Abstract
Since donor T-cells' allorecognition of host antigens is a prerequisite for the onset of graft-versus-host disease (GVHD), blocking their cellular signaling pathways can decrease the severity of GVHD. We hypothesized that epigallocatechin-3-gallate (EGCG), due to its strong affinity to macromolecules, would adhere to surface molecules of donor T cells, inhibit their allorecognition, and attenuate GVHD in the recipient. We tested the hypothesis by treating donor splenocytes with EGCG in both in vitro and in vivo murine GVHD models. EGCG treatment decreased the proliferation of donor cells in MLR cultures and secretion of IL-2 and INF-γ. It also reduced the epitope detection of CD3∊, CD4, and CD28 but did not downregulate the protein expression of these molecules, suggesting blockage of cell surface stimulatory signals. Similarly, EGCG treatment did not decrease mRNA expression for some of these molecules but decreased mitogen-induced cell proliferation, indicating that EGCG did not interfere the transcription of these genes but affected cell proliferation pathways. Furthermore, EGCG-treated donor splenocytes, when transplanted into immunocompromized recipient mice, decreased of proliferation, and the treatment extended the recipients' survival at least during the early stage of GVHD. These results strongly suggest that EGCG attenuates GVHD by both blocking specific cell surface molecules and affecting the donor T-cell proliferation pathways.
Introduction
Graft-versus-host disease (GVHD), in which immunocompetent donor lymphocytes attack recipient tissues after allogenic transplantation, often causes significant morbidity and mortality, particularly in immunocompromised recipients. An ordinary therapeutic remedy for GVHD is the application of immunosuppressive drugs that inhibit donor T-cell activation, which underlies this disease (4,5). Although these pharmacological agents prevent the disease by inhibiting T-cell signaling and activation, they sometimes cause significant side effects on the recipients (2,17,25).
Studies on transplantation medicine have shown that certain plant-derived chemicals can decrease the severity of transplantation-associated problems such as GVHD and graft rejections (8,26,27). Among these phytochemicals, a green tea polyphenol, epigallocatechin-3-gallate (EGCG), has been found to protect certain tissue transplants from allograft rejection. In fact, immersing peripheral nerve tissues in an EGCG-containing solution before transplanting them prevented graft rejection in recipient rats (11).
Because EGCG has a strong affinity to cell surface macromolecules (19) and a potential to impair receptor—ligand interactions (1), it is hypothesized to adhere to the surface molecules of donor T cells and inhibit allorecognition and GVHD in transplant recipients. In our previous study, we found in an in vitro graft rejection model that EGCG treatment of donor splenocytes in mixed lymphocyte cultures significantly reduced recipient T-cell proliferation response (12). In the present study, we tested the immunocamouflage hypothesis by using both in vivo and in vitro murine GVHD models and further examined the influence of EGCG on various parameters of allorecognition. The results demonstrate that EGCG treatment of donor T cells inhibits allorecognition and attenuates the severity of an early stage GVHD in a murine allograft context.
Materials and Methods
Mice
For both in vitro and in vivo studies, female BALB/c (H-2Kd) and C57BL/6 (H-2Kb) mice (Japan SLC, Shizuoka, Japan) between 5 and 11 weeks of age were maintained under specific pathogen-free (SPF) conditions. All experiments were approved by the local review board of Kyoto University and were conducted in accordance with national and international guidelines of laboratory animal care.
EGCG Treatment of Splenocytes
Spleens were aseptically removed from mice, and cell suspensions were prepared by dissecting the spleens with forceps and filtering splenocytes through 50-μm nylon filter mesh in Hanks balanced salt solution [HBSS; +1% fetal calf serum (FCS)]. The cells were centrifuged at 300 × g for 5 min, resuspended in 10 ml RPMI-1640 (Sigma Aldrich, Tokyo, Japan) containing 0, 200, or 400 μM EGCG/107 cells, and incubated at 4°C for 1 h. The cells were washed twice with RPMI-1640 (10% FCS) before being used in experiments.
