Abstract
T-cell dysregulation plays an important role in the pathogenesis of immunoglobulin A nephropathy (IgAN). Adipose-derived stem cells (ASCs) have been reported to be able to prevent tissue damage through immune-modulating effects. To evaluate the effects of ASCs in high IgA ddY (HIGA) mice, ASCs were isolated from HIGA mice with different stages of IgAN before and after disease onset. ASCs were injected at a dose of 5 × 106 cells/kg body weight through the tail vein every 2 weeks for 3 months. Although the administered ASCs were rarely detected in the glomeruli, 24-h proteinuria was markedly decreased in all ASC-treated groups. Although glomerular deposition of IgA was not significantly different among groups, mesangial proliferation and glomerulosclerosis were dramatically decreased in most ASC treatment groups. In addition, levels of fibrotic and inflammatory molecules were markedly decreased by ASC treatment. Interestingly, ASC therapy significantly decreased Th1 cytokine activity in the kidney and caused a shift to Th2 responses in spleen T-cells as determined by FACS analysis. Furthermore, conditioned media from ASCs abrogated aggregated IgA-induced Th1 cytokine production in cultured HIGA mesangial cells. These results suggest that the beneficial effects of ASC treatment in IgAN occur via paracrine mechanisms that modulate the Th1/Th2 cytokine balance. ASCs are therefore a promising new therapeutic agent for the treatment of IgAN.
Introduction
Immunoglobulin A nephropathy (IgAN) is the most common type of glomerulonephritis in the world and more common in Asia than the rest of the world (11,47). Although IgAN is considered a benign disease, almost 20–40% of patients experience chronic renal failure (2,21). Although the deposition of IgA immune complexes in the mesangium is the major pathological finding in IgAN, the exact pathogenesis of IgAN is not yet known.
In previous studies, increases in IgA-containing B lymphocytes, IgA-specific T-helper cells, and decreased activity of IgA-specific suppressor T-cells were observed in IgAN patients (16,36,37). These results implicate lymphocyte dysregulation in IgAN. In one recent study, administration of purified IgA antiphosphorylcholine and the specific pneumococcal C-polysaccharide antigen-induced renal injury in B-cell-deficient mice, but not in both B-cell and T-cell-deficient mice (7). Taken together, these data suggest an important role for T lymphocytes in the development of IgAN. Corticosteroids and other immunosuppressants are the major agents used to treat IgAN. Their therapeutic effect is related to their ability to regulate T-cell immunity and the Th1/Th2 balance. In a recent study, bone marrow transplantations from IgAN mice to quiescent mice induced IgAN (40). However, stem cell therapy does not currently have a defined role in the treatment of IgAN.
Mesenchymal stem cells (MSCs) have been studied in the context of nephrology (33). MSCs can differentiate into various cell types, migrate to sites of tissue injury, and enhance repair by secreting antifibrotic and proangiogenic factors (31,33,39). Over the past few years, adipose tissue has attracted much research attention because adipose-derived stem cells (ASCs) from adipose tissue contain greater numbers of MSCs than bone marrow-derived stem cells from bone marrow (3). Undifferentiated, self-renewable ASCs can differentiate into osteoblasts, chondrocytes, myocytes, adipocytes, cardiomyocytes, ehdothelial cells, or hepatocytes (3,31,34,40). ASCs have been demonstrated to have significant potential to rescue tissues from damage in various disease models through their ready availability, proangiogenic effects, antiapoptotic factor secretion, capacity for multilineage differentiation, and immune-modulating effects (18,30,38).
Because IgA nephropathy is an immunological disease and ASCs have immune-modulating effects, ASCs may be effective at treating this disease. However, no previous study has examined the effects of ASCs in IgAN. In this study, we investigated the effect of ASCs on renal function and renal inflammation in high IgA ddY (HIGA) mice, an animal model of IgAN. A previous report suggested that bone marrow cells at different stages of IgAN such as preonset or postonset IgAN could have different effects We therefore isolated ASCs from HIGA mice at different stages of IgAN before and after disease onset and examined the effects of different sets of ASCs (40). Additionally, to define the molecular mechanisms underlying the observed effects of the ASCs, we evaluated the Th1/Th2 cytokine balance. Furthermore, we performed in vitro experiments in primary cultured HIGA mesangial cells to determine whether the therapeutic effect of ASCs was mediated via engraftment of ASCs or via paracrine mechanisms.
Materials and Methods
Animal Experiments
We purchased female 8-week-old HIGA mice (high IgA ddy mice, Japan SLC, Inc., Hamamatsu, Japan) for the experiments. Mice were divided into four groups, and each group contained 10 mice. All mice were provided with a standard diet and water. They were maintained in a temperature (23 ± 2°C) and humidity-controlled (55 ± 5%) room with a 12-h light–dark cycle. At 2-week intervals, we measured 24-h urine microalbuminuria, and at the age of 24 weeks, all mice showed microalbuminuria and we started treatment. Group 1 (control) mice were injected with Dulbecco's modified Eagle's medium (DMEM) (vehicle). Group 2 mice were injected with preonset adipose-derived stem cells extracted from HIGA mice before disease manifestation (preonset ASC group). Group 3 mice were injected with postonset adipose-derived stem cells extracted from HIGA mice after disease manifestation (postonset ASC group). Group 4 mice were injected with adipose-derived stem cells from humans (human ASCs). ASCs were injected through the tail vein every 2 weeks for 3 month at a dose of 5 × 106 cells/kg body weight. During treatment, 24-h urinary excretion of protein was measured at 2-, 4-, 10-, and 12-week intervals after ASC injection, and at the end of the study period, urinary levels of neutrophil gelatinase-associated lipocalin (NGAL) were measured. All urine samples were centrifuged at 14,000 rpm for 5 min, and clear supernatant was used for urine protein and NGAL assays. For urinary protein measurements, urine samples were precipitated with an equal volume of 30% trichloroacetic acid (TCA), dissolved in 1 M NaOH, then assayed using the Bio-Rad protein assay (Hercules, CA, USA). The results are expressed as the protein:creatinine ratio. Urinary albumin and NGAL concentrations were determined using a competitive ELISA (Shibayagi, Shibukawa, Japan) and mouse NGAL sandwich ELISA kit (BioPorto Diagnostics, Grusbakken, Gentofte, Denmark), respectively. Urinary creatinine levels were measured by a creatinine colorimetric detection kit (Assay Designs, Philadelphia, PA, USA). Serum concentrations of IgA at the end of the study period were determined using a mouse IgA ELISA kit (Life Diagnostics, Inc., Philadelphia, PA, USA). Serum creatinine levels were determined using a modified Jaffe method. Concentrations of Th1 and Th2 cytokines in the serum and urine were determined using Millipore's MILLIPLEX™ mouse cytokine/chemokine kit (Millipore Corp., St. Charles, MO, USA) according to the manufacturer's instructions with a Luminex 200 system (Luminex Corporation, Austin, TX, USA). After 3 months of treatment, surviving mice were killed under anesthesia by IP injection of sodium pentobarbital (50 mg/kg), and spleen and kidney tissues were weighed and subsequently snap-frozen in liquid nitrogen. All the animal experiments were conducted in accordance with the National Institute of Health guidelines and with the approval of the Korea University Institutional Animal Care and Use Committee.
