Abstract
Tendon stem/progenitor cells (TSPCs) were recently identified within tendon tissues. The aim of this study was to investigate TSPC-seeded knitted silk—collagen sponge scaffold for functional shoulder repair. The multidifferentiation potential, proliferation, and immune properties of TSPCs were investigated in vitro, while the efficacy of TSPC-seeded knitted silk—collagen sponge scaffolds in promoting rotator cuff regeneration was evaluated in vivo within a rabbit model. TSPCs, which exhibited universal stem cell characteristics (i.e., clonogenicity, high proliferative capacity, and multidifferentiation potential), nonimmunogenicity, and immunosuppression, proliferated well on our scaffold in vitro. Implantation of allogenous TSPC-seeded scaffolds within a rabbit rotator cuff injury model did not elicit an immunological reaction, but instead increased fibroblastic cell ingrowth and reduced infiltration of lymphocytes within the implantation sites at 4 and 8 weeks postsurgery. After 12 weeks, the allogenous TSPC-treated group exhibited increased collagen deposition and had better structural and biomechanical properties compared to the control group. This study thus demonstrated that the allogenous TSPC-seeded knitted silk—collagen sponge scaffold enhanced the efficacy of rotator cuff tendon regeneration by differentiating into tenocytes, and by secreting anti-inflammatory cytokines that prevent immunological rejection. Hence, allogenous TSPC-seeded knitted silk—collagen sponge scaffolds can be a clinically useful application for tendon tissue engineering.
Keywords
Introduction
Shoulder rotator cuff tendon injury is one of the most common types of tendon injuries encountered during overhead activities and sports (10,34), and increases in frequency with age. Currently, there are numerous treatment modalities for repairing rotator cuff injuries, including tendon-to-bone fixation (11), suture techniques (28), tendon prostheses (48), autografts (53), allografts (60), and xenografts (32). However, these therapeutic options still have considerable drawbacks, such as high rates of recurrent tearing (20), inevitable donor site morbidity (5,6), poor graft integration (41), and immunological reaction (7,44). Due to the various shortcomings of these current treatment modalities, more optimal techniques for repairing rotator cuff tendon injuries need to be developed.
In recent years, tissue engineering constructs have been developed to aid the regeneration of tendon/ligament injury (14,55). Transplanted cells are a crucial component of tissue engineering and should ideally be able to influence the regeneration of injured tendons by enhancing cell proliferation and deposition of appropriate extracellular matrix (56). Bone marrow mesenchymal stem cells (BMSCs) and embryonic stem cell-derived mesenchymal stem cells (ESC-MSCs) have been widely utilized as a cell source in tendon/ligament tissue engineering due to their multipotency and high proliferative capacity (16,22,25,26,45). However, optimal regeneration was not achieved with these cells (9,16,30). Recently, a new specific cell type named tendon stem/progenitor cells (TSPCs) was identified within the tendon tissues of many species (9,51,65). These cells not only exhibit universal stem cell characteristics similar to BMSCs (52), but also displayed strong expression of tendon-related genes and proteins, such as collagen I, tenascin C, scleraxis (Scx), and tenomodulin (TNMD) (9). Our previous work also showed that tendon-like tissues can be formed when TSPCs were cultured on aligned nanofibers or transplanted into the dorsal surface of immunocompromised mice (9,64). These results thus suggest that tendon-derived TSPCs have great potential for the repair and regeneration of tendon injuries. However, the transplantability and efficacy of TSPCs in promoting the healing of tendon injury has not yet been evaluated in vivo.
Inadequate cell numbers and donor site morbidity (i.e., pain, muscle atrophy, and tendonitis) (61) may still limit the clinical application of TSPCs in regenerating tendon tissue in vivo. Previous studies have shown that many different types of adult stem cells, such as BMSCs, adipose-derived stem cells (ADSCs), and periodontal ligament stem cells (PDLSCs) possess immunosuppressive properties (42,57). Based on these previous data, we set out to determine whether TSPCs possess similar immunosuppressive properties, by evaluating their ability to elicit an immune reaction and contribute to tendon regeneration within an allogenous environment.
A crucial mainstay of ligament and tendon tissue engineering, besides the seed cells, are scaffolds fabricated from various materials such as collagen, silk, acellular matrix, biodegradable synthetic polymers, and composite materials (2,14,22,23,27,32,46,47,58). Our group has developed a knitted silk—collagen sponge scaffold (15), and previous studies have shown that this scaffold possess sufficient mechanical strength and optimal internal spaces, as well as can mimic natural extracellular matrix (ECM) by degrading slowly while being replaced by new tissue (15,54). However, the structure and function of the tissue engineered tendon was still much inferior to that of natural tendon. These results thus suggest that biomaterials alone or biomaterials incorporated with chemokines still cannot initiate full regeneration and repair of tendon. Hence, this study developed a cell-seeded knitted silk—collagen sponge scaffold to aid repair of rotator cuff tendon injury.
The aim of this study was to investigate TSPC-seeded knitted silk—collagen sponge scaffold for functional shoulder repair. The results showed that allogenous TSPC-seeded knitted silk—collagen sponge scaffolds improved the structural and biomechanical properties of the repaired rotator cuff tendon without any observable immunological rejection. Hence, allogenous TSPCs-seeded knitted silk—collagen sponge scaffolds can be a clinically useful application for tendon tissue engineering.
