Abstract
The aim of the current study was to characterize the anti-HLA antibodies before and after pancreatic islet or pancreas transplantation. We assessed the risk of anti-donor-specific antibody (DSA) sensitization in a single-center, retrospective clinical study at Geneva University Hospital. Data regarding clinical characteristics, graft outcome, HLA mismatch, donor HLA immunogenicity, and anti-HLA antibody characteristics were collected. Between January 2008 and July 2014, 18 patients received islet transplants, and 26 patients received a pancreas transplant. Eleven out of 18 patients (61.1%) in the islet group and 12 out of 26 patients (46.2%) in the pancreas group had anti-HLA antibodies. Six patients (33.3%) developed DSAs against HLA of the islets, and 10 patients (38.4%) developed DSAs against HLA of the pancreas. Most of the DSAs were at a low level. Several parameters such as gender, number of times cells were transplanted, HLA mismatch, eplet mismatch and PIRCHE-II numbers, rejection, and infection were analyzed. Only the number of PIRCHE-II was associated with the development of anti-HLA class II de novo DSAs. Overall, the development of de novo DSAs did not influence graft survival as estimated by insulin independence. Our results indicated that pretransplant DSAs at low levels do not restrict islet or pancreas transplantation [especially islet transplantation (27.8% vs. 15.4.%)]. De novo DSAs do occur at a similar rate in both pancreas and islet transplant recipients (mainly of class II), and the immunogenicity of donor HLA is a parameter that should be taken into consideration. When combined with an immunosuppressive regimen and close follow-up, development of low levels of DSAs was not found to result in reduced graft survival or graft function in the current study.
Introduction
Isolated pancreas or simultaneous kidney–pancreas transplantation is a widely accepted therapy to treat type 1 diabetes mellitus (T1D) with or without end-stage renal failure for selected patients. Islet transplantation is an alternative therapy that is emerging to achieve insulin independence or improve glycemic control for patients with “brittle” T1D; “brittle” describes a severe form of diabetes characterized by fluctuation of blood sugar levels that can affect quality of life and lead to hospitalization. Islet transplantation often requires several infusions from different donors. Both islet and pancreas transplantation procedures expose the recipients to multiple human leukocyte antigen (HLA) mismatches, which is a risk factor for the development of anti-HLA antibodies. Pretransplantation anti-HLA antibodies are associated with acute antibody-mediated rejection that could lead to rapid graft loss or reduced long-term graft survival (1,18). The development of anti-HLA antibodies after transplantation (de novo anti-HLA antibodies) is also associated with reduced graft survival. In isolated pancreas or simultaneous kidney–pancreas transplantation and in islet transplantation, several reports have previously demonstrated that the development of anti-HLA antibodies after transplantation is a risk factor for graft function and graft survival (1,2,10,14). However, these studies usually include several centers with different immunosuppressive strategies and do not compare both procedures (pancreas vs. islet transplantation), with regard to the presence of anti-HLA antibodies, as an independent risk factor for graft function and graft survival.
In this single-center study, we have compared recipients of islet or pancreas transplantation and characterized the anti-HLA antibody specificities before and after transplantation to assess the specific risk of anti-HLA sensitization with a focus on the immunogenicity of donor HLA through HLA mismatches, HLA eplet determination, and predicted indirectly recognizable HLA epitopes (PIRCHE).
Materials and Methods
Patient Cohort
This study is a single-center, retrospective clinical study. It included all recipients of islet or pancreas transplants (including combined transplantation, i.e., kidney–pancreas, kidney–lung, etc.) performed between January 2008 and July 2014 at Geneva University Hospital. We collected demographic and graft outcome data. Data were collected regarding pancreas or islet rejection episodes, insulin independence status, mortality, and fasting C peptide and hemoglobin A1c (HbA1c) levels. The ethical committee of the Geneva University Hospital approved the study (No. 6-208), which is in accordance with the regulations of the Geneva University Hospital.
