Abstract
Pancreatic islet transplantation is a promising treatment option for patients severely affected with type 1 diabetes. This report from CITR presents pre- and posttransplant human leukocyte antigen (HLA) class I sensitization rates in islet-alone transplantation. Data came from 303 recipients transplanted with islet-alone between January 1999 and December 2008. HLA class I sensitization was determined by the presence of anti-HLA class I antibodies. Panel-reactive antibodies (PRA) from prior to islet infusion and at 6 months, and yearly posttransplant was correlated to measures of islet graft failure. The cumulative number of mismatched HLA alleles increased with each additional islet infusion from a median of 3 for one infusion to 9 for three infusions. Pretransplant PRA was not predictive of islet graft failure. However, development of PRA >20% posttransplant was associated with 3.6-fold (p < 0.001) increased hazard ratio for graft failure. Patients with complete graft loss who had discontinued immunosuppression had significantly higher rate of PRA ≥ 20% compared to those with functioning grafts who remained on immunosuppression. Exposure to repeat HLA class I mismatch at second or third islet infusions resulted in less frequent development of de novo HLA class I antibodies when compared to increased class I mismatch. The development of HLA class I antibodies while on immunosuppression is associated with subsequent islet graft failure. The risk of sensitization may be reduced by minimizing the number of islet donors used per recipient, and in the absence of donor-specific anti-HLA antibodies, repeating HLA class I mismatches with subsequent islet infusions.
Introduction
Pancreatic islet transplantation is an emerging β-cell replacement approach to achieve insulin independence or improve glycemic control for patients with “brittle” type 1 diabetes (T1D). While insulin-independent rates have been —60% at 1 year, islet graft function decreases over time with insulin independence rates declining to ~13% at 5 years (1,8,10,22,26). Many factors could contribute to either early or late loss of islet graft function, including posttransplant inflammation (13), potential toxicity of immunosuppressive drugs (24), recurrence of autoimmunity (25,28), β-cell exhaustion (19,27), and alloimmune rejection. Several reports (4—7,9,15,16,18, 20,21) have shown the development of alloimmune responses in islet transplant recipients with detection of antibodies against islet donor human leukocyte antigen (HLA) class I and II molecules. HLA sensitization can be explained by the absence of HLA matching at the time of selection of donor pancreas and the use of multiple islet infusions to achieve a sufficient engrafted islet mass to support insulin independence, practices that may expose an islet recipient to a number of mismatched HLA alleles.
The effect of HLA sensitization on islet graft function is not sufficiently clear (6). The presence of anti-HLA antibodies pretransplant has been associated with worse islet graft functional outcomes in some (4,16), but not all (7), studies; however, the negative report did not determine donor specificity of the detected alloantibodies. The development of de novo anti-HLA antibodies posttransplant has also been associated with islet graft failure (15,18,20), although some islet transplant recipients who develop positive panel-reactive antibodies (PRA) may continue to have well-functioning islet grafts (2, 7). Regardless of the mechanism of islet graft deterioration, patients who discontinue immunosuppressive drugs following complete or partial islet graft loss seem to be at high risk for HLA sensitization (5,7,9,21).
The Collaborative Islet Transplant Registry (CITR) contains data on islet transplantation contributed from 28 North American medical institutions, three European, and two Australian centers between January 1999 and December 2008. As of April 2009, the registry contained data from 412 islet transplant recipients of 828 infusions derived from 905 donor organs (26). This report describes the analysis of HLA class I sensitization among 303 islet-alone transplant recipients. The role of multiple islet infusions, the number of HLA mismatches, and immunosuppression withdrawal on the development of HLA class I sensitization were analyzed, as well as the effect of HLA sensitization on islet graft failure. Currently the data on HLA class II PRA are incomplete. Efforts are being made to complete data collection on sensitization to HLA class II and a comprehensive analysis will be the focus of future reports.
Materials and Methods
Islet transplant centers contributing to the CITR obtained local IRB approval and written informed consent from all patients whose data were entered. The data are rigorously monitored to comply with Code of Federal Regulations (CFR) requirements. This study reports an analysis of data collected from 303 recipients transplanted with islets alone between January 1999 and December 2008. Patients receiving allogeneic islet-alone transplants generally are required to have had type 1 diabetes mellitus for more than 5 years, be between 18—65 years of age, and have extreme difficulties with glycemic control indicated by either hypoglycemia unawareness with episodes of severe hypoglycemia, or marked glycemic lability with consistently elevated HbA1c levels (1,26). Information on donor HLA phenotyping was collected from the United Network for Organ Sharing (UNOS) for North American patients, and data on all recipients were provided by the transplant centers.