Materials and Methods
Cell Isolation and Culture
The isolation and culture protocols for tendon stem/ progenitor cells were based on a previous study by our group (64). Briefly, rotator cuff (RC) tendons of female New Zealand white (NZW) rabbits (6 months old) were collected, washed in phosphate-buffered saline (PBS), and cut into 1—2-mm3 pieces. The tendon pieces were then digested with 0.25% (w/v) collagenase (Sigma-Aldrich Inc., St. Louis, MO, USA) at 37°C overnight. Single cell suspensions were cultured in growth medium consisting of Dulbecco's modified Eagle's medium (DMEM, low glucose; Gibco-BRL Inc., Long Island, NY, USA) supplemented with 10% (v/v) fetal bovine serum (FBS; Invitrogen Inc., Carlsbad, CA, USA) and 1% (v/v) penicillin-streptomycin (Gibco-BRL Inc.). Fresh culture medium was replenished once every 3 days.
Monoclonal Selection and Colony Forming Unit (CFU) Assay
The isolated cells were transferred to fresh growth medium comprised of DMEM supplemented with 20% (v/v) FBS (Invitrogen Inc.) and 1% (v/v) penicillin-streptomycin (Gibco-BRL Inc.). After two passages, fibroblast-like cells were reseeded at a very low density (2 cells/cm2) on 6-cm dishes to form colonies. After 12 days of culture, some of the dishes were stained with 1% (w/v) crystal violet solution (Sigma-Aldrich Inc.) in methanol, and the number of colonies (with diameter >2 mm) was counted. Cells isolated from rabbit rotator cuff tendon-derived colonies were named as rTSPCs, and after another two to four passages in ex vivo culture they were utilized for both in vitro and in vivo experiments.
Evaluating the Multipotent Differentiation Potential of rTSPCs
The multipotent differentiation potential of rTSPCs towards the adipogenic, osteogenic, and chondrogenic lineages was evaluated in vitro according to established protocols (9,64,65). For adipogenic differentiation, rTSPCs were induced under the influence of 1-methyl-3-isobutylxanthine, dexamethasone, insulin, and indo-methacin. For osteogenic differentiation, rTSPCs were induced by treatment with β-glycerol phosphate, dexamethasone, and ascorbic acid. For chondrogenic differentiation, rTSPCs were induced by treatment with transforming growth factor-β1 (TGF-β1). Positive induction of adipogenesis, osteogenesis, and chondrogenesis was confirmed by oil red staining, alkaline phosphatase (ALP) staining, and Safranin O staining, respectively.
Flow Cytometry Analysis of Major Histocompatibility Complex Class II (MHC-II) Expression
rTSPCs (1 × 106) were incubated with mouse anti-rabbit MHC-II antibodies (AbD Serotec Inc. Raleigh, NC, USA) for 0.5 h at 4°C. After washing, the cells were incubated with fluorescein isothiocyanate (FITC)-conjugated rabbit anti-mouse immunoglobulin G (IgG) (BD Biosciences Pharmingen Inc., San Diego, CA, USA) for 45 min on ice. Subsequently, the washed samples were analyzed with a Coulter Epics XL flow cytometer (Beckman-Coulter Inc., Brea, CA, USA) (39).
One-Way Mixed Lymphocyte Culture (MLC)
MLC assay was used to evaluate the immunogenicity of rTSPCs. Peripheral blood mononuclear cells (PBMCs) were collected from two different animals (NZW rabbit, 6 months old) by Ficoll-Paque Plus (1.077 g/ml; Amersham Biosciences Inc., Uppsala, Sweden) and cultured with lymphocyte culture medium [RPMI-1640 supplemented with 20% (v/v) FBS and 1% (v/v) penicillin-streptomycin]. PBMCs and rTSPCs were exposed to 25 μg/ml of mitomycin C (Kyowa Inc., Tokyo, Japan) in the dark at 37°C for 0.4 h, then washed twice, and utilized as stimulators (PBMC1 and rTSPC1). Untreated PBMCs were used as responders (PBMC2).
Mitomycin C-treated peripheral blood mononuclear cells (PBMC1, 1 × 106) or rTSPCs (TSPC1, 1 × 106) were cocultured with responding PBMCs (PBMC2, 1 × 105) in 0.2 ml of lymphocyte culture medium, within the wells of a 96-well round-bottomed plate at 37°C for 3 days. The results of the MLC assay was evaluated according to methods utilized in previous studies (8,39), and the results were expressed by the formula: stimulation index = [the proliferative response of PBMC1 (/rTSPC1)-treated groups — the proliferative response of untreated groups]/(the proliferative response of untreated groups) x 100% ± standard deviation (SD). All experiments were performed in triplicate.
Immunosuppression Assay
The two-way MLC assay was used to evaluate the immunosuppressive properties of rTSPCs. Responding PBMCs (PBMC2, 5 × 104) and stimulating PBMCs (PBMC1, 5 × 104) were cocultured with or without stimulating rTSPCs (TSPC1, 8 × 103) within a 96-well plate at 37°C and 5% CO2 for 3 days. The percentage inhibition of allogeneic proliferation was calculated by the formula: percent of maximal response = [the proliferative response of groups without (with) TSPC1]/(the proliferative response of groups without TSPC1) x 100% (8,39). All experiments were performed in triplicate.
Scaffold Fabrication
Raw silk fibers (Bombyx mori) were purchased from Zhejiang Cathaya International Inc. (Hangzhou, China). The knitted silk—collagen sponge scaffold was fabricated as previously described (15), by forming sponge of collagen I within the opening of a knitted silk mesh. The knitted silk—collagen sponge scaffolds were cross-linked by dehydrothermal treatment. For the in vitro study, the knitted silk—collagen sponge scaffold were cut into 10 × 10-mm pieces, while for the in vivo study, they were cut into 20 × 10-mm pieces, and then folded into 10 × 5-mm pieces.
