Abstract
Generation of tumor specific alloreactive CD4+ T cells is important to circumvent tumor tolerance. Here, we generate allorestricted peptide-specific CD4+ T cells by coculture of lymphocytes and autologous monocytes bearing allogeneic HLA-DR15 molecule associated with its restricted peptide. Binding of a dimeric HLA-DR15/IgG1-Fc fusion protein (the dimer) to HLA-DR15 negative (HLA-DR15-ve) monocytes made the monocytes coated with the allogeneic epitope. An increased proliferation of CD4+ T cells and induction of Th1 cells appeared after coculturing of HLA-DR15-ve lymphocytes and the autologous monocytes loaded with the dimer. The cocultural bulks showed an increased frequency of the specific dimer-stained CD4+ T cells and the expanded CD4+ T cells exhibited an elevated IFN-γ production in response to specific TCR ligand. Tumor rejection effects of the allorestricted E7-specific CD4+ T cells raised by the coculture were observed in nude mice challenged with human cervical cancer cell SiHa expressing both HLA-DR15 and E7 antigens, as the tumor avoidance and life span of the mice were improved after adoptive transfer of the CD4+ T cells. This study may help to develop strategies to separate graft-versus-leukemia or graft-versus-tumor reaction from graft-versus-host disease, and add to the pool of human high-avidity TCRs specific for tumor or virus antigens.
Introduction
T cells play a major role in allograft rejection and graft-versus-host disease (GVHD); both CD4+ and CD8+ T cells are involved in the process (21–23,31). T-cell receptors (TCRs) are formed by antigenic peptide associated with major histocompatibility complex (MHC) class I or class II molecule, which are recognized by CD8+ and CD4+ T cells, respectively (21). Direct T-cell allorecognition is the principal player in vigorous response to allogeneic cells that causes acute rejection (49). The structure basis for T-cell direct recognition has been actively investigated (10,26,41,53). Current belief in the field is that allogeneic T-cell responses, like syngeneic T-cell responses, are largely peptide specific, and that TCRs interact with allogeneic MHC in a manner that is almost indistinguishable from conventional recognition of antigenic peptide presented by self-MHC (31,38,49). Alloreactive T cells of peptide-dependent and allogeneic MHC-restricted properties are referred to as allorestricted T cells (10,41,53).
It has been observed that CD4+ but not CD8+ T cells are essential for allorejection (7,26), since CD4+ T cells are generally regarded as helper cells by facilitating other immune cells (12) and effector cells of graft rejection (47). It is well established that leukemia patients can benefit from graft-versus-leukemia reaction (GVLR) (9,54), and evidences of graft-versus-tumor reaction (GVTR) have also been reported in many solid tumors (6,8,14), in which donor immune cells mount a response against recipients' leukemia or tumor cells. Both CD4+ and CD8+ T cells are required for GVLR (5), although most studies focus on the role of CD8+ T cells in GVLR or GVTR and reveal CD8+ T cell is an effective means (24,43,45). Based on the importance of CD4+ T cells in both nominal and allogeneic responses, we propose allorestricted CD4+ T cells would be responsible for GVLR or GVTR. The aim of this study was to establish a strategy to generate allorestricted CD4+ T cells and examine their tumor rejection effects.
In the previous study, we developed a strategy to generate allorestricted CD8+ T cells specific for a given peptide/MHC complex (51). Briefly, an allogeneic MHC class I molecule associated with its restricted peptide is attached to monocyte; the latter “presents” the alloepitope to and induces autologous lymphocytes to generate the alloepitope-specific cytotoxic T lymphocytes (CTLs). This strategy was used in the present study to generate allorestricted CD4+ T cells. A dimeric human leukocyte antigen (HLA)-DR15/IgG1-Fc (fragment crystallizable) fusion protein (the dimer) was constructed with divalent TCR ligands and an Fc region of immunoglobulin G receptor 1 (FcγRI) binding moiety. Binding of the Fc fragment to FcγRI attached the peptide/HLA-DR15 complex as an alloepitope onto HLA-DR15-ve monocytes. Coculturing of HLA-DR15-ve lymphocytes and the dimer-loaded autologous monocytes expanded CD4+ T cells of peptide/HLA-DR15 complex-specific properties. In nude mice challenged with human cervical cancer cell SiHa expressing both HLA-DR15 and early 7 (E7) antigens of human papilloma virus (HPV), adoptive transfer of the allorestricted E7-specific CD4+ T cells exhibited tumor rejection effects (Fig. 1). This study provides a practical and flexible protocol to raise allorestricted CD4+ T cells of desired specificity, which would largely add to the pool of human TCRs specific for tumor or viral antigens in addition to adoptive transfusion.

Schematic illustration of major steps in this work. (A) Via the interaction of the fragment crystallizable (Fc) part and IgG Fc receptor type I (FcγRI), binding of a dimeric human leukocyte antigen (HLA)-DR15/IgG1-Fc fusion protein (the dimer) to HLA-DR15-ve monocytes makes the monocytes loaded with the allogeneic epitope. (B) The E7/HLA-DR15 dimer-loaded monocytes “present” the allogeneic epitope to autologous lymphocytes when coculturing in vitro. (C) The expanded CD4+ T cells exhibit allorestricted and E7-specific properties. (D) Tumor rejection effects of the allorestricted E7-specific CD4+ T cells are observed in nude mice challenged with human cervical cancer cell SiHa expressing both HLA-DR15 and E7 antigen.
