Abstract
Natural regulatory T cells (Tregs) may have a great therapeutic potential to induce tolerance in allogeneic cells and organ transplantations. In mice, we showed that alloantigen-specific Tregs (spe-Tregs) were more efficient than polyclonal Tregs (poly-Tregs) in controlling graft-versus-host disease (GVHD). Here we describe a clinical-grade compliant method for generating human spe-Tregs. Tregs were enriched from leukapheresis products with anti-CD25 immunomagnetic beads, primed twice by allogeneic mature monocyte-derived dendritic cells (mDCs), and cultured during 3 weeks in medium containing interleukin 2 (IL-2), IL-15, and rapamycin. After 3 weeks of culture, final cell products were expanded 8.3-fold from the initial CD25+ purifications. Immunophenotypic analyses of final cells indicate that they were composed of 88 ± 2.6% of CD4+ T cells, all expressing Treg-specific markers (FOXP3, Helios, GARP, LAP, and CD152). Spe-Tregs were highly suppressive in vitro and also in vivo using a xeno-GVHD model established in immunodeficient mice. The specificity of their suppressive activity was demonstrated on their ability to significantly suppress the proliferation of autologous effector T cells stimulated by the same mDCs compared to third-party mDCs. Our data provide evidence that functional alloantigen Tregs can be generated under clinical-grade compliant conditions. Taking into account that 130 × 106 CD25+ cells can be obtained at large scale from standard leukapheresis, our cell process may give rise to a theoretical final number of 1 × 109 spe-Tregs. Thus, using our strategy, we can propose to prepare spe-Tregs for clinical trials designed to control HLA-mismatched GVHD or organ transplantation rejection.
Keywords
Introduction
Graft-versus-host disease (GVHD) is one of the major complications of allogeneic hematopoietic stem cell transplantation (HSCT). This life-threatening complication is due to the fact that donor T cells, present within the transplant, recognize recipient major and/or minor histocompatibility antigens, become activated, and attack target organs of GVHD. Although a multiplicity of nonspecific immunosuppressive agents, such as calcineurin inhibitors and steroids, have been extensively used to diminish the intensity of GVHD, more than 50% of severe GVHD patients are refractory to these current therapies (36). Moreover, the long-term use of such agents impairs immune reconstitution and diminishes T-cell-mediated graft-versus-leukemia (GVL) responses. For these reasons, the development of specific cell-based therapies may offer alternative approaches to prevent and/or to control severe GVHD.
Natural regulatory T cells (Tregs) are defined by constitutive expression of high levels of the transcription factor forkhead box P3 (FOXP3), which is critical for their development and suppressive function (4,17,24,28), high levels of the interleukin 2 (IL-2) receptor α chain [cluster of differentiation 25 (CD25)], and, in humans, low or no expression of the IL-7 receptor α chain (CD127) (33,47). Tregs represent, in mice and humans, a small population of CD4+CD25+ cells (3-5% of total CD4+ T cells) that play a key role in the control of autoreactive T cells (26,43) and possibly in the protection of fetuses during pregnancy in mammals (3,12). Interestingly, using a murine model of GVHD, we and others have demonstrated that cotransferred donor Tregs with effector T cells (Teffs) dramatically suppress GVHD without impairing engraftment, GVL effect, and response against third party of alloantigens (15,25,49,53). Of note, a better effect was observed when using Tregs specific for recipient alloantigens compared to polyclonal Tregs (10,53,54). In patients, Treg-based therapy for GVHD has been reported using freshly isolated Tregs (13,55) or Tregs from umbilical cord blood isolated with anti-CD25 immunomagnetic beads and expanded using anti-CD3-and anti-CD28-conjugated beads (9). Different attempts have been carried out to generate in vitro allogeneic-specific Tregs (spe-Tregs). Recently, Tregs have been cocultured in vitro with allogeneic dendritic cells (DCs) (57) or CD40 ligand-activated B cells (40) and selected by fluorescence-activated cell sorting (FACS) on either the expression of CD4+CD127low/-CD25+(40) or activation markers such as CD69 and CD71 (41) or CD137 (44), or on the basis of their cell division measured by carboxyfluorescein succinimidyl ester (CFSE) loss (30,58). Although all these reports have provided evidence that human spe-Tregs can be generated in vitro, the methods used to either select (FACS selection) and/or expand such spe-Tregs are not based on the use of clinically approved methods and reagents according to the European regulations.
In the present study, we successfully designed a strategy to obtain functional spe-Tregs using an initial immunomagnetic CD25+ selection of Tregs, followed by their priming with allogeneic monocyte-derived DCs and in vitro expansion. Our data were generated with clinical-grade reagents allowing then to scale-up our findings in view of future clinical trials.
Materials and Methods
Human Samples and Ethical Approval
Leukapheresis samples were freshly collected from 37 healthy donors (13 females and 24 males, median age 39.5 years; range: 22-65) at Pitié Salpêtrière hospital (Etablissement Français du Sang, Paris, France). Approval for these studies was obtained from the institutional review boards of the Université Pierre et Marie Curie and the Centre National de la Recherche Scientifique. Written informed consent was provided according to institutional guidelines and to the Declaration of Helsinki.
