Abstract
Mesenchymal stem cells have been isolated from various adult human tissues and are valuable for not only therapeutic applications but for the study of tissue homeostasis and disease progression. Subcutaneous adipose depots have been shown to contain large amounts of stem cells. There is little information that has been reported to date describing the isolation and characterization of mesenchymal stem cells from visceral adipose tissue. In this study, we describe a mesenchymal stem cell population isolated from mediastinal adipose depots. The cells express CD44, CD105, CD166, and CD90 and are negative for hematopoietic markers CD34, CD45, and HLA-DR. In addition, the cells have a multilineage potential, with the ability to differentiate into adipogenic, osteogenic, and chondrogenic cell types. The biological function of visceral adipose tissue remains largely unknown and uncharacterized. However, the proximity of adipose tissue to the heart suggests a potential role in the pathogenesis of cardiovascular disease in obesity. In addition, with the ability of fat to regulate metabolic activity in humans, this novel stem cell source may be useful to further study the mechanisms involved in metabolic disorders.
Introduction
Mesenchymal stem cells (MSCs) are stem cells that have extensive proliferative capacity and multilineage potential. They arise from progenitor cell reservoirs and have been isolated from various tissues including bone marrow (14), muscle (17), and adipose tissue (3,19,20). MSCs isolated from various tissues share common characteristics, including the ability to (1) form fibroblastic like colonies; (2) proliferate extensively; (3) be directionally differentiated into adipocytes, osteocytes, and chondrocytes; and (4) express several common cell surface markers.
Adipose depots contain an abundant source of stem cells isolated using enzymatic digestion, filtration, and centrifugation of the stromal vascular fraction (SVF). The stromal vascular fraction can be further expanded in cell culture and differentiated into multiple cell types. Most studies of adipose-derived stem cells have derived the cells from subcutaneous adipose depots. Baglioni et al. described a population of adipose-derived stem cells derived from visceral omental adipose depots and found that there was no significant differences between visceral or subcutaneous adipose-derived stem cell populations (1).
In this study, we describe the isolation and characterization of a multipotent stem cell population from mediastinal adipose depots. Our data provide the first evidence of a stem cell population in mediastinal adipose depots. These stem cells can be readily expanded, have clonogenic capacity, and can be differentiated into multiple cell types.
Materials and Methods
Mediastinal Adipose Tissue Procurement
Mediastinal adipose tissues were obtained from five patients undergoing coronary artery bypass grafting surgeries [three males and two females; mean ± SE age, 67.4 ± 4 years (range 62–72 years)] at the University of Utah under IRB-approved protocol numbers 35241 and 35242.
Derivation and Cloning of Mediastinal Adipose-Derived MSCs
Under aseptic conditions, mediastinal adipose tissue was placed into a 50-ml conical tube containing 25 ml of Hank's basic salt solution (HBSS) (Hyclone, Rochester, NY) and washed by shaking vigorously for 5–10 s. This was repeated three times to remove erythrocytes and leukocytes. The tissue was then cut into 3-mm pieces and digested with 0.5% collagenase A type I (Sigma Aldrich, St. Louis, MO) in a shaking water bath at 37°C for 30 min. Collagenase digestion was stopped by addition of XcyteMPL™ supplement (JadiCell, Salt Lake City, UT) to a final concentration of 10%. Digested tissue was then filtered through a 100-μm cell strainer. The cell suspension was then centrifuged at 200 × g for 5 min, followed by an additional wash with 10% of XcyteMPL™ supplement. Cells were plated into a 225-cm2 flask and grown in Dulbecco's modified Eagle's medium (DMEM) low glucose, 10% XcyteMPL™ supplement (JadiCell, Salt Lake City, UT), 1x Glutamax, and 1x minimum essential medium and nonessential amino acids (MEM-NEAA; Life Technologies) and cultured in 5% CO2 incubators at 37°C. After 2 days, the nonadherent cells were discarded, and the attached cells were cultured and expanded. At 70% confluency, the cells were detached using TrypLE (Life Technologies). Cloning of the cells was accomplished by plating the cells at a concentration of one cell per well in 96-well plates (Corning, Acton, MA).
Growth Kinetics
To generate a growth curve and determine population doublings (PD), cells were cultured in 25-cm2 flasks, harvested, counted, and replated when they reached 80% confluency. Cultures were terminated after the cells failed to double after 2 weeks in culture. Population doublings was calculated as previously described (6).
Karyotyping
Karyotyping was performed by Cell Line Genetics (Madison, WI) using standard cytogenetic protocols. Briefly, G-banding was applied to karyograms generated from at least 20 metaphases of cells in log phase.