Preparation and Culture of Cell-Seeded Scaffold
Before use, all knitted silk—collagen sponge scaffolds were treated with 75% ethanol (v/v) for 1 h and washed three times with sterile PBS. For the in vitro study, the knitted silk—collagen sponge scaffolds were cut into 10 × 10-mm pieces, and were seeded with 1 × 105 rTSPCs in 24-well culture plates. The cell-seeded scaffolds were cultured in a 5% CO2 incubator at 37°C for 14 days, and the culture medium was replaced once every 3 days. Cellular proliferation was quantified on days 1, 4, 7, and 14. Scanning electron microscopy (SEM) imaging of cellular morphology was carried out on days 1, 7, and 14. Extraction of cellular mRNA for real-time polymerase chain reaction (RT-PCR) analysis was carried out on days 1, 7, and 14.
For the in vivo study, the knitted silk—collagen sponge scaffold was cut into 20 × 10-mm pieces. Subsequently, 6 × 105 allogenous rTSPCs [prestained with DiI (1,1′-dioctadecyl-3,3,3′3′-tetramethylindocarbocyanine perchlorate)] were uniformly suspended in type I collagen solution (6 mg/ml), prior to being loaded onto each scaffold and cultured for 8 h to enable cell adhesion before implantation. The culture medium was replaced by serum-free medium on the day before the allogenous TSPC-seeded scaffolds were to be implanted.
Proliferation Assay
The proliferation of rTSPCs within a 10 × 10-mm piece of knitted silk—collagen scaffold (initial seeding density of 105 cells/scaffold) was assayed by the cell counting kit-8 (CCK8, Dojindo Laboratories Inc., Kumamoto, Japan) on day 1, 4, 7, and 14 after seeding, according to the manufacturer's instructions, with absorbance readings being taken at 450 nm (43).
SEM Imaging
At 1, 7, and 14 days after seeding, the cell morphology and distribution were imaged with SEM. The cell-seeded scaffolds were immersed in OsO4 for 40 min and then washed three times in PBS. After dehydration in increasing concentrations of acetone (30—100%, v/v), the samples were coated with gold, then viewed under a Hitachi S-3000N scanning electron microscope (Hitachi Corp., Tokyo, Japan) at an accelerating voltage of 15 kV (64).
Animal Model
Fourteen female rabbits (6 months old) weighing 2.5—2.7 kg were used in this study. The rabbits were subjected to general anesthesia by administration of chloral hydrate, and their rotator cuff tendons were subsequently exposed by releasing parts of the trapezius and deltoid muscles through a longitudinal incision over the shoulder. A portion of the rotator cuff tendon was fully removed to create a rectangular defect of approximately 10 × 5 mm in area (13,14). On the left limbs, the defects were implanted with the control scaffold (knitted silk—collagen sponge + collagen gel containing PBS) using a nonresorbable suture (control group, n = 14). On the right limbs, the defects were implanted with the cell-seeded scaffold (knitted silk—collagen sponge + collagen gel containing allogenous rTSPCs, folded into 10 × 5 mm) (TSPC-treated group, n = 14). After surgery, the rabbits were allowed free cage activity and were housed at constant temperature with a 12-h dark—light cycle with unrestricted access to water and fed a standard diet.
After euthanasia, three rotator cuff tendons of rabbits from each experimental group were used for histological evaluation and gene expression analysis at 4, 8, and 12 weeks time points. Five samples containing the humerus head and rotator cuff tendon from both experimental groups were utilized for mechanical testing and three randomly selected rotator cuff tendons were imaged by transmission electron microscopy (TEM) to assess the collagen fibril diameter after 12 weeks posttreatment. Treatment of animals was in accordance to standard guidelines approved by the Zhejiang University Ethics Committee (ZJU2010105001).
Cell Labeling and Detection
The allogenous rabbit TSPCs utilized for in situ repair of rotator cuff were prestained with DiI. Briefly, the TSPCs were incubated with 5 μl/ml of Dil (Sigma-Aldrich Inc.) at 37°C for 20 min, and then washed with PBS. The Dil-stained cells were observed under fluorescence microscopy (IX71; Olympus Inc., Tokyo, Japan) at an excitation wavelength of 543 nm. Subsequently, the cells were seeded onto the knitted silk—collagen sponge scaffolds, as described earlier.
To evaluate the survival of implanted TSPCs within the rotator cuff defect, a noninvasive CCCD tracking system was used to image the implantation sites (containing the humerus head and rotator cuff tendon) at 4 and 12 weeks postsurgery (16). The fluorescent imaging system produces a composite pseudocolor image representing light intensity (blue signifies least intense and red most intense). The specific excitation wavelength of Dil was at 550 nm, and the control excitation wavelength was at 475 nm to eliminate background. The fluorescent image was superimposed in real time over the white-light image of the same sample. Additionally, samples were also harvested and histological sections were prepared. The positively stained cells within the histological sections of the TSPC-treated group were observed under fluorescence microscopy, while DAPI was used to stain the nuclei of all cells (64).
Histological Assessment
Specimens were fixed, dehydrated, and embedded within paraffin blocks. Histological sections (8 μm) were prepared using a microtome, and subsequently deparaffinized with xylene, hydrated using decreasing concentrations of ethanol, and then subjected to hematoxylin and eosin (H&E) staining and Masson trichrome staining. General histological scoring was performed using a blinded semiquantitative scoring system based on six parameters (fiber structure, fiber arrangement, rounding of nuclei, vascularity, inflammation, cell population) of H&E staining. Zero to 3 points were allotted to each of these parameters, which 0 being normal and 3 being maximally abnormal (14). The quantity and ratio of fibroblast-like cells and lymphocytes within the neotissue (internal space of scaffold but not including silk and collagen itself) were assessed by a modification of methods utilized in a previous study. Briefly, the number of lymphocytes was counted based on the presence of lymphocytes, while counts of fibroblast-like cells (including rTSPCs, tenocytes and fibroblasts) were performed based on the presence of spindle-shaped cells and collagen deposition in selected high power fields (HPFs) (54,63).