Panel Reactive Antibodies (PRA)
HLA sensitization in islet transplant patients is reported as percent PRA against class I HLA. PRA was measured either by the complement-dependent cytotoxicity assay using a reference panel consisting of lymphocytes with known HLA specificity, enzyme-linked immunosorbent assay (ELISA) using antigen coated trays, FLOW PRA, which is based on antigen-coated fluorescent beads, and/or Luminex solid phase assays (11). PRA provides the percent of cells that were either killed by patient's serum or the percent of cells that showed binding of immunoglobulins to HLA proteins. We analyzed positive PRAs in this study as either 1—19% or ≥ 20%.
Statistical Analysis
Descriptive statistics are presented as numbers for qualitative variables and mean ± SD or median with range for quantitative variables. Boxplots are used to summarize data. The “star” in the boxplot represents the mean value while the lower, middle, and upper lines of the box represent the 25th, 50th, and 75th percentile, respectively. The square roots of pretransplant and 2-year posttransplant PRA were plotted. McNemar's test was used to determine if islet transplantation was associated with developing a posttransplant PRA ≥ 20%. Univariate analysis was performed using chi-square test to compare number of PRA ≥ 20% patients with pretransplant PRA, number of islet infusions, and with reexposure to HLA class I mismatches. Proportional hazards analysis of time to graft failure was performed with fixed and time-varying covariates. A value of p < 0.05 was considered statistically significant. All analyses were performed in SAS 9.2 for Windows (SAS Institute Inc., Cary, NC).
Results
The number of patients analyzed was 303 from 27 centers; 81 patients received one, 144 patients received two, and 78 patients received three or more infusions. The mean age of the recipients was 44 ± 9.7 years and included 108 males and 195 females (Table 1). Fourteen out of 303 patients had a pretransplant PRA ≥ 20% and 37/303 patients had low levels (1—19%) of PRA. The registry does not have alloantibody specificity data, and so cannot determine whether unacceptable antigens were avoided and/or if prospective cross-matches were negative before proceeding with the islet transplantation in these 51 PRA positive patients.
Patient Demographics, Islet Transplant Data, and Panel Reactive Antibodies (PRA) Status

The median number of class I (A and B) mismatches at first islet infusion was 3 with a range of 0 to 8. A mismatch of 5 to 8 class I antigens reflects infusion of combined islet preparations from two donor pancreata. The median number of mismatches increased to 6 (range 2—12) for the second infusion and 9.5 (range 6—15) for the third infusion (Table 2). Results were similar for the number of class II (DR and DQ) mismatches. The median number of mismatches for the combination of all HLA alleles reported (A, B, DR, DQ) are 5 for one infusion, 11 for two infusions, and 17 for three infusions (Table 2).
Cumulative Number of Mismatched Alleles for Human Leukocyte Antigen (HLA) Class I and II (Median With Range) Where Patient and Donor HLA Are Known

The prevalence of a class I PRA ≥ 20% before and after one (A) or two (B) or three (C) islet infusions is shown in Figure 1. The follow-up period was 6 months and yearly after 1 year for up to 3 years. At first infusion 289 patients were <20% for class I PRA. During the follow-up period after a single islet infusion, 4/102 at 6 months, 4/70 at 1 year, 4/42 at 2 years, and 4/25 at 3 years had a PRA ≥20%. At the time of a second islet infusion 4/197 patients were PRA ≥20%, and subsequently 2/123 were ≥20% at 6 months, 6/109 at 1 year, 9/81 at 2 years, and 8/58 at 3 years. Similarly 1/69 patients who received a third islet infusion was ≥20% for class I PRA at the time of infusion, and then 1/40 were ≥20% at 6 months, 0/38 at 1 year, 0/28 at 2 years, and 4/23 at 3 years.

Changes in panel reactive antibodies (PRA) status during postislet transplant period. Percent class I PRA after first (A, N = 289), second (B, N = 214), and third (C, N = 76) infusions in islet-alone transplant patients whose pretransplant (Pre) PRA was below 20%. The follow-up period included 6 months (M6), 1 years (Y1), 2 years (Y2), and 3 years (Y3) postislet infusion, censored at reinfusion.
Figure 2 shows the development of class I PRA (≥20%) at 2 years post-final infusion when compared to the pretransplant status after one, two, or three islet infusions in 161 patients under immunosuppression. Of 152 patients with pretransplant class I PRA <20% (Q1 and Q3) and assayed at 2 years post-final islet infusion, 15 (9.9%) developed a PRA ≥20%. The increase was significant (p < 0.001) and the one case showing ≥20% PRA during pretransplant period (Q3) was determined to have resolved with a decrease in PRA to 0%. After two islet infusions the increase in number of patients with ≥20% PRA (13/83) was more when compared to one infusion (2/42). Surprisingly among 27 patients with three infusions, there was no evidence of increase in PRA.