Materials and Methods
Mice, Cells, and Peptides
Balb/c athymic nude mice were purchased from Chinese Shanghai Laboratory Animal Center (China). The mice were bred and maintained under specific pathogen-free conditions and treated in accordance with Tongji Medical College (China) animal care guidelines. Animal experiments are approved by the Ethical Committee of Tongji Medical College. Human cervical cancer cell lines SiHa (ATCC HTB-35), HeLa (ATCC CCL-2), and prostatic cancer cell line DU145 (ATCC HTB-81) were purchased from American Type Culture Collection. The E003 was a lymphoblastoid cell line established with Epstein-Barr virus transforming from human B lymphocytes of a healthy individual (35). All cell lines and human peripheral blood used in this study are shown in Table 1. The Ethical Committee approved sampling of the human blood, and informed consent was obtained from donors. HLA-DR15-restricted peptides, E748–62 (DRAHYNIVTFCCKCD) of HPV E7 protein origin (47,50), NS3141–155 (NRKGKVIGLYGNGVV) of Dengue virus NS3 protein origin (27), were synthesized by a peptide synthesizer and purified to >90% homogeneity by reverse-phase HPLC, which are named as E7 and NS3, respectively.
Cells Used in This Study

expression
non-expression
not tested.
Typed by allele sequence-specific primers (PCR-SSP) shown by Bunce et al. (4) and HLA-DR15 allele-specific antibody (Abcam, ab31258).
NS3 antigen expression is determined by RT-PCR. The sequences of primers of NS3 were designed according to Kurane et al. (27) in which the specific amino acid sequence of NS3 was presented.
Preparation, Detection of the HLA-DR15 Dimer, Peptide Pulsing, and Loading Monocytes with the Dimer
Preparation and detection of the dimeric HLA-DR15/ IgG1-Fc fusion protein were based on the protocol previously published (2,52). To pulse the dimer with peptide, 5 μg dimer was incubated with 180 μM peptide in 100 μl PBS (pH 7.4) at 37°C for 24–48 h. The dimers pulsed with E748–62 or NS3141–155 are named E7/HLA-DR15 dimer or NS3/HLA-DR15 dimer, respectively. The dimer was loaded onto HLA-DR15-ve monocytes based on the protocol previously published (51).
Coculture of the Dimer-Loaded Monocytes with Autologous Lymphocytes, and Proliferation Assay
Peripheral blood monocyte (PBMC) samples of three HLA-DR15-ve donors were separated into adherent (i.e., monocyte) and nonadherent [i.e., lymphocyte or peripheral blood lymphocyte (PBL)] populations. The HLA-DR15 dimer-loaded monocytes were cocultured with autologous lymphocytes to raise alloreactive CD4+ T cells. The monocytes (2 × 105 cells/well) and the lymphocytes (1 × 106 cells/well) were plated into 24-well plates in 2 ml of 10% fetal calf serum (FCS) RPMI-1640 medium and incubated at 37°C, 5% CO2 for 7 days. On day 8, the coculture was re-stimulated with 2 × 105 cells/well of the dimer-loaded monocytes and incubated for further 7 days.
Lymphocyte proliferation was assayed with fluorescent dye dilution as previously reported (51). Briefly, on day 7 of the coculturing, lymphocytes prestained with carboxyfluorescein succinimidyl ester (CFSE; Sigma, C5041) were incubated with anti-CD3-phycoerythrin (PE) (BD Biosciences, 555340) and anti-CD4-PE-cyanine 5 (Cy5) (BD Biosciences, 555348). Three-color analysis was performed with CFSE, anti-CD3-PE, and anti-CD4-PE-Cy5 using a FACSCalibur flow cytometer (BD Biosciences). The data were analyzed with the “Proliferation Wizard” module in ModFit LT Macintosh software (BD Biosciences).
Sorting of Alloreactive CD4+ T Cells
On day 14, CD4+ T cells were sorted from the cocultural bulks by negative immunomagnetic sorting with CD4+ T Cell Isolation Kit II (Miltenyi Biotec, 130-091-155) following the manufacturer's protocol. Viability of isolated CD4+ T cells was over 97%, assessed by trypan blue exclusion. The E7/HLA-DR15 and NS3/HLA-DR15 dimers were included in the coculture and the raised alloreactive CD4+ T cells are named E7/allo-CD4+ T cells and NS3/allo-CD4+ T cells accordingly.
