscope, and nonadherent cells were removed by washing with phosphate-buffered saline (PBS). The cell medium was then changed everyday for 4–5 days until the cells reached confluence, which was defined as passage 0. When the cells were 100% confluent, they were subcultured in RAEM and designated as passage 1. The immunophenotype of the h-AECs was then analyzed using flow cytometry (FACS Caliber, Becton Dickinson, Mountain View, CA, USA) and immunocytochemistry. Cell viability, as estimated by trypan blue exclusion assay, was above 95% prior to cell transplantation (30). No evidence of bacterial, fungal, or mycoplasmal contamination was observed in the cells tested. The procedure for h-AECs preparation was performed under good manufacturing practice conditions (RNLBIO, Seoul, South Korea).
Isolation and Culture of Human Adipose Tissue and Human Cord Blood-Derived MSCs
Human adipose tissues were obtained by simple liposuction of abdominal subcutaneous fat from healthy donors following informed consent. In brief, the subcutaneous adipose tissue was digested with 4.0 ml RTase (RNLBIO, Seoul, South Korea) per 1.0 g fat under gentle agitation for 1 h at 37°C. The adipose MSCs were prepared and maintained in the same media and conditions as those used for the h-AECs cultures. Cord blood samples were obtained from the umbilical vein immediately after delivery, with informed consent, as approved by the Boramae Hospital Institutional Review Board (IRB) and the Seoul National University IRB. Human cord blood-derived MSCs were obtained according to published protocols (29,31).
Rat Model of Acute MI
Male athymic nude rats (NIH-rnu, Taconic, Germantown, NY, USA), 8 weeks of age and weighing 250–300 g, were used in this study. The animals were cared for according to the US National Institute of Health guidelines for the care and use of laboratory animals, and the Hanyang University Institutional Animal Care and Use Committee approved all protocols.
To standardize infarct size, MI induction was performed by the cryo-injury method (11). Rats were anesthetized with a mixture of ketamine hydrochloride (100 mg/kg) and xylazine hydrochloride (10 mg/ kg). The anesthetized rats were ventilated with a rodent ventilator (Model 683, Harvard Apparatus, South Natick, MA, USA). A left thoracotomy was performed in the fourth intercostal space, and the pericardium was removed. Cryo-injury was performed using a metal probe (8.0 mm in diameter) cooled by immersion for over 15 min in liquid nitrogen. The cooled metal probe was put into contact with the left ventricle free wall for 10 s, and this procedure was repeated three times at the same site, at 1-min intervals.
Cell Transplantation
To trace the fates of the transplanted cells, cultured h-AECs and MSCs were labeled with chloromethyl-benzamidodialkylcarbocyanine (Cell Tracker™: CM-DiI; Molecular Probes, Eugene, OR, USA), according to the manufacturer's protocol (37). In brief, 2.0 mg/ml CM-DiI was diluted into Hank's balanced salt solution to a final concentration of 5.0 μg/ml. The cells were incubated in the diluted CM-DiI solution for 5 min at 37°C, followed by 15 min at 4°C. Then the cells were washed with PBS. After overnight culturing, the cells were examined by fluorescence microscopy using a rhodamine filter to determine labeling efficiency.
Ten days after MI insult, rats were randomly assigned into four groups (n = 8 rats for each group). The AEC, CBC, and ATC groups received transplantation of h-AECs, cord blood-derived MSCs, and adipose tissue-derived MSCs, respectively. The saline group was injected with saline as a control. Cells (1 × 106 cells) in 100 μl medium or saline were injected at the border toward the center of the infarcted myocardium using a 30-gauge needle.
Immunohistochemical Analysis
For immunofluorescence staining, heart tissues obtained 4 weeks after cell injection were embedded in Tissue-Tek OCT (Sakura Finetek USA, Inc., Torrance, CA, USA) compound and snap frozen in liquid nitrogen. Frozen tissue sections (4.0 μm thick) were stained with antibodies against myosin heavy chain (MHC; Serotec, NA, USA), using fluorescein isothiocyanate (FITC)-conjugated anti-goat IgG as a secondary antibody. Immunofluorescent staining of the sections was analyzed using a fluorescent microscope (Nikon Eclipse E800, Tokyo, Japan). Images were captured using a microscope-mounted digital camera (Leica DFC350 FX, Germany). Detection of myocardial differentiation was based on presence of colocalization of CM-DiI and MHC (11).
