Abstract
Development of a tissue-engineered neo-kidney augment (NKA) requires evaluation of defined, therapeutically relevant cell and cell/biomaterial composites (NKA constructs) for regenerative potential in mammalian kidney. Previous work identified primary renal cell populations that extended survival and improved renal function in a rodent model of chronic kidney disease (CKD). This study extends that work toward the goal of developing NKA by (i) screening in vivo inflammatory and fibrotic responses to acellular biomaterials delivered to healthy rodent renal parenchyma, (ii) evaluating the functionality of renal cell/biomaterial combinations in vitro, (iii) generating NKA constructs by combining therapeutically relevant cell populations with biocompatible biomaterial, and (iv) evaluating in vivo neokidney tissue development in response to NKA constructs delivered to healthy rodent renal parenchyma. Gelatin and hyaluronic acid (HA)-based hydrogels elicited the least inflammatory and fibrotic responses in renal parenchyma relative to polycaprolactone (PCL) and poly(lactic-
Introduction
The de novo regeneration of laminarly organized, tubular organs in mammals has been demonstrated for multiple organs, including bladder, trachea, gastrointestinal tract, lung, and vessels using cell/biomaterial composites as a foundational technology (4). However, regeneration of solid organs presents a unique challenge: how to elicit the coordinated organization of multiple, highly specialized cell types into complex three-dimensional microarchitectures within a defined parenchymal matrix and functional connection to vascular, lymphatic, and nervous systems. The kidney illustrates these challenges well. Numerous specialized cell types including podocytes, mesangial cells, endothelial cells, fibroblasts, epithelial cells, and multiple stem and progenitor cell populations are organized across the renal parenchyma into discrete, specialized functional units or nephrons, which serve to selectively filter electrolytes from the vasculature (40).
Numerous studies on regeneration of renal architecture and function following acute kidney injury point to tubular epithelial cells as playing a key role in restoring function (11,20). Heterogenous, tubular cell-enriched primary kidney cell isolates prepared from the medulla, cortex, and cortico-medullary junction compartments of kidneys based on differential buoyant density (1,29) increased survival and enhanced renal function in the 5/6 nephrectomy rodent model of chronic kidney disease (CKD) when treatment was administered after a progressive, chronic disease state was established (17). These selected, therapeutically bioactive primary renal cell populations are referred to as selected regenerative renal cells (SRRCs).
Applying the cell/biomaterial composite technology to a solid organ requires the identification of biomaterials that should ideally induce minimal inflammatory and fibrotic responses and elicit establishment of a vascular bed shortly after implantation to facilitate survival of implanted cells as well as infiltration by host-derived cells and tissues (6,21). Biopolymer gels based on naturally occurring hydrogels such as gelatin or hyaluronic acid (HA) have been widely used for encapsulating mammalian cells (14). Beads or particles composed of polycaprolactone (PCL) or poly(lactic-
We and others have been successful in regenerating three-dimensional tissue structures for hollow organs by combining ex vivo expanded autologous cells with three-dimensional scaffolds (15,16). Injectable cell/biomaterial composites were also used to regenerate adipose tissue following subcutaneous injection (12). These successes formed the basis for the experiments reported here, which evaluated the feasibility of extending the foundational technology of cell/biomaterial composite-based regeneration from tubular organs and soft tissue to solid organs by: (i) screening in vivo inflammatory and fibrotic responses to acellular biomaterials delivered to healthy rodent renal parenchyma, (ii) evaluating the functionality of renal cell/biomaterial combinations in vitro, (iii) generating neo-kidney augment (NKA) constructs by combining therapeutically relevant renal cell populations with biocompatible biomaterial(s), and (iv) evaluating in vivo neotissue development in response to NKA constructs delivered to healthy rodent renal parenchyma. We show that gelatin or HA-based hydrogels elicited minimal inflammatory and fibrotic responses, facilitated neovascularization, were biocompatible with primary renal cell populations, and maintained the tubular epithelial functional phenotype of seeded cells during in vitro maturation. In addition, NKA constructs composed of renal cells and gelatin-based hydrogels were delivered to the renal parenchyma of healthy rodent kidneys and elicited neokidney tissue formation at 1 week postimplantation. Finally, we explored a potential mechanism by which NKA constructs might influence renal responses after delivery to diseased tissue: transforming growth factor-β (TGF-β) induced epithelial-to-mesenchymal transition (EMT). Conditioned medium from SRRC, harvested from either two-dimensional (2D) cultures or NKA constructs matured for 3 days, attenuated the in vitro response of immortalized proximal tubule epithelial HK2 cells to TGF-β at concentrations previously demonstrated to induce EMT (9,13). Taken together, these data provide preliminary evidence that a regenerative response was induced in vivo by NKA constructs and are suggestive that NKA constructs have the potential to facilitate regeneration of neokidney tissue, thereby guiding the extension of these results to established mammalian models of CKD.
