Abstract
Early vascularization of a composite in a critical bone defect is a prerequisite for ingrowth of osteogenic reparative cells to regenerate bone, since lack of vessels does not ensure a sufficient nutritional support of the bone graft. The innovation of this study was to investigate the direct and indirect effects of endothelial progenitor cells (EPCs) and cotransplanted mesenchymal stem cells (MSCs) on the in vivo neovascularization activity in a critical size defect at the early phase of endochondral ossification. Cultivated human EPCs and MSCs were loaded onto β-TCP in vitro. A critical-sized bone defect (5 mm) was created surgically in the femoral diaphysis of adult athymic rat and stabilized with an external fixateur. The bone defects were filled with β-TCP, MSCs seeded on β-TCP, EPCs seeded on β-TCP, and coculture of MSCs and EPCs seeded on β-TCP or autologous bone of rat. After 1 week, the rats were sacrificed. Using quantitative CD34 immunohistochemistry as well as qualitative analysis of vascularization (staining of MHC and VEGF) in decalcified serial sections were performed by means of an image analysis system. Fluorescence microscopy analyzed the direct effects and indirect effects of human implanted EPCs for vessel formation at bone regeneration site. Formation of a primitive vascular plexus was also detectable in the β-TCP, MSC, or autologous bone group, but on a significantly higher level if EPCs alone or combined with MSCs were transplanted. Moreover, highest amount of vascularization were detected when EPCs and MSCs together were implanted. Early vascularization is improved by transplanted EPCs, which formed new vessels directly. Indeed the indirect effect of EPCs to vascularization is much higher. Transplanted EPC release chemotactic factors (VEGF) to recruit EPCs of the host and stimulate vascularization in the bone defect. Transplantation of human EPCs displays a promising approach to improve early vascularization of a scaffold in a critical bone defect. Moreover, coculture of EPCs and MSCs demonstrate also a synergistic effect on new vessel formation and seems to be a potential osteogenic construct for in vivo application.
Keywords
Introduction
More than one million cases of reconstructive surgeries with autologous bone grafting for critical bone defects are carried out wordwide annually. But since the amount of the autologous bone graft remains limited and conditions such as osteoporosis precludes its use, alternatives are aggressively being explored. Bone tissue engineering aims at combining a biomaterial with osteoconductive properties such as β-tricalcium phosphate (β-TCP) and potential osteoinductive substances with viable osteoprogenitor cells, which are expected to be responsible for the osteogenesis process. One prerequisite for tissue regeneration is a readily available population of cells that are both highly renewable and differentiable. These cells are commonly known as stem or precursor cells such as mesenchymal stem cells (MSCs) or endothelial progenitor cells (EPCs) (14).
The ability of MSCs to form bone has been confirmed in various models in vivo (12,25,34). The treatment of large bony defects with the application of scaffolds loaded with bone marrow MSCs has also been undertaken in animal models (2,8,15,19). However, the size of the bone defect may limit the ingrowth of bone-forming cells, since lack of vessels does not ensure a sufficient nutritional support for the bone graft. Early vascularization of the scaffold in the critical bone defect may be necessary for ingrowth of osteogenic reparative cells to regenerate bone in vivo. Optimal bone healing is dependent on adequate vascularization and therefore requires the development of new blood vessels. It might be beneficial to first generate a new vascular network in order to successfully regenerate large bone defects. Growth and development of new blood vessels in the adult is not restricted to angiogenesis but encompasses both vasculogenesis and angiogenesis (9). In the field of vascular biology, a major finding has been the identification of EPCs, which are able to initiate neovascularization (5). These progenitor cells represent a small population with the capacity to proliferate, migrate, and differentiate into cells that line the lumen of blood vessels (23). In general, two types of EPCs are described. “Late EPCs” from the bone marrow, which are positive for CD133/CD34/VEGFR-2, whereas circulating “early EPCs” are positive for CD34/VEGFR-2/CD31/VE-cadherin/von Willebrandt factor (vWF) and leukocyte markers such as CD45 and CD14. Early EPCs demonstrate only weak proliferation but are a rich source of vascular endothelial growth factor (VEGF) (26). It has been demonstrated in a variety of animal models that early EPCs improve the vasculogenesis and are incorporated to a significant extent into new formed vessels (4,32). Prior to the discovery of EPCs, new vessel formation was believed to occur by proliferation of existing endothelial cells. These findings have overturned the previous dogma that vasculogenesis can only occur during embryogenesis and have introduced the notion of cellular therapy as a new approach to therapeutic angiogenesis. Mobilized and incorporated EPCs were detected in ischemic heart tissue (1), fracture healing (24), and distraction osteogenesis (21). Neovascularization activity at the fracture site peaked at day 7 postfracture, the early phase of endochondral ossification (24).
