Abstract
The supply of human hepatic stem cells (hHpSCs) and other hepatic progenitors has been constrained by the limited availability of liver tissues from surgical resections, the rejected organs from organ donation programs, and the need to use cells immediately. To facilitate accessibility to these precious tissue resources, we have established an effective method for serum-free cryopreservation of the cells, allowing them to be stockpiled and stored for use as an off-the-shelf product for experimental or clinical programs. The method involves use of buffers, some serum-free, designed for cryopreservation and further supplemented with hyaluronans (HA) that preserve adhesion mechanisms facilitating postthaw culturing of the cells and preservation of functions. Multiple cryopreservation buffers were found to yield high viabilities (80–90%) of cells on thawing of the progenitor cells. Serum-free CS10 supplemented with 0.05% hyaluronan proved the most effective, both in terms of viabilities of cells on thawing and in yielding cell attachment and formation of expanding colonies of cells that stably maintain the stem/progenitor cell phenotype. Buffers to which 0.05 or 0.1% HAs were added showed cells postthaw to be phenotypically stable as stem/progenitors, as well as having a high efficiency of attachment and expansion in culture. Success correlated with improved expression of adhesion molecules, particularly CD44, the hyaluronan receptor, E-cadherin, β4 integrin in hHpSCs, and β1 integrins in hepatoblasts. The improved methods in cryopreservation offer more efficient strategies for stem cell banking in both research and potential therapy applications.
Introduction
On a cellular level, hypoxemia is initiated the moment a tissue is removed from a living organism; it leads quickly to tissue and organ damage. Freezing cells is a method to arrest cells in a storage form to enable stockpiling and use in off-the-shelf strategies for numerous experimental and therapeutic programs (43). This strategy has created interest in mechanisms by which cells are able to survive freezing. The need for successful cryopreservation methods is especially acute in sourcing of human cells. Human tissues are difficult to obtain, and their availability is irregular, making their utilization in experimental or clinical programs problematic.
Freezing cells without special conditions is lethal. Standard buffers and procedures have been developed to enable cells to survive cryopreservation. Strategies have been identified beginning as early as 1949 with preserving spermatozoa to survive prolonged freezing in buffers with glycerol (29). Several factors have been investigated for cryopreservation techniques, including the use of cryopreservation agents (21,27), encapsulation techniques (5,9,13), pre- and posttreatment of cells (40), and regulation of freezing temperature (11,31). Current challenges for cryopreservation of adherent cell types, like hepatic cells, are cell loss, viability, and high variability in cell adherence after thawing (1,2,40). Entrapment of hepatocytes within alginate beads has protective effects (24) and enables the hepatocytes to be transplanted into rats after freezing while still maintaining their viability (3). However, release of the cells from the alginate beads can be a variable in success during postthawing.
Human hepatic stem cells (hHpSCs) are multipotent stem cells found in livers from donors of all ages (50) and give rise to hepatoblasts (hHBs) that are bipotent maturing into hepatocytes and cholangiocytes (44). The hHpSCs have been shown to have the ability to alleviate liver dysfunctions clinically and to be able to do so without the need for immunosuppression of the patients (18). When isolated from livers by immunoselection or by culture selection methods, they form colonies capable of self-replication in culture (25) and of differentiation to mature cells in culture and in vivo (34,48). One of the hHpSCs' prominent surface markers is epithelial cell adhesion molecule, EpCAM. Similar to that found for cryopreserved hepatocytes (8,34), hepatic progenitors have significant reduction in attachment to culture dishes after thawing due to loss of cell adhesion molecules, specifically β1-integrin (ITGB1) and E-cadherin (CDH-1), shown in studies to be significantly reduced after cryopreservation (41).
Hyaluronan (HA), a form of glycosaminoglycan present in all stem cell niches, is highly porous, nonrigid unless heavily cross-linked, and without chemical sites for binding signaling molecules, making it neutral as a scaffold for stem cells and progenitors and with respect to growth versus differentiation effects on cells (35,42,48,51). Early lineage stage cells, whether epithelial or mesenchymal, have receptors for hyaluronans (14,45,46). We hypothesized that HA could provide a mechanism for protecting the cell adhesion mechanisms of the hepatic progenitors during cryopreservation and enable one to cryopreserve stem cells with retention of their stem cell phenotype. This would be invaluable, since the cryopreserved cells would retain their extraordinary expansion potential as stem cells. In this study, we evaluated cryopreservation conditions, including different isotonic media with and without the presence of HAs, and with and without supplements that improve survival of endodermal stem cells and progenitors to determine the best freezing method for cell banking.