Isolation and Culture of Adipose-Derived Stem Cells
Additional female HIGA mice were also purchased for harvesting ASCs. ASCs were isolated and cultured from the inguinal fat pad of HIGA mice. To obtain preonset ASCs, the inguinal fat pad was isolated from 9-week-old HIGA mice (n = 6) that did not show microalbuminuria (6.74 ± 0.91 μg/mg creatinine). To obtain postonset ASCs, the inguinal fat pad was isolated from 29-week-old HIGA mice (n = 21) with microalbuminuria (28.36 ± 4.50 μg/mg creatinine). Fat pads were minced into small pieces and digested in phosphate-buffered saline (PBS; Hyclone, Boston, MA, USA) containing 1% bovine serum albumin (BSA) and 0.025% collagenase for 80 min at 37°C with intermittent shaking. Isolated ASCs were cultured in DMEM containing 10% fetal bovine serum (FBS) and 1 ng/ml basic fibroblast growth factor (bFGF) and used at passage 3. Human ASCs were isolated from lipoaspirates of human subcutaneous fat tissue obtained from healthy females who provided informed consent. Lipoaspirates were washed at least three times with PBS and then processed in the same manner as described above. Since human ASCs have an equivalent immunophenotype and immunoregulatory properties similar to those of murine ASCs, we also investigated the potential therapeutic action of human ASCs in a mouse model of IgAN.
Characterization of Adipose-Derived Stem Cells
ASCs were characterized by determining surface marker expression by FACS analysis, a differentiation potency assay, and an immunosuppressive potency assay. Briefly, ASCs at passage 3 were harvested by trypsin-EDTA digestion and washed with phosphate-buffered saline (PBS) that contained 1% fetal bovine serum (FBS). Cells were incubated with monoclonal antibodies for 30 min at 4°C, then washed, and analyzed. Antibodies against CD14, CD29, CD34, CD44, CD45, CD73, and CD105 were used (BD Pharmingen, Franklin Lakes, NJ, USA). Isotype-matched negative controls were used to define background staining. The data were analyzed by collecting 10,000 events on a BD FACSCanto II flow cytometer system using fluorescence-activated cell sorter (FACS) Diva software (BD Biosciences, Franklin Lakes, NJ, USA). To assess the differentiation potency of HIGA mice-derived ASCs, expanded cells were plated at a density of 5,000 cells/cm2. When cells were 100% confluent, they were treated with the following differentiation medium: DMEM Ham's F-12 supplemented with 10% FBS, 33 μmol/l biotin, 17 μmol/L pantothenate, 10 μmol/L insulin, 1 μmol/L dexamethasone, 0.1875 mmol/L isobutylmethylxanthine (IBMX), and 0.2 mmol/L indomethacin. After 3 days of adipogenic induction period, cells were fed with the same medium, but without IBMX and indomethacin, for an additional 2–5 days. Adipogenic differentiation was assessed in the presence of intracellular lipid droplets by observing cellular morphology and Oil Red O staining. To assess the immunosuppressive potency of ASCs from HIGA mice and control BALB/c mice, they were plated onto 48-well plates (3×104 cells/well) and allowed to adhere to the plate for 2 h. BALB/c spleen cells (9×105 cells/well) were added to wells containing ASCs in the presence of the immune activator, Concanavalin A (5 μg/ml; Sigma, St. Louis, MO, USA). Mixtures were incubated for 48 h at 37°C in a CO2 incubator. After 48 h, supernatants were collected and analyzed for the proinflammatory cytokines interferon (IFN)-γ by ELISA (R&D Systems, Minneapolis, MN, USA). Remaining spleen cells were treated with 5-ethynyl-2′-deoxyuridine (Edu; Invitrogen, Eugene, OR, USA) for 20 h, harvested, then stained with anti-Edu antibody (Invitrogen), and analyzed by flow cytometry. Proliferation inhibition (%) was calculated as follows: [(% of EdU-positive cell in the spleen cells treated with ConA)–(% of EdU-positive cells in the spleen cells treated with ConA and ASCs)] / (% of EdU-positive cell in the spleen cells treated with ConA)x 100.
Localization of Injected Adipose-Derived Stem Cells
To determine whether injected ASCs were engrafted in the kidney, we labeled passage 2 ASCs with 1,1′-dioctadecyl-3,3,3′3′-tetramethylindocarbocyanine per-chlorate (DiI; Molecular Probe, Eugene, OR, USA) followed by passage through a 40-μm mesh filter. DiI-labeled ASCs were injected through the tail vein at the dose of 106 cells/kg body weight. After 20 h of injection of ASCs, multiple organs were harvested for detection of DiI-labeled ASCs. To assess the localization of ASCs, half of the tissues were snap-frozen, and 4-mm-thick tissue sections were used for immunofluorescence microscopy. For FACS analysis, the remaining tissues were meshed, and single cell suspensions were passed through a nylon mesh (100 μm; BD Bioscience) and collected into PBS. Erythrocytes were lysed with 160 mM NH4Cl and subsequently washed with PBS and DiI-expressing cells were assessed by FACS analysis.