Thus, the combination of MSCs with their osteogenic properties and EPCs with their angiogenic characteristics seeded onto a scaffold has a benefit for the healing of a large bone defect, as we demonstrated in a previous study (28).
The innovation of this study was to investigate the direct and indirect effects of cotransplanted human EPCs and MSCs on the in vivo neovascularization activity in a critical size bone defect at an early phase of endochondral ossification (7 days). Therefore, we evaluated if human transplanted EPCs were able to form vessels directly or if they acted indirectly by releasing chemotactic factors (VEGF) and recruiting endogenous EPCs from the host (rat) in order to improve early vascularization.
Materials and Methods
Scaffold
A commercially available bone graft substitute was studied: Synthetic β-tricalcium phosphate (Chronos®, Synthes, Oberdorf, Switzerland) with a size of 0.7–1.4 mm, 60% porosity, a pore size of 100–500 μm, low mechanical stability, and moderate biodegradability. These small granules exhibit a high surface area; thus, a high amount of cells is seeded. Furthermore, cells migrate easily in the core of the scaffold (14).
Fibronectin Coating of β-TCP
Sterile bone graft granules for all experimental groups were incubated for 60 min in a fibronectin solution (10 μg/ml, Sigma, Deisenhofen, Germany) in PBS without Mg2+ and Ca2+ (PBS-/-). The supernatant was then removed and replaced by PBS-/-. The granules were immediately placed as a dense single layer in a 24-well plate (Nunc, Wiesbaden, Germany) using sterile forceps.
Cell Culture
EPCs were isolated according to the procedure as described previously (14). Briefly, peripheral blood mononucleated cells (PBMCs) were isolated from the buffy coat by density gradient centrifugation (30 min, 900 x g) with Ficoll (1.077 g/ml, Biochrom, Berlin, Germany). The PBMCs were washed twice with cold PBSw/o (10 min, 900 x g), and each 4 × 106 cells/cm2 were cultivated on a fibronectin-coated (10 μg/ml, Sigma), 24-well culture dish in 1 ml of endothelial basal medium (EBM, Cambrex, Verviers, Belgium) supplemented with endothelial growth medium-2 (EGM-2) singlequots at 37°C, 5% CO2. After 48 h, nonadherent and weak adherent cells were removed, the medium was exchanged, and the cells were cultivated for additional 72 h. A parallel preparation was performed to evaluate the percentage of endothelial cell-like differentiated cells. EPCs were identified as previously described (13). In brief, cells presenting double-positive fluorescence [1,1 dioctadecyl-3,3,3,3-tetramethyl-indocarbocyanine-labeled acetylated low-density liproprotein (DiLDL), UEA-lectinfluorescein isothiocyanate (FITC); Sigma] were considered to be EPCs. For the experiments, the cells were detached by incubation (10 min) with accutase (PAA Laboratories, Linz, Austria), were washed once with MesenCult + supplements (Cell Systems, St. Katharinen, Germany), and were subsequentely adjusted to a density of 2.5 × 105 cells in 100 μl.
MSCs were obtained from the iliac crest bone marrow aspirate of a voluntary trauma patient undergoing pelvic surgery using Ficoll density gradient centrifugation (30 min, 1,100 x g, d = 1.073 g/ml, Biochrom, Berlin, Germany) as previously described (30). Patients were informed and signed a consent form. The study was confirmed by the local ethics committee (249/07).
The cells in the interphase were collected and washed twice using PBS (10 min, 900 x g) containing 2% fetal bovine serum (FBS), resuspended in 3 ml MesenCult + supplements (Cell Systems), and were counted using a Neubauer chamber.