Mixed Lymphocyte Reaction (MLR)
Splenocytes derived from BALB/c mice were used as “stimulator” cells and those from C57BL/6 as “responder” cells. In both one-way and two-way MLRs, prior to coculture, the responder cells were treated with EGCG as described earlier. In one-way MLR, cells from BALB/c mice were treated with 20 μg/ml mitomycin C (MMC)(MP Biomedicals, Aurora, OH) in RPMI-1640 (10% FCS) at 37°C for 30 min to arrest their proliferation before EGCG treatments. In two-way MLR, both stimulator and responder populations were left capable of responding. Each cell population was resuspended in RPMI-1640 (10% FCS, 100 U/ml penicillin, and 100 μg streptomycin) to a final concentration of 5.0 × 106 cells/ml. The stimulator and responder cells were mixed at a 1:1 ratio (100 μl each) and incubated in triplicate in 96-well plates at 5% CO2 and 37°C for 72 h. Cell proliferation was estimated by counting a small aliquot of cells from each MLR well with a trypan blue exclusion method. After the period of MLR incubation, supernatants from the cultures were assayed for interleukin-2 (IL-2) and interferon-γ (INF-γ) production using cytokine quantification ELISA kits (eBioscience, San Diego, CA) according to the manufacturer's protocol. To examine the toxicity of EGCG on unstimulated cells, C57BL/6 splenocytes were treated with EGCG before culturing in 96-well plates for 72 h, and the proportion of live cells was determined for small samples taken every 24 h.
Flow Cytometric Analysis of Cell Surface Molecules
Flow cytometric analyses of cell surface molecules were performed using a series of fluorescence-labeled monoclonal anti-mouse antibodies and corresponding isotype-matched control antibodies. These antibodies are to detect cluster of differentiation 2 (CD2), CD3∊, CD4, CD11a, CD 28, CD49d (eBioscience) and T-cell receptor (TCR)αβ (BD Biosciences Pharmingen, San Diego, CA). The splenocytes were treated with red blood cell lysis buffer (eBioscience) for 5 min, and the remaining mononuclear cells were treated with EGCG and washed with RPMI (10% FCS) and then with a staining buffer (eBioscience) at 4°C. Each splenocyte sample was reconstituted in a 50-μl aliquot in the staining buffer and subjected to antibody labeling for 15 min at 4°C. After washing the samples twice with the staining buffer, a minimum of 10,000 events per sample was collected and analyzed on a FACScan flow cytometer (Becton Dickinson, Flanklin Lakes, NJ).
Immunoblot Analysis
To examine how EGCG treatment influences the production of cell surface proteins, immunoblot analyses were performed. Splenocytes of C57BL/6 were treated with EGCG in PBS at 4°C for 1 h. The cells were washed once with PBS and incubated in RPMI-1640 (10% FCS, 100 U/ml penicillin, and 100 μg streptomycin) at 37°C for 24 h. Harvested cells were washed once in cold PBS and lysed in RIPA buffer (BD Biosciences Pharmingen) to prepare protein extracts from three treatment groups (i.e., cells treated with 0, 200, or 400 μM EGCG/107 cells). The samples were electrophoresed on a 4—20% sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) gel for 1 h (60 V, 15 mA) and transferred to a nitrocellulose membrane for 1 h (4 V, 20 mA). After the membrane was blocked with a blocking solution (Blocking One, Nacalai Tesque, Japan) at room temperature (RT) for 1 h, primary antibodies against mouse TCRαβ (Santa Cruz Biotechnology, Santa Cruz, CA), CD3∊ (BD Biosciences Pharmingen), CD28 (eBioscience), and CD49d (Cedarlane Laboratories, Ontario, Canada) were applied on the membrane and incubated at RT for 1 h on a swirl shaker. The membrane was washed three times with TBS and 0.5% Tween 20 for 10 min and treated with horseradish peroxidase-conjugated secondary antibodies for 1 h at RT. The three times washed membrane was then treated with Chemi-lumi One Western blotting kit (Nacalai Tesque), and banding images were developed on X-ray films.