Materials and Methods
Biomaterials
Biomaterials were prepared as beads (homogenous, spherical configuration) or as particles (heterogenous population with jagged edges). Hydrogels: Gelatin beads (Cultispher S and Cultispher GL) manufactured by Percell Biolytica (Åstorp, Sweden) were purchased from Sigma-Aldrich (St. Louis, MO) and Fisher Scientific (Pittsburgh, PA), respectively. Cross-linked HA and HA/gelatin (HyStem™ and Extracel™ from Glycosan BioSystems, Salt Lake City, UT) particles were formed from lyophilized sponges made according to the manufacturer's instructions. Gelatin (Sigma) particles were formed from cross-linked, lyophilized sponges.
PCL was purchased from Sigma-Aldrich (St. Louis, MO). PLGA 50:50 was purchased from Durect Corp. (Pelham, AL). PCL and PLGA beads were prepared using a modified double emulsion (W/O/W) solvent extraction method. PLGA particles were prepared using a solvent casting porogen leaching technique.
All beads and particles were between 65 and 355 μm when measured in a dry state.
Cell Isolation, Preparation, and Culture
Cadaveric human kidneys were procured through National Disease Research Institute (NDRI) in compliance with all NIH guidelines governing the use of human tissues for research purposes. Canine kidneys were procured from a contract research organization (Integra). Rat kidneys (21-day-old Lewis rats) were obtained from Charles River Labs (MI).
The preparation of selected regenerative renal cell populations from whole rat, canine, and human kidney has been previously described in detail (1,17,29). Briefly, kidney tissue was dissociated enzymatically, centrifuged through a density gradient to recover the cells and remove debris, and cultured. Fractionation of cells was then achieved using a four-step OptiPrep (Axis Shield, Norton, MA) density gradient.
Cell/Biomaterial Composites (NKA Constructs)
For in vitro analysis of cell functionality on biomaterials, a uniform layer of biomaterials (prepared as described above) was layered onto one well of a six-well low attachment plate (Costar #3471, Corning). Human SRRCs (2.5 × 105 per well) were seeded directly onto the biomaterial. For studies of adherence of canine cells to biomaterials, 2.5 × 106 SRRC were seeded with 50 μl packed volume of biomaterials in a nonadherent 24-well plate (Costar #3473, Corning). After 4 h on a rocking platform, canine NKA constructs were matured overnight at 37°C in a 5% CO2 incubator. The next day, live/dead staining was performed using a live/dead staining assay kit (Invitrogen) according to the manufacturer's instructions. Rat NKA constructs were prepared in a 60-cc syringe on a roller bottle apparatus with a rotational speed of 1 rpm.
For the transcriptomic, secretomic, and proteomic analyses described below, NKA constructs were matured for 3 days. Cells were then harvested for transcriptomic or proteomic analyses and conditioned media was collected for secretomic profiling.
Functional Analysis of Tubular Cell-Associated Enzyme Activity
Canine NKA constructs (10 μl loose packed volume) in 24-well plates were evaluated using an assay for leucine aminopeptidase (LAP) activity adapted from a previously published method (37). Briefly, 0.5 ml of 0.3 mM l-leucine
Transcriptomic Profiling
Poly-adenylated RNA was extracted using the RNeasy Plus Mini Kit (Qiagen, CA). Concentration and integrity were determined by UV spectrophotometry. cDNA was generated from 1.4 μg isolated RNA using the SuperScript VILO cDNA Synthesis Kit (Invitrogen). Expression levels of target transcripts were examined by quantitative real-time polymerase chain reaction (qRT-PCR) using commercially available primers and probes (Table 1) and an ABI-Prism 7300 Real Time PCR System (Applied Biosystems, CA). Amplification was performed using TaqMan Gene Expression Master Mix (ABI, Cat #4369016) and TATA Box Binding Protein gene (TBP) served as the endogenous control. Each reaction consisted of 10 μl Master Mix (x2), 1 μl primer and probe (x20), and 9 μl cDNA. Samples were run in triplicate.
TaqMan Primer/Probes Used for Transcriptional Profiling of NKA Construct Prototypes

Secretomic Profiling
Conditioned medium from human NKA constructs was collected and frozen at −80°C. Samples were sent to Rules Based Medicine (TX) and Aushon Biosystems (MA) for biomarker concentration quantitation. Returned results for a given biomarker concentration in conditioned media were normalized relative to the concentration of the same biomarker in conditioned media from control cultures (2D culture without biomaterial) and expressed as a unitless ratio.
Proteomic Profiling
Protein from three independent replicates was extracted from cell/biomaterial composites and pooled for analysis by 2D gel electrophoresis. All reagents were from Invitrogen. Isoelectric focusing (IEF) was conducted by adding 30 μg of protein resuspended in 200 μl of ZOOM 2D protein solubilizer #1 (Cat #ZS10001), ZOOM carrier ampholytes pH 4–7 (Cat #ZM0022), and 2M DTT (Cat #15508-013) to pH 4–7 ZOOM IEF Strips (Cat #ZM0012). Following electrophoresis for 18 h at 500 V, IEF strips were loaded onto NuPAGE Novex 4–12% Bis-Tris ZOOM IPG well gels (Cat #NP0330 BOX) for SDS-PAGE separation and electrophoresed for 45 min at 200 V in MES buffer (Cat #NP0002). Proteins were visualized using SYPRO Ruby protein gel stain (Cat #S-12000) according to the manufacturer's instructions.