Each 4 × 106 cells were seeded in a 25-cm2 culture flask. The cells were expanded over five passages. Subsequently, the cells were detached by incubation with accutase, washed, and resuspended in a medium consisting of 90% fetal calf serum (FCS; PAA Laboratories) and 10% dimethyl sulfoxide (DMSO; Sigma). Aliquots were stored in liquid nitrogen until use.
For the experiments, a portion of the cryoconserved cells were thawed and expanded over two additional passages. The MSCs were then detached by an incubation of 10 min with accutase. After one wash (10 min, 300 g), the cells were resuspended in MesenCult + supplements. The cell suspension was then divided. One part was adjusted to a density of 2.5 × 105 cells in 100 μl and was used subsequently for the experiments. The other portion was used for the confirmation of surface characteristics ascertained to MSCs as described earlier by our group (28). The cryoconservation of the cells ensured that, in all groups, MSCs from one donor in a comparable quality were used.
Major Histocompatibility Complex-1 (MHC-1) Expression on MSCs and EPCs In Vitro
Detection for human characteristics of MSCs and EPCs in vitro were performed by using FACS analysis. Detached MSCs, respectively, detached DiLDL prestained EPCs, were resuspended in PBS-/-, which was supplemented with 2 mM EDTA (Sigma) and 0.5% FCS (PAA Laboratories). Each 100-μl cell suspension was incubated with 7.5 μl of FITC-conjugated monoclonal antibodies against human leukocyte antigen (HLA)-ABC (MSCs, BD Pharmingen, Heidelberg, Germany), MSCs received additionally anti-CD90-phycoerythrin (PE; BD Biosciences, Heidelberg, Germany) for 15 min in the dark. After one wash with above-mentioned buffer, the cells were subjected immediately to flow cytometry. A dual laser four-color FACScalibur (BD Biosciences) was used. At least 10,000 events were recorded. For analysis, the software CellQuest pro (BD Biosciences) was applied.
Composite Scaffold: β-TCP Seeded with EPCs and MSCs
Scaffolds were placed into individual 48-well plate without having direct contact with the surrounding well border. The scaffolds were loaded with 5 × 105 cells of either a mixture composed of 50% MSCs and 50% EPCs or 5 × 105 MSCs alone or 5 × 105 EPCs alone. The cells were dripped in a volume of 200 μl to the bone graft layer and were incubated for 10 min at 37°C. The medium containing the nonadhering cells was then removed and rinsed once again over the bone graft layer, followed by incubation as indicated above. This procedure was repeated three times. Then the granules were gently transferred to another well containing 500 μl MesenCult + supplements. The remaining cells in the supernatant and at the bottom of the initial seeding well were isolated and counted, and the seeding efficiency was calculated [(initial cell number – remaining cell number)/initial cell number x 100%].
Animals and Cell Transplantation
Experimental Setting

β-TCP, β-tricalcium phosphate; MSCs, mesenchymal stem cells; EPCs, endothelial progenitor cells.
A general anesthesia with 1.0 ml of a mixture of Ketavet (100 mg/ml) and Rompun (20 mg/ml) was given intraperitoneally. In order to fix the monolateral external fixator to the anterolateral aspect of the right femur, the right leg of the rat was shaved and cleaned with antiseptic fluid. A lateral longitudinal incision over the femur was made under aseptic conditions. The fascia was cut, and the muscles were separated between the quadriceps femuris and the hamstrings. The bone cortex was drilled using a 0.8-mm bit, and four self-tapping pins were placed at right angles to the surface of the femoral shaft. Four pins were fixed to the external fixateur. An osteotomy was made midshaft between the second and the third pin using an oscillating saw and a critical bone defect of 5 mm was created (Fig. 1). The composite scaffold was implanted into the osseous defect zone, and the wound was closed with continuous subcutaneous stiches using a 4/0-monofilament nylon suture.
A 5-mm critical size defect in the right femur of a rat is stabilized with an external fixateur. The gap is filled with β-tricalcium phosphate (β-TCP) granules. Scale bar: 5 mm.
The animals were fed a standard laboratory diet ad libidum and were housed double in type III cages (800 cm2) in temperature controlled rooms (22°C) having a 12-h light cycle.