Detection of CD4+ T-Cell Subtypes
On day 14, the cocultural bulks were detected for CD4+ T helper 1 (Th1), Th2, Th17, and Treg (regulatory T) cells by identification of intracellular expression of interferon-γ (IFN-γ), interleukin-4 (IL-4), IL-17, and forkhead box P3 (Foxp3), respectively. For IFN-γ, IL-4, and IL-17 staining, the cocultural bulks were treated with 20 ng/ml phorbol 12-myristate 13-acetate (PMA; Sigma, P1585), 1 μg/ml ionomycin (Sigma, 10634), 2 μM monensin (eBiosciences, 00-4505-51) for 6 h. The suspended cells were initially stained with anti-CD3-APC-Cy7 (Biolegend, 300426) and anti-CD8-PE-Cy5 (Biolegend, 300910), then fixed and permeabilized with Fixation & Permeabilization Kit (eBiosciences, 88-8823-88), and finally stained accordingly with anti-IFN-γ-fluorescein isothiocyanate (FITC) (eBioscience, 11-7319-81), anti-IL-4-PE-Cy7 (eBioscience, 25-7049-82), and anti-IL-17-PE (Biolegend, 512306) for 45 min. For Foxp3 staining, the cells were initially stained with anti-CD3-APC-Cy7, anti-CD4-PE-Cy5, and anti-CD25-PE-Cy7 (eBiosciences, 25-0259-41), then with anti-Foxp3-PE (eBiosciences, 12-4777-41) and Foxp3 Staining Buffer Set (eBiosciences, 00-5523-00). Fixation, permeabilization, and staining were based on the manufacturer's recommendations. The data were analyzed using the FACSCalibur and CellQuest software (BD Biosciences).
Specific Peptide/HLA-DR15 Dimer Staining Assay and IFN-γ ELISPOT Assay
On day 14, the cocultural bulks were incubated with the peptide/HLA-DR15 dimer at 50 μg/ml for 1 h at 37°C, washed, and stained with FITC-conjugated goat anti-human IgG1 at 4°C for 30 min. Anti-CD3-PE and anti-CD4-PE-Cy5 were added subsequently for a further 30-min incubation followed by a final wash and samples were fixed with PBS containing 2% formaldehyde. Three-color analysis was performed with anti-IgG1-FITC, anti-CD3-PE, and anti-CD4-PE-Cy5 by using the FACSCalibur and CellQuest software.
For IFN-γ ELISPOT assay, the negative immunomagnetically sorted CD4+ T cells were enriched immunomagnetically with the peptide/HLA-DR15 dimer-coated beads, the latter was prepared by incubation of 50 ml Protein A MicroBeads (Miltenyi Biotec, 130-071-001) with the dimer (50 ml at 50 mg/ml) for 30 min. The peptide/HLA-DR15 dimer-bound cells were tested as responders for IFN-γ production by ELISPOT using Human IFN-γ precoated ELISPOT kit (Millipore, ELI-016-H). The enriched E7/allo-CD4+ T cells and NS3/ allo-CD4+ T cells were included for responders, two panels of cells for stimulators. One panel of stimulators was autologous monocytes loaded with the E7/HLA-DR15 or NS3/ HLA-DR15 dimer, the other was SiHa, HeLa, E003, and DU145. The SiHa, HeLa, and DU145 cells were incubated with 500 U/ml IFN-γ (eBioscience, 14-8319-80) at 37°C 5% CO2 for 48 h to induce the expression of HLA-DR molecules; since over 90% of the cells were stained with anti-HLA-DR-FITC (BD Biosciences, 555811), the HLA-DQ and HLA-DP molecules should be simultaneously expressed on the membrane of the cells (40). Responders (1 × 105) were incubated with 2 × 104 monocytes or 1 × 104 tumor cells or 1 × 104 lymphoblastoid cells in an ELISPOT well at 37°C 5% CO2 for 32 h. All samples were run in triplicate with spots imaged using CTL Analyzers LLC (CTL Analyzers Company, USA).
Tumor-Challenged Mice, Adoptive Transfer of Alloreactive CD4+ T Cells, and Histological Examination
SiHa cells (1 × 106 or 2 × 106) in 100 ml PBS were injected SC into the left flank of 4-week-old female nude mice. Primary tumor was estimated by palpation and use of a caliper in two perpendicular diameters every 3–4 days after tumor inoculation. A tumor of 3 mm in diameter was scored as a tumor take; mice were considered to be tumor free until a palpable mass (>3 mm) persisted for longer than 4 days. Mice were sacrificed for humanitarian reasons when primary tumor reached 10 mm in mean diameter, when ulceration, bleeding, or both developed, or when mice became distressed (29).
Adoptively transferred CD4+ T cells included E7/ allo-CD4+ T cells, NS3/allo-CD4+ T cells (nonspecific control), and CD4+ T cells cocultured with autologous monocytes alone (negative control); injection of PBS served as blank control. The CD4+ T cells from random individuals were chosen, without regard to alleles on DQ and DP loci, for the absence of HLA-DR15 allele. These cells then alloresponded to allo-MHC molecules of the SiHa cells besides HLA-DR15. PBMCs from each donor were handled and used as E7/allo-CD4+ T cells, nonspecific control, and negative control. CD4+ T cells (1 × 107) in 100 ml PBS were transferred SC into the left flank of nude mice 1 day after the tumor inoculation (30).