HLA Antibody Analysis
Before transplantation, the monitoring of anti-HLA antibodies was performed when the patients were registered on the transplantation waiting list, then three times a year until transplantation, and finally, on the day of the transplantation. Subsequent detections were performed at hospital discharge, every 6 months during the first year after the transplant procedure and once a year afterward, and whenever major clinical events such as rejection or infection occurred. Sera of the patients were analyzed for the presence of anti-HLA class I and class II antibodies by solid phase assays on Luminex, using the LABscreen Mix assay for HLA class I and HLA class II following the recommendations of the manufacturer (One Lambda, Thermo Fisher Scientific, Canoga Park, CA, USA). For all positive individuals, sera collected before and after transplantation were subsequently tested for anti-HLA class I- and class II-specific antibodies using the Luminex single-antigen beads (One Lambda). Briefly, color-coded microspheres coated with the major HLA class I and II antigens were incubated with 10 μl of serum for 30 min at room temperature (RT) in the dark. After three washes, samples were incubated with 100 μl of 1:100 phycoerythrin-conjugated goat anti-human immunoglobulin G (IgG; One Lambda). Finally, after two washes, fluorescence signal intensity for each microsphere was measured using a LABScan 100 flow analyzer (One Lambda). The cutoff for positive samples was the normalized background (NBG) ratio recommended by the manufacturer, which was calculated by HLA Fusion software (One Lambda). A mean fluorescence intensity (MFI) above 1,000 was considered as positive. Donor-specific antigens were classified as either immunogenic HLA or nonimmunogenic HLA based on the presence of anti-donor-specific antibodies (DSAs) in the recipient. Immunogenic HLA was further subdivided into preformed DSAs and de novo DSAs.
HLA Typing
HLA-A, -B,-C, -DRB1, and -DQB1 typing of the patients and their donors was performed either by polymerase chain reaction using sequence-specific oligonucleotide (PCR-SSO) DNA typing (LABType HD; One Lambda) or PCR using sequence-specific primer (PCR-SSP) typing (Olerup, Vienna, Austria).
HLA Eplet Determination
The HLA matchmaker program was used to calculate and summate the number of mismatched eplets between the recipient and pancreas or islet donors at the HLA-A, -B, -DRB1, and -DQB1 loci (www.epitopes.net). Highresolution determination was deduced from the HLA medium resolution typing performed by SSO and SSP with the most frequent allele and haplotype for the given population of recipient and donors from Switzerland.
Identification of Predicted Indirectly Recognizable HLA Epitopes (PIRCHE)
For all mismatched HLA-A, -B, -DRB1, and -DQB1 molecules of the donor, we determined the number of donor-derived epitopes that can be presented by recipient HLA-DRB1 molecules (PIRCHE-II), as described previously (5). Briefly, we predicted at which position a mismatched HLA-derived peptide may bind to the binding groove of recipient HLA-DRB1 by using the netMHCII-pan-3.0 algorithm (8). The algorithm predicts the binding affinity of the nonameric binding core of the mismatched HLA-derived peptide to recipient HLA-DRB1 mole cules, considering IC50 binding values <1,000 nM as relevant HLA-DRB1 binders. The relevant HLA-DRB1 binders that differed at least one amino acid from the recipient HLA were classified as PIRCHE-II. Only unique donor-derived peptide–HLA complexes were considered PIRCHE-II. In our analysis, the major part of the HLA amino acid sequence was included; only leader peptide sequences were excluded from the HLA amino acid sequence. The PIRCHE-II algorithm is available online via https://www.pirche.org.
Statistical Analysis
Data are represented using median and interquartile range. The Mann–Whitney test was used to compare quantitative data. Graft survival was assessed by Kaplan–Meier curves and compared with the log-rank test. Mann–Whitney
Results
Study Population
Forty-five islet or pancreas transplants were performed in 43 patients between January 1, 2008 and July 1, 2014. Eighteen patients received islet transplants, and 26 received a pancreas transplant (among whom 1 patient received two pancreas transplantations and 1 patient received both pancreas and islet transplantations). All patients were transplanted with a negative complement-dependent cytotoxicity (CDC) crossmatch. Patient characteristics are shown in Table 1. Among the 27 pancreas transplantations, 18 (66.7%) were simultaneous pancreas–kidney (SPK) transplantations, 6 (22.2%) were pancreas-after-kidney (PAK) transplantations, 2 (7.4%) were pancreas transplant alone (PTA), and 1 (3.7%) patient received a simultaneous pancreas–intestine (SPI) transplant. Except for one patient who received basiliximab (Novartis, Basel, Switzerland), each patient (96.3%) received anti-thymocyte globulin (ATG; Sanofi, Paris, France) as part of the induction therapy. Maintenance therapy was composed of short-term steroids for each patient, calcineurin inhibitors for 26 patients (100%), and antimetabolites for 21 out of 26 patients (80.8%).