Square root of class I PRA at 2 years post-final islet infusion by square root of pretransplant class I PRA. Reference lines drawn at √20 on both the x and y axis separate the graph into four quadrants. N = 33, 52, and 19 for recipients of 1, 2, and 3 islet infusions, respectively. A large number of observations are at the 0,0 point of the graph; N = 40, 70, and 27 for recipients for 1, 2, and 3 infusions in Q1 area, 1, 0, and 0 in Q2, 2, 13, and 0 in Q3, and 2, 6, and 0 in Q4, respectively.
The cumulative incidence of class I PRA ≥20% among 250 islet transplant patients under immunosuppression is shown in Figure 3. At 1 year post-first infusion 3% of patients developed a class I PRA ≥20% and the incidence increased to 9% at 2 years, 13% at 3 years, 26% at 5 years, and 33% at 6 years. As shown in Figure 4, median PRA among patients with functioning islet grafts was zero for all four follow-up periods; however, 4/180 patients at 6 months, 4/152 at 1 year, 6/102 at 2 years, and 5/75 at 3 years had a class I PRA >20%. Among patients with complete graft loss median PRA was 5% at 6 months, 0% at 1 year, 4.5% at 2 years, and 25% at 3 years posttransplant. In an analysis of PRA as a predictor of time to initial graft failure, elevated baseline PRA was not a risk factor as failure rates were 100/252 (39.7%), 20/37(54.1%), and 3/14(21.4%) for those with 0% PRA, 1—19, and ≥20, respectively. Among those with % PRA 0—19 at baseline, recent elevation of PRA ≥20% during follow-up was a risk factor for graft failure (HR = 3.6, p < 0.0014).
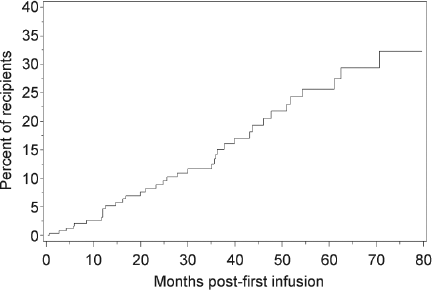
Posttransplant development of class I PRA in the presence of immunosuppression. Cumulative incidence of de novo development of >20% class I PRA is shown in 250 patients for a period of 6 years following first islet infusion.
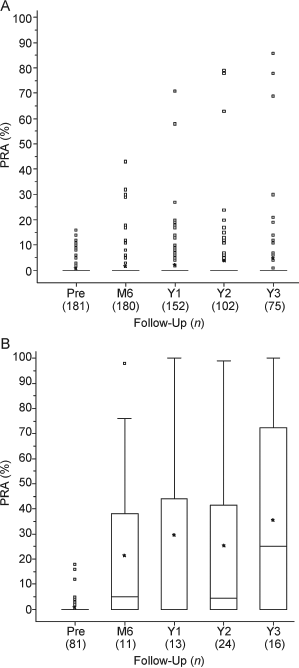
PRA status in islet transplant recipients with different graft outcome. Percent class I PRA in patients without (A, N = 181) or with (B, N = 81) complete graft loss post-last infusion is shown. The follow-up period included pretransplant of infusion 1 (Pre), 6 months (M6), 1 years (Y1), 2 years (Y2), and 3 years (Y3) post-last islet infusion.
We analyzed development of posttransplant PRA ≥20% by baseline and treatment characteristics (Table 3). Among 217 patients with 0% PRA before islet transplant, 13% (28/217) developed ≥20% PRA, whereas 38% of patients with 1—19% PRA at baseline showed development of ≥20% PRA. This data showed that presence low levels PRA at first infusion will significantly increase (p < 0.001) the possibility of development class I PRA to ≥20%. However multiple islet infusions did not increase the possibility of development of class I PRA of ≥20% significantly when compared to single infusion (15% vs. 20%; p < 0.26). HLA class I sensitization (≥20% PRA) among patients reexposed to repeat HLA donor mismatches (8%; n = 39) was less than those patients who were not reexposed (20%; n = 35). However, this difference did not reach statistical significance (p < 0.12) as shown in Table 3.
Posttransplant PRA >20% by Baseline and Treatment Characteristics

Patients with pretransplant PRA <20% and at least one known posttransplant PRA measurement.
Restricted to recipients of two islet infusions.