Echocardiographic Examination and Positron Emission Tomography (PET) Imaging
Echocardiographic examination was conducted, as previously described (11). Briefly, a blinded investigator performed transthoracic echocardiography 4 weeks after cell injection. M-mode traces were obtained at the papillary muscle level targeting the area of cryo-injury using an echocardiography system (128 XP, Acuson, Mountain View, CA, USA) equipped with a 7.0-MHz transducer. The left ventricular end systolic and diastolic diameters were measured using the American Society for Echocardiography leading-edge method from at least three consecutive cardiac cycles (16). The left ventricular volume and ejection fractions were calculated using the Teichholtz formula (22).
Fluorine-18 fluorodeoxyglucose (18F-FDG) PET imaging studies were performed using a dedicated small animal PET scanner (micro-PET® R4 scanner, Concorde Microsystems, Inc., Knoxville, TN, USA) with lutetium oxyorthosilicate (LSO) crystals, a 350–750 keV energy window, and 6-ns timing windows (6,39). Rats were injected with 1.0 mCi/0.2 ml of 18F-FDG via the tail vein and subsequently anesthetized by isoflurane inhalation anesthesia (2% in 100% oxygen, Forane Solution; Choongwae Pharma Co., Seoul, South Korea). The infarcted rat myocardium was scanned for 20 min. The acquired three-dimensional list mode data were reconstructed into temporally framed sinograms using Fourier rebinning and the ordered subsets expectation maximization algorithm (10). A polar map was generated from the 20-min static 18F-FDG PET image. The 18F-FDG PET image pixel size was multiplied by a scaling factor to adapt the rat heart size to approximate human dimensions. This adjustment was necessary because geometric assumptions were made for the automated myocardial wall detection by the commercial software (QPS, Siemens E-Soft) (6).
Analysis of Myocardial Protein and Gene Expression in MI Tissue
Western blot analyses were conducted as previously described (11). Four weeks after cell injection, the rats were sacrificed, and MI-injured heart tissue was obtained from the center of the infarct area (n = 3 rats for each group), which was clearly demarked by pale coloring. The samples were homogenized on ice in 0.1% Tween-20 homogenization buffer with a protease inhibitor. Then, 40-μg protein was transferred into sample buffer, separated using a 7.5% sodium dodecyl sulfate-polyacrylamide gel and blotted onto a polyvinylidene fluoride membrane (0.45-μm pore size, Bio-Rad Laboratories, Hercules, CA, USA). After blocking in 5% skim milk solution for 120 min, the membrane was incubated with primary antibody at a dilution of 1:200. The membrane was incubated with peroxidase-labeled secondary antibody at a dilution of 1:1,000. Positive protein bands were visualized using an ECL kit (GE Healthcare, Buckinghamshire, UK). All primary antibodies were rabbit polyclonals against cardiac α-actin, MHC, and troponin I (Abcam, Cambridge, UK). A mouse polyclonal antibody against glyceraldehyde-3-phosphate dehydrogenase (GAPDH) (Abcam, Cambridge, UK) was used as a protein loading control.
Total RNA was extracted from MI heart tissue (n = 3 rats for each group) with Trizol reagent (Invitrogen, Carlsbad, CA, USA) for use in reverse transcription-polymerase chain reaction (RT-PCR). RT-PCR was performed with 5.0 μg pure total RNA using SuperScript II reverse transcriptase (Invitrogen, Carlsbad, CA, USA). Synthesized cDNA was amplified by PCR using the following primer sets: cardiac homeobox transcription factor Nkx-2.5, sense 5′-CAACTTCGTGAACTTTGGCG-3′, antisense 5′-GGATCGGAGAAAGGTCCCAG-3′; GATA binding protein 4 (GATA-4) transcription factor, sense 5′-AGAGAGTGTGTCAACTGCGGG-3′, antisense 5′-TTTGAATCCCCTCCTTCCG-3′; and GAPDH, sense 5′-CCTTCATTGACCTCAACTACA-3′, antisense 5′-CT-GGCGTCTTCACCACCATG-3′. RT-PCR was conducted for 30 cycles of denaturing (94°C, 30 s), annealing (60°C, 30 s), and extension (72°C, 60 s), with a final extension at 72°C for 7 min. PCR products were visualized by electrophoresis on 2% (w/v) agarose gels. The RT-PCR product sizes for Nkx-2.5, GATA-4, and GAPDH are 251, 307, and 209 base pairs, respectively.