Characteristics

Among the 18 islet-transplanted patients, 6 (33.3%) received an islet transplant alone (ITA), 5 (25.8%) a simultaneous islet–kidney (SIK) transplant, 4 (22.2%) an islet-after-kidney (IAK) transplant, 2 (11.1%) an islet-after-lung# or -liver (IAL) transplant, and 1 (5.6%) a simultaneous islet–lung–liver (SILL) transplant. The first transplant induction included ATG therapy for 13 patients (72.2%) or basiliximab for 2 patients (11.1%). Maintenance therapy included steroid for 3 patients (16.7%), calcineurin inhibitors for 13 patients (72.2%), and antimetabolite for 16 patients (88.9%).
Anti-HLA Antibodies
Eleven out of 18 islet transplant recipients (61.1%) and 12 out of 26 pancreas transplant recipients (46.2%) had pretransplant anti-HLA antibodies (Fig 1). Among the islet transplant recipients, 5 patients (27.8%) were transplanted across DSAs (3 recipients across anti-HLA class I antibodies only, 1 recipient across anti-HLA class II antibodies only, and 1 recipient across anti-HLA class I and II antibodies). Among the pancreas transplant recipients, 4 patients (15.4%) were transplanted across DSAs (3 recipients across anti-HLA class I antibodies only and 1 recipient across anti-HLA class II antibodies only). Except for 1 patient in the pancreas transplant group, all preformed DSAs were still detected after transplantation. The specificities and the MFI of pretransplant DSAs are indicated in Table 2. Six patients (33.3%) developed de novo DSAs directed against HLA antigens of the islets, and 10 patients (38.4%) developed DSA antibodies against HLA antigens of the pancreas. The specificities and the MFI of de novo DSAs are indicated in Table 2.

Risk of anti-HLA sensitization after islet or pancreas transplant.
First Mean Fluorescence Intensity of Donor-Specific Antibodies (DSAs)

Preformed are in italics. *Transplant complicated by rejection(s).
Clinical Parameters in Relationship with the Occurrence of De Novo DSAs
The following parameters were included in a statistical analysis to establish factors that could predict the development of anti-HLA antibodies after transplantation: recipient gender, recipient age, number of infections occurring after transplantation, number of rejections (including cellular and humoral rejection), HLA mismatch between donors and recipients, eplet mismatch for HLA class I and/or class II, and immunosuppressive therapy (Table 3). There were no statistically significant differences for these factors when comparing patients who developed de novo DSA to those who did not, neither in the islet-transplanted patients nor in the pancreas-transplanted patients. As shown in Figure 2, the development of de novo DSAs did not influence graft survival as estimated by insulin independence.

Pancreas and islet graft survival among patients with and without donor-specific antibodies.
Evaluation of Common Risk Factors of Alloimmunization After Islet or Pancreas Transplant

Values are expressed as median (range).
De Novo Anti-HLA Class II DSA Development Is PIRCHE-II Dependent
In order to determine whether anti-HLA antibody formation after islet and pancreas transplantation is related to the number of T helper epitopes, we evaluated the number of PIRCHE-II in the two transplantation settings. First, we determined whether PIRCHE-II numbers differed between pancreas transplantation and islet transplantation. Since the de novo DSA group is rather small, the number of PIRCHE-II in the nonimmunogenic HLA group was analyzed. For nonimmunogenic anti-HLA class I (Fig. 3A), the PIRCHE-II numbers were higher for islet transplantation compared to pancreas transplantation (

Comparison of the number of PIRCHE-II for nonimmunogenic HLA class I (A) and nonimmunogenic HLA class II (B) between pancreas transplantation and islet transplantation groups. (C) The nonimmunogenic HLA class I group was further subdivided into single transplantations or into transplantations combined with another organ type. The reported
To investigate whether this difference is due to simultaneous transplantations, pancreas and islet transplantations were further subdivided into pancreas transplantation alone, pancreas transplantation combined with another type of organ transplantation, islet transplantation alone, and islet transplantation combined with another type of organ transplantation (Fig. 3C). For both pancreas transplantation and islet transplantation, no difference was observed between a single transplantation and a combined transplantation (