Confocal Microscopy
NKA constructs prepared from human or rat SRRC were matured for 3 days and then fixed in 2% paraformaldehyde for 30 min. Fixed NKA constructs were blocked and permeabilized by incubation in 10% goat serum (Invitrogen) in D-PBS (Invitrogen) + 0.2% Triton X-100 (Sigma) for 1 h at room temperature (RT). For immunofluorescence, NKA constructs were labeled with primary antibodies (Table 2) at a final concentration of 5 μg/ml overnight at RT. Labeled NKA constructs were washed twice with 2% goat serum/D-PBS + 0.2% Triton X-100 and incubated with goat or rabbit tetramethyl rhodamine iso-thiocyanate (TRITC) conjugated anti-mouse IgG2A (Invitrogen) secondary antibody at 5 μg/ml. For double labeling with DBA (
Primary Antibodies Used for Confocal Microscopy of NKA Constructs

Analysis of TGF-β-Mediated EMT in HK2 cells
HK2 cells (ATCC) were cultured in 50:50 media in fibronectin or collagen (IV)-coated culture dishes (BD Biosciences). For EMT assays, HK2 cells were seeded in 24-well collagen (IV)-coated plates at 70–80% con-fluency with 50:50 media or conditioned media collected from either 2D human SRRC cultures or NKA constructs made with human SRRCs that were matured for 3 days prior to media collection. TGF-β induction was initiated by adding 10 ng/ml TGF-β to the culture media 3 days prior to isolating RNA from the cells for the EMT assay. EMT was monitored by qRT-PCR by analyzing the relative expression of E-cadherin (an epithelial marker) and calponin (mesenchymal marker) at the end of the 3-day incubation period. RNA was prepared from harvested HK2 cells for TaqMan qRT-PCR analysis as described above. Statistical analysis was performed comparing E-cadherin and calponin expression in cultures grown with conditioned medium versus 50: 50 media. Statistical significance was determined using the Sidak corrected alpha level of 0.0253 for each individual marker.
In Vivo Implantation of Acellular Biomaterials and NKA Constructs
Lewis rats (6–8 weeks old) were purchased from Charles River (Kalamazoo, MI). All experimental procedures were performed under PHS and IACUC guidelines of the Carolinas Medical Center. Under isoflurane anesthesia, female Lewis rats (approximately 2–3 months old) underwent a midline incision, and the left kidney was exposed. Packed biomaterials (35 μl) (acellular biomaterial or NKA construct) were introduced by microinjection into the renal parenchyma. Two injection trajectories were used: (i) from each pole toward the cortex (referred to as cortical injection), or (ii) from the renal midline toward the pelvis (referred to as medullary injection). Rats were sacrificed at 1, 4, or 8 weeks postinjection. No early deaths occurred. Study design for the acellular implantation study is presented in Table 3.
Study Design for Evaluating Acellular Biomaterials in Healthy Adult Lewis Rat Kidneys

ND, not done.
Renal Histology
Representative kidney samples were collected and placed in 10% buffered formalin for 24 h. Sections were dehydrated in ascending grades of ethanol and embedded in paraffin. Sections (5 μm) were cut, mounted on charged slides, and processed for hematoxylin and eosin (H&E), Masson's trichrome, and Periodic Acid Schiff (PAS) staining in accordance with standard staining protocols (30). Digital microphotographs were captured at total magnification of x20, x100, and x400 using a Nikon Eclipse 50i microscope fitted with a Digital Sight (DS-U1) camera. Renal morphology changes were assessed by commonly used (30) severity grade schemes, to which descriptive terms (minimal, mild, moderate, marked/severe) were applied to describe the degree of glomerulosclerosis, tubular atrophy and dilatation, tubular casts, and interstitial fibrosis, and inflammation observed.
Results
Responses of Mammalian Kidney Tissue to Injection of Acellular Biomaterials Into the Renal Parenchyma
A panel of acellular biomaterials was initially screened for potential use in renal cell/biomaterial composites by direct injection into healthy rat kidneys (Tables 3 and 4). Tissue responses were evaluated by measuring the degree of histopathology parameters (inflammation, fibrosis, necrosis, calcification/mineralization) and biocompatibility parameters (biomaterial degradation, neovascularization, and neotissue formation) at 1 and 4 weeks postinjection (Fig. 1). At 1 week postimplantation, the host tissue responses elicited by each biomaterial tested were generally similar; however, gelatin hydrogels appeared to elicit less intense histopathological and more biocompatible responses (Fig. 1A). At 4 weeks postimplantation, the severity of histopathology parameters in tissues injected with HA or gelatin particles were qualitatively reduced compared to 1 week postimplantation. Gelatin particles were nearly completely resorbed and less giant cell reaction was observed than in tissues that received HA particles (Fig. 1B). In most cases where biomaterials were injected via the medullary injection trajectory (e.g., deeper into the medulla/pelvis), undesirable outcomes including obstruction leading to hydronephrosis, inflammatory reactions of greater severity, and renal arteriolar and capillary microembolization leading to infarction was observed (data not shown).