Immunocytochemistry
Cells were fixed with 4% paraformaldehyde in 1x phosphate-buffered saline (PBS) for 30 min at room temperature. Cells were then rinsed three times with 1x PBS and blocked with 5% normal donkey serum overnight at 4°C. Cells were incubated at 4°C with primary antibody diluted in 5% donkey serum with 1x PBS overnight. Cells were then washed three times with blocking solution. Cells were incubated with secondary antibody in blocking solution for 2 h at room temperature. Cells were counterstained with DAPI (Molecular Probes, Carlsbad, CA) and mounted with antifading mounting solution. Primary antibodies used were homing cell adhesion molecule (H-CAM/CD44) clone F10-44-2 (Millipore, Billerica, MA), CD90 clone F15-42-1 (Millipore), STRO-1 clone STRO-1 (Millipore), CD146 clone P1H12 (Millipore), CD19 clone FMC63 (Millipore), and CD14 clone 2D-15C (Millipore). Secondary antibodies cyanine 3 (Cy3) and fluorescein isothiocyanate (FITC) were used (Millipore). For negative controls, incubation without primary antibody and with corresponding specific nonimmune immunoglobin (Millipore) in blocking buffer was used.
Flow Cytometry
Cells were collected, resuspended in MEM + HEPES (Life Technologies) with 2% bovine serum albumin (BSA; Sigma Aldrich), and counted. The directly conjugated antibodies used were human leukocyte antigen (HLA)-DP DQ DR (BD Biosciences), CD90, LIN, CD166, STRO-1, stage-specific embryonic antigen-4 (SSEA-4), CD44, CD106, CD73, CD117, CD105, HLA-ABC, CD86, CD63, CD9, CD80 (Biolegend), CD45, CD133, and CD34 (Miltenyi Biotech). After staining, the cells were fixed with 4% paraformaldehyde and analyzed using a FACSCanto II analyzer (BD Biosciences).
In Vitro Cell Differentiation
Adipogenic Differentiation
Cells were plated in six-well dishes at a density of 50,000 cells per well. At 90% confluency, StemPro® Adipogenesis Differentiation medium (Life Technologies) was added. The medium was changed every 3 days, and after 21 days, the cells were fixed with 4% paraformaldehyde (PFA). Control cells were maintained in 10% culture medium. Oil Red O (0.3%; Sigma Aldrich) was used for staining to detect intracellular lipid accumulation.
Osteogenic Differentiation
Cells were plated in six-well dishes at a density of 50,000 cells per well. At 90% confluency, StemPro® Osteogenesis Differentiation medium (Life Technologies) was added. The medium was changed every 3 days, and after 21 days, the cells were fixed with 4% PFA. Control cells were maintained in 10% culture medium. Alizarin Red S (2%; Sigma Aldrich) was used for staining to detect de novo formation of bone matrix.
Chondrogenic Differentiation
Cells were harvested, and 500,000 cells/15 ml tube were pelleted and induced with StemPro® Chondrogenesis Differentiation medium (Life Technologies). Control cells were maintained in 10% culture medium. Cell pellets were stained with 1% Alcian Blue (Sigma Aldrich) to detect the secretion of sulfated glycosaminoglycans.
Results
Expansion and Characterization of Mediastinal Adipose-Derived Stem Cells
During surgery for coronary artery bypass grafting, the mediastinum is routinely exposed. The visceral mediastinal adipose encountered was removed and studied to determine if a stem cell population was present. After enzymatic digestion, the primary cell cultures were fed every 3 days. In order to define a clonal population of cells, we derived cell lines by single-cell plating in 96-well plates. Confluency was reached at 6 days, and the cells exhibited MSC-like morphology (Fig. 1A). Karyotyping of the clonal cell population at passage 7 showed normal diploid cells without chromosomal aberrations (Fig. 1B). Growth kinetics of the clonal cell population demonstrated that the population could be propagated for greater than 20 passages (Fig. 1C).

(A) Mediastinal adipose-derived stem cells after single cell cloning exhibit mesenchymal stem cell (MSC) like morphology, magnification at 10x. (B) Mediastinal adipose-derived stem cells retain a normal karyotype at passage 7. (C) Growth kinetics of mediastinal adipose-derived stem cells demonstrate that the cells can be expanded for greater than 20 passages.
In order to characterize the cells derived from the mediastinum, we performed immunocytochemistry (ICC) and flow cytometry. ICC analysis confirmed that the cells were positive for CD44, CD90, and CD146 and negative for STRO-1, CD19, and CD14 (Fig. 2). Flow cytometry was performed on the mediastinal adipose-derived cells at passage 3. This analysis revealed that the mediastinal adipose-derived cells were positive for CD90, CD166, CD44, CD73, CD105, HLA-ABC, CD63, and CD9 and negative for STRO-1, CD45, CD133, CD34, CD106, HLA-DR, CD117, CD86, and CD80. The cells expressed low levels of SSEA-4 and LIN (Fig. 3). We found that cells derived from mediastinal adipose tissue expressed CD9 and CD63, both of which are part of the tetraspanin family, involved in cell motility and correlated with exosome production (7,10,12). CD9 has also been found to play a role in the proliferative and proangiogenic action of adipose-derived stem cells (9).