Generation of DCs
Monocytes were enriched from the adherent fraction of mononuclear cells (MNCs) (32) or purified using CD14 immunomagnetic beads (Miltenyi Biotec, Paris, France) as previously described (14). DCs were generated from monocytes using good manufacturing practice (GMP)-grade cytokines and reagents and an already approved procedure for DC-based clinical trials (46). Briefly, monocytes were cultured for 8 days in RPMI medium (Invitrogen Corporation, Paris, France) containing 10% human AB serum (hSAB purchased from Etablissement Français du Sang, Paris, France), 2 mM L-glutamine, 100 U/ml penicillin, and 100 ug/ml streptomycin (all from Invitrogen), 1,000 IU/ml granulocyte macrophage colony-stimulating factor (Gentaur, Kampenhout, Belgium), and 200 IU/ml IL-4 (Miltenyi Biotec) in order to generate immature DCs (iDCs) as previously described (14). Mature DCs (mDCs) were obtained using a slightly modified method described by Feuerstein et al. (16). Briefly, tumor necrosis factor (TNF)-α (10 ng/ml; Peprotech, Neuilly, France), JL-1β (100 UI/ml; Invitrogen), IL-6 (1,000 UI/ml; Peprotech), and prostaglandin E2 (PGE-2; 1 μg/ml, Prostin® E2; Pfizer, Paris, France) were added to the culture at day 6. At day 8, iDCs and mDCs were harvested, characterized, and frozen until use. The maturation status of mDCs, compared to iDCs, was assessed by flow cytometry on the expression of human leukocyte antigen (HLA)-DR, CD40, CD80, CD83, and CD86 (data not shown) as previously described (14).
Selection of Tregs
Tregs were enriched from MNCs either by FACS (FACS Aria®; BD Biosciences, Pont de Claix, France) or by magnetic-activated cell sorting (MACS; Miltenyi Biotec). For FACS purification, CD25+ cells were selected by MACS using anti-CD25 microbeads (Miltenyi Biotec), then stained with CD4, CD25, CD127, and CD8 directly labeled monoclonal antibodies (mAbs) according to the manufacturer's instructions (see Table 1) and sorted into CD4+CD25highCD127low/-CD8- cells. Thus, highly purified Tregs (>99%) were obtained. For MACS purification, Tregs cells were obtained by using anti-CD25 microbeads (Miltenyi Biotec), and three consecutive magnetic selections were applied to the cells to reproduce the 3.1 enrichment-selective program performed at large scale with the CliniMACS device (Miltenyi Biotec) as described elsewhere (20).
List of Monoclonal Antibodies
CD3, cluster of differentiation 3; PerCP, peridinin–chorophyll–protein complex; FITC, fluorescein isothiocyanate; APC, allophycocyanin; PE, phycoerythrin; HLA, human leukocyate antigen; FOXP3, forkhead box P3; GARP, glycoprotein A repetitions predominant (garpin); LAP, latency-associated peptide.
Cell Division Kinetics of Tregs
FACS-sorted Tregs (0.5-5 × 106 cells) were labeled with 1 μM CFSE (Invitrogen). The CFSE-labeled Tregs were cocultured with either allogeneic mDCs or iDCs at a 10:1 ratio. Cocultures were performed in complete culture medium that corresponds to Roswell Park Memorial Institute (RPMI)-1640 medium containing 10% hSAB, 2 mM L-glutamine, 100 U/ml penicillin, and 100 ug/ml streptomycin (Invitrogen). Tregs were stimulated by IL-2 (600 UI/ml, Proleukin®; Novartis, Rueil-Malmaison, France), or IL-15 (10 ng/ml; Miltenyi Biotec), or by both. As control, CFSE-labeled Tregs were also polyclonally stimulated in the presence of anti-CD3/anti-CD28-coated Dynabeads (Invitrogen) and IL-2 (see below). Cells were daily collected from day 3 to day 6, and cell division-based CFSE dilution was monitored by flow cytometry.
Culture of Poly-Tregs and Allo-Tregs
Poly-Tregs were cultured as described elsewhere (20). Briefly, MACS-sorted Tregs seeded at 5 × 105 cells/ml in complete culture medium containing 100 μxM rapamycin (Sigma-Aldrich, Saint Quentin Fallavier, France) and 600 UI/ml IL-2 were stimulated by anti-CD3/anti-CD28-coated Dynabeads (Invitrogen) at day 0 and day 15 of culture, cultures being maintained until day 21.
Spe-Tregs were prepared as follows. MACS-sorted Tregs were seeded at 106 cells/ml in complete culture medium containing 100 μM rapamycin, 600 UI/ml IL-2, 10 ng/ml IL-15, and cultured with 105 allogeneic mDCs. Ten days later, medium was changed, and Tregs were restimulated with the same source of allogeneic mDCs at the same ratio and expanded under the same culture conditions until day 21. In some experiments, CD4+CD25-CD127+ Teff cells were purified by FACS from the CD25- cell fraction and used as controls. Teffs were cultured using either the same antigen-specific stimulation as for spe-Tregs or under polyclonal activation in order to generate spe-Teffs and poly-Teffs, respectively. For all culture conditions, medium changes were performed every 3 days. At the end of culture, CD3/CD28 Dynabeads were removed with a magnet (Invitrogen); immunophenotypic and functional characterizations of Tregs were then performed.
