Abstract
Ischemic preconditioning (IPC) confers tissue resistance to subsequent ischemia in several organs. The protective effects are obtained by applying short periods of warm ischemia followed by reperfusion prior to extended ischemic insults to the organs. In the present study, we evaluated whether IPC can reduce pancreatic tissue injury following cold ischemic preservation. Rat pancreata were exposed to IPC (10 min of warm ischemia followed by 10 min of reperfusion) prior to ~18 h of cold preservation before assessment of organ injury or islet isolation. Pancreas IPC improved islet yields (964 ± 336 vs. 711 ± 204 IEQ/pancreas; p = 0.004) and lowered islet loss after culture (33 ± 10% vs. 51 ± 14%; p = 0.0005). Islet potency in vivo was well preserved with diabetes reversal and improved glucose clearance. Pancreas IPC reduced levels of NADPH-dependent oxidase, a source of reactive oxygen species, in pancreas homogenates versus controls (78.4 ± 45.9 vs. 216.2 ± 53.8 RLU/μg; p = 0.002). Microarray genomic analysis of pancreata revealed upregulation of 81 genes and downregulation of 454 genes (greater than twofold change) when comparing IPC-treated glands to controls, respectively, and showing a decrease in markers of apoptosis and oxidative stress. Collectively, our study demonstrates beneficial effects of IPC of the pancreas prior to cold organ preservation and provides evidence of the key role of IPC-mediated modulation of oxidative stress pathways. The use of IPC of the pancreas may contribute to increasing the quality of donor pancreas for transplantation and to improving organ utilization.
Keywords
Introduction
Restoration of β-cell function is a desirable goal for patients with insulin-requiring diabetes. Currently, β-cell replacement is achieved in selected cases of diabetes by transplantation of vascularized pancreata or as isolated islet grafts (islet transplantation) (47). Despite the steady increase in organ donation following brain death over the last two decades, pancreas recovery rates remain unsatisfactory and thus far lower than those of other solid organs, with an underutilization of potentially transplantable glands (www.optn.org) (25,52). The shortage of donor pancreata particularly impacts clinical islet transplantation, where the number of islets obtained from a donor pancreas remains quite variable and not completely reproducible (55); approximately 50% of human glands processed with the intent to transplant yield islets deemed adequate for transplantation (51), and generally islets obtained from more than one donor pancreas are needed to attain good metabolic control after implant (20).
Multiple variables can influence both the yield and the quality of islets obtained from a single gland, including donor and organ characteristics, as well as the methods used for pancreas recovery and preservation (3,27, 41,49,51). It has been recognized that the duration of pancreas cold ischemia inversely correlates with the number of islets recovered from a donor pancreas (27,41,49). The ischemic damage endured by the exocrine pancreas may be exacerbated at the time of pancreatic digestion. This digestion, during the islet isolation process, results in the induction of stress-activated inflammatory pathways and reduces the mass of functionally transplantable islets (49). Implementation of strategies aimed at reducing organ ischemic injury may allow for a better utilization of donor pancreata for transplantation. Several approaches have been explored in recent years that have generally relied on modifications of organ procurement techniques or of the procurement solution used for organ storage leading to improved islet recovery rates from deceased donor pancreata (3,30).
Ischemic preconditioning (IPC) is emerging in recent years as a means to induce organ resistance to subsequent noxious stimuli, including ischemic injury. Initially described by Murry et al. in the myocardium (39) and then extended to other organs (liver, kidney, brain, etc.) (8,9,17,19,62), IPC is a cytoprotective surgical procedure consisting of exposing organs to brief periods of warm ischemia (inflow occlusion) followed by briefs period of warm reperfusion.
The effects of IPC have not yet been studied extensively on the pancreas or on the islets of Langerhans. In rodent models of acute pancreatitis, IPC of the pancreas resulted in a decrease in the severity of pancreatic damage via a reduction of inflammation and coagulation (11,45,64).