In vivo evaluation of acellular biomaterials at 1 (A) and 4 (B) weeks postimplantation. Trichrome x10: low-power image of kidney cross section showing biomaterial aggregate. Trichrome x40: close-up of biomaterial aggregate. Hematoxylin and eosin (H&E) x400: high magnification image of biomaterial aggregate to evaluate extent of cell/tissue infiltration. Each kidney was injected at two locations as described in Materials and Methods. PCL, polycaprolactone; PLGA, poly(lactic-
Histological Outcomes Following Acellular Implantation of Biomaterials Into Healthy Adult Lewis Rat Kidneys

ND, not done.
Assessing Functional Phenotype of Therapeutically Relevant Renal Cell Populations with Biomaterials
Therapeutically relevant renal cell populations (SRRC) that extended survival and increased renal function in a rodent model of chronic kidney disease after direct injection into renal parenchyma have been characterized (17) and methods for their isolation, characterization, and expansion have been developed and translated across multiple species (29). To assess whether SRRCs adhere to, remain viable, and retain a predominantly tubular, epithelial phenotype when incorporated into NKA constructs, transcriptomic, secretomic, proteomic, and confocal immunofluorescence microscopy analyses were conducted on NKA constructs produced from SRRCs and various biomaterials.
Adherence and Viability
Canine-derived SRRCs were seeded onto gelatin beads, PCL beads, PLGA beads, HA particles, and HA/gelatin particles as described (three NKA constructs per biomaterial). Cell distribution and viability were assessed 1 day after seeding by live/dead staining (Fig. 2). SRRCs adhered robustly to naturally derived, hydrogel-based biomaterials such as gelatin beads and HA/gelatin particles (Fig. 2A, D), but showed minimal adherence to synthetic PCL (Fig. 2B) or PLGA beads (not shown). Cells did not adhere to HA particles (Fig. 2C) but showed evidence of bioresponse (i.e., spheroid formation). Functional viability of the seeded SRRCs on hydrogel-based NKA constructs was confirmed by assaying for leucine aminopeptidase, a proximal tubule-associated hydrolase (data not shown).

Live/dead staining of neo-kidney augment (NKA) constructs seeded with canine-selected regenerative renal cells (SRRCs). Green indicates live cells; red indicates dead cells. (A) Gelatin beads; (B) PCL beads; (C) HA/gelatin particles; and (D) HA particles. Viable cells may be observed on all hydrogel-based NKA constructs.
Transcriptomic Profiling
The gene expression profiles of human SRRC in hydrogel-based NKA constructs (three NKA Constructs per biomaterial) and parallel 2D cultures of SRRCs were compared by quantitative transcriptomic analysis (Table 5). Transcripts examined fell into four principal categories: (i) tubular: aquaporin 2(AQ2), E-cadherin (ECAD), erythropoietin (EPO), N-cadherin (NCAD), cytochrome P450, family 24, subfamily A, polypeptide 1—aka vitamin D 24-hydroxylase (CYP), cubilin; (ii) mesenchymal: calponin (CNN1), smooth muscle myosin heavy chain (SMMHC); (iii) endothelial: vascular endothelial growth factor (VEGF), platelet endothelial cell adhesion molecule (PECAM); and (iv) glomerular: podocin nephrin. Overall, tubular marker expression was comparable between hydrogel-based NKA constructs and 2D SRRC cultures. Expression of the endothelial markers (VEGF and PECAM) was significantly increased in hydrogel-based NKA constructs compared to 2D SRRC culture. In contrast, the glomerular marker podocin exhibited significant variation among NKA constructs. Podocin levels in HA/gelatin-based NKA constructs were most comparable with those observed in 2D SRRC cultures. Conversely, podocin transcript was significantly overexpressed in gelatin hydrogel-based NKA construct. The glomerular marker nephrin was overexpressed from HA/gelatin hydrogel-based NKA construct relative to 2D SRRC culture or gelatin-hydrogel-based NKA construct. Interestingly, mesenchymal marker (CNN1 and SMMHC) expression was significantly downregulated (
Transcriptomic Profiles of NKA Constructs

Secretomic Profiling
NKA constructs were produced with human SRRC and gelatin or HA/gelatin hydrogel. Secretomic profiling was performed on conditioned media from NKA constructs matured for 3 days and compared with parallel 2D cultures of human SRRC (Table 6) by calculating the ratio of analyte expression of NKA constructs (3D culture) to 2D culture (3D:2D ratio). For both hydrogel-based NKA constructs seeded with SRRCs, the average fold 3D:2D ratios (representing the mean of the 3D:2D ratio across all analytes examined) was close to 1 (1.4 ± 1.2 and 0.82 ± 0.46 for gelatin hydrogel-based NKA construct and 1.3 ± 0.78 and 0.83 ± 0.45 for HA/gelatin hydrogel-based NKA construct), suggesting that when broadly evaluated, the seeding process and 3 days of maturation on these biomaterials had little impact on the overall, global secretomic profile of SRRCs. This result notwithstanding, as shown in Table 6, the 3D:2D ratios of individual analytes may deviate from 1.0 for both hydrogel-based NKA constructs. For example, IL-6 presents a 3D:2D ratio of 10.35 from gelatin hydrogel-based NKA construct and a 3D:2D ratio of 1.47 on HA/gelatin hydrogel-based NKA construct.