Materials and Methods
Liver Sourcing and Processing
Liver tissue was provided by an accredited agency (Advanced Biological Resources, Alameda, CA) from fetuses between 14 and 20 weeks of gestational age that were obtained by elective terminations of pregnancy. The research protocol was reviewed and approved by the Institutional Review Board for Human Research Studies at the University of North Carolina. All processing and cell enrichment procedures were conducted in a cell wash buffer composed of a basal medium (RPMI 1640, GIBCO, Carlsbad, CA) supplemented with 0.1% bovine serum albumin (BSA Fraction V; Sigma-Aldrich, St. Louis, MO), insulin, and iron-saturated transferrin (both at 5 μg/ml; Sigma-Aldrich), trace elements (300 pM selenious acid and 50 pM ZnSO4), and antibiotics (AAS; Invitrogen, Carlsbad, CA). Liver tissue was subdivided into 3-ml fragments (total volume ranged from 2 to 12 ml) for digestion in 25 ml of cell wash buffer containing type IV collagenase (300 U/ml) and deoxyribonuclease (3 mg/ml; Sigma-Aldrich) at 32°C with frequent agitation for 15–20 min. This resulted in a homogeneous suspension of cell aggregates that were passed through a 70-gauge mesh and spun at 170G for 5 min before resuspension in cell wash solution. Erythrocytes were eliminated by slow-speed centrifugation (20G, 5 min, 4°C). Estimated cell viability by trypan blue exclusion was routinely >95%.
Hepatic Stem Cell Culture Conditions
The fetal human liver cells were suspended into a serum-free medium, Kubota's medium (KM), tailored for progenitors from endodermal tissues (19). Fresh and thawed fetal liver cell mix were plated at 4,000 to 8,000 cells/cm2 on tissue culture-treated plastic (Becton-Dickinson, Franklin Lakes, NJ). The low cell density used and the conditions in KM are not conducive to survival of mature liver cells but only of stem cells and progenitors. Use of serum-free conditions was essential to keep the hHpSCs and angioblasts stable in their initial lineage stage that provides the required paracrine signaling, enabling them to self-replicate. Cells were plated with KM with 10% fetal bovine serum (FBS) for at least 6 h and, at most, 24 h to facilitate attachment and placed under serum-free conditions thereafter. Serum-free KM was changed every 3–4 days. It is known that cryopreservation with dimethyl sulfoxide (DMSO) causes a decrease in adhesion molecules and attachment, either mechanically or through downregulation of gene expression (12,20). Plates for seeding of thawed cells were coated with collagen III at 1 μg/cm2 (Sigma-Aldrich, type X) to facilitate stem cell attachment by more closely resembling the normal microenvironment of the stem cells (25). Typical plates had single cells and small clusters of cells that adhered within the initial 24 h. Colonies began to appear after 1–2 weeks.
Freezing Conditions
Freshly isolated human hepatic progenitors were allowed to recover at 4°C for 2 h prior to freezing in KM. Samples were frozen at 2 × 106 cells/ml in cryopreservation vials (Nunc, Rochester, NY). Cryopreservation solution conditions consisted of either KM culture medium supplemented with 10% DMSO or CryoStor™-CS10 (Biolife Solutions, Bothell, WA) and with 0%, 0.05%, or 0.10% hyaluronans (Biotime, Alameda, CA) by weight (KM, KM + 0.05% HA, KM + 0.1% HA, CS10, CS10 + 0.05% HA, CS10 + 0.1% HA). A final condition consisted of KM supplements added to CS10 (CS10 + SUP). Cells were allowed to equilibrate in the cryopreservation buffer for 10 min at 4°C.
We have shown previously that higher concentrations of HA, especially if cross-linked, will result in rigidity that can induce differentiation of the stem cells by means of mechanical forces (22). Thus, we focused on samples frozen in a low percentage of hyaluronan (HA) that was not cross-linked. In general, the hyaluronans increase the sample viscosity, but HA without crosslinking does not form a rigid gel. Samples were prepared by mixing three parts: cells in freezing medium, gel in freezing medium, and freezing medium to adjust volume and component ratios. A summary of step-by-step freezing and thawing methods, with temperatures, can be seen in Figure 1. Samples were frozen using a Cryomed 1010 control rate freezer (Forma/Thermo Scientific, Marietta, OH).