Cell Survival and Migration Analysis
One week after implantation of β-TCP alone, β-TCP seeded with MSCs, β-TCP seeded with EPCs, β-TCP seeded with EPCs and MSCs, or autologous bone into a critical size defect of the femur in athymic rat, the degree of vascularization was observed.
To evaluate early vascularization, we analyzed all bone composites for engraftment of human cells. One week after surgery, the rats were killed with an overdose of pentobarbital (150 mg/kg IP) and weighed. Femura were dissected free, and the distraction device was removed. All bones were examined macro- and microscopically for signs of infection or tumors. Blinded with respect to treatment, all pins were checked for pin fixation, and only in cases where all four pins had to be twisted in order to be removed, the rat was included. Bones were taken, frozen, and stored at −80°C. The formalin-fixed femur (20 h in Zinc-Formal-Fixx, 4%, Thermo Electron, Pittsburgh, PA, USA) was subjected to decalcification (14 days a buffer containing 0.25 M Trizma base, both obtained from Sigma).
Histology and Immunohistology
Histology and immunohistology for qualitative determination of ingrowth behavior in decalcified serial sections [staining of anti-rat CD34 (R&D Systems AF4117) MHC-1 and VEGF] as well as quantitative analysis of vascularization were performed using an image analysis system.
Paraffin-embedded sections (5 μm) of the decalcified specimens were taken parallel to the long axis of the femur and incubated with an anti-rat CD34 antibody. As secondary antibody, a rabbit anti-goat (414331F) was applied, and the sections were incubated with a 3-amino-9-ethylcarbazole (AEC, Lab Vision, Dreieich, Germany). Evaluation of new vessel formation in a standardized picture frame was performed using the software Image J.
Histomorphometric assessments were performed with blinded specimens examined in random order. The area for measurement was chosen in the callus next to the cortex, at the surface of the osteotomy cut. Areas of CD34 staining were marked by an independent observer blinded to the group setup, and the number of pixels within the area was evaluated.
For the evaluation of human MHC-1-positive cells, the tissue samples were incubated with monoclonal mouse anti-human MHC-1 (Santa Cruz Biotechnology, Heidelberg, Germany) for 24 h at 4°C to identify the human cells within rat's bone. Analysis were performed using light microscopy (Axioobserver Z1, Zeiss, Göttingen, Germany) in combination with a computer-supported imaging picture analysis system (Axiovision; Zeiss) and qualified.
Immunofluorescence Staining
To detect human transplanted EPCs at the early vascularization of a critical size model using athymic rat, double immunohistochemistry was performed 1 week after surgery with the mouse anti-human anti-rat vWF 1:100 1 h/RT (US Biologocal#V2700) and rabbit anti-human MHC-1 (SantaCruz, Heidelberg, Germany, #sc25619) antibodies (n = 6 in each group). The secondary antibodies for each immunostaining are as follows: FITC-conjugated goat anti-mouse IgG (Abcam, Berlin, Germany, #ab5838) for vWF and cyanine 3 (Cy3)-conjugated goat anti-rabbit IgG 1:20, 1h/RT (Abcam, #ab6939) for MHC-1 staining. DAPI (Vector, Burlingame, CA, USA, #H-1500) was applied for 5 min for nuclear staining. Fluorescence was visualized with an Axiovert 100 M microscope (Zeiss) equipped with a digital camera (Axiocam; Zeiss) and imaging software (Axiovision, Zeiss).
All morphometric studies were performed by an experienced examiner who was blinded to each experimental group.
Statistics
Results are presented as median and interquartiles (median, 25% quartile, 75% quartile). Kruskal–Wallis test with Dunn's post hoc test for multiplicity was used for comparisons between the groups. A value of p <0.05 indicates statistical significance.
Results
Human EPCs and MSCs Express MHC-1 Using FACS Analysis
Nearly all MSCs express MHC-1 on their surface (Fig. 2A, B). A concomittant staining with anti-CD90 identifies the MSCs. EPCs express also MHC-1 (Fig. 2C, D). A concomittant DiLDL staining identifies the EPCs.