Histological Analysis for Estimation of MI Area
Four weeks after cell injection, heart tissues (n = 3 rats for each group) were fixed in 10% buffered formaldehyde and embedded in paraffin, and the sections were obtained with a 1.0-mm interval vertical to the long axis of the heart from apex to base. For comparison, a section at the center of the cryo-injury and its surrounding four sections (total of five sections) were selected and stained with Masson's trichrome. Each stained section was scanned and computerized using a digital image analyzer (Image-Pro plus, USA). The infarct area was measured as the percent ratio (%) of the injured area, divided by the whole LV area, and averaged over the five sections in each rat (11) (Fig. 1).
Experimental scheme. PET, positron emission tomography; RT-PCR, reverse transcription-polymerase chain reaction.
Statistical Analysis
Numerical values are expressed as the mean ± SD, unless otherwise indicated. Comparisons of parameters among the groups were performed with a one-way ANOVA followed by post hoc Tukey test using the statistical package SPSS 17.0 (SPSS, Inc., Chicago, IL, USA). A value of p < 0.05 was considered statistically significant.
Results
Characteristics of h-AECs
The h-AECs prepared in this study were cuboidal with 5–10 μm diameters, which is the typical appearance for epithelial cells (Fig. 2A). The h-AECs showed a homogeneous population of cells and nearly all cells showed persistent expression of pan-cytokeratins (R&D System, USA), which are specific markers of epithelial cells (Fig. 2B).
Characteristics of h-AECs. (A) The culture-expanded h-AECs showed a homogeneous population of cuboidal cells with 5–10 μm diameters. (B) Nearly all cells showed persistent expression of pan-cytokeratins, which are specific markers of epithelial cells. Pan-CK-positive cells were visualized with green fluorescence of Alexa-488 staining. (C) Immunophenotypic characterization of h-AECs. FACS histogram for surface markers demonstrated that h-AECs were positive for CD9, CD29, CD49f, CD90, CD105, and (HLA)-ABC and negative for CD31, CD34, CD45, and HLA-DR. Scale bars: 50 μm (A) and 100 μm (B). hAECs, human amniotic epithelial cells; pan-CK, pan-cytokeratins; FACS, fluorescence-activated cell sorting; CD9, cluster of differentiation 9; HLA, human leukocyte antigen.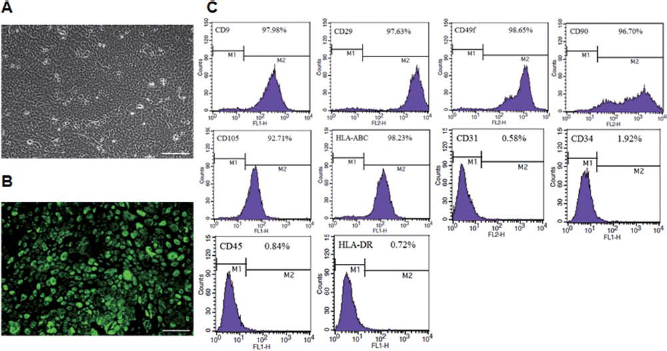
For immunophenotypic characterization of h-AECs, surface protein expression was examined by flow cytometry. The results of the FACS analysis showed that the immunophenotype of the h-AECs was positive for CD9, CD29, CD49f, CD90, CD105, and human leukocyte antigen (HLA)-ABC but negative for CD31, CD34, CD45, and HLA-DR (38). Representative results of the FACS assay from the h-AECs are shown in Figure 2C.