Schematic of cryopreservation and thawing methods.
Viability
Cell viability was determined by trypan blue exclusion assays. Samples were thawed quickly and then slowly diluted with culture media on ice at approximately 1 ml/min, achieving a dilution of DMSO less than 0.5%. After counting of the cells, the cells were plated in culture media as previously described.
Colony Counting
Hepatic stem cell and progenitor colonies began to appear between 1 and 2 weeks after plating, and tight cell clusters were observed by looking at the plate with the naked eye but were easily identified by inspection at 10x with a light microscope. Any size colony was counted as one, whether large ones at >3,000 cells or small ones at <200 cells. Each well of six wells was evaluated using 10x magnification for colonies and counted after 2–3 weeks of culture. Observations of colony number, size, and morphology were noted.
Quantitative Real-Time PCR (qRT-PCR)
Gene-specific primer sequences for quantitative gene expression analysis of differentiation markers (32–34,49) were designed and validated for PCR doubling efficiency and priming specificity using cDNA templates assembled from total RNA of relevant cell lines positively expressing each gene of interest extracted with the RNeasy Mini Plus kit (QIAGEN, Valencia, CA). Quantitative real-time PCR (qRT-PCR) measurements were performed with an Applied Biosystems® 7500 Real-Time PCR System (Carlsbad, CA) available in the Functional Genomics Core Facility at UNC at Chapel Hill, NC. Primers used can be found in Table 1. All measurements of relative expression were normalized with respect to glyceraldehyde 3-phosphate dehydrogenase (GAPDH) by the absolute quantification method described elsewhere (32–34,49).
Primer Sequences Used for Quantitative RT-PCR

Immunohistochemistry
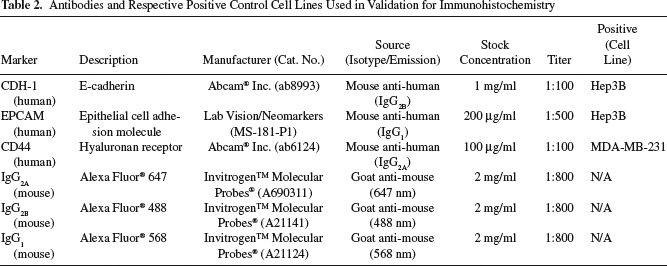
By 24 h after plating, cells were fixed for 2 h with 4% buffered paraformaldehyde (PFA). Plates were then washed with PBS; colonies were identified and bordered using a PAP-PEN and blocked by using PBS + 0.1% Triton + 10% goat serum. Primary antibodies (Abcam, Labvision/Neomarkers, Cambridge, MA) were applied and allowed to incubate overnight at 4°C, followed by another wash, and then the application of secondary (Alexa Fluor, Invitrogen Life Technologies, Carlsbad, CA) antibodies at room temperature for 2–4 h. Once tagged, a mounting medium containing 4′,6-diamidino-2-phenylindole (DAPI) was applied, and slides were imaged using confocal fluorescent microscopy. The hepatoblastoma cell line, Hep3B (ATCC HB-8084, Manassas, VA), was used as a positive control cell line for CDH-1 and EpCAM, and the metastatic breast adenocarcinoma cell line, MDA-MB-231 (ATCC HTB-26, Manassas, VA), was used as a positive control for CD44. Samples stained with only secondary antibodies were used as negative controls. A summary of antibodies and respective positive control cell lines used for immunohistochemistry (IHC) can be seen in Table 2. Cells, stained and tagged with secondary antibodies, coupled to fluoroprobes, were identified using fluorescent microscopy. Negative controls were used to normalize backgrounds and eliminate image acquisition noise. Microscopy was performed at the Microscopy Core Laboratory, a CORE laboratory of UNC at Chapel Hill. An Olympus IX 81-ZDC Inverted Fluorescence Microscope (Center Valley, Philadelphia, PA) was used, with phase contrast, and fluorescent filters at 350, 488, 568, and 647 nm. It was equipped with a fully motorized stage, computer controlled by Improvision's Velocity software (Perkin Elmer, Waltham, MA), and a Hammatsu ORCA RC camera (Bridgewater, NJ) and QImaging RETIGA 4000R color camera (Surrey, British Columbia, Canada).