In the present study, we have evaluated the effect of IPC of the pancreas in a rodent model of cold ischemic preservation (49) through the assessment of multiple molecular and functional parameters. We observed that IPC of the pancreas prior to cold preservation can improve islet yields and recovery possibly via the modulation of the levels of oxidative stress mediators in the gland. Additionally, IPC of the pancreas resulted in the differential expression of multiple molecular markers assessed by gene expression that help shedding new light on the mechanisms underlying IPC-mediated cytoprotection in the pancreas.
Materials and Methods
Animals
Lewis rats (males, 260–280 g) and athymic nu/nu mice (males, 20–22 g)(both from Harlan, Indianapolis, IN) were housed in virus antibody-free rooms in isolated cages with free access to autoclaved water and food at the division of Veterinary Resources of the University of Miami (UM). All animal procedures were performed under protocols approved and monitored by the IACUC.
Pancreatectomy
As described previously (49), surgery in donor rats was performed under general anesthesia, using an intramuscular mixture of ketamine hydrochloride 80–100 mg/kg (Ketaset®; Wyeth-Fort Dodge) and xylazine 5–10 mg/kg (Anased®; Akron) in saline solution. The common bile duct (CBD) was cannulated with a polyethylene (PE-50) catheter secured by a suture and the pancreas was dissected en-block with adjacent stomach, duodenum, and spleen. The abdominal viscera were perfused in a retrograde fashion with ice-cold University of Wisconsin (UW; ViaSpan®; DuPont) preservation solution, via 24-gauge catheter cannulation of the abdominal aorta. Prior to UW infusion, the infrahepatic portal vein and inferior vena cava were incised to provide outflow tracts, and the suprahepatic inferior vena cava and aorta were occluded. Frozen saline slurry was loaded into the abdomen to reduce the temperature of the pancreas during organ perfusion and retrieval. The explanted pancreas was separated from its vascular pedicle and placed into a sterile jar containing UW. The jar was kept on ice for a cold ischemic preservation of 18 h before islet isolation was performed.
Ischemic Preconditioning
The ischemic preconditioning procedure was performed prior to CBD cannulation and en-block dissection. This entailed placement of micro-bulldog clamps on the celiac trunk (CT) and the superior mesenteric artery (SMA) (Fig. 1). The arterial supply to the pancreas (and adjacent organs) was occluded for 10 min (warm ischemia). The clamps were subsequently removed and 10 min of reperfusion allowed before initiating the pancreatectomy.

Technique for the induction of ischemic preconditioning (IPC) of the rat pancreas. (A) Retroperitoneal vessels are identified. Along the infrarenal abdominal aorta (AA) the emergence of the celiac trunk (CT) and superior mesenteric artery (SMA) are isolated and prepared for inflow occlusion. In the image are indicated the inferior vena cava (IVC), the liver, the right kidney (RK), duodenum (D), and pancreas. (B) Microvascular clamps are applied on CT and SMA during the inflow occlusion phase of ischemic preconditioning (IPC).
Islet Isolation
As reported previously (49), the dissociation enzyme solution (Liberase®; Roche) was injected through the CBD catheter after the ampulla of Vater was clamped. The distended gland was dissected free from nonpancreatic tissue. Pooled glands were subjected to digestion at 37°C for 21 min. This was followed by purification on Euroficoll discontinuous density gradients (Mediatech). Islets were cultured in supplemented CMRL media (Gibco-Invitrogen) at 37°C and 5% CO2 (10).
Islet Counts
The zinc-binding dye dithizone (Sigma-Aldrich) was used to stain islet samples (29), which were subsequently scored for size range (54). An algorithm was then utilized to convert islet counts to the “ideal” 150-μm islet (islet equivalent; IEQ) and then multiplied for the dilution factor to estimate the total IEQ obtained for each condition (54).
Islet Recovery After Culture
After isolation, islet aliquots were cultured in supplemented CMRL media at 37°C, 5% CO2. After overnight culture, counts allowed for the estimation of the percent islet recovery over the amount plated on the day of isolation, which is a surrogate measure of islet quality (23,49).