Antibodies and Flow Cytometry Analysis
The list of murine anti-human mAbs directly conjugated to fluorescein isothiocyanate, phycoerythrin (PE), PE-cyanine 7 (PE-Cy7), allophycocyanin, and peridinin–chorophyll–protein complex is described in Table 1, and the mAbs were used according to the manufacturer's instructions. Cell surface staining and intracellular staining (FOXP3 and Helios) were performed according to the manufacturer's instructions. Matched mouse isotype mAbs were used as negative controls. Flow cytometry analyses were performed on either a FACSCalibur® or a LSR II® (both from BD Biosciences). Data were analyzed with FlowJo® software (Treestar, Ashland, OR, USA).
Suppression T-Cell Assay
The suppressive activity of Tregs was assayed in round-bottomed 96-well tissue culture plates (Corning, Avon, France) before and after culture. After culture, CD3/CD28 Dynabeads were removed, cells were washed twice in phosphate-buffered saline, and rested in RPMI-1640 medium containing 10% hSAB and IL-2 (600 UI/ml) for 24 h before performing the suppression assay. Then Tregs were mixed at various cell ratios (1:1; 1:2; 1:4; 1:8; 1:16, 1:32, 1:64, 1:128) with 5 × 104 autologous CD4+CD25- cells (responder cells) in the presence of 104 stimulator cells corresponding to either the original DCs used to expand spe-Tregs (DCs referred to as from donor a, DCa) or another batch of allogeneic DCs, used as a third party (DCs referred to as from donor b, DCb) in RPMI medium containing 10% hSAB. Autologous CD4+CD25- cells were obtained by depleting CD25+ cells from MNCs using 10 μl of CD25 microbeads per 107 cells and followed by CD4+ cell selection using the untouched CD4+ T-cell Isolation kit II (Miltenyi Biotec). CD4+CD25- cells were used immediately or frozen in liquid nitrogen until use. After 4 days, cell proliferation was determined by incorporation of 2 μCi (0.037 MBq) of tritiated thymidine ([3H]TdR; Amersham, Little Chalfont, Buckinghamshire, UK) for 16 additional hours and measured using a ß-counter (counter-WALLAC, Ramsey, MN, USA). Results expressed in counts per minute (cpm) represent the mean ± SEM of triplicates.
Quantitative PCR of FOXP3 Demethylation
The percentage of the Treg-specific demethylated region (TSDR) within the FOXP3 locus in spe-Tregs and poly-Tregs after culture was measured by Epiontis GmbH (Berlin, Germany), as a subcontractor, using quantitative PCR (qPCR) as previously described (60). As controls, the percentage of the TSDR was measured on cultured spe-Teffs and poly-Teffs. Briefly, genomic DNA was isolated using the DNeasy blood and tissue Kit (Qiagen, Valencia, CA, USA). After bisulfite conversion of genomic DNA, PCR products were generated with methyl- and non-methyl-specific primers for FOXP3 TSDR. Comparison of the number of demethylated TSDR to methylated TSDR copies allows an accurate measurement of Tregs (60).
Murine Experiments
Twelve- to 20-week-old nonobese diabetic/severe combined immunodeficient/IL-2 receptor γ chain (NOD/SCID/γC-/-; NSG) male mice (NOD.Cγ-Prkdcscid IL2rgtm1Wjl/Sz, stock 61/4 005557; The Jackson Laboratory, Bar Harbor, ME, USA) were bred in our own animal facilities in specific pathogen-free conditions (accreditation number from the Veterinary services: A75-13-10) with an enriched fat regimen and addition of Bactrim (Roche, Boulogne-Billancourt, France) in drinking water every other week. The colony was regularly checked for γ-c deficiency by PCR according to the Jackson Laboratory protocol. Experiments were performed according to the European Union guidelines and approved by our institutional review board (CRRA Ile de France No. 3).
Xenogeneic GVHD Model in NSG Mice and Control by Tregs
To induce a xenogeneic GVHD, human CD3+ T cells were first purified using the CD3 microbeads-II kit (Miltenyi Biotec). The CD3+ T cells (107 cells/mouse) were intravenously injected into NSG mice in the presence of allogeneic DCs (2 × 106 cells/mouse). The latter are helpful to enhance the GVHD clinical signs and to accelerate the mortality, compared to our previously published xeno-GVHD model (51). Under these conditions, NSG mice developed a xenogeneic GVHD within 4-5 weeks. In order to evaluate the ability of spe-Tregs or poly-Tregs to control xenogeneic GVHD, spe-Tregs or poly-Tregs (5 × 106/mouse) were concomitantly injected with autologous CD3+ T cells referred to as Teffs. Mouse survival and symptoms of GVHD including hunched back, diarrhea, and body weight were monitored daily. Mice were sacrificed when they lost ≥30% body weight. Each group contained three mice.