Analysis of Spleen IgA-Bearing B-cells and the Th1/Th2 Profile in Experimental Animals
To assess changes in IgA-bearing B-cells and the Th1/Th2 cytokine profiles of spleen cells, spleens were removed from the mice at the end of the study period. Spleen cells were isolated from each tested HIGA mice. Cells were treated with 160 mM NH4Cl (Sigma-Aldrich, St. Louis, MO, USA) to lyse erythrocytes and subsequently washed three times with RPMI 1640. To ascertain IgA-bearing B-cells, spleen cells were stained with Alexa Fluor 647-CD45R and fluorescein isothiocyanate (FITC)-IgA monoclonal antibody (MoAb) (BD Bioscience). Samples were acquired using a FACS Canto II (BD) and analyzed using CellQuest software (BD). To assess the production of Th1/Th2-associated cytokines, spleen cells were stimulated with phorbol 12-myristate 13-acetate (PMA, 0.2 ng/ml) and ionomyin (1 ng/ml, Sigma) for 24 h. Brefeldin A (10 ng/ml, Sigma) was added to the culture to inhibit protein secretion. After stimulation, cells were stained with phycoerythrin (PE)-CD4 MoAb, and intracellular staining was subsequently performed with fix/permeabilization buffer and peridinin-chlorophyll protein cyanine 5.5 (PerCP-Cy5.5)-interleukin-4 (IL-4) or FITC-IFN-γ MoAb (BD Bioscience). Samples were acquired using a FACS Canto II (BD) and analyzed using CellQuest software (BD). In addition, to determine the cytokines secreted from spleen cells, spleen cells (3×106 cells/well) were cultured in 48-well plates for 72 h with or without stimulation. Cell culture supernatants were stored at −20°C until analysis. IFN-γ, tumor necrosis factor (TNF)-α, IL-4, and IL-10 ELISA kits were obtained from R&D Systems. ELISA assays were performed according to the manufacturer's instructions.
Analysis of Gene Expression by Real-Time Quantitative PCR
Total RNA was extracted from renal cortical tissues and experimental cells with Trizol reagent. Quantitative gene expression was performed according to the method described previously (23). The nucleotide sequences of all of the primers used in this study are shown in Table 1. PCR was performed on a LightCycler® 1.5 system (Roche Diagnostics Corporation, Indianapolis, IN, USA) using SYBR Green technology. In 32-well, real-time PCR plates, 10 μl SYBR Green master mix was added to 1 μl of RNA (corresponding to 50 ng of total RNA) and 900 nM of forward and reverse primers for a total reaction volume of 20 μl. Real-time RT-PCR was performed by incubating samples for 10 min at 95°C, followed by 30 cycles of denaturation for 10 s at 95°C and annealing with extension for 30 s at 60°C. The expression of each gene relative to the β-actin level (relative gene expression number) was calculated by subtracting the threshold cycle number (Ct) of the target gene from that of β-actin and raising two to the power of this difference. The specificity of each PCR product was evaluated by melting curve analysis. In this experiment, each sample was run in triplicate, and the corresponding non-reverse-transcribed mRNA samples were used as negative controls. The mRNA levels of each sample were normalized to that of β-actin mRNA.
Primer Sequences for Real-Time Quantitative PCR

HIGA, high IgA ddY; MCP-1, monocyte chemoattractant protein-1; PAI-1, plasminogen activator inhibitor-1, TGF-β1, transforming growth factor-β1; Col-IV, type IV collagen; TNF-α, tumor necrosis factor-α; IFN-γ, interferon-γ; IL-2, interleukin-2; FOXP3, Forkhead box P3; CCR3, chemokine receptor type 3; CXCR3, C-X-C chemokine receptor type 3.
Histopathological Analysis
Kidney tissues embedded in paraffin were cut into 4-mm-thick slices and stained with periodic acid–Schiff stain. A semiquantitative score (GSI) was used to evaluate the degree of glomerulosclerosis on periodic acid–Schiff (PAS)-stained sections according to the method described previously (23). The severity of sclerosis for each glomerulus was graded from 0 to 4+ as follows: 0, no lesion; 1+, sclerosis of <25% of the glomerulus; 2+, sclerosis of 25–50%; 3+, sclerosis of >50–75%; and 4+, sclerosis of >75%. The mesangial proliferation index (MPI) was determined by calculating the total cell number in each glomerulus except endothelial cells and epithelial cells under a high-power field containing 30–40 glomeruli, and an average score was calculated. For immunohistochemical staining, renal tissue was sliced into 4-μm-thick sections and transferred to a 10 mmol/L citrate buffer solution at pH 6.0 and heated at 80°C for 12 min to retrieve antigens for transforming growth factor (TGF)-β1 staining. Alternatively, sections were transferred to a Biogenex Retrievit (pH 8.0) (InnoGenex, San Ramon, CA, USA) and microwaved for 10 min for antigen retrieval before plasminogen activator inhibitor-1 (PAI-1) staining. For type IV collagen and F4/80 staining, slides were treated with trypsin (1 tablet per 1 ml H2O) for 20 min for antigen retrieval. To block endogenous peroxidase activity, 3.0% H2O2 in methanol was applied for 20 min after washing in water, followed by slide incubation at room temperature for 60 min with 3% bovine serum albumin/3% normal goat serum (type IV collagen), 10 min with 0.1% BSA in 1% powerblock (Biogenex HK085-5K), 15 min with 10% powerblock (PAI-1), or 30 min with 20% normal sheep serum (TGF-β1). Slides were then incubated overnight at 4°C with a rabbit polyclonal anti-TGF-β1 antibody (1:100; Santa Cruz Biotechnology, Santa Cruz, CA, USA), a rabbit polyclonal anti-type IV collagen antibody (1:150; BioDesign International, Sarco, ME, USA), a rabbit polyclonal anti-PAI-1 antibody (1:60; American Diagnostica, Stamford, CT, USA), or a rat anti-mouse F4/80 [epidermal growth factor-like module-containing mucin-like hormone receptor-like 1 (EMR1)] antibody (1:20; Serotec, Inc., Raleigh, NC, USA). Slides were then incubated at room temperature with the respective secondary antibodies for 30 min. For coloration, slides were incubated with a mixture of 0.05% 3,30-diaminobenzidine containing 0.01% H2O2 and counterstained with Mayer's hematoxylin. Negative control sections were stained under identical conditions with a buffer solution substituted for the primary antibody. To evaluate immunohistochemical staining for type IV collagen, TGF-β1, and PAI-1, glomerular fields were graded semiquantitatively under a high-power field containing 30–40 glomeruli and an average score was calculated as described previously (23). Infiltrating macrophages in the interstitium were counted and expressed as the number of macrophages per high-power field. To determine the extent of IgA deposition in the glomeruli, frozen 4-mm-thick renal sections were fixed in cold acetone, and slides were stained with fluorescein isothiocyanate-conjugated rat anti-mouse IgA antibody (1:50, BD Biosciences, San Diego, CA, USA) for 1 h at 37°C and then examined under an immunofluorescence microscope (LSM 5 PASCAL; Carl Zeiss Microimaging, Berlin, Germany). The score for IgA staining was semiquantitatively assessed. Briefly, a score that reflected both changes in the extent and intensity of staining was given. All fields were graded according to a 4-point scale, with a grade of 0 representing absent; grade 1, weak positive staining; grade 2, moderate positive staining; grade 3, strong positive staining. More than 30 glomeruli were evaluated under high power fields, and average scores were calculated by a pathologist in a blinded manner.