Nude mice were injected SC with 9 × 106 SiHa cells and palpable tumor (8 mm in diameter) developed 10 days later and 1 × 107 CD4+ T cells were transferred into the tumor (30). Six days later, the mice were sacrificed and tumors were subjected to H&E staining for histopathology, Paraffin-embedded section was stained with anti-human CD4 (BD biosciences, 555344) and HRP-goat anti-mouse IgG for immunohistochemistry.
Statistical Analysis
Data were analyzed using SPSS 13.0. Statistical significance was determined by ANOVA analysis. All pair-wise comparisons between group means were performed by LSD if comparing three groups or Tukey if comparing more than three groups in the post hoc tests. Tumor survival data were analyzed with the Kaplan-Meier method. The log-rank test was used to compare survival curves and tumor avoidance for different groups on univariate analyses. A value of p < 0.05 was considered to be statistically significant.
Results
Proliferation of CD4+ T Cells and Induction of CD4+ Th1 Cells by Coculturing
The prepared HLA-DR15 dimer consisted of divalent TCR ligands and an FcγRI binding moiety, which could be loaded onto HLA-DR15-ve monocytes effectively (data not shown). The purified dimer was pulsed with the HLA-DR15-restricted peptide E7 (of HPV E7 protein origin) or NS3 (of Dengue virus NS3 protein origin), to form E7/HLA-DR15 or NS3/HLA-DR15 dimer, respectively.
PBMCs of each HLA-DR15-ve donor were separated into PBLs and monocytes by adhesion; the coculture was set up by incubation of the HLA-DR15-ve PBLs with the dimer-loaded autologous monocytes. After 7 days of incubation, the cocultural bulks were tested for CD4+ T-cell proliferation (Fig. 2A, B). The PBLs cocultured with the monocytes alone (negative control) showed a background proliferation, and those with the dimer-loaded monocytes exhibited a vigorous CD4+ T-cell proliferation. Although the E7 peptide alone induced CD4+ T-cell proliferation, its proliferation index was significantly lower than that of the dimer-loaded monocytes. The reason is probably that the E748–62 peptide can bind to other HLA-DR alleles with lower affinity than HLA-DR15 (48,50) and the formed epitopes would be able to stimulate T-cell proliferation with lower intensity.
The cocultural bulks primed with the E7/HLA-DR15 dimer-loaded monocytes were tested for CD4+ T-cell subtypes (Fig. 2C, D). Th1 cell increased significantly after coculturing, whereas Th2, Th17, and Treg remained unchanged, compared to those with the monocytes alone (unprimed). The results suggested coculturing was able to promote Th1 response, which is essential for allograft rejection.
Allorestricted Peptide-Specific Properties of the CD4+ T Cells Expanded by Coculturing
To test the specificity of the expanded CD4+ T cells, the E7/HLA-DR15 or NS3/HLA-DR15 dimer-loaded monocytes were used to prime autologous lymphocytes, and the cocultural bulks were stained with the specific dimer (Fig. 3A). The cocultural bulk primed with the E7/HLA-DR15 dimer-loaded monocytes showed higher frequency of the E7/HLA-DR15 dimer-stained cells than that with the NS3/HLA-DR15 dimer-loaded monocytes (7.57 ± 1.46% vs. 0.73 ± 0.38%, n = 3, p < 0.001), and relatively low frequency of the dimer-stained cells when the irrelevant NS3/HLA-DR15 dimer was used (1.07 ± 0.43% vs. 7.57 ± 1.46%, n = 3, p < 0.001). Similarly, coculture with the NS3/HLA-DR15 dimer-loaded monocytes expanded NS3/HLA-DR15 dimer-stained CD4+ cells instead of E7/HLA-DR15 dimer-stained cells. These observations suggested that alloreactive CD4+ T cells expanded by coculturing should be peptide specific.

Coculturing induces effective proliferation of CD4+ T cells and an increase of T helper 1 (Th1) cells. (A, B) HLA-DR15-ve lymphocytes showed an elevated proliferation after cocultured with either the E7/HLA-DR15 or NS3/HLA-DR15 dimer-loaded autologous monocytes, compared to that cocultured with the monocytes alone (negative control), or the monocytes plus the E7 peptide. Allogeneic monocytes (HLA-DR15+ve) served as positive control. Proliferation index (PI) and the percentage of proliferated cells were measured by carboxyfluorescein succinimidyl ester (CFSE) dilution assay. (A) The proliferated cells are seen as the subpopulation with lower CFSE intensity (M2) than the parent cells without proliferation (M1). Various peaks with different gray represent generations of the proliferated cells. The data show the results of sample 1. (B) The mean values (±SD) of three samples are represented with markers showing the difference compared with the negative control or E7 peptide control (*p < 0.05; ***p < 0.001). (C, D) The percentage of CD4+ Th1 cell increased significantly after primed with the E7/HLA-DR15 dimer-loaded monocytes, compared to that with the monocytes alone (unprimed). CD4+ Th1, Th2, Th17, and Treg cells were identified by expression of interferon-γ (IFN-γ), interleukin-4 (IL-4), IL-17, and forkhead box P3 (Foxp3) with intracellular staining assay, respectively. (C) A representative of three HLA-DR15-ve samples is shown for the CD4+ T-cell subtyping. Plots in a1–a3 and b1–b3 are gated on CD3+CD8- cells, plots in a4 and b4 are gated on CD3+CD4+ cells. (D) The mean values (±SD) of three HLA-DR15-ve samples are represented with markers showing the difference between groups (**0.001 < p < 0.01).