Histopathological Examination
Livers, lungs, and skin were preserved in Bouin's fixative (Polysciences, Inc., Europe GmbH, Eppelheim, Germany) and embedded in paraffin. For these organs, 3-μm-thick sections were stained with hematoxylin and eosin (Sigma-Aldrich) for histological examination. One pathologist analyzed slides in a blinded fashion to assess the intensity of GVHD. GVHD lesions were scored in each liver sample according to a semiquantitative scoring system (22) using seven parameters (inflammatory cell infiltrate within the portal tracts, bile duct injury, venous endothelialitis, periportal hepatocyte necrosis, acidophilic bodies, confluent lobular necrosis, sinusoidal lymphocytosis). The total score was the sum of those parameters, which were scored as follows: 0 as normal, 1 as focal and rare, 2 as focal and mild, 3 as diffuse and mild, 4 as diffuse and moderate, and 5 as diffuse and severe. GVHD lesions in the lung were evaluated as previously described (11) according to the following score, which is the sum of a periluminal (around airways and vessels) lymphocytic infiltrate index (0: absence; 1: lymphocytic cuff of 1-3 cells in diameter; 2: lymphocytic cuff of 4-10 cells in diameter; 3: lymphocytic cuff >10 cells in diameter), an alveolar/interstitial lymphocytic infiltrates index (0: absence; 1: increased number of cells only visible at high magnification; 2: interstitial thickening; 3: interstitial thickening and alveolar infiltrates), and an index of lung injury extension quantitated according the percentage of tissue involved (1: 5-25%, 2: 25-50%, 3: >50%). GVHD lesions in the skin were evaluated according to the sum of three parameters as described by Kaplan et al. (27) that are vacuolization of the basal layer of the epidermis, necrotic keratinocytes, and lymphocytic infiltrate within the dermis. Each of those parameters was scored as follows: 0 as normal, 1 as mild, 2 as moderate, 3 as severe.
Statistical Analysis
When data were compared according to purification methods from different leukapheresis samples, statistical analyses were performed using the unpaired Student's t-test with Welch's correction when appropriate. Data were analyzed with GraphPad Prism software (GraphPad Software, Inc., San Diego, CA, USA), and expressed as mean ± SEM. Kaplan–Meyer survival curves were constructed, and log-rank comparison of the groups was used to calculate p values. Statistical significance was taken at the 5% level (p < 0.05), the degree of significance was indicated as: p < 0.05, p < 0.01, p < 0.001.
Results
Determination of Culture Conditions for the Activation and Stimulation of Treg Cell Division
We first studied different conditions for the emergence and/or activation of alloantigen-specific Tregs from a polyclonal Treg population. As iDCs may be more tolerogenic than mDCs (30), we compared their ability to activate Tregs. CD4+CD25highCD127low/- Tregs were purified by FACS (purity > 99%) and labeled with CFSE. Then Tregs were cocultured with either iDCs or mDCs in complete culture medium containing IL-2, IL-15, or both cytokines. Indeed, IL-15, whose α chain receptor is expressed by activated Tregs (62), enhances Treg survival, FOXP3 expression, and Treg suppressive function particularly when associated with rapamycin (58). As positive control, CFSE-labeled Tregs were also polyclonally activated with anti-CD3/anti-CD28 Dynabeads and IL-2.
Figure 1A shows the cell proliferation of Tregs quantified by CFSE staining dilution. Early dividing cells were detected as soon as 3 days of stimulation in all conditions. However, mDCs exhibited higher ability to trigger Treg cell divisions than iDCs, whatever the cytokines used in the culture medium. Treg activation was further documented by FACS analysis based on FOXP3 intensity assessed on mean fluorescence intensity and percentage (Fig. 1B). FOXP3 expression was upregulated between day 0 and day 6 in all conditions. When Tregs were cocultured with mDCs, IL-2 and IL-15 were able to upregulate FOXP3 expression at comparable levels, while combination of IL-2 and IL-15 strongly enhanced FOXP3 expression. When Tregs were cocultured with iDCs, FOXP3 was not so well maintained. Furthermore, IL-15 alone or combined with IL-2 was not as good as IL-2 alone to preserve FOXP3 expression. Surprisingly, FOXP3 expression was also maintained in the presence of mDCs and even iDCs without adding cytokines. This can be explained by the production of low amounts of IL-2 by DCs as already reported by others in mice and humans (19,37,61). For further experiments, these findings were taken into account, and alloantigen-specific Tregs were cultured in the presence of mDCs, IL-2, and IL-15.

Culture conditions for the activation and cell division of Tregs. FACS Tregs were labeled with CFSE and stimulated with allogeneic iDCs or mDCs in the presence of IL-2, IL-15, or both cytokines. mDCs were obtained by adding 10 ng/ml TNF-α, 100 UI/ml IL-1β 1,000 UI/ml IL-6, and 1 μg/ml PGE-2 at day 6 of culture. As control, CFSE-labeled Tregs were polyclonally stimulated with anti-CD3/anti-CD28-coated Dynabeads and IL-2. (A) Proliferation was daily monitored on the basis of CFSE dilution and analyzed by flow cytometry from day 3 to day 6. Data presented as CD4/CFSE dot plots gated on CD3+CD4+ cells are representative of three separate experiments. (B) FOXP3 expression of Tregs was monitored by flow cytometry at day 0, day 3, and day 6. Histograms show FOXP3 expression (filled curves) presented as percentage and mean of fluorescence intensity (in brackets), and isotype controls (black lines). Data are representative of three separate experiments.