Immunohistochemistry
A series of 8-μm-thick sections was utilized for immunohistochemical staining. Mouse anti-rabbit monoclonal antibodies against collagen I (1:200 dilution; Abcam Inc., Cambridge, MA, USA) and collagen III (1: 200; Santa Cruz Biotechnology Inc., Santa Cruz, CA, USA) were used to detect the expression of these proteins within the repaired rotator cuff tendon. The primary antibody was omitted in the control experiment as previously described (21).
Determination of the Collagen Content of Repaired Tendons
A collagen quantitative assay kit (Jiancheng Inc., Nanjing, China) was used to quantify the amount of deposited collagen in the repaired rotator cuff tendon, according to the manufacturer's instructions.
Biomechanical Evaluation
Mechanical testing was performed on rotator cuff tendon specimens (n = 5 for the TSPC-treated group, n = 5 for the control group) using an Instron tension/ compression system with Fast-Track software (Model 5543, Instron, Canton, MA, USA) as described previously (15,54). The humerus head was secured by a specially designed restraining jig and the tendon end was pinched with a clamp. Specimen orientation and alignment were maintained within the line of the applied tensile force. After applying a preload of 1 N with a test speed of 5 mm/min, the rotator cuff—humerus head complex underwent preconditioning for a total of 20 cycles. This was followed by load to failure testing at an elongation rate of 5 mm/min. The biomechanical parameters of the rotator cuff—humerus being measured included stiffness (N/mm), modulus (MPa), maximum force (N), stress at failure (MPa), and emerge (mJ).
RNA Isolation and RT-PCR
Total cellular mRNA was isolated by lysis in TRIZOL (Invitrogen Inc.) followed by a one-step phenol chloroform-isoamyl alcohol extraction, according to the manufacturer's instructions. Real-time PCR analysis of the expression of five genes including collagen I, collagen III, biglycan (BGN), tenascin C (TNC), and decorin (DCN) was carried out using the Brilliant SYBR Green QPCR Master Mix (TakaRa Bio Inc., Shiga, Japan) with a Light Cycler apparatus (ABI 7900HT, Applied Biosystems Inc., Carlsbad, CA, USA), as previously described. The primer sequences used in this study are listed in Table 1. At least three experimental replicates were performed for each real-time PCR run, and the results are presented as target gene expression normalized to glyceraldehyde-3-phosphate dehydrogenase (GAPDH) (64).
List of Primer Sequences Utilized for Real-Time Polymerase Chain Reaction

Statistical Analysis
All quantitative data sets are expressed as mean ± SD. Student's t-test was performed to assess statistical significance of results between sets. Values of p < 0.05 were considered to be significantly different.
Results
Isolation and Characterization of TSPCs
Clonogenicity, self-renewal, and multipotency are three commonly used criteria for defining stem cells. In our study, 4 h after seeding a single-cell suspension of tenocytes, a subpopulation of tenocytes attached onto the culture plate. The attached cells remained initially quiescent for the first 3—5 days, and then started proliferating to form colonies. Ten to 12 days later, colonies formed from single cells were visualized with methyl violet staining (Fig. 1A, inset of Fig. 1A). A small subpopulation (15.55 ± 2.4%) of tenocytes explanted from rotator cuff tendon formed colonies (Fig. 1A inset). The rabbit rotator cuff tendon-derived stem cells exhibited similar fibroblast-like morphology (Fig. 1B) as BMSCs. Subsequently, the multidifferentiation potential of these cells was evaluated (Fig. 1C—E). The adipogenic differentiation assay showed that induced TSPCs exhibited accumulation of lipid droplets, as confirmed by oil red staining after 3 weeks of induction (Fig. 1E). The osteogenic differentiation assay showed that most of the TSPCs had the capacity to undergo osteogenesis, as confirmed by positive ALP staining of mineralized calcium deposits (Fig. 1C). Chondrogenic differentiation of hTS-PCs was confirmed by positive Safranin O staining after 4 weeks of mass culture and induction (Fig. 1D).

Isolation and characterization of rTSPCs. Morphology of colony formation (A). Inset of (A) shows the methyl violet staining of colonies formed by rTSPCs. Morphology of TSPCs at P1 (B). Inset of (B) shows the morphology of TSPCs at P0. ALP staining shows osteogenic differentiation of TSPCs (C). Safranin O staining shows chondrogenic differentiation of TSPCs (D). Oil red O staining shows adipogenic differentiation of TSPCs (E). Flow cytometry analysis of the expression of MHC-II (F). TSPCs do not elicit allogeneic PBMCs proliferation (G). TSPCs inhibit ongoing PBMCs proliferation (H). rTSPCs, rabbit tendon stem/ progenitor cells; ALP, alkaline phosphatase; P1, passage 1; P0, passage 0; MHC-II, major histocompability complex class II; PBMC, peripheral blood mononuclear cells. Scale bars> 50 μm (C, D, E), 500 μm (A, B, inset of B).