RNA Isolation and Semiquantitative RT-PCR
C57Bl/6 splenocytes (5.0 × 106 cells) were treated with 0, 200, and 400 μM EGCG at 4°C for 1 h. After washing with PBS once, total RNA was extracted from the cells by RNeasy Mini Kit (Qiagen, Inc.), and DNA contaminants were removed by RNase-free DNase I Set (Qiagen, Inc.). The RNA (50 ng) was reverse transcribed using Super Script III (Invitrogen, Inc.) with random primers (Invitrogen), and synthesized cDNAs were treated with RNase H (Wako Chemicals Co.) to remove RNA contaminants. PCR was performed with 50 ng cDNA with temperature cycling conditions: one cycle of 95°C for 2 min, followed by 28—40 cycles of 94°C for 30 s, 45—59°C for 30 s, and 72°C for 45 s. The number of cycles and annealing temperatures were adjusted for detecting the amount of amplicons that increased linearly. Primer pairs used to detect murine genes were: CD3∊; (GenBank accession No. NM_007648, amplicon size 235 bp, forward primer, GAT GCG GTG GAA CAC CT, reverse primer, ACT GTC CTC GAC TTC CGA GA); CD4 (GenBank accession No. NM_013488, amplicon size 222 bp, forward primer, AGG AAG TGA ACC TGG TGG TG, reverse primer, TCC TGG AGT CCA TCT TGA CC); CD28 (GenBank accession No. NM_007642, amplicon size 166 bp, forward primer, CGG GAA TGG GAA TTT TAC CT, reverse primer, GTA AGG CGG AGG GTA CAT GA); GAPDH (GenBank accession No. NM_008084.2, amplicon size 223 bp, forward primer, AAC TTT GGC ATT GTG GAA GG, reverse primer, ACA CAT TGG GGG TAG GAA CA). Amplified PCR products were analyzed by gel electrophoresis with 2% agarose gel and 0.001% ethidium bromide.
Mitogen Stimulation
C57Bl/6 splenocytes were treated with EGCG as described earlier, and triplicate cultures of the splenocytes containing 3.0 × 105 cells/ml were incubated for 72 h at 37°C in the presence of phytohemagglutinin (PHA; 0.5 μg/ml; Sigma-Aldrich) or a combination of phorbol 12-myristate 13-acetate (PMA; 50 μg/ml; Sigma-Aldrich) and ionomycin (2 μg/ml; Sigma-Aldrich). Cell proliferation was assessed by counting cells with a trypan blue exclusion method.
In Vivo Murine Experiments
BALB/c mice (5—6 weeks old) and C57BL/6 mice (8—9 weeks old) were used for in vivo experiments. To make immunocompromized animals, BALB/c mice were lethally irradiated (6.3 Gy) by a 137cesium irradiator. These immunocompromized animals were housed in a SPF condition and fed with sterilized water and food ad libitum. Then the animals were intraperitoneally injected with C57BL/6 splenocytes (5 × 106 cells) either untreated or treated with 200 μM EGCG/107 cells. The mice were examined daily for mortality for a period of 17 days. Another set of animals that had been similarly treated was sacrificed on day 7 to examine the proliferation of grafted donor splenocytes. The splenocytes were stained with donor-specific fluorescein isothiocyanate (FITC)-labeled anti-mouse major histocompatibility complex 1 (MHC 1; H-2Kb) monoclonal antibody (Caltag Laboratories, Burlingame, CA) and analyzed on a FACScan flow cytometer.
Statistical Analysis
Statistical analyses were performed on a computer using StatView 5.0.1 (SAS Institute, Cary, NC) and STATISTICA 6.1 (StatSoft, Inc., Tulsa, OK) software for Windows. In most experiments, one-way ANOVA was performed and the significance of differences between groups was examined by Fisher's PLSD test. For analyzing the proportion of live cells from splenocyte cultures with respect to the effects of time and EGCG dosage, two-way ANOVA was performed to calculate the significance of each effect. For the data on mouse survival, Kaplan-Meier survival curves were constructed, and log-rank values were compared between treatment groups.