Generation of Attoantigen-Specific Tregs Under Clinically Compliant Conditions
For clinically compliant conditions, we designed a protocol where Tregs were purified by MACS based on CD25+ selection using CD25-magnetic beads as previously described (20). Since preliminary data, also confirmed by others (39), indicated that two allogeneic stimulations would be preferable to a single one in order to enrich antigen-specific Tregs, MACS-purified Tregs were then stimulated twice (at day 0 and day 10) with allogeneic mDCs. Tregs were cultured in complete culture medium supplemented with IL-2, IL-15, and rapamycin up to day 21. As a negative control of specificity, MACS-purified Tregs were generated under polyclonal stimulation (poly-Tregs) as previously described (20). Treg expansion under polyclonal or antigen-specific stimulation is shown in Figure 2A. While poly-Tregs were expanded 39.6 ± 10.5-fold after 21 days of culture, the number of spe-Tregs obtained at 3 weeks was only 8.3 ± 0.1-fold the initial number of Tregs at the onset of cultures. Interestingly, large-scale experiments performed in our clinical cell therapy center using two blood mass leukapheresis products obtained from healthy donors allowed us to obtain ~130 × 106 cells containing ~70-80% of CD4+CD25highFOXP3+ Tregs after CD25+ selection using the CliniMACS® device (Miltenyi Biotec). With eightfold expansions of the initial number of Tregs, we would be able to produce ~1 × 109 spe-Tregs.

Cell expansion and functional study of spe-Tregs and poly-Tregs. (A) Cell expansion of spe-Tregs and poly-Tregs after 21 days of culture. MACS Tregs (106 cells) were seeded in culture and expanded either under polyclonal stimulation or under alloantigen stimulation using allogeneic mDCs as described in the Materials and Methods section. Data from four independent experiments performed with Tregs from four different healthy donors are shown. (B) Suppressive activity of spe-Tregs and poly-Tregs on the proliferation of autologous CD4+CD25- T cells stimulated by allogeneic mDCs after 21 days of culture. Tregs generated from MACS-sorted CD25+ cells were expanded under alloantigen stimulation in the presence of allogeneic mDCs (donor a, DCa, left panels) or under polyclonal stimulation (right panels). For suppressive activity, spe-Tregs or poly-Tregs were mixed at different ratio with autologous effector cells and stimulated either with the same DCa () or a third party of allogeneic mDCs (referred to as donor b, DCb,) for 4 days. Cell proliferation was determined by incorporation of tritiated thymidine ([3H]TdR) and percentage of suppression calculated as follows: % suppression = [1 - (Tregs + Teffs + DC/Teffs + DC)] × 100. Results are presented as the mean ± SEM of six separate experiments for left panels and of four to five separate experiments for right panels. Statistical analysis was performed using an unpaired Student t-test: **p < 0.01.
Functional analysis of MACS-purified Tregs expanded under specific conditions showed that spe-Tregs exhibited a stronger and significant suppressive activity, particularly at low Treg/Teff ratio (p < 0.001), when stimulated by DCa initially used to prime the Tregs compared to a third-party DC (DCb) stimulation (Fig. 2B, left panel). Interestingly, DCa-stimulated Spe-Tregs only began to decrease their suppressive function below a 1:32 Treg/Teff ratio and were still suppressive at a 1:128 Treg/Teff ratio compared to DCb-stimulated Spe-Tregs (data not shown). As expected, poly-Tregs stimulated by DCa or by DCb exert a comparable suppressive activity (Fig. 2B, right panel). These data demonstrate that the initial population of MACS-sorted Tregs, which was polyclonal, can be enriched into spe-Tregs after two rounds of stimulation by allogeneic mDCs. It is important to point out that we have used clinical-grade reagents for the production of spe-Tregs, and subsequently, our culture conditions can be easily scaled up in view of clinical applications.
Immunophenotypic Characterization of Alloantigen-Specific Tregs
The strategy we set up to generate spe-Tregs was based on the purification of Tregs using anti-CD25 immunomagnetic beads. In the initial mononuclear cell fraction, Tregs defined as CD4+CD25highCD127low/- cells or CD4+CD25highFOXP3+ cells represented 3-4<% of CD3+CD4+ cells (Table 2), and CD3+CD4+ cells represented 59.5 ± 4.6% of total MNCs.
Identification of Tregs in CD3+CD4+-Gated Cells Before and After CD25+ Selection by MACS

Data presented are means ± SEM of at least nine separate experiments. Tregs, regulatory T cells; MACS, magnetic-activated cell sorting; MNCs, mononuclear cells.
After CD25+ selection, CD25+ cells represented 95.5 ± 0.9% of total cells and contained 91.7 ± 3.6% of CD3+CD4+ cells. Contaminating cells were B cells (CD19+ cells, 5.3 ± 0.7%), natural killer cells (CD3-CD56+ cells, 0.7 ± 0.2%), CD8+ cells (1.4±0.3%), and monocytes (CD14+ cells, 2.1 ± 0.2%). Tregs (CD4+CD25highCD127low/- cells or CD4+CD25high FOXP3+ cells) within CD4+CD3+ cells represented 74-79% of the cells (Table 2). Helios has also been described as a transcription factor expressed by natural Tregs (18,50). We found that CD4+FOXP3+Helios+ cells represented before and after CD25+ enrichment 3.3 ± 0.4% and 69.7 ± 1.3% of the cells, respectively (Table 2). Interestingly, 88.3 ± 1.6% of CD4+CD25high were CD127low/-FOXP3+Helios+, suggesting that Helios and FOXP3 are both good markers of Tregs.