Immunogenicity and Immunosuppressive Properties of TSPC In Vitro
Previous studies showed that MSCs are MHC-II negative and are able to suppress lymphocyte proliferation in vitro (39). Hence, we investigated the expression of MHC-II on the TSPC surface by flow cytometry analysis and the results showed that TSPCs lacked expression of MHC-II (Fig. 1F). MLC assay was used to evaluate the immunogenicity of rTSPCs. Autologous lymphocytes served as the negative control while allogeneic PBMCs served as the positive control. The results demonstrated that allogeneic TSPCs decreased lymphocyte proliferation compared with allogeneic PBMCs (Fig. 1G). To determine the suppressive effects of TSPCs on activated lymphocytes, the two-way MLC assay was carried out. The results showed that TSPCs can partially inhibit the proliferation of lymphocytes in response to allogeneic PBMCs (Fig. 1H).
Cell Proliferation and Morphology on Scaffold
Rabbit TSPCs were seeded on our knitted silk—collagen sponge scaffold (Fig. 2) and the cell—biomaterial interaction was characterized. Results of the proliferation assay showed that TSPCs initially exhibited fast proliferation, and then reached a plateau on day 7 (Fig. 2B—E). SEM imaging of the TSPCs-seeded scaffold showed that TSPCs attached and spread well on the collagen sponge (Fig. 2B). TSPC aggregates started to overlap with adjacent cell aggregates on day 7 and a thick cell sheet was formed by day 14 (Fig. 2C, D).

Cell proliferation and morphology on scaffold. The SEM image of the interface of scaffold shows that spaces between knitted silk fibers are filled with collagen microsponge (A, left). (A, right) shows the surface of the scaffold. The SEM images showed the adhesion, proliferation, and cell sheet formation by rTSPCs cultured on the knitted silk—collagen sponge scaffold for 1, 7, and 14 days (B, C, D). Proliferation of rTSPCs on the knitted silk—collagen sponge scaffold (E). Real-time PCR analysis of COL I (F) and COL III (G). BGN (H) and TNC (I) gene expression by TSPCs cultured on scaffolds for 7 and 14 days. The results of the target gene expression were normalized to the housekeeping gene, GAPDH. *Significant differences between two time points (7 and 14 days) (p < 0.05). SEM, scanning electron microscopy; rTSPCs, rabbit tendon stem/progenitor cells; PCR, polymerase chain reaction; COL I, collagen I; COL III, collagen III; BGN, biglycan; TNC, tenascin; GAPDH, glyceraldehyde 3-phosphate dehydrogenase. Scale bars: 500 μm (A—D).
Expression of Tendon-Related Genes
We compared the gene transcript levels of collagen I, collagen III, biglycan (BGN), and tenascin (TNC) of TSPCs cultured on knitted silk—collagen sponge scaffolds with RT-PCR on days 7 and 14 (Fig. 2F—I). From days 7 to 14, the data showed that the transcript levels of collagen I, collagen III, BGN, and TNC of TSPCs were increased by 1.13 (p < 0.05), 1.63 (p < 0.05), 0.27, and 2.46-fold, respectively. This suggests that implanted TSPCs may increase ECM production during the repair process.
Gross Morphology of Regenerated Rotator Cuff Tendons
Rotator cuff defects implanted with silk—collagen scaffold (Fig. 3A, B and inset of Fig. 3A) with or without seeded TSPCs were observed to heal with continuity of neotissues without inflammation or scaffold dislocation. Nevertheless, it was observed that the TSPC-treated group had larger midsubstance and more neotissue ingrowth compared to the control group. The neotendons in both groups can easily be distinguished from native tendon at 4 and 8 weeks postsurgery, due to the repair sites exhibiting amber coloring. At 12 weeks postsurgery, the neotendons in both groups exhibited a cord-like shape filled with fibrous tissue. Although the shape, luster, and color of regenerated tendons were similar to that of normal uninjured rotator cuff tendons, the regenerated tendons still did not exhibit the typical shiny-white smooth surface appearance associated with native rotator cuff tendons (Fig. 3C, D).
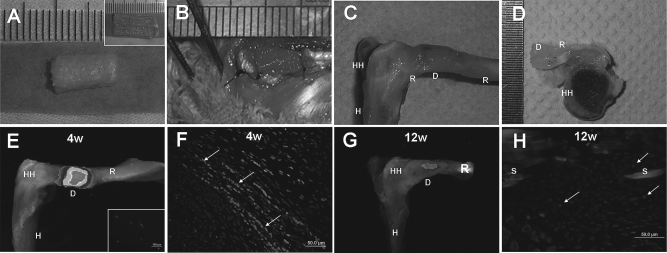
Rotator cuff regeneration model and CCCD analysis. Gross views of TSPCs-seeded knitted silk—collagen sponge scaffold (A). Scaffolds were sutured to the rotator cuff tendon defect (B). Gross and cross-sectional view of repaired rotator cuff tendon (C, D). CCCD analysis demonstrated a positive orange fluorescence signal in the repair sites, indicating the survival of implanted TSPCs at 4 and 12 weeks postsurgery (E, G). Inset of (E) shows the DiI-stained TSPCs. The fluorescence image of repaired tendon showing surviving TSPCs at the tendon defect at 4 and 12 weeks postsurgery, with white arrows showing the DII-stained TSPCs (F, H). rTSPCs, rabbit tendon stem/progenitor cells; H, humerus; HH, head of humerus; R, rotator cuff tendon; D, TSPCs (prestained with DiI), seeded scaffolds; 4W, 4 weeks postimplantation; 12W, 12 weeks postimplantation; S, knitted silk. Scale bars: 50 μm (F, H), 100 μm (inset of E).