Mesangial Cell Culture
A part of the renal cortex from 8-week-old HIGA mice was obtained immediately after surgical nephrectomy and glomeruli were isolated using a differential sieving method. Mesangial cells (MCs) were cultured in DMEM containing 10% FBS. Aggregated IgA or IgG was prepared by heating monomeric mouse IgA (Bethyl Laboratories, Inc., Montgomery, TX, USA) or IgG, respectively, for 150 min at 63°C as described previously (15). After cooling at room temperature, the proteins were centrifuged at 11,000 x g for 5 min to remove insoluble precipitant. The supernatants were then used as heat-aggregated IgA or IgG. Subconfluent MCs were serum-starved for 24 h, and mesangial cells were treated with or without IgA aggregates or IgG aggregates at a final concentration of 100 μg/ml for 24 h. This dose of IgA aggregates was selected based on a previous study in mesangial cells (15). To evaluate whether the beneficial effect of ASCs were mediated by paracrine effects, conditioned media obtained from culture of preonset ASCs, postonset ASCs, or human ASCs was added to the culture medium at a 20% vol/vol ratio in some wells in the presence or absence of aggragated IgA or IgG. Secreted Th1/Th2 cytokines were measured in culture supernatants using Millipore's MILLIPLEX™ mouse cytokine/ chemokine kit, and supernatant levels of Th1/Th2 cytokines were expressed relative to the total protein concentration. All experiments were performed in triplicate, and cells were harvested at 24 h to extract total RNA and protein.
Statistical Analysis
Nonparametric analyses were used because of small sample sizes. Multiple comparisons were done by a Kruskal–Wallis test with Bonferroni correction to compare more than two groups, followed by a Mann–Whitney U test, using a microcomputer-assisted program with SPSS for Windows 10.0 (SPSS, Chicago, IL, USA). A value of p < 0.05 was considered statistically significant.
Results
Biochemical and Physical Parameters in HIGA Mice
Mice were divided into four groups, and each group initially contained 10 mice. However, some mice were dead after injection of ASCs in early period of experiment. Then we analyzed the size distribution of ASCs by FACS and separated ASCs with a diameter of 10–20 μm by FACS due to size variations in ASCs. Twenty-six mice survived until the end of the study, and data from these mice were analyzed (Group 1: n = 7, Group 2: n = 5, Group 3: n = 6, and Group 4: n = 8). Table 2 summarizes various biochemical and physical parameters of the experimental animals. Serum creatinine levels were not significantly different among groups, except in mice treated with human ASCs, where it was significantly decreased. The urine albumin to creatinine ratio was lower in all ASC treatment groups than the control group, but this difference was only significant in the postonset ASC and human ASC treatment groups. There was no significant difference in urinary excretion of NGAL, body weight, kidney weight, or spleen weight between groups. Although serum IgA levels tended to be lower in all ASC treatment groups than the control group, this difference was only significant in the group treated with human ASCs because of interindividual variation. Table 2 also summarizes the plasma levels of representative Th1/ Th2 cytokines. There was a tendency toward decreased TNF-α and IFN-γ (Th1 cytokines) levels and increased IL-4 and IL-10 (Th2 cytokines) levels in the treatment groups, but these changes were not statistically significant due to inter-individual variation.
Various Biochemical Parameters and Plasma Levels of Cytokine in HIGA Mice

Cr, creatinine; ACR, albumin to creatinine ratio; NGAL, neutrophil gelatinase-associated lipocalin; P, plasma; TNF-α, tumor necrosis factor-α; IFN-γ, interferon-γ; IL-2, interleukin-2. Group 1, DMEM control group; Group 2, preonset adipose stem cell (ASC)-treated group; Group 3, postonset ASC-treated group; Group 4, human ASC-treated group. Data are shown as median (interquartile).
p < 0.05 versus Group 1.
Characterization and Localization of ASCs
Figure 1A shows the surface immunophenotypes of ASCs as confirmed by FACS analysis. As described previously for ASCs, ASCs from HIGA mice were positive for CD29, CD44, CD73, and CD105 and negative for CD14, CD34, and CD45. Although ASCs from HIGA mice showed less than 50% of CD73- and CD105-positive cells, this may be due to the different strains of experimental mice. In addition, ASCs from HIGA mice showed adipogenic differentiation potential as determined by the presence of intracellular lipid droplets by Oil Red O staining (Fig. 1B). Furthermore, an immunosuppressive potency assay performed by culturing normal mouse spleen cells also showed that ASCs from HIGA mice had marked anti-inflammatory and antiproliferative effects similar to control BALB/c mice (Fig. 1C). To determine the localization of the ASCs 20 h after injection, immunofluorescent microscopy and FACS analysis were performed in three different mice. As shown in Figure 2, most of the DiI-labeled ASCs were localized in the lung, heart, liver, and spleen; we did not find significant engraftment of ASCs in the kidney.

(A) Characterization of adipose-derive stem cells (ASCs) by flow cytometric analysis. Positive cell populations of ASCs for each surface marker are shown in the table together with previous study results. (B) Differentiation potency assay of ASCs. Adipogenic differentiation was assessed by Oil Red O staining after treatment of ASCs with differentiation medium. (C) Assay to determine the immunosuppressive potency of ASCs from high IgA ddY (HIGA) mice and control BALB/c mice. BALB/c spleen cells were cocultured with ASCs in the presence of Concanavalin A, and supernatant interferon gamma (IFN-γ) levels and cell proliferation were measured. Scale bars: 20 and 100 μm.