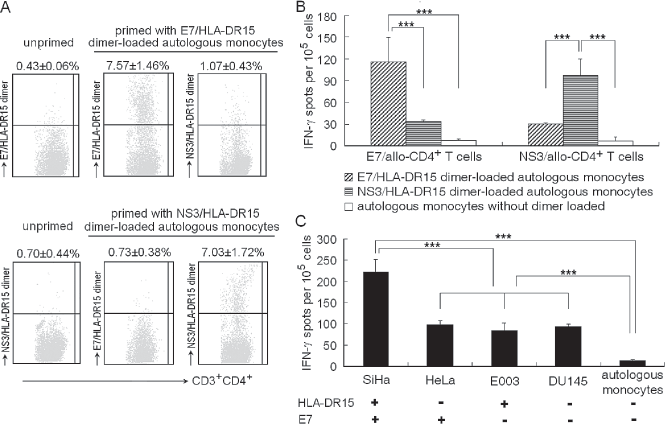
Coculturing expands allorestricted peptide-specific CD4+ T cells. (A) Specific binding of the peptide/HLA-DR15 dimer to the cocultural bulks. The cocultural bulks primed by the monocytes loaded with E7/HLA-DR15 dimer (upper panels) or NS3/ HLA-DR15 dimer (lower panels) were incubated with the dimer as the primary and fluorescein isothiocyanate (FITC)-conjugated anti-human IgG1 as secondary reagent. Coculture with the monocytes alone (unprimed) serves as negative control, and cocultural bulks stained with mismatched dimer as nonspecific control. Dimers used are indicated on the left of each figure. The mean values (±SD) of dimer-stained CD4+ T cells of three samples are represented on the top of each figure. The cocultural bulks primed with the E7/HLA-DR15 dimer-loaded monocytes were more frequently stained by the E7/HLA-DR15 dimer than that by irrelevant NS3/ HLA-DR15 dimer (n = 3, p < 0.001). The dimer staining for the cocultural bulks primed by the NS3/HLA-DR15 dimer-loaded monocytes also exhibited a peptide-dependent fashion. (B, C) The expanded CD4+ T cells exhibited T-cell receptor (TCR) ligand-specific IFN-γ production. The E7/allo-CD4+ T cells or NS3/allo-CD4+ T cells were enriched with the dimer-coated beads from the cocultural bulks. (B) Responders were the E7/allo-CD4+ T cells and the NS3/allo-CD4+ T cells; stimulators were autologous monocytes loaded with the E7/HLA-DR15 dimer or NS3/HLA-DR15 dimer. (C) Responders were the E7/allo-CD4+ T cells; stimulators were SiHa, HeLa, E003, and DU145 cells. The HLA-DR15 and E7 antigen expression patterns of the stimulators are indicated on the bottom of the figure: +, present; -, absent. IFN-γ production was determined by ELISPOT and shown as the mean (±SD) number of spots/105 responder CD4+ T cells. The mean (±SD) of three samples is represented with markers showing the difference between groups (***p < 0.001).
IFN-γ production in response to specific TCR ligand was examined by ELISPOT to verify the specificity of the expanded CD4+ T cells. To reduce the background and enhance the sensitivity of the assay, the dimer-stained CD4+ T cells were enriched with the peptide/ HLA-DR15 dimer-coated beads from the cocultural bulks. The enrichment gave about 60% of the dimer-stained CD4+ T cells. The enriched E7/HLA-DR15 dimer-bound CD4+ T cells demonstrated an increased frequency of IFN-γ spots when stimulated by autologous monocytes loaded with the E7/HLA-DR15 dimer, compared with that by the NS3/HLA-DR15 dimer-loaded monocytes (115.67 ± 33.53 vs. 33.33 ± 3.05, n = 3, p < 0.001). The enriched NS3/HLA-DR15 dimer-bound CD4+ T cells also showed the NS3/HLA-DR15-specific IFN-γ response (Fig. 3B). More importantly, the enriched E7/HLA-DR15 dimer-bound CD4+ T cells exhibited a more intensive response of IFN-γ production to SiHa cells than that to HeLa, E003, or DU145 cells (Fig. 3C). Since SiHa cells express both HLA-DR15 and E7 antigens, whereas HeLa or E003 cells express either one of the antigens, and DU145 cells express neither one of them, the observations indicated the alloreactive CD4+ T cells raised by the coculture were allorestricted and peptide specific. It should be noted that the enriched E7/HLA-DR15 dimer-bound CD4+ T cells showed a relatively obvious response to HeLa, E003, or DU145 cells, compared with that to autologous monocytes alone. This was probably due to T-cell alloresponse to the allo-MHC molecules other than the E7/HLA-DR15 complex, because nonspecific CD4+ T cells may not have been completely excluded from the responders, even after conducting careful enrichment, although there exists the possibility that the virus-specific T cells are cross-reactive to HLA class II mismatches (28,46).