Islet Transplantation
A single intravenous streptozotocin (200 mg/kg; Sigma) injection was used to render nude mice diabetic. Glycemia was monitored using portable glucometers (OneTouchUltra2; Lifescan) on whole blood samples obtained from the tail vein. Nonfasting glycemic values ≥ 350 mg/dl in three consecutive readings confirmed the occurrence of diabetes after induction and achievement of normoglycemia (defined as the first of at least three nonfasting glycemic values ≤200 mg/dl on 3 consecutive days) after islet transplantation. Islets were implanted under the left kidney capsule of diabetic mice under general anesthesia obtained with isofluorane (Aerrane®, Baxter) 2–3% mix with oxygen, as described previously (49). Nephrectomy of the graft-bearing kidney was performed in all animals achieving normoglycemia to exclude residual function of the native pancreas, which was confirmed by prompt resumption of hyperglycemia (48). Postoperative analgesia was obtained by subcutaneous injections of buprenorphine (Buprenex®; Reckitt-Benckiser) for the first 48 h. An intraperitoneal glucose tolerance test was performed to assess the metabolic performance of transplanted islets. Briefly, after overnight fasting, animals received an intraperitoneal glucose bolus (2 mg/kg of body weight) and glycemia was measured with portable glucometers on whole blood (tail vein) for 120 min in conscious animals. The area under the curve (AUC) of glycemic values after bolus was calculated for individual animals to evaluate glucose clearance between experimental groups (22).
NADPH-Dependent Oxidase Assay
Pancreata were either subjected or not subjected to IPC. Pancreas biopsies were snap-frozen immediately after pancreatectomy and stored at −80°C until assayed. The nicotinamide adenine dinucleotide phosphate (NADPH)-dependent superoxide activity was measured by lucigenin-enhanced chemiluminescence in tissue homogenates (60). Briefly, pancreatic tissue was suspended in phosphate-buffered saline (PBS) containing 2 mM of diethylenetriamine pentaacetic acid with the addition of protease inhibitors (pepstatin, aprotonin, leupeptin, and phenylmethanesulphonylfluoride, all from Sigma-Aldrich). Tissue was homogenized and sonicated (two 5-s pulses using a cup-horn sonicator probe at ~50% power). Protein concentrations were determined by the BCA Protein Assay (Pierce). The reaction was initiated by addition of NADPH (100 μM final concentration) and dark-adapted lucigenin (5 μM final concentration; Sigma). Light emission was recorded over several minutes using a microtiter plate luminometer (Centro-LB960; Berthold, Germany). Data were expressed as relative light units (RLU) normalized to protein content and corrected for by a sample blank. Each experiment was performed in duplicate. The specificity of lucigenin-enhanced chemiluminscent superoxide detection was confirmed by adding the flavoprotein NADPH oxidase inhibitor diphenyleneiodonium (10 μM) and the respiratory mitochondrial chain inhibitor rotenone (50 μM) to block specific superoxide pathways (Sigma) (32,60).
Gene Arrays
Pancreatic biopsies were stored in RNAlater® (Ambion/Applied-Biosystems) immediately after pancreatectomy following or not following IPC treatment (n = 5 animals per group). Specimens were processed for RNA isolation and gene arrays by Genome Explorations USA (Memphis, TN). Briefly, total RNA was isolated using RNA STAT-60 (Tel-Test; Friendswood, TX) from ~100 mg of frozen pancreatic tissue. Immediately prior to cDNA synthesis, RNA sample purity and concentration were determined by dual-beam UV spectrophotometry (OD260/280); RNA integrity was determined by capillary electrophoresis (RNA-6000 Nano Lab-on-a-Chip kit and a Bioanalyzer 2100; Agilent). The RNA was processed and labeled according to standard reverse transcription in vitro transcription methods. First- and second-strand cDNA were synthesized from total RNA (2.0 μg) using T7-oligo(dT)primer (5′-GGCCAGTG AATTGTAATACGACTCACTATAGGGAGGCGG-3′) and cRNA was synthesized, labeled with digoxigenenin-11-UTP (Roche) and purified using the NanoAmp RT-IVT Labeling Kit (Applied-Biosystems). Labeled cRNA (10 μg) was fragmented and hybridized to the Rat Genome Survey Microarray for 16 h at 55°C (Chemiluminescence Detection Kit; Applied-Biosystems), which contains 60-mer oligonucleotide probes representing approximately 27,000 rat genes in public and Celera databases. Arrays were washed and stained with anti-digoxigenin-AP Fab fragments (Roche). Chemiluminescent signals were measured using the AB1700 Microarray Analyzer (Applied-Biosystems). Analysis of expression data, quality control metrics, and statistical analysis were performed in Bioconductor R using the ABarray package (Applied-Biosystems). Additional gene annotation, gene ontology and pathway analysis was performed using the Celera Panther database.