Antibodies and Respective Positive Control Cell Lines Used in Validation for Immunohistochemistry
Statistical Analysis
Results were presented as mean ± standard error of the mean. Statistical analysis of data was performed by a one-way ANOVA. Significant findings were followed with pair-wise t tests corrected for multiple comparisons using the step-down Bonferroni method.
Results
Establishment of Steps and Conditions to Minimize Damages From Freezing
After cell isolation and freezing preparation shown in Figure 1, a controlled freezing rate program for freezing cryovials was established to maximize cell survival. The freezing program was modified after Hubel et al. (16), tailored for our cell population, and can be seen with the chamber and sample temperatures with respect to time in Figure 2. A sharp drop in temperature when the chamber reaches −8°C minimizes liquid–ice phase entropy seen within samples during freezing. This freezing program also allowed for reproducible results with all samples.

Controlled rate freezing program minimizes liquid-ice phase entropy preventing internal ice damage and allows for repeatable freezing. (A) Graph shows chamber temperature in relation to sample temperature [10% dimethyl sulfoxide (DMSO)]. (B) Freezing program rates used for the Cryomed 1010 system. The sharp drop in temperature occurring when the chamber reaches –8°C minimizes the liquid–ice phase entropy typically seen within samples during freezing. The consistent freezing program also allows for repeatable freezing between samples.
Effect of Freezing Conditions and Hyaluronans on Cell Viability and Colony Counts
Immediately after thawing, the cell viability of thawed cells was assessed. As shown in Figure 3, all of the cryopreservation buffers tested resulted in remarkably high viabilities of the cells on postthawing, with viabilities ranging from 79% to 87%. There were no significant differences among the buffers and media assayed. The best viabilities were observed in samples of cells frozen in CS10 + SUP or CS10 with 0.05% or 0.1% hyaluronan. Greatest colony formation counts after thaw were seen in samples frozen in cryo-specific media with hyaluronans (CS10 + 0.05% HA).

(A) Percent cell viability of cryopreserved hepatic stem/progenitor cells. (B) Colony counts after 3 weeks of culture for each condition and normalized to freshly isolated cell samples. Results are reported as mean ± standard error of the mean. Viability (n > 6), colony counts (n > 3). KM, Kubota's medium with 10% DMSO and 10% fetal bovine serum (FBS). CS10, CryoStor; CS10 + SUP, CryoStor10 with KM supplements. The percentages, 0.05% and 0.10%, refer to the percentage of hyaluronan (HA) supplemented in each sample. Higher viabilities were observed in conditions frozen in CS10.
Effect of Culture and Freezing Media with and Without Hyaluronans on Expression of Cell Adhesion Molecules
The expression of key cell–cell and cell–matrix adhesion factors was examined. A summary of the genetic expression profiles of cell adhesion molecules in cryopreserved samples can be seen in Figure 4. The highest expression of integrin β1 postthaw was seen in samples frozen in CS10 + 0.05% HA (0.130 ± 0.028, n = 28). This is significantly different when compared to expression in freshly isolated cells (0.069 ± 0.007, n = 24, p < 0.01). Even more interestingly, no significant difference was found between integrin β4, expressed by the hHpSCs but not the hHBs, in freshly isolated versus cryopreserved samples. CD44, expressed by both the hHpSCs and the hHBs, was seen at higher expression levels in HA-containing samples when compared to fresh samples, and those frozen in traditional culture media (KM). CDH-1 expression in cells frozen in CS10 + 0.1% HA (0.049 ± 0.006, n = 20) and CS10 + 0.05% HA (0.064 ± 0.003, n = 16) showed significant increases in expression when compared to freshly isolated cells (0.037 ± 0.005, n = 36, p < 0.05).

Relative mRNA expression normalized to GAPDH expression. Mean ± standard error of the mean. Significance ∗p>0.05 when compared to fresh samples. KM, Kubota's medium with 10% DMSO and 10% FBS; CS10, CryoStor; CS10 + SUP, CryoStor10 with KM supplements. The percentages, 0.05% and 0.10%, refer to the percentage of HA supplemented in each sample. The highest expression of integrin β1 (ITGB1) cadherin-1 (CDH-1) was seen in samples frozen in CS10 + 0.05% HA. There was a trend of higher expression of CD44 in samples frozen with HA. CDH-1 expression in cells frozen in CS10 + 0.05% HA and + 0.10% HA showed significant increases in expression when compared to freshly isolated cells.