In Vivo h-AECs Differentiation Into Cardiomyocyte-Like Cells
The labeling efficiency CM-DiI is more than 95% in vitro prior to cell transplantation. Four weeks after cell transplantation, fluorescence microscopy detected h-AECs within the infarcted region of the rat myocardium. Approximately 3% of transplanted h-AECs, which were labeled with CM-DiI fluorescent dye prior to transplantation, were positive for the specific cardiac marker MHC. Coexpression of CM-DiI and MHC shows that transplanted h-AECs differentiated into cardiomyocytic lineage, that is, cardiomyocyte-like cells, in vivo (Fig. 3). The majority of h-AECs were found in the periphery of the infarct, while few cells were found in the center tissue.
Differentiation of h-AECs into myocardial cells in vivo. Infarcted rat left ventricle 4 weeks after h-AECs transplantation labeled with CM-DiI. (A) Masson's trichrome stain. (B) A higher magnification of the rectangle in A. (C–F) Fluorescent photographs of the rectangle area in B. CM-DiI, red in C; MHC, green in D; DAPI nuclear stain, blue in E. F is a merged image of C, D, and E and shows CM-DiI-labeled h-AECs expressing MHC. Arrows are for localizing the same cells. Insets show images of boxed regions in C, D, E, and F. IM, infarcted myocardium. Scale bars: 1.0 mm (A), 100 μm (B), 50 μm (C–F), and 5 μm (in the insets of C–F). CM-DiI, Cell tracker™; MHC, myosin heavy chain.
Induction of Myogenesis
Western blot analysis demonstrated that cardiac markers, including MHC, α-actin, and troponin I, were detected in the center of the infarcted myocardium, the necrotic core. These cardiac markers were more highly expressed in the cell-transplanted groups, including the AEC group, than in the saline group (Fig. 4A). RT-PCR analysis showed that transcription factors Nkx-2.5 and GATA-4 were expressed in the necrotic core of the MI. All cell-transplanted groups, including the AEC group, expressed higher levels of Nkx-2.5 and GATA-4 than of the saline group (Fig. 4B).
Expression of cardiac-specific genes and proteins. (A) Western blot analysis of MHC, α-actin, and troponin I in infarcted myocardium. MHC, α-actin, and troponin I proteins were more highly expressed in all cell-implanted groups, as compared to the saline group. GAPDH was used as a loading control. (B) RT-PCR analysis of Nkx-2.5 and GATA-4 expression in infarcted myocardium. Nkx-2.5 and GATA-4 were more abundantly expressed in all cell-implanted groups than in the saline group. GAPDH was used as a loading control. CBC, cord blood-derived mesenchymal stem cells; AEC, amniotic epithelial cells; ATC, adipose tissue-derived mesenchymal stem cells; GAPDH, glyceraldehyde-3-phosphate dehydrogenase.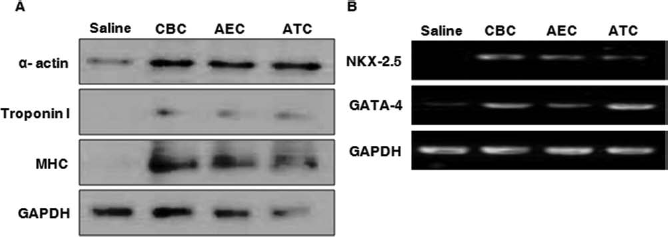
Improvement of Cardiac Function
Echocardiography 4 weeks after cell transplantation showed that transplanted h-AECs attenuated the left ventricular dilation and improved cardiac function compared with the saline group (n = 8 rats for each group). The left ventricular ejection fraction (LVEF) in the AEC group was significantly increased compared with that in the saline group (43.1% ± 5.5% vs. 34.5% ± 4.3%, p < 0.05). LVEF in the CBC (45.6% ± 4.3% vs. 34.5% ± 4.3%, p < 0.05) and ATC (43.1% ± 4.9% vs. 34.5% ± 4.3%, p < 0.05) groups was also higher than in the saline group. No significant difference in LVEF was found among the cell-transplanted groups themselves (43.1% ± 5.5% vs. 45.6% ± 4.3%, p = 0.73; 43.1% ± 5.5% vs. 43.1% ± 4.9%, p = 1.00; 45.6% ± 4.3% vs. 43.1% ± 4.9%, p = 0.72) (Fig. 5).