After 21 days of culture, final cell products corresponding to spe-Tregs and poly-Tregs were composed of 88 ± 2.6% and 94.1 ± 3.6% of CD3+ cells, respectively. The fact that spe-Tregs cell products only contained 88% of CD3+CD4+ cells was due to the presence of 4.9 ± 1.4% CD3+CD8+ cells, 3.5 ± 0.3% CD3-CD56+cells, and some remaining DCs. Further immunophenotypic analyses of spe-Tregs and poly-Tregs within gated CD4+CD3+ cells showed that Treg populations expressed at similar and high levels FOXP3, Helios, glycoprotein A repetitions predominant (garpin, GARP), latency-associated peptide (LAP), CD152, and CD278 (Fig. 3). As control, CD4+CD25-CD127+ effector cells purified by FACS were cultured under similar conditions in order to generate spe-Teffs and poly-Teffs, and their phenotype was studied (Fig. 3). Interestingly, Helios, GARP, LAP, and CD152 could strongly statistically discriminate (unpaired Student t-test, p < 0.001) spe-Tregs from spe-Teffs (Fig. 3A). Similar statistical differences were observed when poly-Tregs and poly-Teffs were compared, except for CD152 that was upregulated when Teffs were expanded under strong polyclonal activation (Fig. 3B). The ectonucleotidase CD39, albeit important for the Treg function, was not discriminative after culture. Indeed, CD39 only expressed by ~40% of human Tregs (8,45) is also expressed by activated Teffs (41). As others (1,2), we observed that CD25 and CD127 could not anymore be discriminative markers between Tregs and Teffs after ex vivo expansion. It is also known that activated human Teffs express FOXP3 (2,48,59). Thus, 20-25% of spe-Teffs and poly-Teffs expressed FOXP3, albeit at lower intensity levels than Tregs. The highly conserved region within the human FOXP3 gene, named TSDR, is fully demethylated in Tregs only, and can discriminate Tregs from activated FOXP3+ conventional T cells (5). Therefore, we analyzed the number of demethylated TSDR in comparison to methylated TSDR copies in spe-Tregs and poly-Tregs as well as in spe-Teffs and poly-Teffs obtained after 3 weeks of culture, as previously described (60). As shown in Table 3, spe-Tregs and poly-Tregs expanded from MACS-sorted CD25+ cells exhibited a similar average demethylation level of 80.3 ± 5.5% (range 70.1-95.2%). Using the same culture conditions as for Tregs, less than 0.2% of expanded spe-Teffs and poly-Teffs were demethylated in the FOXP3 TSDR. Altogether these data confirm that spe-Tregs that exhibited a similar immunophenotype than poly-Tregs can be generated and easily distinguished from cultured Teffs.

Immunophenotypic analysis of spe- and poly-Tregs comparatively to spe- and poly-Teffs after 3 weeks of culture. Spe-Tregs and poly-Tregs were cultured as described in Materials and Methods for 3 weeks. Spe-Teffs and poly-Teffs generated from FACS-sorted CD4+CD25-CD127+ cells were cultured under the same conditions. At 3 weeks of culture, aliquots from each culture were stained with different combinations of mAbs and analyzed by FACS. Data presented are means ± SEM from three to five separate experiments, represent the percentage of positive cells within gated CD3+CD4+ cells. (A) Immunophenotypic analysis of spe-Tregs (■) and spe-Teffs ( ). (B) Immunophenotypic analysis of poly-Tregs (
). (B) Immunophenotypic analysis of poly-Tregs ( ) and poly-Teffs (
) and poly-Teffs ( ). Statistical analysis was performed using an unpaired Student t-test: **p < 0.01; ***p < 0.001.
). Statistical analysis was performed using an unpaired Student t-test: **p < 0.01; ***p < 0.001.
Analysis of the FOXP3 TSDR in Tregs and Teffs After 3 Weeks of Culture

Teffs, effector T cells.
Control of Xeno-GVHD by Allogen-Specific Tregs Using Humanized NSG Mice
In order to test and to compare the ability of spe-Tegs versus poly-Tregs to control GVHD, we set up a model in which immunodeficient NSG mice were infused intravenously (IV) with human purified effector CD3+ T cells (10 × 106/mouse) and allogenic DC (2 × 106/mouse) in the presence or not of spe-Tregs or poly-Tregs (5 × 106/mouse) that were autologous to effector T cells. Clinical signs of GVHD were daily monitored on several parameters (see Materials and Methods). It is important to note that the same model has been previously used with success to study the regulatory function of ex vivo expanded human Tregs (i) in xeno-GVHD (21,38) or (ii) in other models of allogeneic immune response (41). It is likely that this model reflects more closely the effect of human Tregs on human T cells in vivo and the consequences on the disease process, compared to pure murine models of GVHD. As expected, mice injected with effector CD3 T cells developed a GVHD within 4-5 weeks. Animals were sacrificed for ethical reasons in case of strong clinical signs of GVHD or at 120 days. Kaplan–Meyer survival curves (Fig. 4A) showed that 66-77% of mice treated by Tregs were alive at 120 days, while only 22% of mice survived in the control group (nontreated animals). Thus, both spe-Tregs and poly-Tregs can significantly (p < 0.05) control xeno-GVHD induced by human effector T cells and allogeneic DCs. Histopathological examinations of different tissues (Fig. 4B) showed that spe-Tregs significantly diminished GVHD scores in the skin (p < 0.01), the lung (p < 0.01), and the liver (p < 0.05), while poly-Tregs only significantly diminished GVHD scores in the skin (p < 0.05). These data indicate that spe-Tregs exert, as good as poly-Tregs, a suppressive activity that can significantly (p < 0.05) control a xeno-GVHD induced by human effector T cells.