Discussion
This report of CITR data represents the largest analysis to date of HLA class I sensitization in islet transplant recipients. The results show that i) multiple islet infusions increased the number of HLA mismatches to which transplant recipients were exposed; ii) multiple islet infusions did not further increase the risk of sensitization over a single islet infusion; iii) the proportion of patients developing anti-HLA class I antibodies increased over time even in the presence of immunosuppression; and iv) the development of a posttransplant PRA ≥20% was associated with subsequent islet graft failure. The limited pool of donor organs precludes efficient islet transplant allocation according to HLA matching. Hence, it is expected that multiple infusions will expose recipients to increased numbers of HLA mismatches. Our results confirm this expected outcome as the majority of islet recipients who received two islet infusions were exposed to at least six HLA class 1 mismatches. Data from the Edmonton group (4,5) showed that the median number of class I mismatches was 3 with a range of 0—7, and for both class I and II the number of mismatches in their patient population ranged from 1—19 with a median of 8.5; however, there was no relationship between the number of mismatches and the development of HLA antibodies. Development of approaches that can achieve insulin independence after single islet transplantation will minimize patient's exposure to HLA mismatches which in turn may decrease the risk for allosensitization.
Another important issue for the transplantation of islets mismatched for HLA is the effect of pretransplant sensitization on subsequent islet graft function and survival. The inadvertent transplantation of islets to recipients presensitized against donor-specific HLA can result in rapid islet graft loss (16), and the presence of a pretransplant class I PRA >15% was associated with islet graft failure that was largely explained by the presence of islet donor-specific antibodies (4). In the present report, we did not find a significant relationship between a positive PRA pretransplant and subsequent islet graft failure; however, donor specificity data were not available. Most centers now perform prospective cross-matches, and in the cases of positive PRA pretransplant, avoid unacceptable antigens by excluding donors with the same specificities as anti-HLA antibodies regardless of the cross-match (21).
De novo HLA sensitization may also be one of the factors that are associated with loss of islet graft function. The adverse effect of HLA sensitization in recipients of kidney (12,14), heart (3), and lung (23) transplants has been reported earlier. Among pancreas transplant recipients (simultaneous pancreas kidney), 21% developed anti-HLA antibodies (6), and the presence of anti-HLA antibodies correlated with poor pancreas graft survival. Islet grafts may be more “sensitive” to humoral immunity when compared to solid organ grafts and may be more rapidly destroyed in the presence of alloantibody (17). In the first report associating islet graft loss with sensitization to mismatched donor HLAs (18), patients who developed anti-HLA antibodies had significantly impaired islet graft function when compared to nonsensitized patients. Subsequent reports have also demonstrated a temporal relationship with the development of islet donor specific anti-HLA antibodies and subsequent islet graft functional deterioration and failure (15,20), although there have also been cases reported where the development of donor specific anti-HLA antibodies was not related to any deterioration in islet graft function (2) or long-term survival (7). Nonetheless, based on results from a larger pool of patients, Campbell et al. (5) have reported that 26 out of a total of 98 (27%) islet transplant patients developed de novo donor specific anti-HLA antibodies while on maintenance immunosuppression that was associated with significantly worse islet graft function when compared to the patients without sensitization. The data reported here extend these findings by demonstrating a significantly increased risk for islet graft failure following the development of a PRA ≥20% posttransplant.
Our data also confirm earlier reports (5,7,9,21) of increased HLA sensitization among patients with failed islet grafts who discontinued their immunosuppression. Development of HLA sensitization among these patients remains a concern because of the potentially prolonged waiting time for subsequent organ transplants (e.g., pancreas or kidney), if needed. A final consideration is that the type of immunosuppression may have a major effect on anti-HLA antibody production. While the Edmonton group showed that 27% of patients treated with glucocorticoid-free immunosuppression develop de novo anti-HLA antibodies (5), the Geneva group (9) demonstrated that 0/27 patients receiving low-dose glucocorticoids as part of their immunosuppression for previous or simultaneous kidney transplants developed de novo anti-HLA antibodies, whereas 2/8 patients receiving Edmonton immunosuppression and 2/3 patients during withdrawal of immunosuppression became sensitized. These results suggest that glucocorticoid-free immunosuppression may not sufficiently control the development of alloimmune response to transplanted islets.
In summary, the present report demonstrates that the development of anti-HLA class I antibodies posttransplant represents a significant risk for subsequent islet graft failure.
Footnotes
Acknowledgments
Expert comments by Dr. T. Mohanakumar, Washington University in St, Louis are gratefully acknowledged. The authors declare no conflicts of interest on this study.
The Collaborative Islet Transplant Registry (CITR) is funded by a grant from the National Institute of Diabetes, Digestive and Kidney Diseases, National Institute of Health, USA and by a supplemental grant from the Juvenile Diabetes Research Foundation International. Additional data are made available through cooperative agreements with the US United Network for Organ Sharing and the Administrative and Bioinformatics Coordinating Center of the City of Hope. The following institutions and individuals contributed to the reporting and/or analysis of the data included in this manuscript.