Human cells express major histocompatibility complex-1 (MHC-1) using fluorescence-activated cell sorting (FACS)-analysis. Mesenchymal stem cells (MSCs) express MHC-1:(A) isotype control and (B) MHC-1 staining, a concomitant staining with anti-CD90 identifies the MSCs. Nearly all MSCs express MHC-1 on their surface. EPCs also express MHC-1. (C) isotype control and (D) MHC-1 staining. A concomittant low density lipoprotein (DiLDL) staining identifies the endothelial progenitor cells (EPCs). PE, phycoerythrin; FITC, fluorescein isothiocyanate.
EPCs and EPCs/MSCs Loaded β-TCP Improve Early Vascularization
After 1 week, areas of neovascularization in the critical bone defect of male athymic rats treated with β-TCP, β-TCP + MSCs, β-TCP + EPCs, β-TCP + EPCs + MSCs, or autologous rat bone (Fig. 3A–E) were detected by CD34 histomorphochemistry.
Representative CD34-stained histological sections of the defect site of male athymic rats treated with (A) β-TCP, (B) β-TCP + MSCs, (C) β-TCP + EPCs, (D) β-TCP + EPCs + MSCs, and (E) autologous rat bone. Due to the process of decalcification, the β-TCP was completely solved and left a gap corresponding to the area of the β-TCP-granule. Scale bar: 50 μm.
Histomorphometric analysis was performed, resulting in a comparison of area fractions in a standardized picture frame (Fig. 4). Values denote median ± quartiles, Kruskal–Wallis test, p < 0.05.
Vascularization area fractions (%). After 1 week, histomorphometric analysis using CD34 histomorphochemistry was performed, resulting in a comparison of area fractions in a standardized picture frame. Values denote median ± quartiles. Kruskal–Wallis test, p < 0.05. Compared to the β-TCP and β-TCP + MSC group, a significant higher amount of vascularization was observed in the β-TCP + EPC and in the β-TCP + MSC + EPC group (+#). Also a significantly higher amount of early vascularization was observed in the coculture group and EPC group compared to the autologous group (*). Moreover, we found significant higher amount of vascularization in the coculture group compared to the β-TCP + EPC group (§). No difference between the β-TCP and the β-TCP + MSC group was detectable.
Compared to the β-TCP group [2.2, 1.3, 3.5 (% of total pixel)] and β-TCP + MSCs group [3.01, 1.61, 6.31 (% of total pixel)], respectively (Fig. 4), a significant higher amount of vascularization (+#) was observed in samples derived from animals that received β-TCP loaded with either EPCs [8.12, 3.7, 11.62 (% of total pixel), p < 0.05] or coculture of EPCs and MSCs [11.76, 8.2, 15.6 (% of total pixel), p < 0.05].
Also a significant higher amount of early vascularization (*) was observed in the coculture group and EPC group compared to the autologous group [2.57, 1.5, 4.65 (% of total pixel)]. Moreover, we found significant more vessel formation in the coculture group compared to the β-TCP + EPC group (§). No difference between the β-TCP and the β-TCP + MSC group was detectable.
Contribution of Transplanted Human Cells
Using MHC-1 staining, we detected human cells (MSCs, EPCs) after transplantation in the bone defect. Neither in the TCP nor in the autologous group, MHC-1 staining was positive (Fig. 5A–E). Interestingly, MHC-1 staining was easily detectable if MSCs alone or in coculture with EPCs were transplanted. MHC-1 staining was also obvious when EPCs were transplanted, but to a lower degree. One week after cell transplantation, we observed human cells in the area around TCP gaps (decalcified histological section), but also within the center of the bone defect (Fig. 5B–D).