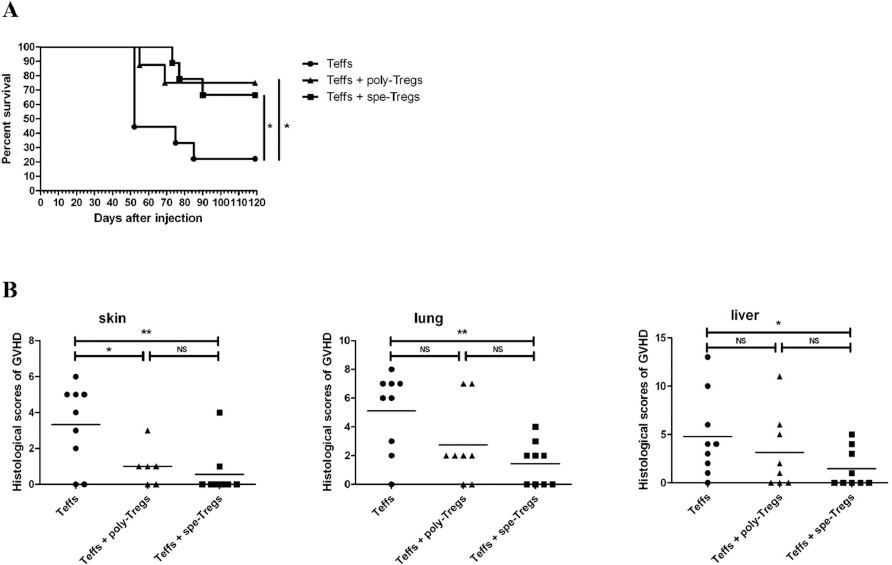
Control of xeno-GVHD by spe-Tregs and poly-Tregs. Effect of spe-Tregs and poly-Tregs on the control of GVHD in a model of xenogeneic transplantation. Xenogeneic GVHD was induced by adoptive transfer of 107 CD3+ human T cells (Teffs) into NOD/SCID/γC-/- mice. Mice received either Teffs alone (●), or cotransfer of Teffs + 5 × 106 spe-Tregs (■) or cotransfer of Teffs + 5 × 106 poly-Tregs (▲). Mice also received concomitantly 2 × 106 of allogeneic mDCs initially used to stimulate Tregs. Clinical symptoms of GVHD were monitored daily up to 120 days. (A) Kaplan–Meier survival curve for mice grafted with Teffs alone (n = 9) or Teffs + Tregs (Teffs/Tregs ratio: 2:1, n = 9). Data were obtained from a pool of three independent experiments. Log-rank comparison of the groups was used to calculate p values. (B) Histopathological examinations on the skin, lung, and liver of each animal were performed by a pathologist in a blinded fashion, and GVHD lesions were as described by Kaplan et al. (27), Cooke et al. (11), and Hill et al. (22), respectively (see Materials and Methods section). Statistical analyses were performed using an unpaired Student t-test with Welch's correction: *p < 0.05; **p < 0.01.
Discussion
Treg-based therapy may offer alternative approaches to prevent and/or control GVHD after HSCT, to control graft rejection after organ transplants, or to treat autoimmune diseases (7,42). Today, very few clinical trials designed to treat GVHD after HSCT are ongoing to test the therapeutic potential of Tregs (7). These clinical trials are based on the injection of freshly isolated Tregs or Tregs generated after an ex vivo polyclonal expansion (9,13,55). The production of antigen-specific Tregs in humans will be worthwhile inasmuch as we and others clearly demonstrated in mice that allo-Tregs are more efficient than poly-Tregs to control GVHD while preserving the GVL effect (10,49,53,54). Recent progress has been made regarding the generation of clinical-grade allospecific Tregs using either CD40 ligand-stimulated B cells (40) or immortalized human B-cell lines (31) as antigen-presenting cells. Consequently, two clinical trials based on the production of Ag-specific Tregs are starting in kidney and liver transplantation (NCT02145325 and NCT01624077). Our study poses the basis for the development of allospecific Tregs in HSCT.