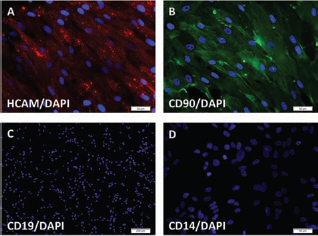
Immunocytochemistry of mediastinal adipose-derived stem cells and counterstained with DAPI. (A) Cells are positive for homing cell adhesion molecule (HCAM). (B) Cells are positive for CD90. (C, D) Cells are negative for CD19 and CD14.
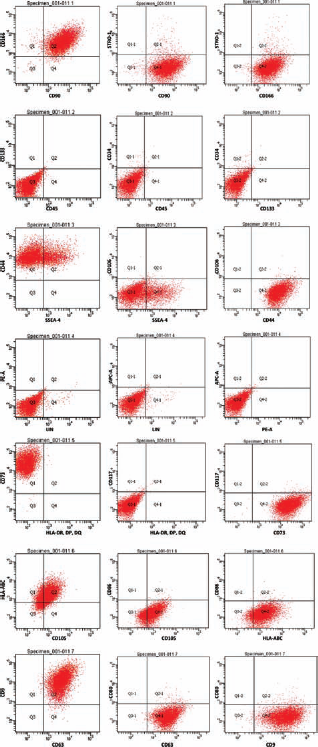
Flow cytometry of mediastinal adipose-derived cells. Cells are positive for CD90, CD166, CD44, CD73, CD105, human leukocyte antigen (HLA)-ABC, CD63, and CD9, but negative for STRO-1, CD45, CD133, CD34, CD106, HLA-DR, CD117, CD86, and CD80.
Differentiation of Mediastinal Adipose-Derived Stem Cells
MSCs have the ability to differentiate into cell types of the mesodermal lineage. We directionally differentiated mediastinal adipose-derived stem cells for 21 days into adipogenic, osteogenic, and chondrogenic lineages using commercially available differentiation protocols. The cells induced to adipogenic differentiation displayed lipid vacuoles (Fig. 4A) and positive staining for Oil Red O (Fig. 4B). Chondrogenic differentiation was confirmed by Alcian Blue staining of sulfated proteoglycans (Fig. 4C). Osteogenic differentiation was confirmed by Alizarin Red S staining of calcium deposits (Fig. 4D). These data clearly demonstrate that mediastinal adipose-derived stem cells have the ability to be directionally differentiated into multilineage cell types.

Directional differentiation of mediastinal adipose-derived stem cells. (A) Phase contrast of fat droplets formed after cells were induced to undergo adipogenesis, magnification set at 10x. (B) Oil Red O staining of fat droplets after adipogenesis induction. (C) Alcian Blue staining of the cells after chondrogenesis induction. (D) Alizarin Red S staining of the cells after osteogenesis induction.
Discussion
Adipose tissue is a dynamic endocrine organ that plays a critical role in metabolism regulation and physiological homeostasis (15,16). Mesenchymal stem cells have been routinely isolated from subcutaneous adipose depots for a number of years (2,13), but little information has been reported to date, describing the isolation of mesenchymal stem cells from visceral adipose depots. In this study, we describe stem cells isolated from mediastinal adipose tissue. We characterized the cells and demonstrated that they possess fundamental stem cell properties, including clonogeneity, multipotentiality, and self-renewal.
Mediastinal adipose-derived stem cells are similar to MSCs derived from subcutaneous adipose depots and bone marrow based on their growth kinetics, cell surface marker profile, and multilineage potential (5,8,11). They express typical mesenchymal stem cells markers, such as CD44, CD73, CD90, and CD105, and are negative for hematopoietic markers CD14, CD34, and CD45. Multilineage differentiation demonstrated that the cells were capable of undergoing adipogenesis, osteogenesis, and chondrogenesis. These mediastinal adipose-derived stem cells can be expanded for greater than 20 passages and express cell surface markers similar to subcutaneous adipose-derived stem cells, with the exception of CD9 and CD63. Previous reports have demonstrated that levels of CD9 play a role in adipose-derived stem cell proliferation, attachment, and in vivo engraftment (9).
Our data provide the first evidence of a stem cell population derived from mediastinal adipose depots. With the proximity of these adipose depots to the heart (4,18), this cell source may be a novel means to study the mechanisms underlying the development of cardiovascular disease in obesity and related metabolic disorders.
Footnotes
Acknowledgment
The authors declare no conflict of interest.