Tumor Rejection Effects of Allorestricted E7-Specific CD4+ T cells in SiHa Cell-Challenged Nude Mice
With negative immunomagnetic sorting, the CD4+ T cells were sorted from the cocultural bulks primed by the E7/HLA-DR15 dimer-loaded monocytes (i.e., E7/allo-CD4+ T cells) or the NS3/HLA-DR15 dimer-loaded monocytes (i.e., NS3/allo-CD4+ T cells) for adoptive transfer, the transferred cells reached 97% of CD4+ T cells. Nude mice inoculated with SiHa cells were used to check the tumor inhibition effects of the CD4+ T cells. Nude mice inoculated with 1 × 106 SiHa cells developed palpable solid tumors. Transferring of both the 1 × 107 E7/allo-CD4+ T cells and the 1 × 107 NS3/allo-CD4+ T cells or even the 1 × 107 CD4+ T cells cocultured with the autologous monocytes alone were able to prevent the mice from developing tumors, whereas the mean life span of the mice treated with PBS was 83.00 ± 6.54 days (Fig. 4A, B). When the tumor load increased to 2 × 106 SiHa cells, it was the E7/allo-CD4+ T cells that showed the tumor inhibition effects (Fig. 4C, D). Mice (62.5%; 5 of 8) injected with the E7/allo-CD4+ T cells rejected tumor and survived without palpable tumor on day 140. On the other hand, all of the mice in the other three groups developed tumors. The mean values of life span of the mice treated with the CD4+ T cells cocultured with autologous monocytes alone and the NS3/ allo-CD4+ T cells were 80.12 ± 5.22 and 86.25 ± 4.23 days, respectively; the difference was nonsignificant compared to that of mice treated with PBS (75.87 ± 4.48 days, p = 0.599 or p = 0.182, respectively). The tumor avoidance and life span of the mice transferred with the E7/allo-CD4+ T cells were significantly improved (Fig. 4C, D). After injection of the CD4+ T cells into the palpable solid tumor, biopsies showed more human CD4+ T cell infiltration with the E7/allo-CD4+ T cells transferred than those with the NS3/allo-CD4+ T cells (Fig. 5). These results showed antitumor immunity ignited by the allorestricted tumor-specific CD4+ T cells was more effective than that by nonspecific CD4+ T cells.

Allorestricted E7-specific CD4+ T cells ignite potent tumor rejection effects in SiHa cell-challenged nude mice. The mice were challenged by SiHa cells and treated as the following four groups: transfer of the 1 × 107 E7/allo-CD4+ T cells as specific group, the 1 × 107 NS3/allo-CD4+ T cells as nonspecific control, 1 × 107 CD4+ T cells cocultured with autologous monocytes alone as negative control and PBS as blank control. (A, B) In 1 × 106 SiHa cell-challenged mice (7 mice per group), the inoculated tumor cells were rejected by the E7/allo-CD4+ T cells, the NS3/allo-CD4+ T cells, and even CD4+ T cells cocultured with autologous monocytes alone, the tumor avoidance and life span curves of the three groups are overlapping, whereas the mice treated with PBS all developed tumors. (C, D) In 2 × 106 SiHa cell-challenged mice (8 mice per group), tumor avoidance (C) and life span (D) of the mice were improved only by adoptive transfer of the E7/allo-CD4+ T cells. Mice (62.5%; 5 of 8) treated with the E7/allo-CD4+ T cells rejected tumors and survived for at least 140 days, while all mice (8 of 8) of the rest three groups developed tumors. The life span of the mice treated with the CD4+ T cells cocultured with autologous monocytes alone or with the NS3/allo-CD4+ T cells showed nonsignificant difference compared to that of mice treated with PBS (p = 0.599 or p = 0.182, respectively). The differences in tumor avoidance (p < 0.001) and life span (p < 0.001) were significant between the E7/allo-CD4+ T cells group and the rest of the groups as evaluated by the log-rank test.