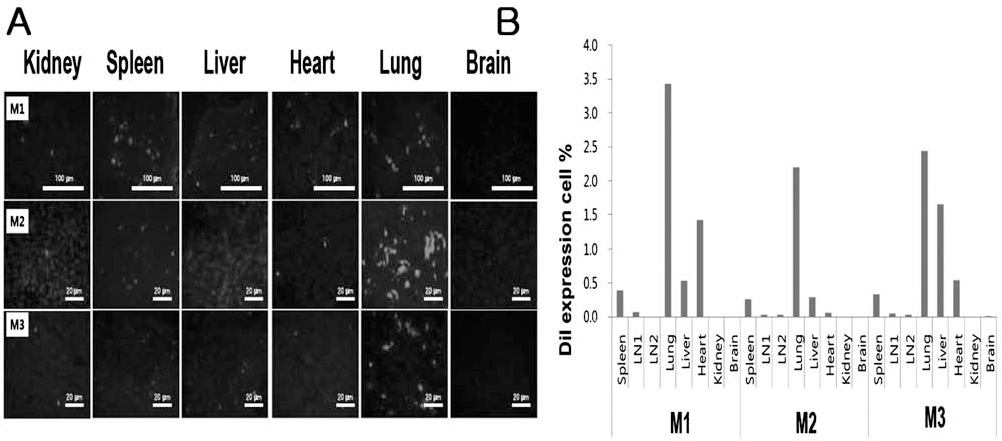
(A) Localization of DiI-labeled ASCs 20 h after intravenous injection as determined by immunofluorescent microscopy. (B) Flow cytometric analysis of DiI-expressing cells to localize DiI-labeled ASCs 20 h after intravenous injection. M1, mouse 1; M2, mouse 2; M3, mouse 3. Scale bars: 20 and 100 μm.
Effects of ASCs on Proteinuria and Renal Histological Changes in HGA Mice
Although we did not observe engraftment of the administered ASCs in the glomeruli, all ASC treatment groups showed dramatically improved proteinuria and renal structural changes. Interestingly, a 24-h urinary protein excretion was markedly decreased in all ASC treatment groups even after 1 month of treatment, and the antiproteinuric effects of ASC treatment persisted throughout the entire study period (Fig. 3A). Representative renal histological findings are shown in Figure 3B, and as shown in Figure 3C, the mesangial proliferation index was significantly decreased in all ASC treatment groups irrespective of whether the ASCs were obtained preonset or postonset IgA nephropathy. The glomerulosclerosis index also showed a significant improvement in the groups treated with preonset ASCs and human ASCs (Fig. 3C). However, mesangial IgA deposition was not significantly different among groups (Fig. 3B).
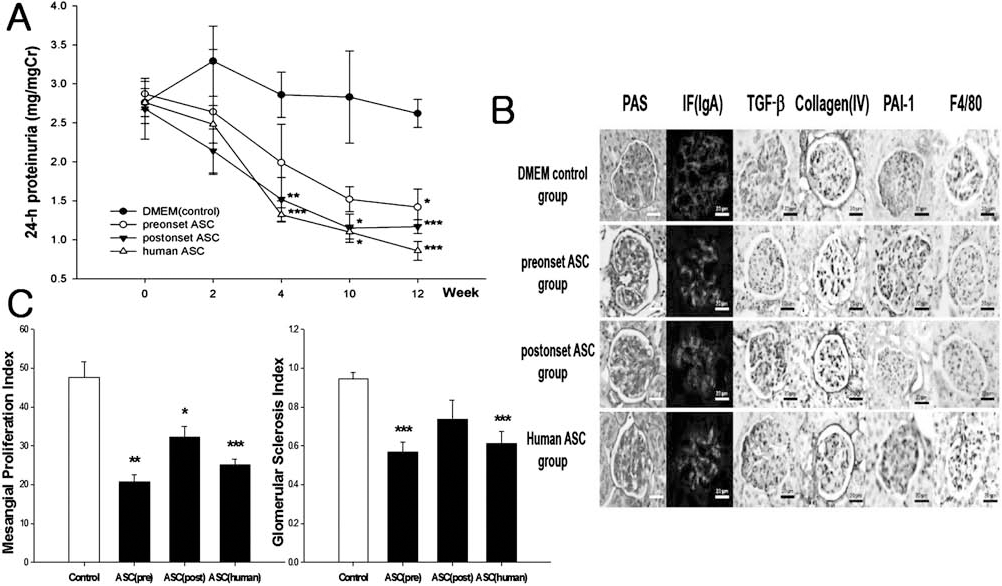
Effects of ASC treatment on renal function in HIGA mice. (A) Twenty-four-hour proteinuria in HIGA mice. Twenty-four-hour urine was collected, and the amount of proteinuria was expressed as the protein-to-creatinine ratio. Comparisons were performed among groups at the same time points. (B) Representative renal histological findings in HIGA mice: periodic acid–Schiff (PAS) stain, immunofluorescence (IF) stain for IgA, transforming growth factor (TGF)-β1 stain, type IV collagen stain, plasminogen activator inhibitor-1 (PAI-1) stain, F4/80 stain. (C) Effects of ASC treatment on renal histological changes including mesangial proliferation index and glomerular sclerosis index in HIGA mice. ASCs, adipose-derived stem cells. Data are means ± SEM. ∗p < 0.05, ∗∗p < 0.01, ∗∗∗p < 0.001 versus DMEM control group. Scale bar: 20 μm.
Anti-inflammatory Effects of the ASCs in HIGA Mice
Because the ASCs markedly improved renal structure, we next investigated the molecular mechanism underlying the positive effects of ASC treatment. We examined the expression of profibrotic and proinflammatory molecules, such as TGF-β1, type IV collagen, PAI-1, and monocyte chemotactic protein-1 (MCP-1) in the kidney. As shown in Figure 4A, the expression of all of these genes was significantly decreased in the treatment groups except for type IV collagen in the postonset ASC treatment group. In accordance with the gene expression changes, immunostaining of these markers in the kidney also revealed a marked decrease in expression of these molecules in the preonset ASC and postonset ASC treatment groups (Fig. 4B).