In Vivo Tracking of Implanted DiI-Labeled TSPCs
In order to evaluate whether the implanted allogeneic TSPCs contributed to the regeneration of rotator cuff tendon, we traced the fate of TSPCs in vivo. The TSPCs were stained with DiI before the cell-seeded scaffold was implanted into the rotator cuff defects and we examined the samples at 4 and 12 weeks postsurgery. Image tracking showed positive DiI labeling at the repair site after 4 weeks postsurgery (Fig. 3E), and the presence of DiI-labeled TSPCs was confirmed with fluorescence microscopy (Fig. 3F). Imaging with fluorescence microscopy confirmed that the DiI-labeled TSPCs contributed to tendon regeneration at the injury site, as seen by the spindle-shaped distribution of the labeled cells and their alignment along the axis of the rotator cuff tendon. The number of implanted TSPCs greatly decreased after 4 weeks postsurgery. However, we can still find some surviving implanted cells at the injury site, as confirmed by the small animal fluorescent imaging system and fluorescence microscopy (Fig. 3G, H).
Histology and Immunohistochemistry of Rotator Cuff Tendon Repair
After 4 weeks postsurgery, it was observed that allogeneic TSPC implantation did not elicit an immunological reaction, and no inflammation was evident at any time point. In contrast, the TSPC-treated group had more ingrowth of fibroblastic cells (605 ± 147 vs. 424 ± 126/HPF) and less infiltration by lymphocytes (14 ± 7 vs. 25 ± 16/HPF) compared to the control group (Fig. 4). In the TSPC-treated group, the surface and interface of the scaffold were composed of about 2.3% lymphocytes, whereas in the control group, 5.6% of cells were lymphocytes (Fig. 4). After 8 weeks postimplantation, the number of fibroblast-like cells and lymphocytes decreased in both groups. However, the TSPC-treated group still had slightly more fibroblastic cells (399 ± 11 vs. 322 ± 114/HPF) and less lymphocytes (3 ± 2 vs. 10 ± 3/HPF, p < 0.05) compared to the control group (Fig. 5). Additionally, the TSPC-treated group had denser ECM within the repaired tissue compared to the control group (Figs. 4 and 5). After 12 weeks postimplantation, bundles of ripe collagen fibers were formed at the interface of the knitted silk—collagen sponge scaffold, which more closely resembled normal tendon fibers (Fig. 6).

Regenerated rotator cuff tendon at 4 weeks postimplantation. Typical H&E staining (A, D), Masson trichrome staining (B, E), and polarized light microscopy images (C, F) of TSPC-treated group and control group. The quantity and ratio of fibroblastic and lymphocytes within the scaffold (G). Histological evaluation (H) and collagen content (I) of the regenerated rotator cuff tendon. H&E, hematoxylin and eosin staining; Control, knitted silk—collagen sponge scaffold-treated group; TSPC, rabbit tendon stem/ progenitor cell scaffold-treated group; 4W, 4 weeks postimplantation; F, fibers; S, silk; IC, immune cells; C, continuous fibers; N, number. Scale bars: 50 μm (A, B, D, E), 100 μm (C, F, and insets).

Regenerated rotator cuff tendon at 8 weeks postimplantation. Typical H&E staining (A, D), Masson trichrome staining (B, E), and polarized light microscopy images (C, F) of TSPC group and control group. The quantity and ratio of fibroblastic cells and lymphocytes within the scaffold (G). Histological evaluation (H) and collagen content (I) of the regenerated rotator cuff tendon. H&E, hematoxylin and eosin staining; Control, knitted silk-collagen sponge scaffold-treated group; TSPC, rabbit tendon stem/ progenitor cell scaffold-treated group; 8W, 8 weeks postimplantation; F, fibers; S, silk; IC, immune cells; C, continuous fibers; N, number. Scale bars: 50 μm (A, B, D, E), 100 μm (C, F, and insets). *p < 0.05 versus control group.

Regenerated rotator cuff tendon at 12 weeks postimplantation. Typical H&E staining (A, B), Masson trichrome staining (C, D), and polarized light microscopy images (E, F) of TSPC-treated group and control group. Histological evaluation (G) and collagen content (H) of the regenerated rotator cuff tendon. H&E, hematoxylin and eosin staining; TSPC, rabbit tendon stem/ progenitor cell scaffold-treated group; 12W, 12 weeks postimplantation; C, continuous fibers. Scale bars: 50 μm (A, B, C, D), 100 μm (E, F, and insets).
The histology score showed that the TSPC-treated group exhibited more physiological native tendon structures (the histology score of native/normal tendon is zero) than that of the control group at 4, 8, and 12 weeks postimplantation [10.4 ± 0.67 vs. 12.0 ± 0.86 (Fig. 4); 8.8 ± 1.28 vs. 10.7 ± 0.27, p < 0.05 (Fig. 5); 5.8 ± 1.70 vs. 6.7 ± 0.92, (Fig. 6)]. Polarized light microscopy of H&E staining showed that continuous fibers started forming at the implantation sites after 8 weeks postimplantation, and that the fibers were more homogeneous in the TSPC-treated group compared to the control group (Figs. 4, 5, and 6). Immunohistochemical staining of the ECM within the regenerated tissue showed that the TSPC-treated group had higher concentrations of collagen I at 8 weeks postimplantation, and higher concentrations of collagen I and collagen III at 12 weeks postimplantation (Fig. 7).

mmunohistochemical staining, real-time PCR analysis, and mechanical properties of regenerated rotator cuff tendon at 8 and 12 weeks postimplantation. Histological observation of repaired tendons by immunohistochemical staining specific for collagen I (A, B, D, E) and collagen III (C, F). Real-time PCR analysis of gene expression of COL I, COL III, and DCN in vivo at 12 weeks (G, H, and I). Mechanical properties of regenerated rotator cuff tendons, including the stiffness (J), maximum force (K), modulus (L), stress at failure (M), and emerge (N) of TSPC-treated groups were higher than that of control group. PCR, polymerase chain reaction; COL I, collagen I; COL III, collagen III; DCN, decorin. &p < 0.05 vs. control group. Scale bars: 50 μm (A, B, C, D, E, F), 100 μm in insets.