Statistical Analysis
Data were analyzed using Microsoft® Excel®, SigmaPlot®-v9.0 (Systat) and Prism®v4.00 (GraphPad). Comparisons between two experimental groups were done using paired or unpaired (whenever appropriate) Student's t-test. Survival curves were compared using log rank test. Data are presented as means ± SE. Statistical significance was considered for p < 0.05.
Results
Ischemic Preconditioning Increases the Islet Yields Following Cold Pancreas Preservation
Islets were obtained in parallel, side-by-side isolations (n = 9 individual isolations per group) from pools of rat pancreata that were subjected or not subjected to IPC prior to 18-h cold preservation (from 4.7 ± 0.1 and 6.3 ± 0.8 donors, respectively). Significantly higher islet yields on a per donor basis were obtained from pancreata exposed to IPC (964 ± 112 IEQ/donor; n = 42) when compared to controls (711n = 68 IEQ/donor; n = 57; p = 0.004) (Fig. 2A).
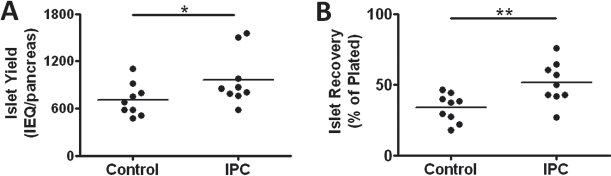
Effects of ischemic preconditioning (IPC) on islet yield and recovery. (A) Islet yields after isolation (n = 9 per group) from glands exposed to 18 h of cold preservation in University of Wisconsin (UW) solution without (Control, n = 57 pancreata) or after IPC treatment (IPC; n = 42 pancreata). (B) Islets obtained from pancreata exposed to 18-h cold ischemia without (Control) or with IPC treatment (IPC) were plated soon after isolation with loss estimated following overnight culture. Data are representative of 9 independent experiments. Paired Student's t-test: *p = 0.004; **p = 0.0005. IEQ, islet equivalents.
Pancreatic Ischemic Preconditioning Improves Islet Recovery After Overnight Culture
Assessment of the islet counts following the first postisolation overnight culture is a surrogate marker of islet quality (23,49). Islet recovery after overnight culture (calculated as the percent of islets counted on day 1 compared to that plated on day 0) was significantly lower in the control group (33.7 ± 3.3%; n = 9), when compared to the IPC group (51.3 ± 4.9%; n = 9; p = 0.0005) (Fig. 2B).
Preserved In Vivo Potency of Islets Isolated from Ischemic Pancreata
Rat pancreata exposed to 18-h cold ischemia from IPC-treated and control donors were processed to isolate islets. After isolation, the islets were cultured overnight and then separated into 100 IEQ aliquots. The aliquots were transplanted into chemically induced diabetic nude mice in order to assess the effects of pancreatic IPC on islet function in vivo (Fig. 3A). Reversal of diabetes in mice after transplantation of islets obtained from control (n = 10 mice) and IPC-treated (n = 9 mice) pancreata occurred in 90% and 77.7% of recipients with a median of 2 and 3 days, respectively (p = NS). Additionally, glucose clearance during intraperitoneal glucose tolerance test showed better islet potency in the IPC group when compared to control (Fig. 3B), with the AUC of glycemic values 9,495 ± 1,292 versus 12,090 ± 2,006 mg min dl−1, respectively.