First, using CFSE-labeled FACS-sorted Tregs, we studied different culture conditions in order to induce cell division of Tregs when primed by mature or immature allogeneic monocyte-derived DCs in the presence of IL-2 and IL-15. Under these conditions, we observed that mDCs associated with the combination of IL-2 and IL-15 enhanced the cell division of Tregs and upregulated FOXP3 expression. Interestingly, in some experiments, we sorted by FACS early dividing Tregs (CFSElow cells) and showed that this early dividing cell fraction, whose frequency was estimated at 1-1.5% of total Tregs, exhibited a more specific suppressive activity than nondividing Tregs (CFSEhigh cells) (data not shown). These data are in line with those of Veerapathran et al. (58), who found, using similar stimulating conditions and a limiting-dilution analysis, a frequency of alloantigen Tregs corresponding to 0.7-2%. However, CFSE cannot be used as a selectable method for clinical application.
Therefore, we adapted the use of allogeneic mDCs for priming Tregs under culture conditions compliant for clinical applications. Thus, Tregs were selected by MACS, primed twice with allogeneic mDCs, and expanded in culture in the presence of IL-2, IL-15, and rapamycin, all being clinical-grade reagents. The selection or the depletion of Tregs based on CD25 immunomagnetic beads and MACS have been already accepted by regulatory agencies for clinical trials (23,35) as well as the clinical-grade production of monocyte-derived DCs (32,46). Thus, our good manufacturing practice (GMP)-compliant conditions allow generating alloantigen-specific Tregs.
In this setting, we extensively analyzed the immunophenotype of spe-Tregs in comparison to poly-Tregs and Teffs expanded under similar conditions. FOXP3, a master gene of natural Tregs, is also expressed by activated conventional Teffs (48). The transcription factor Helios, known to control expression of FOXP3, may be also useful to discriminate activated Tregs from induced Tregs and from activated conventional Teffs (50), as well as to distinguish thymic-derived Tregs from periphery-induced Tregs (29). Furthermore, it has been reported that CD4+CD25high Helios+ cells encompass mostly Tregs for which the FOXP3 promoter is fully demethylated (5). Taking into account these publications, we first observed that Helios was expressed on ~90% of CD4+CD25highCD127low/-FOXP3+ cells in the initial fraction. Thus, CD4+CD25highCD127low/-FOXP3+Helios+ cells may represent natural Tregs. After 3 weeks of expansion, immunophenotypic analyses of spe-Tregs indicated that CD3+CD4+ cells were mainly FOXP3+Helios+ by contrast to Teffs expanded under similar culture conditions; this indicates that expanded Tregs have a stable immunophenotype. Additionally, we found that CD278, GARP, and LAP, considered as activation and functional markers of Tregs (6,52,56), were highly and significantly expressed both by spe-Tregs and poly-Tregs, but not or weakly by spe-Teffs and poly-Teffs. By contrast, CD39 and CD152 that are usually described as good Treg markers were not discriminative after ex vivo culture. Indeed, we found that polyclonally activated Teffs also expressed these markers as reported by others (41). As expected, no suppressive activity was detected within the cell products containing either spe-Teffs or poly-Teffs (data not shown). Altogether, our data indicate that the final cell product obtained after selection and expansion contained at least 80% of natural spe-Tregs with a stable phenotype as demonstrated by the percentage of TSDR demethylation, and a mild contamination (≤5%) of non-Treg cells. Thus, using GMP-compliant conditions, we have been able to generate alloantigen-specific natural Tregs that express highly FOXP3 and Helios, two specific markers for natural Tregs.
Although we demonstrated that the spe-Tregs obtained are functional both in vitro, even at low Treg/Teff ratio (1:16), and in vivo using a xeno-GVHD murine model, they are only specific for major allogeneic antigens. Thus, our approach may be limited to clinical settings such as HLA-mismatched HSCT or solid organ transplantations. Obviously, the frequency of alloantigen-specific Tregs in the context of matched HSCT would be much lower than that obtained in the present work, and their generation would be more difficult to achieve. However, we recently showed in mice that Tregs specific for minor histocompatibility antigen HY could be obtained and capable of preventing severe GVHD (34). Recently, the generation of human Tregs against minor histocompatibility antigens using very similar conditions has been reported by others (57). Our findings are encouraging for the production of spe-Tregs in order to control GVHD occurring in fully HLA mismatched HSCT or rejection of organ transplantation from unrelated donors. Validation of the generation at large scale of spe-Tregs under these GMP conditions will permit to envision future clinical trials.
Footnotes
Acknowledgments
Y.H. was financially supported by “le fond d'amorçage en Biothérapies de l'Assistance Publique Hôpitaux de Paris.” M.G.-D. was financially supported by Canceropole Ile de France. S.T. was financially supported by a fellowship from the French Ministry of Education and Research and ANR TK-safe. This work was granted by “le fond d'amorçage en Biothérapies de l'Assistance Publique Hôpitaux de Paris” and “Cent Pour Sang La Vie.” M.C. and Y.H. designed and performed research, analyzed the data, and contributed to the writing of the manuscript. C.B. performed research and, more specifically, FACS cell sorting. S.T. performed research and, more specifically, murine experiments. M.G.-D., G.S., and L.K. performed research and, more specifically, cell cultures and suppressive assays. F.C. performed histological analysis. S.M. and J.L.C. initiated the project, obtained funding, and contributed to the writing of the manuscript. F.M.L. directed the work, analyzed the data, and wrote the paper. The authors declare no conflicts of interest.