Immunohistochemistry was performed on cell samples 24 h after thawing in CS10 sample conditions to show the presence and location of critical cell adhesion molecules in cells. EpCAM was used to specifically target hepatic stem/progenitors in the heterogeneous populations that also included nonparenchymal cells. Positive costaining for CDH-1 in EpCAM+ cells, comprising stem cells (hHpSCs) and hepatoblasts (hHBs), is seen in Figure 5. When cells were frozen with HA, there was an increase in CD44-stained cells, and expression was more abundant near parenchymal cell colonies. These adhesion molecules were significantly reduced or lost altogether in the cultures of samples cryopreserved in CS10 or CS10-Sup, not containing hyaluronans. The only condition in which the cells remained viable, attached readily, formed expanding colonies that remained positive for CD44 and other phenotypic traits indicative of hHpSCs was CS10 + 0.05% HA.

Cryopreserved hepatic progenitor cells after 24 h of attachment on collagen III. CS10, CryoStor; CS10 + sup, CryoStor10 with KM supplements. The percentages, 0.05% and 0.10%, refer to the percentage of HA supplemented in each sample. Epithelial cell adhesion molecule (EpCAM) was used to specifically target hepatic progenitors in the heterogeneous population. Staining for CDH-1 showed the presence of key adhesion molecules in the cells. When cells were frozen with HA, there was an increase in CD44+ cells, and expression was more abundant near parenchymal cell colonies when compared to those samples without HA.
Discussion
Cryopreservation of cells requires strategies that protect cell adhesion mechanisms in addition to the use of isotonic buffers, antifreeze proteins, antioxidants, and freezing reagents such as DMSO or glycerol. The extant methods work well for hemopoietic cell subpopulations that have evolved to retain their extracellular matrix components with maturation but switch to matrix components missing cell-binding domains, enabling the cells to float. Thus, their adhesion mechanisms are intact. By contrast, all cells from solid organs are bound together by complex mixtures of extracellular matrix molecules that must be enzymatically altered or destroyed to prepare cell suspensions, making the cells more sensitive to freezing and to other microenvironmental changes. Cryopreservation of liver cells is representative of the greater difficulties in cryopreservation of cells from solid organs, easily damaged by the freezing and thawing processes (17). In addition, cryopreservation conditions for stem and progenitor cells have additional hurdles over those for mature cells, because many factors used in cryopreservation buffers, such as serum, can trigger the cells to lineage restrict to more mature cells (17). We show that these hurdles are overcome by utilizing serum-free cryopreservation buffers that are isotonic; have antioxidant, antifreezing proteins, and DMSO; and are supplemented with HAs, an extracellular matrix component that is dominant in stem cell niches.
Common methods of cryopreservation include the use of specific freezing media and cryopreservation agents to reduce internal ice formation. These agents include glycerol, dimethyl sulfoxide, ethanediol, and propanediol that permeate the cell and sugars and polymers that are non-permeating (21,27,28,40). Glycerol is found in the cells and tissues of many organisms living in freezing conditions including the Arctic, Antarctic, and high mountains (30), but its viscosity makes it difficult to utilize in routine procedures. Bovine or calf serum and DMSO are commonly used as cryoprotectants, but both can initiate differentiation of stem cells and early progenitor cells (7). In addition, serum cannot be used for samples intended for clinical applications due to concerns of possible xenozoonosis contamination.
There are many publications on the cryopreservation of mature hepatocytes and, more recently, ones focused on cryopreservation of diploid cells with their ability to proliferate and function after freezing. Rat small hepatocytes, diploid cells that are ~17–18 μm in diameter, have been maintained at −80°C for over 6 months and with 60% attachment and proliferation of colonies observed after thawing (17). Liver functions that were preserved included albumin production and synthesis of other serum proteins.
The cryopreservation of human hepatocytes shows greatest success with a progressive freezing, with its slow freezing rate, following a 30-min preincubation at 37°C and could potentially be used in cell banking (2). Work has been done also involving the cryopreservation of hepatic stellate cells (26), which provide critical paracrine signaling for the hHBs. Here, both cooling rates and cryoprotectants were investigated and found to have a clear effect on the resulting viability and cell survival. It was found that DMSO as a cryoprotectant gave superior attachment and viability for mature hepatocytes. More extensive studies of freezing mature rat hepatocytes in a controlled rate freezer with programmed steps found better control of viability using these controlled freezing steps (15). The freezing program designed for this study took into account these freezing steps to minimize the latent heat seen in freezing, resulting in a smooth sample freezing rate and better viability.