In vivo potency of islets obtained following 18-h of cold pancreas preservation with or without previous IPC treatment. (A) Time to diabetes reversal of athymic nu/nu (nude) mice induced diabetic by streptozotocin and transplanted with 100 IEQ of islets isolated from untreated glands (Control; n = 10) or IPC-treated glands (IPC; n = 9) exposed to cold ischemia. (B) Mean glycemic values ± SE during an intraperitoneal glucose tolerance test in recipients of rat islets isolated from pancreata treated or not treated with IPC (n = 5 mice per group). The mean area under the curve of glycemic values for control and IPC group was 12,090 ± 2,006 versus 9,495 ± 1,292 mg min dl−1), respectively.
Reduced NADPH-Dependent Oxidase Activity in Pancreas After Ischemic Preconditioning
NADPH-dependent oxidase (NOX) is an enzyme that catalyzes the formation of reactive oxygen species (ROS) (32,59,60). NOX is present in endothelial cells and smooth muscle cells. To evaluate whether IPC could modulate oxidative pathways in pancreatic tissue, biopsies were collected right after the completion of pancreatectomies from glands exposed or not exposed to IPC and processed to determine NOX activity. IPC-treated pancreata expressed significantly lower levels of NOX activity compared to non-IPC pancreata (78.4 ± 19.6 vs. 216.2 ± 19.6 RLU/μg of proteins, respectively; n = 5 pancreata per group; p = 0.003) (Fig. 4).

NADPH-dependent superoxide activity in pancreatic tissue after ischemic preconditioning. Pancreatic biopsies were obtained from rat glands exposed (IPC) or not exposed to IPC (control) prior to pancreatectomy (n = 5 glands per group). Nicotinamide adenine dinucleotide phosphate (NADPH)-dependent superoxide (NOX) activity was measured on tissue homogenates by lucigenin-enhanced chemiluminescence. Data are expressed as relative light units (RLU) normalized to protein content and corrected for by a sample blank. Paired Student t-test: *p = 0.003.
Ischemic Preconditioning Is Associated with a Differential Gene Expression in the Pancreas
To identify potential molecular signatures associated with IPC-mediated cytoprotection in the pancreas, we performed gene expression arrays to assess RNA transcription on control and IPC-treated pancreata that were collected immediately after pancreatectomy. The threshold for statistical significance (p < 0.05) of differential gene expression was set to twofold in either direction. This analysis elicited 81 genes with >twofold increase and 454 genes with >twofold decrease when comparing IPC-treated glands to controls, respectively (n = 5 per group). Table 1 summarizes the most representative genes identified by our analysis (expressed as fold changes when comparing IPC to control) and grouped based on known gene function.
Ischemic Preconditioning (IPC)-Mediated Differential Gene Expression in the Pancreas

Fold change. Positive numbers indicate upregulation and negative numbers downregulation when comparing IPC to control pancreata.
Discussion
The ultimate goal of insulin-requiring diabetes therapies is attaining optimal metabolic control to stabilize and prevent the progression of diabetic complications. Transplantation of pancreatic islets, as either vascularized whole organ or isolated cell clusters, offers the option of achieving restoration of β-cell function with a more physiologic metabolic control than exogenous insulin therapy. One of the limitations of β-cell replacement therapies is the current shortage of pancreata for transplantation obtained from heart-beating donors after cerebral death.