Collagen Content
In this study, we used a collagen assay kit to quantify the amount of deposited collagen within the implanted scaffold, and the results showed that the TSPC-treated group had a higher mean collagen content than that of the control group at 4, 8, and 12 weeks postimplantation [462.5 ± 206.02 vs. 363.8 ± 31.44 μg/mg (Fig. 4); 484.5 ± 198.13 vs. 383.0 ± 192.65 μg/mg (Fig. 5); 524.9 ± 103.39 vs. 521.8 ± 243.05 μg/mg (Fig. 6)].
Gene Expression Analysis
Using real-time PCR analysis, we compared the expression levels of collagen I, collagen III, and decorin of the TSPC-treated group and the control group at the 12 weeks time point. The transcript levels of collagen I, collagen III, and DCN were all higher in the TSPC-treated group compared to the control group (Fig. 7) at 12 weeks postimplantation.
Biomechanical Properties of Repaired Rotator Cuff Tendon
The Instron tension/compression system was used to evaluate the biomechanical properties (including stiffness, maximum force, emerge, modulus, and stress at failure) of our specimens (Fig. 7). All specimens were firmly attached during biomechanical testing, and the load—deformation curves of repaired rotator cuff tendon of both the TSPC-treated group and control group were similar to that of native rotator cuff tendon, including toe region, linear region, microfailure region, and failure region.
The values of stiffness, maximum force, modulus, stress at failure, and emerge were all higher in the TSPC-treated group compared to the control group (64.3 ± 12.9 vs. 41.4 ± 7.0 N/mm, p < 0.05; 89.9 ± 18.5 vs. 64.1 ± 16.8 N, p > 0.05; 67.8 ± 14.6 vs. 44.3 ± 12.1MPa, p < 0.05; 8.3 ± 1.5 vs. 5.9 ± 1.0 MPa, p < 0.05; 107.9 ± 31.2 vs. 79.6 ± 38.4 mJ, p > 0.05) (Fig. 7). The maximum force and emerge in the TSPC-treated group were 40% and 35% higher than that of the control group, respectively, even though differences were not statistically significant due to small sampling size (Fig. 7).
Discussion
In this study, we developed a novel allogenous TSPC-seeded knitted silk—collagen sponge to aid the repair and regeneration of rotator cuff tendon injury in vivo for the first time. The results positively indicated that: 1) tissue engineered tendon can be successfully fabricated by seeding TSPCs; 2) TSPCs are nonimmunogenic and are able to suppress the immune response in a similar manner as BMSCs; 3) TSPC-seeded scaffolds promoted the regeneration of rotator cuff injury in vivo, resulting in more physiological ECM structure and better biomechanical function without eliciting an immunological reaction. Collectively, these results demonstrated that allogenous TSPC-seeded scaffolds offer much promise for clinical application of tendon tissue engineering in the near future.
TSPCs Are an Ideal Cell Source for Tendon Tissue Engineering
Selecting the suitable cell type for transplantation, which should ideally possess high proliferative capacity and tissue specificity, is absolutely crucial for successful tissue engineering. Tenocyte, as a tissue-specific cell, has also been used to repair tendon and ligament injuries (12,37,40). However, the limited availability of autologous tendon tissues, low proliferative capacity, and reduced matrix production significantly lowers the efficacy of successful tendon repair and regeneration with tenocytes (56). Recently, several studies identified a subpopulation of multipotent stem cells within the tendon tissues of various species, which were named tendon stem/progenitor cells (TSPCs) (9,51,65). These cells exhibit high proliferative capacity like BMSCs, as well as displayed strong expression of tendon-related genes and factors (9). Wang and colleagues demonstrated that the platelet-rich plasma releasate (PRCR) treatment promoted differentiation of TSPCs into functional tenocytes exhibiting high proliferation rate and abundant collagen production (66). Our previous study also demonstrated that the aligned electrospun nanofiber structure provided a conducive microenvironment for TSPC differentiation (64). These data thus suggest that TSPCs can be an ideal cell source for tendon repair in vivo.
Immunogenicity and Immunosuppressive Properties of TSPCs
The transplantation of allogenous TSPCs opened up a new treatment modality for the successful regeneration of injured tendons, but the risk of immunological rejection may limit clinical application of allogenous TSPCs. In recent years, BMSCs were widely reported to be nonimmunogenic and immunosuppressive (4,8,29,39,49). MSC-derived osteogenic, chondrogenic, and adipogenic cells have also been shown to be nonimmunogenic after differentiation (35). Some adult tissue derived stem cells, such as ADSCs, umbilical cord lining mesenchymal stem cells, and PDLSCs, possessed similar immune properties as BMSCs (17,19,42,57). However, other adult stem cells such as telencephalon tissue-derived neuronal stem cells were rejected upon transplantation (36). These conflicting data mean that adult stem cells derived from different tissues may exhibit different immune properties. Hence, it is necessary to investigate the immune properties of TSPCs, and we carried out a number of assays in vitro, to characterize expression of MHC-II and the influence of TSPCs on resting and activated allogenic PBMCs.