In these studies, we focused on the needs of stem cell and progenitor populations for successful cryopreservation. Kubota's medium (KM), found effective for stem cells and progenitors from liver (34,48), was supplemented with 10% FBS and 10% DMSO, a necessity, since it is not isotonic. It was compared to serum-free CryoStor CS10, an isotonic buffer designed for tissue or cell storage at low or freezing temperatures (38,39). Both serum supplemented KM and serum-free CS10 were found to work equally well in protecting the cells during freezing yielding cells with equivalent viabilities on thawing. However, CS10 proved preferable given its success when serum-free, enabling one to avoid serum that could trigger differentiation of the stem cells, a concern for cells to be used in clinical programs.
Previous studies showed cells cryopreserved in CryoStor remained highly viable on thawing (4). Similarly, hematopoietic stem cells from cord blood had more colony formation units after freezing in CryoStor (37). Human hepatocytes frozen in CryoStor and then transplanted in nonobese diabetic severe combined immunodeficient (NOD-SCID) mice were able to retain function and reconstitute liver tissue (6).
The choice of conditions for cryopreservation of the stem cells proved obvious from the studies on the ability of the thawed cells to attach and grow in culture and most especially on the ability of the stem cells to remain as stem cells after freezing. Thus, though there was colony formation by thawed cells from all of the freezing conditions, only those frozen in Kubota's medium with serum supplementation or in serum-free CS 10 with 0.05% HA gave rise to colonies that attached, expanded, and yet retained a stem cell phenotype These findings correlated with the preservation of key adhesion molecules, particularly CD44 and E-cadherin. Considerable importance has been found for the several adhesion molecules known to be expressed by the early progenitors and needed for attachment and colony formation after thawing (41). These adhesion molecules were shown to be important in the cryopreservation, while others showed that apoptosis can occur in cells detached from their matrix (52). All subpopulations of stem cells and progenitors in the liver express CD44, receptors for hyaluronans (45), as do stem cells and progenitors from most tissues (23,36,47). Therefore, it was logical that hyaluronans would facilitate their cryopreservation and that coexpression of CD44 was seen only in attached colonies from samples frozen with a hyaluronan present in the freezing medium. Similarly, we found that there was indeed a significant increase in the cell–cell adhesion molecule, CDH-1, found on all of the stem/progenitor populations, when cultured with HA.
There are distinctions in the integrin expression in the hHpSCs versus hHBs in vivo. The integrins associated with the hHpSCs are α6β4, assumed to be used to attach to either laminin 111 or laminin 511 (10), whereas those associated with hHBs are multiple a integrins (α1, α5, α6, α9) and always β1 but not β4 (8,34). The integrin β4 expression and, therefore, hHpSCs were especially preserved in KM with HA and in the CryoStor10 with HA. By contrast, there was a significant difference in the expression of integrin β1 among the conditions implicating a greater sensitivity by the hHBs for cryopreservation; the best conditions for preserving this population was CS10 + 0.05% HA.
Conclusions
In summary, the cryopreservation of hHpSCs and retention of their stem cell phenotype can be achieved with serum-free CryoStor buffers in combination with low hyaluronan concentrations (0.05% or 0.1%). The HA supplementation improved cell attachment and colony formation due at least in part to improved expression of key cell adhesion factors in the stem cell and progenitor cell populations. This study provides successful methods in cryopreservation of hepatic stem cells, progenitors, and potentially other types of stem cells. It can be implemented for more efficient banking methods in both research and potential therapy applications.
Footnotes
Acknowledgments
Funding derived from sponsored research grants from Vesta Therapeutics (Bethesda, MD), NC TraCS (UNC CTSA) Grant (#2KR50905), and Gigacyte (Brandon, CT), a grant from the North Carolina Biotechnology Center (NCBC), and an NIH Center grant (CA016086). We acknowledge our research collaborator and friend, Dr. Claire Barbier, and recognize posthumously her invaluable contributions to the performance of this work and preparation of this manuscript. Technical core services were provided by the Nucleic Acids, Histology, and Functional Genomics core facilities. The authors declare no conflicts of interest.