Over the last three decades, the steady increase in success rates of organ transplantation has been paralleled by improved rates of deceased multiorgan donation both in the US (based on United Network for Organ Sharing—UNOS statistics) and abroad. Unfortunately, the number of donor pancreata recovered for transplantation (either as whole organ or as isolated islets) remains unsatisfactory to date (25,52). For instance, from a pool of >8,000 multiorgan donors available in 2006 through UNOS, approximately 2,000 pancreata were recovered with <1,500 used for transplant (www.optn.org). In the US alone, underutilization of pancreata potentially “optimal” for transplantation has been recognized: from the pool of pancreata available over a 4-year period, 22.3% of “optimal” glands were used for whole organ transplant, while from the remaining pool, 48.5% were considered “suitable islet donors” (i.e., 11% “optimal” and 89% “standard”), but only 2.1% of them were eventually used for islet transplantation (52).
Several factors that may influence the quality of pancreata and islets for transplantation have been recognized (3,27,41,49,51,56). The duration of cold ischemia inversely correlates with organ quality, islet yields, and potency obtained from a donor pancreas (3,27,49). Various approaches have been proposed to improve organ preservation and to increase the number of islets obtained from a single donor pancreas. These include the implementation of improved donor selection and organ allocation criteria, pancreas recovery techniques that reduce injury during pancreatectomy (30,51), the use of different preservation solutions (3,43) and oxygen-enhancing moieties during organ preservation (2,16, 26,53), intraductal injection of preservation solutions (38,57) with antioxidants (1) or protease inhibitors (44), among other strategies.
Ischemic preconditioning represents a natural means of inducing organ resistance to subsequent ischemic injury that has been described as efficacious for several organs (8,9,17,19,39,62). The effects of and mechanisms underlying IPC on the pancreas have not been studied extensively. A decrease in the severity of inflammation and coagulation has been described after IPC treatment in a model of induced pancreatitis (11,45,64).
In the present study, we evaluated the effects of IPC prior to cold pancreas preservation injury in a rat model (49). This model of extended cold preservation is associated with measurable reductions in islet yields after isolation and recovery after overnight culture, increased activation of stress signal transduction pathways, as well as impaired in vivo potency, when compared to organs exposed to short ischemia (49). Our current study indicates that IPC can significantly improve pancreas quality after extended cold preservation, as shown by the increased islet isolation yields in our model. Islet recovery after overnight islet culture is a surrogate marker of islet quality based on the fact that if islets are damaged during the isolation process (i.e., due to pancreatic ischemic damage) fewer islets will be recovered (49). Indeed, under control conditions (namely, 18 h of cold preservation without preconditioning), a significantly lower number of islets was recovered when compared to IPC-treated pancreata exposed to the same period of cold preservation. This finding extends our preliminary observations on a smaller sample size (21) and suggests that IPC treatment prior to cold ischemia may lead to less damaged islets after isolation and, therefore, a reduction in islet loss in culture. Islets isolated from control or IPC-treated pancreata that were subjected to 18-h cold preservation were assessed in vivo using a model of sub-optimal islet transplantation into chemically induced diabetic immunodeficient mice (49). We have previously shown that islets obtained from pancreata exposed to prolonged cold ischemic preservation are severely impaired, when compared to those from short ischemic glands in this model (50). In the present study, glucose clearance in response to intraperitoneal challenge showed a trend toward better function in recipients of islets from IPC-treated pancreata with decreased AUC than controls, suggesting improved glucose clearance.