Secretomic Profile of NKA Constructs
Proteomic Profiling
Proteomic profiles of a given cell or tissue are produced by separating total cellular proteins using 2D gel electrophoresis and have been used to identify specific biomarkers associated with renal disease (39). In this experiment, proteomic profiling was used to compare protein expression in human SRRCs in NKA constructs (gelatin or HA/gelatin hydrogel-based, three NKA constructs per biomaterial) and in 2D tissue culture (Fig. 3A). The proteome profiles of total protein isolated from NKA Constructs or 2D cultures of SRRC were essentially identical, providing additional evidence that the seeding process and 3 days of maturation on these biomaterials had little impact on the proteomes expressed by SRRCs.

(A) Proteomic analysis of NKA constructs. NKA constructs were produced with human SRRCs and biomaterials as indicated. Proteins in total protein extracts were separated by 2D gel electrophoresis as described in Materials and Methods. TC, 2D culture of human SRRC cells. (B–D) Confocal microscopy of NKA Constructs produced with human (B) or rat (C, D) SRRC and gelatin hydrogel. (B) E-cadherin (red),
Confocal Microscopy
Retention of the tubular epithelial phenotype of rat and human SRRC (29) in NKA constructs was evaluated by confocal imaging of established biomarkers: E-cadherin and DBA in human NKA constructs (Fig. 3B) and E-cadherin, DBA, N-cadherin, cytokeratin 8/18/19, gamma-glutamyl transpeptidase (GGT-1), and megalin in rat NKA constructs (Fig. 3C, D). Expression of megalin, although detectable, was weak (Fig. 3D). Optical sectioning of confocal images also allowed evaluation of the extent of cell infiltration into the biomaterial after seeding and 3 days of maturation. SRRCs in human and rat NKA constructs exhibited expression of multiple tubular epithelial markers. Optical sectioning revealed minimal cell infiltration of the hydrogel construct, with cells generally confined to the surface of the biomaterial.
In Vivo Responses to Implantation of NKA Construct Prototypes
Based on the in vivo responses to biomaterial injection into renal parenchyma and the in vitro phenotype and functional characterization of SRRCs in NKA constructs described above, gelatin hydrogel was selected to evaluate the in vivo response to NKA construct injection into renal parenchyma in healthy Lewis rats. NKA constructs were produced from syngeneic SRRCs and implanted into two animals for each of three time points, which were sacrificed at 1, 4, and 8 weeks postimplantation. All animals survived to scheduled necropsy when sections of renal tissues were harvested, sectioned, and stained with trichrome, H&E, and PAS. At 1 week postinjection (Fig. 4A), gelatin beads were present as focal aggregates (left panel, circled area) of spherical and porous material staining basophilic and surrounded by marked fibrovascular tissue and phagocytic multinucleated macrophages and giant cells. Fibrovascular tissue was integrated within the beads and displayed tubular epithelial components indicative of neokidney tissue formation. Additionally, tubular and vasculoglomerular structures were identified by morphology (PAS panels). By 4 weeks postinjection (Fig. 4B), the hydrogel was completely resorbed and the space replaced by progressive renal regeneration and repair with minimal fibrosis (note the numerous functional tubules within circled area of 4-week trichrome panel). At 8 weeks postimplantation, evidence of neokidney-like tissue formation was observed, consistent with induction of early events in nephrogenesis. Comparison of the area of regenerative induction (Fig. 5B, D) with adjacent cortical parenchyma (Fig. 5C) showed presence of multiple S-shaped bodies and newly formed glomeruli.

In vivo evaluation of NKA constructs at 1 (A) and 4 (B) weeks postimplantation. Trichrome x10: low-power image of kidney cross section showing biomaterial aggregate. Trichrome x40: close-up of biomaterial aggregate. H&E/PAS (Periodic Acid Schiff) x400: high magnification image of biomaterial aggregate to evaluate extent of cell/tissue infiltration. Each kidney was injected at two locations as described in Materials and Methods.

In vivo evaluation of NKA construct at 8 weeks postimplantation. (A) Trichrome x40: low-power image close up of kidney cross section showing biomaterial aggregate. (B) Higher magnification (trichrome stained, x400) of boxed area allows for evaluation of the extent of cell/tissue infiltration and shows regenerative response induction consistent with neokidney tissue formation: moderate chronic inflammation (macrophages, plasma cells and lymphocytes), moderate numbers of hemosiderin-laden macrophages (chronic hemorrhage due to injection) with marked fibrovascular response (blue stained by Masson's trichrome). (C) Representative of adjacent (normal) kidney parenchyma showing typical cortical glomeruli morphology H&E, x400). (D) H&E-stained section, x400 comparing new glomeruli morphology observed in treatment area versus (C).