Results
EGCG Inhibits Murine MLR Activity
We first examined the effects of EGCG treatment of responder T cells on their MLR activity. EGCG treatment of murine splenocytes significantly decreased T-cell proliferation in both one-way and two-way MLR cultures (Fig. 1). The responder cells (C57BL/6, H-2b) without EGCG treatment recognized the stimulator cells (BALB/c, H-2d) as foreign and showed proliferation, but when the responder cells were treated with 200 or 400 μM EGCG/107 cells, the proliferation was attenuated down to about 50% of the untreated control. In two-way MLR, where both responder and stimulator cell populations were capable of proliferation, EGCG treatment attenuated the proliferation down to about 40% of the untreated control.

Effects of epigallocatechin-3-gallate (EGCG) treatment on T-cell proliferation. Data are the mean ± SD cell concentrations for triplicate samples in one-way (hatched columns) and two-way (solid columns) mixed lymphocyte reaction (MLR) cultures. Differences were significant between untreated (0 μM) and treated groups (200 or 400 μM) (*p < 0.01; **p < 0.0001).
Parallel to this attenuation of T-cell proliferation, the production of cytokines was also attenuated. The amount of IL-2 and INF-γ produced in both one-way and two-way MLR cultures significantly decreased when responder cells were treated with EGCG before coculturing with stimulator cells (Fig. 2. To rule out the possibility that the direct toxicity of EGCG on the T cells might contribute to the reduction of T-cell proliferation, EGCG-treated C57BL/6 splenocytes were incubated alone. For all treatments, the cell viability dropped steadily with time, but EGCG treatment slightly increased the viability (Fig. 3).

Effects of EGCG on interleukin-2 (IL-2) (A) and interferon-γ (INF-γ) (B) production. Data are mean ± SD cytokine concentrations for triplicate samples in one-way (hatched columns) and two-way (solid columns) MLR cultures. Differences were significant between untreated (0 μM) and treated (200 or 400 μM) groups for both cytokines in either one-way or two-way MLR cultures (p < 0.0001).
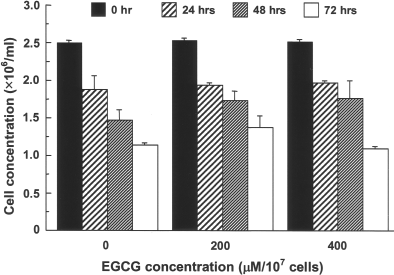
Effects of EGCG on the viability of splenocytes. Splenocytes from C57Bl/6 mice were untreated (0 μM) or treated with 200 or 400 μM EGCG, and the number of live cells was examined after 0, 24, 48, and 72 h of culture (mean ± SD for triplicate samples). The cell viability was significantly higher for 200 μM EGCG than the untreated control (two-way ANOVA, effect of EGCG concentration: p = 0.0038). There was no significant differences between 400 μM EGCG and the untreated control or 200 μM EGCG (p > 0.05).
Reduced Detection of Cell Surface Molecules by EGCG Treatment
To determine the mechanism that EGCG treatment attenuated the MLR activity of T cells, we further analyzed the blocking effects of EGCG on the cell surface molecules of T cells. Antibody-specific detection of epitopes for some cell surface molecules expressed on T cells was reduced by EGCG treatment, but the levels of reduction varied among different cell surface molecules (Table 1). While a moderate to strong reduction was observed for CD3∊, CD4, CD28, and CD49d, no substantial reduction was observed for TCRαβ (Fig. 4, Table 1). Detection of other molecules such as CD2 and CD11a was also reduced but the degree of reduction was lower than that of CD3∊, CD4, CD28, or CD49d (Table 1).