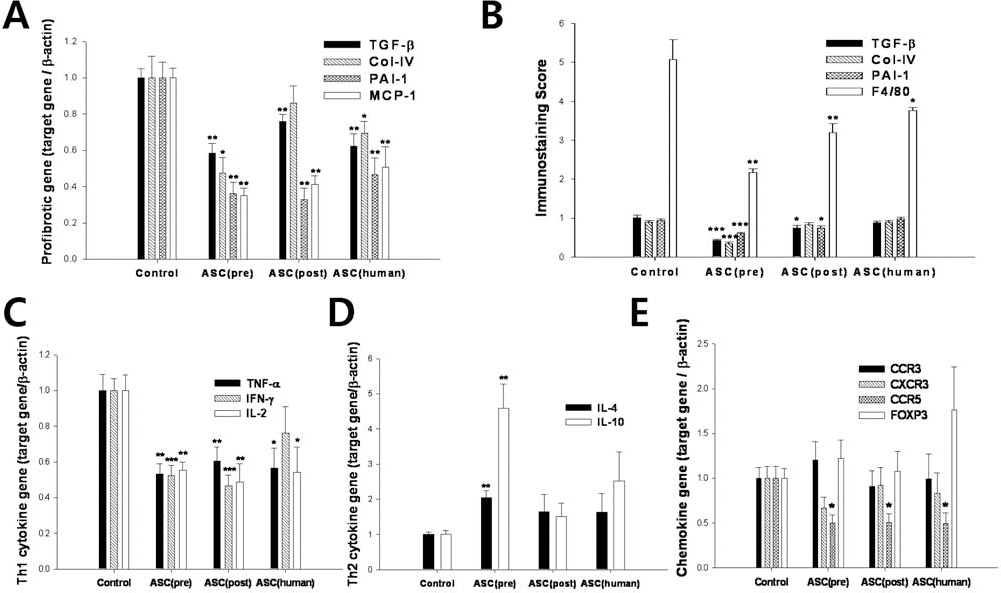
Effects of ASC treatment on the expression of profibrotic, proinflammatory, and Th1/Th2 cytokine molecules in the kidney. (A) mRNA expression of profibrotic genes. (B) Immunostaining score for each molecule. (C) mRNA expression of Th1 cytokine genes. (D) mRNA expression of Th2 cytokine genes. (E) mRNA expression of chemokine genes. ASCs, adipose-derived stem cells; TGF-β transforming growth factor-β; Col-IV, type IV collagen; PAI-1; plasminogen activator inhibitor-1; MCP-1, monocyte chemoattractant protein-1; TNF-α, tumor necrosis factor-α; IFN-γ, interferon-γ; IL-2, interleukin-2. Data are means ± SEM. ∗p < 0.05, ∗∗p < 0.01, ∗∗∗p < 0.001 versus DMEM control group.
Effect of ASCs on the Th1/Th2 Cytokine Balance
Because ASC treatment significantly decreased renal inflammation, we examined changes in the Th1/Th2 cytokine balance after 3 months of ASC treatment. Firstly, we observed changes in spleen T-cell Th1/Th2 cytokine activity after stimulation with PMA. As shown in Table 3, only Th1 cytokine secretion, including TNF-α and IFN-γ, decreased in the preonset and postonset ASC treatment groups. The intracellular cytokine activities in splenic T-cells, determined by measuring the fraction of IFN-γ-and IL-4-positive T-cell by FACS analysis, are shown in Table 3. The ratio of IFN-γ-positive cells to IL-4-positive T-cells was significantly decreased after ASC treatment, which suggests a Th2 shift in spleen T-cells. However, the IgA-bearing B-cell population was not significantly different among groups (data not shown).
Analysis of Cytokine Production of Spleen T-Cells After PMA Stimulation and Cytokine Activity by FACS
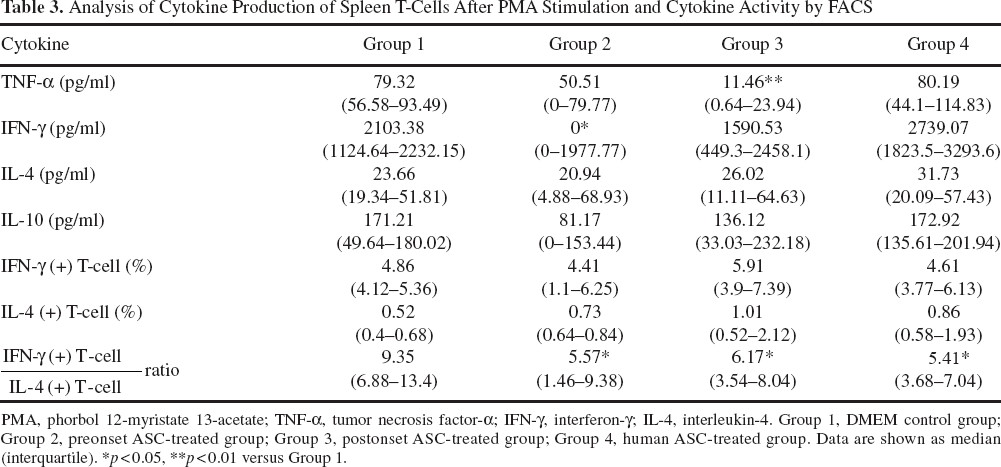
PMA, phorbol 12-myristate 13-acetate; TNF-α, tumor necrosis factor-α; IFN-γ, interferon-γ; IL-4, interleukin-4. Group 1, DMEM control group; Group 2, preonset ASC-treated group; Group 3, postonset ASC-treated group; Group 4, human ASC-treated group. Data are shown as median (interquartile).
∗p < 0.05
∗∗p < 0.01 versus Group 1.
We next examined the changes in Th1/Th2 cytokine levels in the kidney by gene expression analysis and monitoring the urinary excretion of these cytokines. As shown in Figure 4C, the mRNA expression of Th1 cytokines in the kidney was markedly decreased in the ASC treatment groups except for IFN-γ in the human ASC treatment group. In contrast, mRNA expression of Th2 cytokines in the kidney increased after ASC treatment, but this increase was only significant in the preonset ASC treatment group (Fig. 4D). In addition, we monitored chemokine expression in the kidney. Although CCR3, CXCR3, and FOXP3 expression did not show significant changes, CCR5 expression was significantly decreased in the ASC-treated groups (Fig. 4E). We further evaluated the urinary excretion levels of these cytokines, and as shown in Figure 5, urinary excretion of most Th1/Th2 cytokines was decreased in all ASC groups, and among cytokines, TNF-α and IFN-γ levels decreased to the greatest extent after ASC treatment.