Echocardiographic assessment of LVEF 4 weeks after cell transplantation. LVEF in the CBC, AEC, and ATC groups was significantly increased, as compared with that in the saline group, but no significant difference was seen among the cell-implanted groups. Data are the mean ± SD. AEC, amniotic epithelial cells; CBC, cord blood-derived mesenchymal stem cells; ATC, adipose tissue-derived mesenchymal stem cells; LVEF, left ventricular ejection fraction.
Reduction of Myocardial Infarct Area
The cardiac micro-PET defect area was defined by a nonviable myocardium that displayed no signs of metabolism (n = 3 rats for each group) (Fig. 6A). The defect size in the AEC group was significantly reduced, as compared with that in the saline group (25.5% ± 8.8% vs. 47.8% ± 4.5%, p < 0.05). The defect sizes in both the CBC (23.2% ± 7.0% vs. 47.8% ± 4.5%, p < 0.05) and ATC (26.0% ± 10.6% vs. 47.8% ± 4.5%, p < 0.05) group were also significantly reduced, as compared to that of the saline group. Similar to the LVEF results, there was no difference in the size of the defect area among the cell-transplanted groups (25.5% ± 8.8% vs. 23.2% ± 7.0%, p = 0.98; 25.5% ± 8.8% vs. 26.0% ± 10.6%, p = 1.00; 23.2% ± 7.0% vs. 26.0% ± 10.6%, p = 0.97) (Fig. 6B).
PET assessment of defect size at 4 weeks after cell transplantation. (A) Representative short-axis images of rat heart with fluorine-18 fluorodeoxyglucose uptake. The images show that uptake in cell-transplanted groups was reduced, as compared to that in the saline group. (B) Quantitative analysis of defect size. Defect size in the CBC, AEC, and ATC groups was significantly reduced, as compared to that in the saline group, but no significant difference was seen among the cell-implanted groups. Data are shown as the mean ± SD. PET, positron emission tomography; SUV, standardized uptake value.
Cross-sections from cell-transplanted and saline groups were stained with Masson's trichrome and used to quantify the infarct area in each group. The infarct area in the AEC group was significantly reduced, as compared with that in the saline group (32.0% ± 2.0% vs. 39.6% ± 2.0%, p < 0.05). The infarct areas in the CBC (34.3% ± 2.0% vs. 39.6% ± 2.0%, p < 0.05) and ATC (33.3% ± 1.5% vs. 39.6% ± 2.0%, p < 0.05) groups were also significantly reduced, as compared to that of the saline group. Again, no difference among the cell-transplanted groups (32.0% ± 2.0% vs. 34.3% ± 2.0%, p = 0.49; 32.0% ± 2.0% vs. 33.3% ± 1.5%, p = 0.83; 34.3% ± 2.0% vs. 33.3% ± 1.5%, p = 0.91) was found (Fig. 7). In separated experiment, the infarct area at 10 days after cryo-injury was 42.0% ± 2.7% (n = 3).
Quantitative analysis of infarct area in Masson's trichrome-stained sections. Infarct area in the CBC, AEC, and ATC groups was significantly reduced, as compared with the saline group, and no significant difference was seen among the cell-implanted groups. Data are shown as the mean ± SD.
Discussion
In this study, we showed that h-AECs differentiated into cardiomyocyte-like cells in vivo, decreased infarct size, and improved cardiac function in a rat model of myocardial infarction. The beneficial effects were comparable to those seen with cord blood- and adipose tissue-derived MSCs.