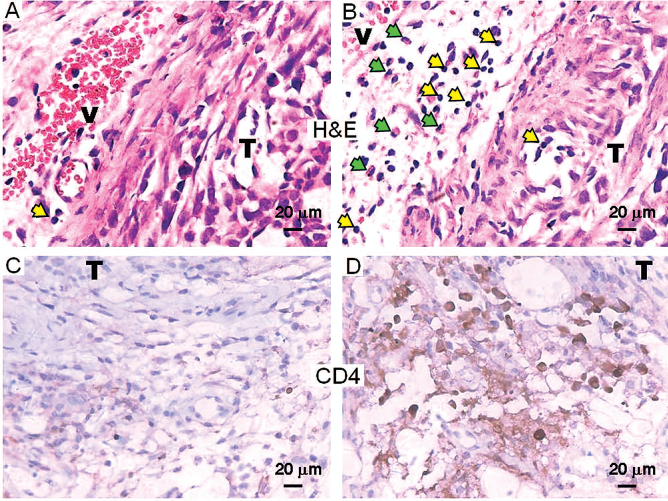
More CD4+ T cells and granulocytes infiltrate into SiHa tumor in mice injected with the E7/allo-CD4+ T cells. The CD4+ T cells (1 × 107) were injected into palpable subcutaneous SiHa tumors and examined 6 days later. (A, B) H&E staining of sections of paraffin-embedded SiHa tumor tissue injected with NS3/allo-CD4+ T cells (A) and with E7/allo-CD4+ T cells (B). V, blood vessel; T, tumor tissue. Yellow arrows indicate lymphocytes infiltrating the perivascular edematous connective tissue. Green arrows indicate granulocytes. (C, D) Immunohistochemistry with anti-human CD4 antibody of sections of paraffin-embedded SiHa tumor tissue injected with the NS3/allo-CD4+ T cells (C) and with the E7/allo-CD4+ T cells (D). Human CD4 is shown in brown. Original magnification 400x.
Adoptive transfer of CD4+ T cells is able to inhibit the growth of human tumor or lymphoblastoid cell line xenograft in mouse (32,36). Although cytolytic activity of the CD4+ T cells is important in the machinery, the allorestricted CD4+ T cells raised by the coculture exhibited little cytotoxicity against target bearing specific peptide/HLA-DR15 complex (data not shown). On the other hand, soluble factors secreted from activated CD4+ T cells, likely acting on the tumor and its microenvironment, are responsible for the enhancement of the in vivo chemotherapeutic antitumor activity (37), and indirect tumor-killing effects of IFN-γ are reported in human tumor xenograft models (17). It is suggested that the allorestricted CD4+ T cells recognized tumor and became activated to ultimately result in tumor rejection via complicated, presently incompletely defined mechanisms, since the allorestricted CD4+ T cells showed TCR ligand-specific IFN-γ production (Fig. 3B, C) as well as more CD4+ T cells and granulocytes were observed in the tumor with the E7/allo-CD4+ T cells transferred than that with the NS3/allo-CD4+ T cells (Fig. 5).
Discussion
CD4+ T cells play a central role in immune responses to a variety of antigens, such as allograft and tumor. GVLR or GVTR after allograft of hemopoietic stem cells suggests the feasibility of exploiting T-cell repertoire of allogeneic donors to circumvent tumor tolerance. Allorestricted CD4+ T cells specific for tumor antigen are expected to mount an effective response to tumor, as CD4+ T cells are essential for allorejection (7,26). A question is how to prepare the allorestricted tumor-specific CD4+ T cells. Coculturing of tumor cells with lymphocytes is probably the most convenient approach to generating tumor-specific T cells, but this method requires the stimulator and autologous effector cells. Because it is very difficult to ensure MHC molecules and their associated endogenous peptides expressed by allogeneic live cells, in a case of coculture of tumor cells and allogeneic lymphocytes, a different set of “irrelevant” endogenous peptides derived from miscellaneous components of allogeneic stimulator cells would be presented by the MHC molecules, leading to potential stimulation of T cells specific for a variety of allogeneic epitopes. One of solutions to the problem is to introduce a single alloepitope into the coculture of stimulator and autologous effector cells.
A dimeric HLA-DR15/IgG1-Fc fusion protein was prepared in our study for the above purpose. The scaffolding of the dimeric HLA-DR15 is Fc fragment of human IgG1 that is able to bind to Fc γRI (11). FcγRI is constitutively expressed on monocytes with high affinity (Kd = 10−8 M) for monomeric IgG1. The binding of monomeric IgG1 to FcγRI stimulates little endocytosis (19), or the non-cross-linked FcγRI-IgG1 is internalized but rapidly recycled to the cell surface without entering the endocytic pathway (16). Our previous study shows the IgG1-Fc containing dimer remains membrane associated on monocyte surface for several days (51). The specificity of the dimer can be manipulated by pulsing antigenic peptide into the groove of MHC molecule. Apparently, binding of the dimer to HLA-DR15-ve monocytes introduces the peptide/HLA-DR15 complex as a single alloepitope. There have been several approaches to making cells to present with a single alloepitope, such as the peptide-pulsed T2 cells (34,39), allogeneic pMHC-coated B cells (41), allogeneic MHC, and antigen RNA cotransfected dendritic cells (53).
Compared with them, our approach has more flexibility and introduces neither genetic material nor foreign protein except the alloepitope, which can be extended to introducing alloepitopes formed by any HLA allele, provided the HLA allele-restricted peptide is well defined.