To specifically dissect the roles of SRRC and biomaterial in modulation of the regenerative process, NKA construct (SRRCs seeded gelatin hydrogel) and acellular gelatin hydrogel was injected into opposing poles of the same kidney (Fig. 6) at 1 week postimplantation. In the proximal pole (injected with NKA construct), the biomaterial was observed as a focal aggregate located within the medulla and surrounded by moderate to marked chronic inflammation consisting of macrophages, plasma cell, lymphocytes, and giant cells with-out evidence of eosinophilic infiltrate or necrosis. The biomaterial was completely infiltrated by a fibrovascular matrix showing moderate to marked neovascularization with focal tubulogenesis observed at the periphery of implant (trichrome, x400). In the distal pole (injected with acellular gelatin hydrogel), the biomaterial was located within the cortex and extending into the outer medulla. There was marked chronic inflammation (slightly greater than in the proximal pole) with less fibrovascular tissue infiltration to that observed in the proximal pole. No evidence of regenerative response induction characterized by neovascularization and tubulogenesis was observed.

In vivo evaluation of SRRC seeded and acellular gelatin hydrogel injected into opposing poles of same kidney at 1 week postimplant. In proximal pole, biomaterial observed as focal aggregate located within the medulla and surrounded by moderate to marked chronic inflammation consisting of macrophages, plasma cells, lymphocytes, and giant cells without evidence of eosinophilic infiltrate or necrosis. Biomaterial was completely infiltrated by fibrovascular matrix showing moderate to marked neovascularization with focal tubulogenesis observed at the periphery of implant (trichrome, x400). In the distal pole, biomaterial was located within the cortex and extending into the outer medulla. There was marked chronic inflammation (slightly greater than in proximal pole) with less fibrovascular tissue infiltration to that observed in the proximal pole. There was no clear evidence of embolic material, infarction or calcification/mineralization within the kidney parenchyma. Degradation of biomaterial was slightly less in the acellular biomaterial but overall considered mild.
Effect of Conditioned Media From NKA Constructs on TGF-β Induced EMT in HK2 Cells
The development of tubulointerstitial fibrosis during the progression of CKD is associated with TGF-β-mediated EMT of tubular epithelial cells (43). Also, attenuation of TGF-β pathways was observed in vivo in a rodent model of progressive CKD where survival was extended and renal function improved by treatment with SSRCs (17). The human proximal tubular cell line HK2 has been well established as an in vitro model system to test the stimulatory or inhibitory effects of small molecules or proteins on TGF-β-induced EMT (9,13). To investigate a potential mechanism by which NKA constructs might affect renal tissue responses postimplantation, conditioned medium collected from NKA constructs produced with SSRCs and hydrogel was evaluated in the HK2 EMT assay system (Fig. 7). When HK2 cells were cultured in 50:50 media (control), ECAD (epithelial marker) was expressed at higher levels than CNN1 (mesenchymal marker). When TGF-β was added to the media for 3 days (TGF-β control), ECAD expression was significantly downregulated with a concomitant upregulation of CNN1, consistent with induction of an EMT event. Conditioned medium from 2D SSRC cultures significantly (
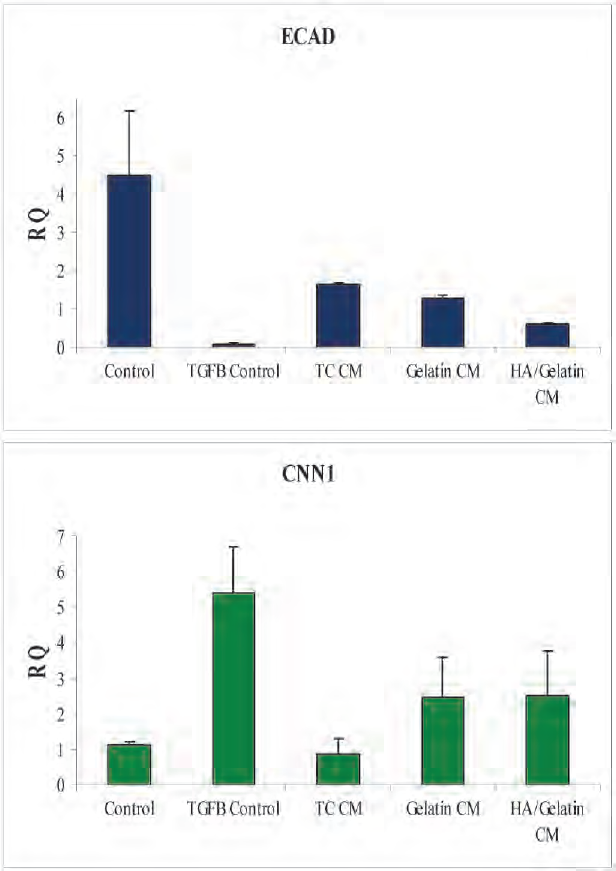
Conditioned medium from NKA constructs attenuates transforming growth factor-β (TGF-β)-induced epithelial-mesenchymal transition (EMT) in HK2 cells in vitro. EMT is monitored by quantitating the relative expression of E-cadherin (ECAD; epithelial) and calponin (CNN1; mesenchymal) markers. HK2 cells were cultured in 50:50 media (control and TGF-β control samples) or conditioned medium (CM) from 2D cultures of human SRRC (TC) or NKA constructs produced from human SRRCs and either gelatin or HA/gelatin as indicated. To induce EMT, 10 ng/ml TGF-β was added to each sample (except control) for 3 days prior to assay.