Our immunological assays demonstrated that TSPCs had negative expression of MHC-II and did not stimulate but in fact actively suppressed allogenous PBMCs. This is similar to what was observed with BMSCs (39), and suggests that TSPCs can also be utilized for allogenous transplantation and tissue engineering in a manner similar to BMSCs. Our in vivo studies also demonstrated that allogenous TSPC-seeded scaffolds not only elicited negligible immune reaction, but also decreased lymphocyte infiltration induced by our xenogenic scaffold. In our present study, we did not detect the expression of MHC-II after maturation of TSPCs. Liu et al. found that osteogenic cells differentiated from MSCs did not express MHC-II under in vitro culture conditions. However, the expression of MHC-II is inducible upon implantation in vivo (39). Our in vivo results also show that the labeled TSPC number decreased after 4 weeks postsurgery, as confirmed by the small animal fluorescent imaging system and fluorescence microscopy. According to our previous finding on tendon tissue engineering with hESCs, this might have been due to the remodeling process of tendon repair as well as immune reactions (16). Hence, the question on how to maintain the nonimmunogenicity and block the way to immunogenicity of TSPCs is the decisive question for the future application of TSPCs. The feasibility of such allogenous TSPC implantation in vivo will be further investigated in preclinical studies with animal models in our future study, and the findings would be more convincing if two strains of rabbits were used and more rabbits were added.
Allogenous TSPC-Seeded Knitted Silk—Collagen Sponge Scaffold Enhances the Efficacy of Rotator Cuff Tendon Regeneration In Vivo
Previous studies showed that TSPCs were able to differentiate into different cell lineages (including adipocytes, chondrocytes, and osteocytes) in vitro and formed tendon-like, fibrocartilage-like, and bone-like tissues in vivo upon ectopic implantation (9,65). However, our in vivo study demonstrated that the TSPC-treated group yielded better results both structurally and functionally. Firstly, our scaffold may play an important role in the healing process as both our current and previous in vitro studies proved that the collagen—silk scaffold supported adhesion, proliferation, and tenocyte lineage differentiation of stem cells (15). Secondly, the host shoulder joint can serve as an autologous bioreactor, while mechanical loading on cell-seeded scaffolds might modulate the differentiation of stem cells into tenocyte-like cells (16,33).
ECM, which functions as a lubricator and organizer of collagen fibril assembly (33), plays an important role in modulating the fate of TSPCs (9). Collagen I and collagen III are the major ECM components of normal tendon (3). Many studies showed that the expression of collagen I and collagen III was increased during the early stages of tendon repair and remodeling, and high production levels of collagen III might suggest that the stem cells are secreting signals for the repair process (1,62). The in vivo study demonstrated that the gene transcript levels of collagen I and collagen III in the TSPC-treated group were higher compared to the control group at 12 weeks postsurgery, while immunohistochemical staining of the regenerated tissue ECM indicated that the TSPC-treated group produced more tendon ECM components (including collagen I and collagen III) at 8 and 12 weeks postimplantation. Therefore, the presence of the implanted TSPCs leads to higher expressions levels of genes and proteins (i.e., collagen I and collagen III) that contribute to the regeneration of rotator cuff tendon injury. However in our study, the implanted TSPCs still cannot fully regenerate the natural structure and function of injured tendons in vivo, and optimal induction and implantation methods need to be investigated in future studies.
Due to the remodeling process of tendon repair and immune reactions, the number of labeled TSPCs decreased after 4 weeks. This result indicated that the seeded TSPCs may not contribute to tendon repair after 4 weeks postsurgery. As a small number of TSPCs can survive until 12 weeks postsurgery, it cannot be excluded that TSPCs secrete neotendon ECM as seed cells. Moreover, TSPCs may also secrete growth factors and enhance the endogenous regeneration of injured tendon. In our previous study, we demonstrated that transplanted hESC-MSCs secreted growth factors, such as TGF-β3, growth differentiation factor 5 (GDF5), bone morphogenetic protein 2 (BMP2), basic fibroblast growth factor (bFGF), stromal derived factor-1 (SDF-1), and fractalkine in repaired tendons (16). A number of growth factors are involved in tendon repair and regeneration (24,31,59).
One limitation of this study is that the detailed mechanism of immunosuppression by TSPCs has not been fully explored. Several different mechanisms of BMSC-mediated immunosuppression have been proposed. Devine et al. (18) hypothesized that allogenous MSCs can integrate into bone marrow to influence early immune cell growth, as well as migrate into the thymus to play a role in T-cell selection. Puissant et al. (50) found that ASCs exhibited immunosuppression by releasing soluble factors, such as interleukin-10 (IL-10) and TGF-β. Hence, further studies are needed to elucidate the mechanisms of TSPC-mediated immunosuppression.
In our study, we found that TSPCs exhibited fast proliferation on our knitted silk—collagen sponge scaffold, and the transcript levels of the tendon-related genes were increased with proliferation. Nevertheless, the exact mechanism by which the scaffolds influence ECM assembly is still unclear. Some studies suggested that the geometry of the sponge surface might alter the structure of the TSPC niche through interaction with cell surface receptors and adhesion sites, thus affecting the differentiation of TSPCs, as well as influencing the synthesis and secretion of key tendon ECM components (9,38). The interaction between TSPCs and scaffold will be explored in more depth in our future work.
Footnotes
Acknowledgments
This work was supported by the Major State Basic Research Development Program of China (2012CB966600), the National Natural Science Foundation of China (81125014, 81071461, 81101355, J1103603), Zhejiang Provincial Key Laboratory of Tissue Engineering and Regenerative Medicine, Zhejiang Provincial Natural Science Foundation of China (Z2100086), Zhejiang Innovation Program for Graduates (YK2010013), and Scholarship Award for Excellent Doctoral Student granted by Ministry of Education. The authors thank Chen Han Ming for the SEM imaging and Wang Li for the TEM imaging. The authors declare no conflicts of interest.