After coculturing of HLA-DR15-ve lymphocytes and autologous monocytes loaded with the peptide/HLA-DR15 dimer, an increased CD4+ T-cell proliferation indicated that the introduced alloepitope was able to ignite CD4+ T-cell alloresponse. The allorestricted peptide-specific properties of the expanded CD4+ T cells were observed, as the cocultural bulks were more frequently stained with specific dimer than that with nonspecific dimer and the CD4+ T cells showed an alloepitope-specific response of IFN-γ production. Obviously, the core of the coculture is exploiting T-cell repertoire of HLA mismatched individuals, because in thymus alloreactive T cells have not been negatively selected to ignore peptides bound by allogeneic MHC, and it would be unlikely that alloreactive T cells fail in positive selection, as allelic residuals of MHC molecules lie on the bottom or inner wall of the groove, away from interface of TCR-pMHC interaction. For the CD4+ T cells of HLA-DR15-ve individual, peptide/HLA-DR15 complex would be able to ignite alloresponse, as the alloepitope is encountered by the T cells for the first time.
Therefore, it is possible to prepare alloreactive T cells of defined specificity with the coculture. Interestingly, recent studies have shown that allorestricted T cells bear superior antitumor activity and higher TCR functional avidity compared with self-restricted T cells (41,53).
E7 onco-protein of HPV is essential for immortalization and transformation of human squamous epithelial cell (20). SiHa cell expresses E7 antigen but no NS3 antigen; its E7/HLA-DR15 complex forms specific TCR ligand for the E7/allo-CD4+ T cells. Adoptive transfer of the E7/allo-CD4+ T cells significantly improved life span and tumor avoidance of the 2 × 106 SiHa cell-challenged mice, compared with those of the mice treated with the NS3/allo-CD4+ T cells or the CD4+ T cells cocultured with autologous monocytes alone although alloresponse is likely to take place in the latter two cases. Alloresponse could deal with a tumor load of 1 × 106 cells (Fig. 4A, B), while the tumor-specific T cells could do with that of 2 × 106 cells (Fig. 4C, D), reflecting an additional tumor-rejection effect of the E7/allo-CD4+ T cells. The tumor biopsies showed an increase in CD4+ T cells when the E7/allo-CD4+ T cells were injected. These observations indicated that the allorestricted tumor-specific CD4+ T cells were more effective in tumor rejection, implying the association of GVLR or GVTR with the tumor-specific CD4+ T cells raised in patients after allografting of hemopoietic stem cells, although alloreactive T cells in general can attack tumor for most antigenic peptides occupying grooves of the HLA molecules are derived from normal components shared by both tumor and normal cells. Meanwhile, the murine innate antitumor-reactivity (e.g., impact of murine NK cell activity) could not be formally excluded, as the PBS-treated mice appeared to progress on their primary tumor growth but survive for up to 60 days (Fig. 4A, B).
GVLR or GVTR and GVHD are closely linked to each other by recognition of tumor-associated antigens (TAA) (15), minor histocompatibilty antigens (13), and HLA mismatch, and thus are of complex immunological nature. Tumor peptide-specific, allorestricted CD4+ T cells raised by coculturing of lymphocytes and autologous monocytes bearing the peptide/allo-HLA-DR complex show tumor rejection effects, which provides experimental evidence for a alternative understanding of mechanisms involved in GVLR or GVTR and would help to establish strategies to separate GVLR or GVTR from GVHD for clinical practice. The findings argue for the hypothesis that a constellation of endogenous peptides presented by allogeneic MHC as thousands of different epitopes on tumor cell, most of the peptides would be derived from normal cellular components that elicit alloresponse killing tumor (GVLR or GVTR) as well as normal cells (GVHD), while a fraction of the peptides from tumor-related components, such as the E7 protein, to which alloresponse would account mainly for GVLR or GVTR. Although the allorestricted CD4+ T cells would be sufficient to combat residual cancer cells without additional CD8+ effectors, complete abrogation of GVHD would require a refined screening for the defined specificity due to cross-priming, as virus-specific T cells have been reported to contribute to alloreactivity (1). In the case of cellular immunotherapy, the allorestricted CD4+ T cells adoptively transferred would carry out their function in days (like delayed type hypersensitivity) and be rejected by the host in 1–2 weeks. The rejection would provide natural means of terminating the T-cell effects, and the second wave of the allorestricted T cells from a third, unrelated donor would be expected to work for further 1–2 weeks. On the other hand, many efforts have been made to develop TCR gene therapies (3) and the outcome is promising (18,33), a large number of human TCRs specific for various tumor or viral antigens restricted by different HLA alleles would be required to treat random patients with malignancies or chronic viral infections. For allorestricted T cell gives TCR of higher avidity (41,53), the coculture described above is a practical and flexible protocol to raise allorestricted peptide-specific CD4+ T cells, from which human high-avidity TCRs specific for tumor or viral antigens would be prepared.
Footnotes
Acknowledgments
This study was supported by grants from the National Natural Science Foundation of China (30772040, 30801017, and 30972697), the “973” and the “863” project of the Chinese Department of Science and Technology (2007CB512900, 2008AA02Z113). We thank Drs. Jinsong Pen, Shigang Shan, Xian Wang, and Lifeng Jin for their technical assistance with this work, and Drs. Zheng Tan, Victor Tonje Jeza, and Xiangwei Wu for helpful discussion. The authors declare no conflict of interest.