Discussion
CKD affects more than 19 million people in the US and frequently develops as a consequence of chronic obesity, diabetes, and/or hypertension (38). Patients in Stage 4–5 CKD receive dialysis and a complex drug regimen and the number of kidneys available for transplant is vastly insufficient to meet the need (28). New treatments that delay or reduce dialysis dependency are needed to fill this void.
Kidney tissue is composed of >20 specialized cell types structurally organized into morphologically and functionally distinct compartments that act in concert to filter blood, produce urine, and regulate endocrine function and acid-base and electrolyte balance. Cell–cell interactions are critical to kidney function and are at least partially dependent on spatial and architectural relationships. Regenerative approaches aim to reestablish homeostasis in part through restoration of cellular organization and intercellular communication. Indeed, progress toward this goal has recently been made with the identification of renal cells that demonstrated therapeutic benefit in a rodent model of CKD (17).
While cell-based approaches to extending survival and enhancing renal function in rodents with progressive CKD (17) are promising, additional efforts may be required to enable product delivery and extend application of these approaches to patients with severe fibrosis and end-stage renal disease. Combining bioactive cells with biomaterials could enhance the regenerative response by providing space and context to drive formation of new functional renal mass (6,24). The biomaterial component(s) of NKA constructs could also serve to enhance shelf-life of the product by preserving cell viability and to target delivery of bioactive component(s) to specific regions of the kidney to maximize effectiveness. Structural modifications that can be accomplished in a defined manner with biomaterials may provide an optimized environment for cell–cell interactions in NKA constructs in vitro or complement/enhance the in vivo regenerative response elicited by the cells. These studies represent preliminary work toward these goals.
Biomaterials, similar to those screened in the studies reported here have been widely leveraged for application in tissue engineering and regenerative medicine (4,6,14,24,36). HA has been shown to direct branching morphogenesis of ureteric buds, induction of EMT, upregulation of aquaporin-2, and differentiation of both metanephric mesenchyme and ureteric buds in vitro in a molecular weight- and concentration-dependent manner (32). PLGA particles have been shown to provide a favorable substrate for the adhesion and proliferation of neural stem cells in vitro, as well as to facilitate the regeneration of neural-like tissue upon directed microinjection into defined lesions created within the brains of rats (5). PCL has been evaluated in multiple tissues, including bone (22,31), the vascular system (27,35), the neural system (41), cartilage (18), and controlled release methodologies (2,25,34). PCL is similar in structure to PLGA, but has a much longer degradation time (36).
To the best of our knowledge, the current study represents the first systematic investigation of the responses of mammalian renal parenchyma to implantation of synthetic and natural biomaterials, both acellular and as bioactive renal cell/biomaterial composites (i.e., NKA constructs). We have applied a combination of in vitro functional assays and in vivo regenerative outcomes to functionally screen candidate biomaterials for potential incorporation into a NKA construct prototype. Implantation of acellular hydrogel-based biomaterials into renal parenchyma (Fig. 1) was typically associated with minimal fibrosis or chronic inflammation and no evidence of necrosis by 4 weeks postimplantation. Moderate cellular/tissue in-growth and neovascularization was observed, with minimal remnant biomaterial. In contrast, implantation of acellular PLGA particles or PCL beads into the renal parenchyma triggered fibrotic encapsulation of the biomaterial, with minimal concomitant neovascularization, cell/tissue infiltration, and neokidney tissue regeneration. These outcomes are confirmed by experiments introducing SRRC seeded and acellular biomaterials into opposing poles of the same kidney (Fig. 6).
These results suggest that despite widespread application of PLGA, PCL, and related polymers in tissue engineering and regenerative medicine (23), no assumptions can be made regarding the potential suitability of any biomaterial for implantation within a given organ. Based on these in vivo data, hydrogel-based biomaterials were selected to produce NKA constructs with which to evaluate in vitro biofunctionality and in vivo regenerative potential. In vitro confirmation of material biocompatibility was provided through live/dead analysis of NKA constructs (Fig. 2). Gelatin-containing hydrogels were associated with robust adherence of primary renal cell populations. Conversely, HA-based hydrogels did not provide a substrate for renal cell attachment, with spheroid formation being triggered instead (Fig. 2D). Neither cell attachment nor spheroid formation was observed upon PCL- or PLGA-based biomaterial, consistent with in vivo data (Fig. 1). Phenotypic and functional analysis of NKA constructs produced from bioactive primary renal cell populations and hydrogel biomaterials was consistent with continued maintenance of a tubular epithelial cell phenotype. Transcriptomic, secretomic, proteomic, and confocal microscopy analyses of NKA construct confirmed no significant differences relative to primary renal cells seeded in 2D culture (Tables 5, 6, Fig. 3). Finally, implantation of hydrogel-based NKA construct into the renal parenchyma of healthy adult rodents was associated with minimal inflammatory and fibrotic response (Figs. 4–6) and regeneration of neokidney-like tissue by 8 weeks postimplantation (Fig. 5).