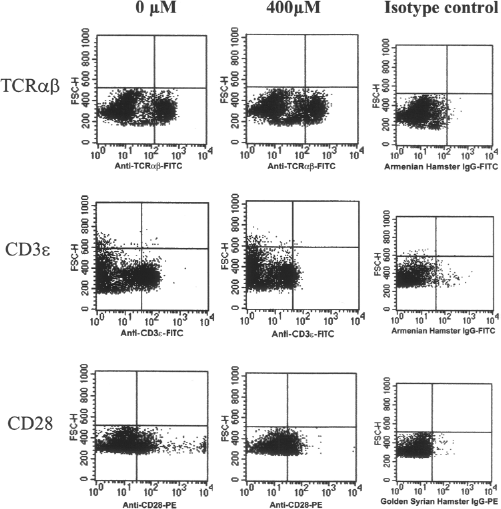
Flow cytometric analysis of cell surface molecules following EGCG treatment. C57Bl/6 splenocytes were either untreated or treated with EGCG (400 μM/107 cells), stained with epitope-specific monoclonal antibodies and analyzed by FACScan. Representative results are shown.
Effect of EGCG Treatment on Detection of Cell Surface Epitopes

Values indicate the mean fluorescence intensity ratio (mean fluorescence of test antibody/mean fluorescence of isotype antibody). Values in parentheses for EGCG-treated triplicate samples (200 and 400 μM/107 cells) represent the average percent reduction in the intensity ratio relative to the untreated control samples (0 μM/107 cells). Differences were significant (*p<0.01) or not significant (p > 0.05) compared with the untreated controls. EGCG, epigallocatechin-3-gallate; TCR, T-cell receptor; CD, cluster of differentiation.
The Production of Cell Surface Molecules Is Not Downregulated by EGCG
To test if EGCG treatment downregulates the production of cell surface molecules of T cells, we further analyzed protein expression for some cell surface molecules by Western blot analysis. The amount of the proteins did not significantly differ between EGCG-treated and untreated splenocytes (Fig. 5). This was apparent not only for TCR (Fig. 5A), but also for other molecules such as CD3∊, CD28, and CD49d (Fig. 5B—D).

Effects of EGCG on the production of cell surface molecules in splenocytes. C57Bl/6 splenocytes were either untreated or treated with EGCG (200 and 400 μM/107 cells) and subjected to Western blot analyses for the expression of T-cell receptor (TCR)β (A), cluster of differentiation (CD)3∊ (B), CD28 (C), and CD49d (D). Densitometric analysis of individual values was presented as relative intensity against β-actin. Data are mean ± SD density ratios for triplicate samples, and the differences between treated and untreated groups for any of these molecules were not significant (p > 0.05).
EGCG Does Not Reduce the mRNA Expression of Cell Surface Molecules
To examine if EGCG affects the cell surface molecules at a transcription level, mRNA expression was analyzed for CD3∊, CD4, and CD28, whose epitope detection was reduced by more than 35% by 200 μM EGCG. The results indicated that EGCG treatment did not significantly change the gene expressions, suggesting that it did not attenuate the transcription of these genes (Fig. 6).
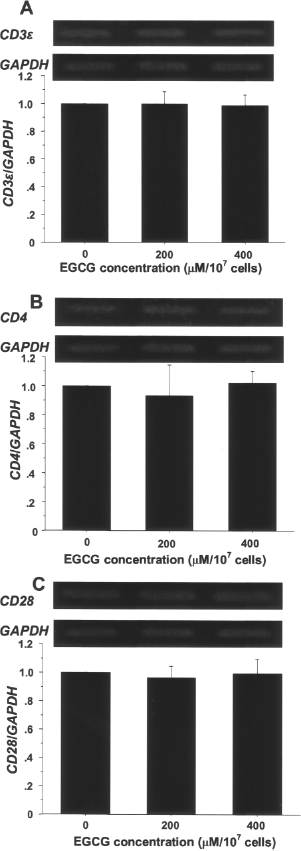
Effects of EGCG on mRNA expressions of CD3∊, CD4, and CD28 in splenocytes. C57Bl/6 splenocytes were either untreated or treated with EGCG (200 and 400 μM/107 cells) and subjected to the mRNA analysis for CD3∊; (A), CD4 (B), and CD28 (C). Densitometric analysis of individual values for EGCG-treated samples in three independent experiments is presented. Data are mean ± SD density ratios for triplicate EGCG-treated samples relative to control, and the differences between treated and control groups for any of these signals were not significant (p > 0.05).