Detection of human cells after xenotransplantation: MHC-1 staining. Detection of transplanted human cells in rat bone by means of MHC-1 immune staining. Positive staining was observed in all samples containing human cells, (B) β-TCP + MSC, (C) β-TCP + EPC, and (D) β-TCP + EPC + MSC, whereas no staining was found in samples without human cells, (A) β-TCP alone and (E) autologous bone. Scale bar: 100 μm.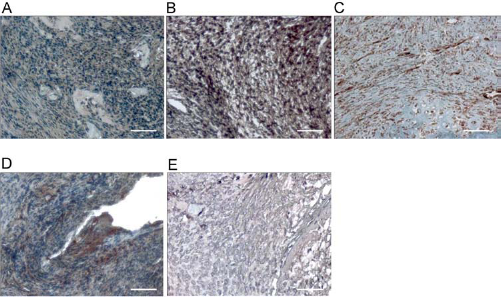
Early Vascularization Is Effected Directly and Indirectly by Human EPCs
Based on the results that transplanted EPCs increased following early vascularization, we performed experiments to identify transplanted human EPC incorporation around the vessel formation. Immunohistochemical staining for vWF, an endothelial marker, and MHC-1, a human cell marker, was performed using tissue samples obtained 1 week after creation of a critical size defect (Fig. 6A–E). No human cells were identified after transplantation of TCP granules (Fig. 6A). After transplantation of human MSCs bone formation took place, but early vascularization was rare (Fig. 6B). vWF marked endothelial cells were more abundantly detected as the rarely double-positive cells for vWF and MHC-1 in bone regeneration area (Fig. 6C) after EPC transplantation. Interestingly, more human MSCs and double-positive EPCs (Fig. 6D) were identified when EPCs were coimplanted with MSCs, which led to an improved vascularization in bone regeneration at week 1. Moreover, transplanted human EPCs directly formed new vessels in the critical size defect (Fig. 6D, white arrows). DAPI staining showed a high amount of cells after transplantation of cocultured EPCs/MSCs. Transplantation of autologous bone (Fig. 6E) did not show improved early vascularization.
Fluorescence microscopy. Immunohistochemical staining for human MHC-1 (red) and vWF (green) using tissue samples of critical bone defect 1 week after surgery when (A) β-TCP, (B) β-TCP + MSC, (C) β-TCP + EPC, (D) β-TCP + MSC + EPC, or (E) autologous bone were implanted. Double-positive cells were rarely identified (D, white arrows), only when EPCs and MSCs were coimplanted. Blue fluorescence indicates DAPI for nuclear staining. Scale bars: 25 μm.
Releasing of VEGF
One week after implantation of β-TCP granules into critical size defects, the degree of VEGF releasing was evaluated (Fig. 7A–E). We observed in animals that received β-TCP loaded with EPCs (Fig. 7C) or both cell types (EPCs/MSCs, Fig. 7D) have a higher amount of VEGF in comparison to animals that received empty β-TCP or β-TCP-loaded with MSCs. A low degree of VEGF release was detected in animals that received autologous bone (Fig. 7E).
Release of vascular endothelial growth factor (VEGF; red staining) to the bone regeneration site. One week after implantation of (A) β-TCP, (B) β-TCP + MSC, (C) β-TCP + EPC, (D) β-TCP + MSC + EPC, or (E) autologous bone into a critical size defects of the femur of the nude athymic rat. Scale bars: 50 μm.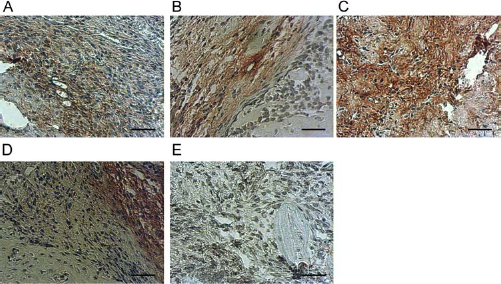
Discussion
In this study, we established a novel role for cell transplantation of early EPCs into a critical size bone defect in vivo. We could demonstrate that EPCs improve directly and indirectly early vascularization of MSC-driven bone regeneration. Seeding efficiency, cell distribution, survival, and differentiation of EPCs, also in coculture with MSCs, seeded onto fibronectin-coated β-TCP were evaluated in vitro as previously described (14). In that former study, a composite scaffold approach as an alternative cell seeding and cell delivery device was developed, and EPCs survived over a period of 10 days on β-TCP, kept their endothelial characteristics also in coculture with MSCs, and still expressed angiogenic factors such as VEGF in a significant amount, but no formation of tube-like structures was observable in vitro (14). In several studies, the application of autologous EPCs resulted in a significant improved vascularization of ischemic regions (5,9,31). It is still however a point of discussion whether the effects are directly due to the transplanted endothelial- like cells generating new vessels or due to secondary effects releasing angiogenic factors such as VEGF and recruiting endothelial cells of the host (33).