A detailed analysis of the biotherapeutic efficacy of rodent SRRC populations has been presented elsewhere (17). Direct microinjection of these bioactive renal cell subpopulations in the rat 5/6 nephrectomy model of CKD resulted in extended survival and enhancement of renal function for 6 months posttreatment. That study also presented evidence that a potential mechanism of action (MOA) by which regenerative outcomes in the treated tissue occurred was consistent with mechanistic models currently under consideration for the therapeutic action of adult derived stem cells (8). Such MOAs leverage paracrine factors secreted by bioactive adult-derived cells to catalyze the mobilization of resident, host-derived stem cell populations, to ameliorate fibrosis and inflammation, to facilitate angiogenesis and neovascularization, and to modulate the immune response. Evidence of paracrine effects after primary renal cell-based treatment in a rodent model of progressive CKD of attenuated fibrotic pathways and mobilization of host-derived stem cell populations were presented elsewhere (3,17). Consistent with the models reviewed by Caplan and the experimental evidence that primary renal cells produce factors that can potentially alter the balance between fibrosis and regenerative pathways in diseased kidney tissue are the secretomic profiles of bioactive renal cell populations (Table 5). Both the cells in 2D culture and those in NKA constructs expressed genes and/or proteins of numerous therapeutically relevant cytokines and growth factors, including platelet-derived growth factor (PDGF)-ββ, vascular endothelial growth factor (VEGF), nerve growth factor (NGF), glial cell line-derived neurotrophic factor (GDNF), the matrix metalloproteinases MMP-1, MMP-9, tissue inhibitor of MMPs-1 (TIMP-1), and monocyte chemotactic protein-1 (MCP-1), which have been shown to mediate such paracrine effects (7,10,42).
Studies are ongoing to assess the in vivo responses to NKA construct implantation in mammalian models of progressive CKD. An important contribution to the transition of animal studies to clinical trials is the availability of in vitro assays that are predictive for an in vivo response. To this end, EMT of renal epithelial tubular cell populations is believed to play a significant role in the development of tubulo-interstitial fibrosis during the progression of CKD. Cytokines, including TGF-β, have been shown to recapitulate EMT within populations of tubular epithelial cells (26,43), and TGF-β-induced EMT of the human primary tubular epithelial cell line HK2 is now a well-established model system to evaluate the impact of small molecule and protein factors on EMT (9,13). Conditioned medium derived from cells alone or from gelatin hydrogel- and/or HA hydrogel-based NKA constructs attenuated TGF-β-induced EMT in HK2 cells, with the conditioned medium from gelatin/HA hydrogel-based NKA constructs generating an effect most comparable to bioactive primary renal cells alone (Fig. 7). Acellular hydrogels implanted into healthy rat renal parenchyma were associated with minimal fibrotic and inflammatory outcomes and concomitant neovascularization, cell and tissue infiltration, and degradation of the biomaterial within 4 weeks postimplantation (Fig. 1). Similarly, implantation of hydrogel-based NKA constructs into healthy rat renal parenchyma was well tolerated and associated with minimal fibrosis and inflammation and moderate cellular in-growth and neovascularization at 4 weeks postimplantation (Fig. 4B). By 8 weeks postimplantation, neokidney-like tissue was observed (Fig. 5), providing preliminary evidence that a regenerative response was induced in vivo.
Depending on the regenerative outcomes induced by NKA constructs implanted into kidneys of animals in a progressive disease state, the association of in vitro assays (e.g., the EMT assay presented in this study) with in vivo response has the potential to identify a clinically relevant assay potentially capable of minimally invasive monitoring of patients posttreatment. The strategy used in this study of screening acellular biomaterials to identify well-tolerated formulations, assessing the viability, phenotype, and function of bioactive cell/biomaterial composites in vitro, and then screening the response of healthy kidney to NKA construct prototypes in parallel with in vitro characterization of potential mechanistic pathways represents a systematic and combinatorial approach to identifying and evaluating candidate regenerative treatments, not only for CKD, but as a model that may be applied to other diseases, organs, and tissues.
Taken together, these data provide preliminary evidence that a regenerative response was induced in vivo by NKA constructs. These studies represent the first in vivo, intrarenal investigations of the biological response of mammalian kidney to implantation of a therapeutically relevant primary renal cell/biomaterial composite. Observed results are suggestive that NKA constructs have the potential to both facilitate regeneration of neokidney tissue and attenuate nonregenerative (e.g., reparative healing) responses, thereby guiding the extension of these results to future studies that will evaluate the treatment effect of NKA constructs in established mammalian models of CKD.
Footnotes
Acknowledgment