Proliferation Arrest in Mitogen-Stimulated Cells by EGCG
To test if EGCG also inhibit the proliferating effects of exogenous mitogens, cell proliferation was assayed on mitogen-stimulated splenocytes. Results indicated that the proliferation of EGCG-treated cells after the stimulation with T-cell mitogens, PHA, or a combination of PMA and ionomycin significantly decreased (Fig. 7).
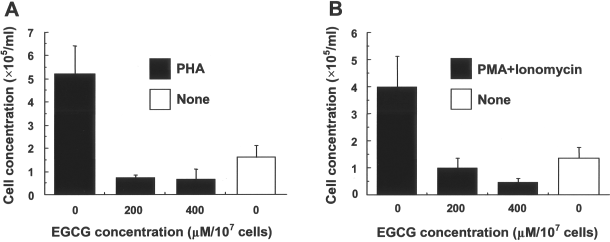
Effects of EGCG on proliferative capacity of mitogen-stimulated splenocytes. C57Bl/6 splenocytes were either untreated or treated with EGCG (200 and 400 μM/107 cells), and then stimulated with phytohemagglutinin (PHA) (A) or a combination of phorbol 12-myristate 13-acetate (PMA) and ionomycin (B), and cell numbers were counted 72 h after cultures. Open columns indicate the response without mitogen. Data are mean ± SD cell concentrations for triplicate samples. (A) Differences were significant between untreated (0 μM) and treated (200 or 400 μM) groups (p < 0.0001) or no mitogen group (p < 0.0002). (B) Differences were significant between untreated and treated groups (p < 0.0004) or no mitogen group (p < 0.0008).
EGCG Treatment Moderately Improved Survival of GVHD Mice
We next investigated the effects of EGCG treatment of donor cells on the survival of GVHD-induced mice. The injection of untreated splenocytes resulted in a 50% survival time of 8 days, while that of EGCG-treated splenocytes extended the survival time to 10 days (Fig. 8A). In groups of immunocopromized mice that were injected with donor splenocytes without EGCG treatment, approximately 18% of the total splenocytes were H-2Kb-positive donor-derived cells (Fig. 8B). In contrast, in mice injected with EGCG-treated splenocytes, only 5% were donor-derived cells.

Survival curve and donor T-cell proliferation in graft-versus-host disease (GVHD)-induced recipient mice. EGCG-treated or untreated C57Bl/6 splenocytes [major histocompatibility complex 1 (MHC 1); H-2Kb) were injected into irradiated BALB/c recipient mice (H-2Kd). (A) Survival curves of recipient mice. Differences were significant between EGCG-treated and control groups (χ2 = 10.2, p = 0.0014). (B) Percent donor T cells (H-2Kb) (mean ± SD) in irradiated recipient mice (H-2Kd) (n = 8/group) assessed 7 days after injection with EGCG-treated or untreated splenocytes (control). Differences were significant between EGCG and control based on unpaired Student t-test at p < 0.0001.
Discussion
In this study, we have shown that EGCG can attenuate allorecognition and severity of an early stage GVHD in both in vitro and in vivo murine allograft contexts. The EGCG treatment of murine splenocytes decreased their proliferation upon stimulation by either coculturing them with stimulator cells in MLR or injecting them into MHC-disparate mice.
To reduce allogenic responses such as GVHD and transplant rejection, tissue matching and pharmacological interventions are employed, but these methods often fail due to the limited availability of tissue grafts in general and side effects caused by the therapeutic agents (2,4,25), respectively. As an alternative method, immunocamouflage, has been attempted, where a donor tissue with its intact cell surface antigenic determinants is coated with nonimmunogenic compounds such as polyethylene glycol (PEG) to block recognition by the host immune system (21,22). Indeed, PEG coating of donor tissues is known to block allorecognition of T cells and prevent GVHD induction (6). In the present study, we hypothesized that a tea polyphenol, EGCG, when applied to donor cells, due to its high affinity for macromolecules, would block T-cell surface allorecognition molecules and decrease the occurrence of GVHD.