Modulation of oxidative stress pathways by subjecting organs to IPC prior to cold ischemic damage has been proposed as one of the putative mechanisms underlying its cytoprotective effects (14,61). We observed that NOX activity in glands obtained soon after the pancreatectomy (following in situ perfusion with cold UW and before initiation of cold preservation) was significantly reduced in IPC compared with control. This suggests a role for the IPC-induced modulation of oxidative stress pathways in the pancreas that may have contributed to the observed improved outcomes in isolated islets. Involvement of the NOX pathway in postischemic oxidative stress has been recognized for the myocardium, where an association between decreased NOX activity and improved myocardial function has been demonstrated after IPC (14). Evidence of increased NOX activity in aging mice has been implicated as source of the oxidative damage that causes cerebrovascular dysfunction (46). Conversely, increased levels of NOX activity have been associated with the favorable effects of IPC in decreasing myocardial infarct (MI) size (5). Additionally, inhibition of NOX activity has been shown to abolish the hepatoprotective effects of IPC (61). The improved outcome associated with the reduction of NOX activity observed in our study may be related to the intrinsic difference between the pancreas and other tissues, as well as the experimental design tested. Indeed, the previous study in the MI model was performed in conditions of warm ischemic damage (5), while ours were done by cold perfusion with preservations solution at the time of organ recovery. Also, the previous study on hepatic cytoprotection were performed using an ex vivo perfusion system to mimic ischemia-reperfusion (61). Notably, in the pathogenesis of atherosclerosis, within the atheroma, low levels of reactive oxygen species promote growth, moderate levels cause senescence, and high levels induce apoptosis of smooth muscle cells (31,32). This observation points to the different role of NOX in different stages of pathophysiological conditions. Although not formally addressed in our study, it is reasonable to assume that the low-to-moderate levels seen in the IPC-treated pancreata, at the very least, do not harm the tissue, whereas the high levels within the control pancreata might be harmful.
Molecular arrays have become accessible research tools to assist in the study of the mechanisms underlying complex biological phenomena. These arrays may also lead to the discovery of potential targets for therapeutic interventions. Because of the lack of in-depth molecular studies in the context of pancreas ischemia and IPC in the scientific literature, we performed a gene array analysis on glands obtained after pancreatectomy. This revealed upregulation of 81 genes and downregulation of 454 genes (using a cut-off of >twofold change) when comparing IPC-treated glands to controls, respectively. Our data indicate that the changes in mRNA synthesis induced by IPC of the pancreas favor cell survival, based on an observed reduction of genes involved in the induction of apoptosis (Table 1). In addition, transcripts involved in cellular differentiation, signal transduction, and metabolic processes were downregulated (Table 1). These observations are in agreement with differential gene expression arrays reported in other models of IPC of the retina and liver, which have demonstrated a controlled arrest of metabolic functions as a possible underlying mechanism of the ischemic tolerance induced by IPC (24,42).
The expression of stress response genes was significantly downregulated by IPC treatment of the pancreas: several chemokine C-C motif ligands (Ccl2, Ccl4, Ccl5, Ccl7) and cellular adhesion transcripts [cadherin 22 (Cdh22), calsyntenin 3 (Clstn3), Lutheran blood group (Lu), myelin-associated glycoprotein (Mag)] that are involved in immunoregulatory processes were differentially expressed (7,37). For example, using the same model of cold pancreas preservation in rats, we have previously reported that CCL4 (macrophage inflammatory protein-1α; MIP-1α) production by islets in vitro after isolation is significantly increased after 18 h of cold ischemic preservation, when compared to short cold preservation (49). Also, increased CCL5 (regulated upon activation, normal T cell expressed and secreted; RANTES) gene expression has observed in rodent islets exposed to cytokines in vitro and was associated with poorer outcome after transplantation (58). The downregulation of Ccl4 and Ccl5 gene expression by IPC in the present study supports a possible anti-inflammatory effect in the pancreas.
The activation of the nuclear factor-κB (NF-κB) pathway in tissues exposed to ischemia and reperfusion injury may lead to both initial cytoprotective effects and to intense and deleterious inflammation upon reperfusion (28). In our study, pancreatic IPC was associated with the upregulation of Nfkbia, which encodes an inhibitor of NF-κB that has been shown to decrease islet cell death (15,18,33). In IPC-treated glands, the expression of Vof16 was increased. The expression of Vof16, encoding for the ischemia related factor Vof-16, has been described in the rat brain following chronic ischemia (63).