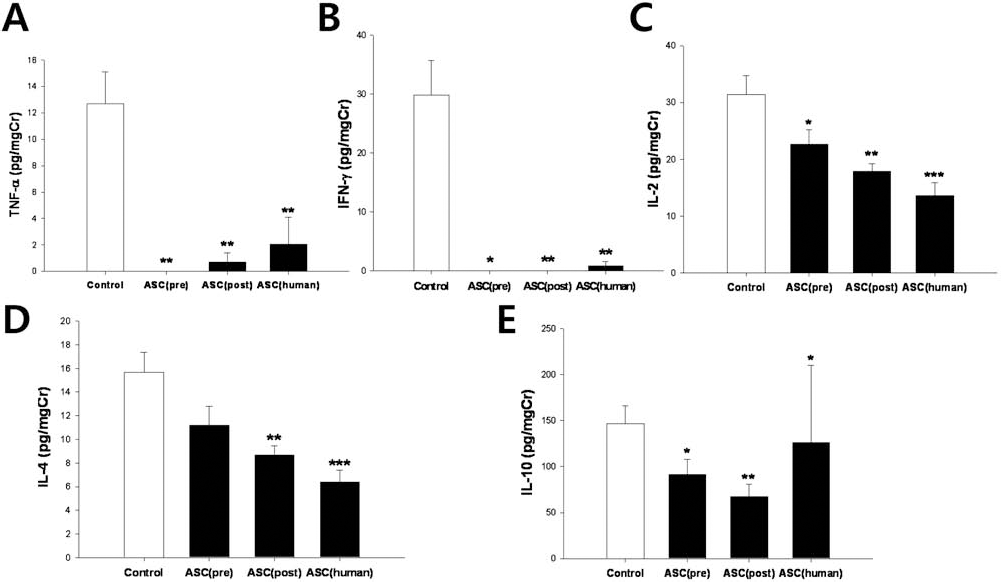
Effects of ASC treatment on 24-h urinary excretion of Th1/Th2 cytokines by HIGA mice. Urinary levels of cytokines were corrected by urine creatinine: ASCs, adipose-derived stem cells; TNF-α, tumor necrosis factor-α; IFN-γ, interferon-γ; IL-2, interleukin-2. Data are means ± SEM. ∗p < 0.05, ∗∗p < 0.01, ∗∗∗p < 0.001 versus DMEM control group.
Mesangial Cell Culture
Because ASC treatment had significant anti-inflammatory effects associated with decreased Th1 cytokine activity in the kideny without engraftment in the glomeruli, we performed in vitro experiments to further define whether the beneficial effects of ASCs were mediated by paracrine mechanisms. Because a previous report suggested that bone marrow cells at different stages of IgA nephropathy such as preonset or postonset IgAN have different effects, we isolated ASCs from HIGA mice with different stages of IgAN before and after disease onset. First, we observed the expression pattern of Th1/Th2 cytokines in HIGA mesangial cells and found that stimulation of cells with IgA aggregates, but not IgG aggregates, resulted in dramatic upregulation of Th1 cytokines (Fig. 6A). Interestingly, administration of conditioned media from all ASCs abrogated IgA aggregate-induced Th1 cytokine upregulation. However, stimulation of cells with IgA aggregates or conditioned media from ASCs did not result in significant changes in Th2 cytokine levels (Fig. 6B). Th1/Th2 cytokine protein secretion showed a similar pattern to the gene expression pattern (Fig. 6C, D). Furthermore, MCP1 secretion increased significantly after IgA aggregate stimulation, and addition of conditioned media from ASCs abolished IgA aggregate-induced MCP1 secretion (Fig. 6E). To exclude nonspecific inhibitory effects of conditioned media, we added conditioned media without IgA aggregates or with IgG aggregates. As shown in Figure 7, conditioned media without IgA aggregates had no significant effects.

Effects of aggregated IgA or IgG and conditioned media from ASCs on Th1/Th2 cytokine and MCP-1 synthesis in cultured HIGA mesangial cells: (A) mRNA levels of Th1 cytokines, (B) mRNA levels of Th2 cytokines, (C) protein levels of Th1 cytokines measured by ELISA, (D) protein levels of Th2 cytokines measured by ELISA, (E) protein levels of MCP-1. ASCs, adipose-derived stem cells; IgG, IgG aggregates; IgA, IgA aggregates; TNF-α, tumor necrosis factor-α; IFN-γ, interferon-γ; IL-2, interleukin-2; MCP-1, monocyte chemoattractant protein-1 protein secretion. Data are means ± SEM. ∗p < 0.05, ∗∗p < 0.01, ∗∗∗p < 0.001 versus control media + IgA aggregates.

Effect of aggregated IgA or IgG and conditioned media from preonset ASCs on Th1/Th2 cytokine synthesis in cultured HIGA mesangial cells: (A) mRNA levels of Th1 cytokines, (B) mRNA levels of Th2 cytokines, (C) protein levels of Th1 cytokines measured by ELISA, (D) protein levels of Th2 cytokines measured by ELISA. ASC, adipose-derived stem cell; IgG, IgG aggregates; IgA, IgA aggregates; TNF-α, tumor necrosis factor-α; IFN-γ, interferon-γ; IL-2, interleukin-2. Data are means ± SEM. ∗p < 0.05 versus contol media + IgA aggregates.
Discussion
We demonstrated that ASC treatment significantly decreased 24-h proteinuria and improved mesangial proliferation and glomerulosclerosis irrespective of the stage of IgA nephropathy, even in mice treated with human ASCs. More importantly, we provided evidence that synthesis of fibrotic and inflammatory molecules was markedly decreased by ASC treatment. We also provided evidence that ASC therapy significantly decreased Th1 cytokine activity in the kidneys and resulted in Th2 polarization of spleen T-cells. Furthermore, we demonstrated that conditioned media from ASCs abrogated aggregated IgA-induced Th1 cytokine production by cultured HIGA mesangial cells.
HIGA mice are a well-established animal model of IgAN (43). In 1985, the first suggestion was made that the Deutschland, Denken, and Yoken (ddy) mouse strain could be an animal model of IgAN (21). Although the penetration of IgA nephropathy in ddy mice was variable, a certain ddy line known as HIGA mice showed a high incidence of IgAN (29). These mice are characterized by mild proteinuria, mesangioproliferative glomerulonephritis with glomerular IgA deposition, and increased IgA levels (43). Furthermore, current therapeutic modalities such as a low protein diet, antiplatelet drugs, angiotensin II receptor blockers, and steroids have been shown to be effective in treating these HIGA mice (43).