EGCG treatment of responder cells in MLR significantly decreased T-cell proliferation (Fig. 1) and the production of IL-2 and INF-γ, two major indicators for T-cell activation and proliferation (Fig. 2). The overall decrease of proliferating T cells was not due to the direct toxicity of EGCG because EGCG-treated cells showed no significant mortality relative to untreated cells (Fig. 3).
Our in vivo experimental results indicated that EGCG treatment moderately extended survival time for the mice injected with EGCG-treated splenocytes compared with those injected with untreated splenocytes (Fig. 8). EGCG treatment did not completely prevent GVHD-enhanced lethality in mice, but it helped them tolerate the high-dose challenge with the splenocytes (i.e., a 2.5x greater cell concentration than what has been reported to induce GVHD in healthy humans). Parallel to the survival experiments, the decreased donor splenocyte population in the recipient mice reflected the attenuating effect of EGCG on its proliferation.
What are the possible mechanisms behind these attenuating effects of EGCG on GVHD? One explanation is a blocking action of EGCG toward T-cell surface molecules as postulated in our present hypothesis. The detection of cell surface molecules involved in allorecognition (CD3∊), costimulations (CD4, CD28), and cell-to-cell adhesion (CD49d) was indeed decreased by EGCG treatment (Table 1). The blocking action was further supported by the immunoblot data for some of these signal molecules, whose production was not significantly influenced by EGCG treatment (Fig. 5). In addition, mRNA expressions of some of these surface molecules were similarly not attenuated by EGCG treatment (Fig. 6), which supports the hypothesis that EGCG camouflages the surface molecules, although EGCG is known to translocate into the nucleus and influence the expression of many genes particularly related to cell proliferation (3).
The weakening of the cell-to-cell signaling through these molecules could lead to incomplete activation of T cells, thereby decreasing GVHD induction (6). While different levels of blocking effects were exhibited among these cell surface molecules (Table 1), the expression of TCR remained essentially unaffected by EGCG treatment, suggesting that its receptor function is expected to be largely intact. In contrast, there was a strong attenuation effect on CD3∊, which is involved in transmitting intracellular stimulatory signals (18). Attenuation occurred also for the costimulatory molecules, CD4 and CD28, which can abrogate costimulatory signals (7,9,14,23,24). These attenuation effects of EGCG are different in levels compared with those of other surface molecules, suggesting that EGCG treatment possibly leads to incomplete stimulation of T cells (16,20). Although cell surface molecule blockage is likely to be a primary effect of EGCG, allorecognition ability of the donor T cells can be restored over time. This is predicted because the EGCG treatment did not significantly influence the gene expression for these molecules. As EGCG will be diluted in vivo over time, newly synthesized molecules will eventually replace the EGCG-attenuated ones; therefore, EGCG is most likely to affect the early stage of GVHD. Thus, future studies need to analyze the detailed dynamics of the cell surface molecules as to how EGCG interacts with the receptors and whether they may be subsequently internalized into the cells under both in vitro and in vivo environments.
EGCG is also a well-known antiproliferating agent that downregulates some of the key intracellular pathways in cell cycle (3,10,15). EGCG treatment conditions and duration used in the present study can arrest cell cycle. Indeed, EGCG treatment significantly reduced mitogen-induced T-cell proliferation (Fig. 7), suggesting that it also interfered with proliferation pathways independent of signals through cell surface molecules. EGCG may influence the production of a number of cytokines that activate not only T cells but also regulatory T cells, the possibility of which needs to be examined in future studies (13).
Treating tissue and/or cell transplants with EGCG can be an inexpensive method to attenuate the responses of early stage GVHD. It can be integrated into clinical transplantation, where a transplant is immersed in a physiological solution containing EGCG before being transplanted to a human recipient. Although further study is necessary to understand the exact mechanisms of the immunosuppressive actions and acquire practical application strategies to prolong the actions, the EGCG treatment will have a potential value to control GVHD.
Footnotes
Acknowledgments
This work was funded by the grants from the Seeds Innovation Research Program by Japan Science and Technology Agency and Grant-in-Aid by the Ministry of Education Science and Sports in Japan. The authors are grateful to Mr. Noriaki Kitazumi for technical assistance. The authors declare no conflict of interest.