In the present study, we demonstrated that human early EPCs and MSCs survived in vivo in a critical size defect of an athymic nude rat after xenotransplantation for at least 1 week. Using quantitative CD34 immunohistochemistry, we showed that the formation of primitive vascular plexus (microvessel) in critical size bone defect is significantly increased after xenotransplantation of EPCs or EPCs/MSCs.
It is known that cytokines such as VEGF are secreted from EPCs and act as prototypical angiogenic growth factors. This well-described chemotactic effect of VEGF on EPCs supports the notion that transplanted EPCs mobilize bone marrow-derived EPCs of the host (6,17,18,24), which in turn contribute to postnatal neovascularization via vasculogenesis (11) and revascularization of injured tissues (7,10). Mobilized EPCs were documented in ischemic heart tissue (1), hindlimb ischemia (20), fracture healing (24), and distraction osteogenesis (21).
Our experiments demonstrate that early vascularization in the critical size bone defect is significantly improved after xenotransplantation of EPCs (especially in coculture with MSCs). These results are mostly due to the indirect effect by releasing angiogenic growth factors. But we also demonstrated that transplanted EPCs (when cocultured with MSCs) were able to form vessels in vivo. This direct effect of EPCs is rarely compared to the indirect manner of releasing cytokines and recruiting endogenous EPCs.
This composite scaffold approach for cell delivery supported early vascularization in vivo, which is needed to support bone-forming cells. Arkudas et al. (3) showed that axial prevascularization of porous matrices using an arteriovenous loop promotes survival and differentiation of transplanted autologous osteoblasts.
Moreover, Kaigler et al. reported that transplanted endothelial cells enhance bone regeneration in a calvarial defect (16). Also heterotopic ossification (33) and orthotopic bone regeneration after 8 weeks (28) was improved by implantation of EPCs and MSCs. Peng et al. described a synergistic enhancement of bone formation and healing by transfected MSCs constitutively expressing VEGF and bone morphogenetic protein-4 (27). In a previous study, we demonstrated that MSCs alone lead to a significant improvement of bone mass formation in comparison to TCP alone. However, xenogenic transplantation of both EPCs and MSCs has a synergistic effect and leads to a superior bone formation in comparison to MSCs alone (28). Thus, b-TCP populated with transplanted human cells appears to be a potential osteogenic construct for in vivo application in a critical size bone defect.
Interestingly, improved new bone formation supported by the neovascularization of EPCs is probably not the only possible mechanism. Our own results from a previous study (28) showed that the highest amount of bone at 1 week was formed by the autologous bone-positive control despite the fact that the associated neovascularization was as low as that recorded in the TCP control group. Additionally, the MSC group also had a low value for the neovascularization and yet had a relatively high degree of new bone formation (28). These observations might suggest that the EPC contribution to osteogenesis is not only due to its effect on neovascularization but in the transdifferentiation of EPCs (22) to osteogenic cells. Here, it has to be mentioned that at least two types of EPCs can be cultured from peripheral blood mononuclear cells: “Early” EPCs, which were derived from myeloid or dendritic cell precursors, still express leukocyte markers such as CD45 and differentiate within 5 days; secondly, “late” EPCs, which were derived from bone marrow stem cells, express stem cell markers such as CD133 but are negative for CD45.
Our data (29) suggest that “early” EPCs could be successfully cultivated on β-TCP-scaffold; they did not lose their endothelial characteristics and remained, at least in part, in a functional state since VEGF gene expression was still detectable in a number of samples even if these cells were cultured under osteogenic conditions in vitro (14). “Late” EPCs showed only a weak adherence to coated β-TCP and adopt osteogenic marker (transdifferentiation) if they were cultivated under osteogenic conditions in vitro (29).
Thus, even “early” EPCs, which we used in the present study showed the lineage typical gene expression under osteogenic conditions in vitro (14), it is at least in part possible that osteogenic differentiation of vascular cells take place in vivo.
In conclusion, human early EPCs promote directly and indirectly early vascularization of MSC-stimulated new bone formation of a composite after xenotransplantation into a critical-sized femoral defect of an athymic nude rat.
Footnotes
Acknowledgment
Project no. 05-S23 was supported by the AO Research Found of the AO Foundation, Switzerland. The authors declare no conflict of interest.