In this study, we examined the potential therapeutic effects of ASC treatment in IgA nephropathy using HIGA mice. Several previous studies have examined the role of stem cell therapy in acute and chronic kidney diseases (9,33,46). However, there is no previous study investigating the therapeutic effects of adipose-derived stem cell therapy in IgA nephropathy. Among different kinds of stem cells, we used adipose-derived stem cells, because ASCs share most of the properties of mesenchymal stem cells and large amounts of these cells can be obtained relatively easily from adipose tissue (34).
ASCs had clear beneficial effects in HIGA mice. The amount of proteinuria and structural changes including mesangial expansion and glomerular sclerosis was dramatically improved by ASC treatment. Although there were several discrepancies among specific results between different types of ASCs, an overall therapeutic effect was clear. Moreover, these beneficial effects coincided with changes in the expression of profibrotic or proinflammatory molecules in the kidney.
The most important finding of our study is that ASCs isolated from different stages of IgAN provided similar renal protective effects. These results are in contrast with a previous bone marrow transplantation study that showed that bone marrow taken from mice with commencing IgAN conferred IgAN with Th1 polarization in recipient-quiescent mice (40). However, this discrepancy can be explained by the fact that whole bone marrow transplanation was used in the previous study, not isolated bone marrow stem cells. Our result is important from a clinical perspective, because it indicates the possibility of autotransplantation of ASCs from patients with IgAN irrespective of the stage of the disease.
Human T lymphocytes can be divided into two groups: Th1 cells and Th2 cells. Th1 cells produce IFN-γ, IL-2, or TNF-α and are involved in cell-mediated immunity. Th2 cells produce IL-4, IL-5, or IL-10 and are involved in allergic reactions and humoral immunity (35). The Th1 and Th2 systems are known to counter-regulate one other (22). Some researchers have suggested that differences in the prevalence of various glomerulonephritis in various areas of the world is based on an imbalance between the Th1 and Th2 systems (19,22). However, conflicting results have been reported concerning the Th1/Th2 ratio in IgAN. Several studies have reported Th2 predominance, but more recent studies have reported Th1 predominance in IgAN (8,13,17,28,31,39,42,45). In our study, the mRNA expression of Th1 cytokines in the kidney was decreased in the ASC treatment groups, whereas expression of Th2 cytokines was increased in the treatment groups. The results of our study are therefore consistent with Th1 predominance in IgAN. To the best of our knowledge, ours is the first demonstration that ASC treatment can be used to treat IgAN by modulating the Th1/Th2 cytokine balance through paracrine mechanisms. These results suggest that ASCs are a valuable therapeutic option for the management of IgAN.
The therapeutic effects of MSCs are due mainly to their regenerating and immune-modulating effects. Multiple mechanisms for the therapeutic effects of stem cells have been suggested such as differentiation of stem cells into organ-specific cells or fusion of stem cells with existing differentiated cells or protection of existing cells by either paracrine or endocrine mechanisms. There is evidence that embryonic stem cells can differentiate into tubular cells or mesonephric ducts (24,48). However, in studies that investigated the therapeutic effect of MSCs in an ischemic renal injury model, MSCs did not differentiate into kidney structures (12,41).
There is a large body of evidence that the therapeutic effects of ASCs, both in vitro and in vivo, are mediated by paracrine mechanisms (5,6,25,44). ASCs secrete multiple angiogenic factors, immune modulating cytokines, and anti-inflammatory factors that eventually provide a protective environment for injured tissue (31,40). The essential role of paracrine secretion in mediating the therapeutic effects of ASCs is supported by recent studies that demonstrated that inhibition of hepatocyte growth factor (HGF) and stromal cell-derived factor-1 (SDF-1) markedly reduced the ability of ASCs to promote angiogenesis in ischemic muscle tissue (5,25). The most compelling evidence that stem cell therapy is effective because of paracrine/endocrine mechanisms comes from the finding that administration of conditioned medium from MSCs protected the kidney from toxic injury independently of MSC tubular engraftment (4). Taken together, these findings suggest that ASCs provide organ protective effects via paracrine and/or endocrine mechanisms.
In agreement with the previous studies by Bi et al. and Kunterl et al., we did not find engraftment of ASCs in the glomeruli (4,26). Therefore, we focused more on the paracrine and immune-modulating effects of ASCs. There are many study results that support the immune-modulating effects of MSCs. MSCs inhibit the division of T-cells, the differentiation and maturation of dendritic cells, and the proliferation of B-cells (10,12,14). MSCs cause Th1 cells to decrease IFN-γ secretion and cause Th2 cells to increase IL-4 secretion, promoting a shift from Th1 to Th2 cells (1). In this study, we performed an in vitro experiment to investigate the paracrine effects of ASCs and observed that conditioned media from ASCs directly suppressed IgA aggregate-induced Th1 cytokine synthesis in HIGA mesangial cells. This result confirms that ASCs have paracrine and immune-modulating effects.
Like many other revolutionary therapies, there are several concerns associated with stem cell therapy. Although the probability is not high, cancer risk is the main concern of stem cell therapy (27). Methodologically, injecting a large amount of MSCs can induce an embolism (32). In our study, most experimental animal deaths occurred immediately after the injection of ASCs, most likely due to embolic events. More long-term studies are required to assess the long-term efficiency and safety of ASC treatment. A major advantage of using adipose-derived stem cells is the potential to easily obtain an autologous preparation of these cells from adipose tissue; furthermore, these cells can be expanded rapidly in vitro.
In conclusion, ASC treatment had a beneficial effect both functionally and structurally in an animal model of IgAN and inhibited profibrotic and proinflammatory processes in the kidney. The therapeutic effects of ASCs were probably mediated by paracrine and immune-modulating mechanisms that promoted a Th1 to a Th2 shift. Further studies are required to clarify whether ASCs can be used to treat IgAN in humans.
Footnotes
Acknowledgments
We thank Anterogen Co., Ltd., Seoul, South Korea, for kindly providing adipose-derived stem cells. This work was supported by a grant from the Brain Korea 21 project and a special grant from Korea University. The authors declare no conflict of interest.