Immunohistochemical analysis is a well-known and proven method to demonstrate that stem cells can differentiate into a cardiomyocyte (1,2,13). h-AECs marked with CM-DiI prior to transplantation were positive for both CM-DiI and cardiac MHC cardiomyocyte markers within the infarct region (Fig. 3). Other kinds of cells are already known to differentiate into cardiomyocytes, but this is the first report of h-AECs differentiating into cardiomyocyte-like cells in vivo. Of note, in our experiment, only 1 × 106 h-AECs were injected and only 3% of surviving h-AECs had MHC, a cardiomyocyte marker. This scale is too small to explain the improved ejection fraction and decreased infarct size (35). Our finding that h-AECs can differentiate into cardiomyocyte-like cells is of scientific interest but is not sufficient to explain the process of recovery from MI.
In the cell-transplanted groups, the infarcted tissue expressed more cardiac markers (α-actin, MHC and troponin I) and cardiac transcription factors (Nkx-2.5 and GATA-4) than the saline group (Fig. 4A, B) and the infarct size was smaller (Figs. 6, 7) and the ejection fraction was greater (Fig. 5) than in the saline group. To evaluate the infarct area, we used high-resolution 18F-FDG PET, which is highly accurate and has been shown to be valuable for in vivo imaging of rat hearts in experimental studies (14). The reduction of histological infarct size, as compared to the control, was about 6–28% in previous cell transplantation studies in an MI animal model (7,22,25,41). In our study, cell transplantation decreased the infarct size by 20% by histology and 40% in vivo by cardiac micro-PET scan. The improvement of LVEF in the rat MI model was reported previously to be about 19–30% (2,23,40), which is comparable with our result of 25% in the AEC group. These data suggest that the beneficial effects of h-AECs on MI were comparable to other kinds of stem cells.
Of note, the effect of h-AECs on infarct size reduction and ejection fraction improvement was equivalent to that achieved with cord blood-derived MSCs and adipose tissue-derived MSCs. This is the first report to show the effect of h-AECs on MI in an animal model. Considering the unique advantages of h-AECs, including their plentiful availability, immune privilege, and anti-inflammatory nature (24), h-AECs could be an optimal candidate as a cell source for myocardial regeneration cell therapy.
In our experiment, the transplanted cell number was small (1 × 106) and the frequency of h-AECs differentiation into cardiomyocyte was low (3%). In 2000, there were reports suggesting that adult stem cells can differentiate into different tissue lineages. However, subsequent studies failed to document this transdifferentiation as a therapeutically relevant mechanism (35). It is unlikely that the therapeutic effect of h-AECs and other cells is due to differentiation into cardiomyocytes. Adult stem cells secrete a number of cytokines and growth factors to regenerate diseased tissue via stimulation of angiogenesis, inhibition of apoptosis, and stimulation of intrinsic stem cells (5,26,27,32). In our expereiment, the tissues from the center of infarct showed increased myocardial gene and protein expression notwithstanding that few transdifferentiated cells were found in the center of infarct. This could be related to the mobilization and/or proliferation of endogenous progenitors at the sites of injury by h-AECs implantation (8). It is more likely that the “trophic” effects of transplanted cells, rather than stem cell “plasticity,” explain the beneficial effect of our cell therapy. Further studies on the trophic effects of cell therapy are required.
MI in animal models resulting from cryo-injury differs from MI resulting from ischemic injury, which is what occurs in clinical MI. Nevertheless, cryo-injury of the myocardium results in the same histological and functional changes (33). Furthermore, the classical is chemic injury model using coronary artery ligation cannot reproduce the same amount of tissue injury, while cryo-injury can. Considering our small sample size for animal models, cryo-injury is a reasonable surrogate for clinical MI.
In summary, we showed that h-AECs can differentiate into cardiomyocyte-like cells in vivo and, regardless of mechanism, regenerate myocardium tissue to improve cardiac function in a rat model of MI. h-AECs, which are easy to acquire, have low immunogenicity and are anti-inflammatory, making them an attractive cell source for myocardial regeneration.
Footnotes
Acknowledgments
This work was supported by the grant for the Medical Research Center (2011-0028261) funded by the National Research Foundation (NRF) of the Ministry of Education, Science and Technology (MEST), Republic of Korea. We thank Kyung-Sun Kang (Seoul National University, Korea) for providing human cord blood-derived mesenchymal stem cells. The authors declare no conflicts of interest.