Pancreatic IPC was associated with a sharp reduction of Ncf1 expression. The cytosolic factor p47-phox, encoded by the neutrophil cytosolic factor (NCF1) gene, is an essential component of the NADPH-oxidase system: Ncf1 transcription is necessary for the activation of NOX (35). This observation is consistent with the observed reduction of NOX activity after IPC in the pancreas. Ncf1 gene deficiency is associated with decreases in atherosclerotic lesion formation via a reduction in NOX-induced production of reactive oxygen species (ROS) (4). Furthermore, the downregulation of transcripts for serum amyloid A4 (Saa4) and macrophage galactose N-acetyl-galactosamine specific lectin 1 (Mgl1), which are expressed in inflammatory and fibrotic diseases (34,65), was observed in IPC-treated glands.
Members of the heat shock protein gene transcript family (Hspa1a, Hspa1b, Hspa4) were upregulated after IPC, suggesting a beneficial stabilization of existing proteins as a cytoprotective mechanism during the subsequent period of ischemic injury (6,36,42). In particular, the overexpression of Hspa1a (heat shock 70kD protein) has been associated with the cytoprotective effects of IPC in other organs (58,66), including a rat model of pancreatitis (64).
Pancreatic IPC was associated with the downregulation of tumor necrosis factor (TNF) receptors (Tnfrsf8, Tnfrsf12a), which are involved in the induction of cellular apoptosis and TNF production (13,40). The expression of five proapoptotic genes [caspase recruitment domain family, member 9 (Card9), caspase 9 (Casp9), granzyme B, G, and C (Gzmb, Gzmg, and Gzmc)] was downregulated, while the expression of Cflar [CASP8 and fas-associated death domain (FADD)-like apoptosis regulator; c-Flip-1], a potent apoptosis inhibitor was upregulated, thus indicating a strict regulation of apoptosis by the IPC. A previous study in rats has shown that pancreatic grafts exposed to IPC have increased cellular apoptosis after transplantation (12). This discrepancy from our results may be explained by the multiple differences in the experimental design, including the fact that we assessed gene expression profiles resulting from the IPC protocol on biopsies obtained immediately after the treatment, whereas the previous study evaluated apoptosis after transplantation, therefore including the effect of surgical and reperfusion injury. Additionally, we have previously demonstrated that ~18 h of cold preservation of the pancreas results in apoptosis induction primarily in the acinar tissue and not in islet cells (49); in the transplant study (12), cold ischemia was 6 h followed by reperfusion injury.
Collectively, our study demonstrates the beneficial effects of ischemic preconditioning of the pancreas prior to cold organ preservation and provides initial evidence of the key role of IPC-mediated modulation of oxidative stress pathways. Additional pathways and differential gene expression patterns identified in our microarray analysis will be of assistance to future studies that aim at a better understanding of IPC-mediated cytoprotection of the pancreas.
The use of IPC of the pancreas may improve the quality of a donor pancreas for transplantation and therefore improve organ utilization. Also, the enhancement of pancreatic preservation may increase the efficiency of islet isolations and allow for the recovery of a higher number of functional islets to attain insulin independence from a single donor organ.
Footnotes
Acknowledgments
This work was possible thanks to the generous support of the Diabetes Research Institute Foundation (www.DiabetesResearch.org). Cell processing and animal procedures were performed at the Pre-clinical Cell Processing and Translational Models Core, partially supported by the Juvenile Diabetes Research Foundation International (to C.R. and A.P.) (4-2004-361; 4-2008-811; 17-2010-5) and by the Diabetes Research Institute Foundation (to A.P.). The University of Miami Institutional Animal Care and Use Committee has an Animal Welfare Assurance on file with the Office of Laboratory Animal Welfare (OLAW), National Institutes of Health (assurance number: A-3224-01, effective 12/4/02) and is accredited by the Association for Assessment and Accreditation of Laboratory Animal Care. The authors are grateful to Drs. Miguel A. Perez-Pinzon, Luca A. Inverardi, David Della Morte, Kunjan R. Dave, Ami P. Raval, Hirohito Ichii, and Christopher A. Fraker for constructive scientific discussions on this study. Special thanks to Sergio San Jose, Weijun An, Yelena Gadea, Irayme Labrada, and Kevin Johnson for their excellent technical assistance. The authors declare no conflicts of interest.