To confirm that anti-HLA antibody formation is not related to mismatched eplets as determined by the HLA matchmaker program, we compared the de novo DSA group and the nonimmunogenic HLA group. For both anti-HLA class I and anti-HLA class II, the number of mismatched eplets did not differ between the de novo DSA group and the nonimmunogenic HLA group (
Finally, we compared the number of PIRCHE-II between HLA mismatches to which de novo antibodies were formed and HLA mismatches to which no antibodies were formed. For HLA class I (Fig. 4A), the number of PIRCHE-II was comparable between the de novo DSA group and the nonimmunogenic HLA group (
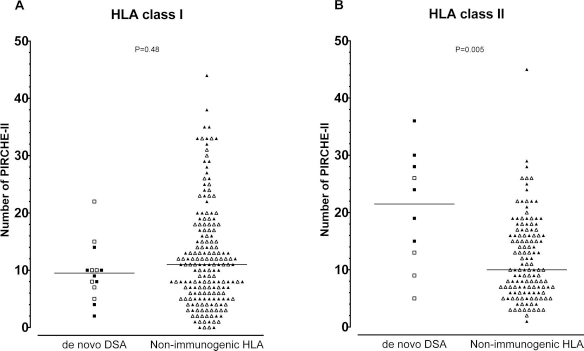
Comparison of the number of PIRCHE-II between HLA mismatches to which de novo DSAs are formed (de novo DSAs) and HLA mismatches to which no anti-HLA antibodies are formed (nonimmunogenic HLA). (A) For HLA class I mismatches, the PIRCHE-II numbers do not differ between de novo DSAs and nonimmunogenic HLA. (B) For HLA class II mismatches, de novo DSAs contain a higher number of PIRCHE-II compared to nonimmunogenic HLA. For (A), the close symbols represent HLA-A, and the open symbols represent HLA-B. For (B), the closed symbols represent HLA-DQB1, and the open symbols represent HLA-DRB1. The reported
Discussion
In this study we compared the sensitization situations in our cohort of islets and pancreas transplant recipients. Our data indicate first, that, before transplantation, islet transplant recipients had more DSAs (preformed anti-HLA DSA) than pancreas transplant recipients (27.8% vs. 15.4%). Second, the development of DSAs posttransplantation was similar in both groups (33.3% in islets vs. 38.4% in pancreas). Third, the presence of pretransplant DSAs and the de novo development of posttransplant DSAs were not associated with a reduced graft survival and a reduced graft function. Finally, we found that the de novo development of posttransplant anti-HLA class II antibodies is related to the number of PIRCHE-II.
In kidney transplantation, DSAs are associated with antibody-mediated rejection (AMR) and a reduced long-term graft survival (16,19). An adverse effect of DSAs developed after other organ transplant modalities such as heart (17) and lung (6) transplants has also been reported. Among pancreas transplant recipients, the presence of anti-HLA antibodies correlated with poor pancreas graft survival (2,11). In 1997, Olack et al. already suggest that anti-HLA sensitization has a significant impact on islet graft function when compared to nonsensitized patients (14). Other reports have also demonstrated an association between DSAs and islet graft deterioration and failure (1). More recently, the collaborative Islet Transplant Registry published the largest cohort of 303 recipients of islet transplantation. In this study, HLA class I sensitization defined by the panel reactive antibodies (% of PRAs) was associated with significantly worse islet graft function when compared to the patients without sensitization (13). However, other reports have indicated that DSAs could not be deleterious for islet function (3) or pancreas function (12). We previously concluded that the addition of islets does not represent a risk factor for the development of anti-HLA antibodies when combined with a kidney transplantation (4). Our current data confirmed this first publication and do not detect a significant difference between islet and pancreas transplantation.
Our data seem to be in contradiction with several reports that show a clear association between increased HLA sensitization among patients with failed islet grafts (1,14) and with worse graft survival in pancreas transplantation (2,11). However, we have to be very cautious when comparing different studies, published at different times, with single or multiple centers, with different immunosuppression protocols, and with different technology with regard to anti-HLA antibody analysis. A major limitation of our work is the small number of patients who were included, which limits conclusions about transplant outcome.
In the present study we evaluated islets and pancreas transplantations under immunosuppression. In islet transplantation, the development of anti-HLA antibodies is strongly associated with immunosuppression withdrawal, which frequently occurs in ITA when the graft is not functional anymore (1,4). In the islet group, a majority of transplants (66.6%) were combined with other organs, which precludes any minimization or withdrawal approach with regard to immunosuppression. Therefore, intensification of the immunosuppressive treatment might be more common in our cohort than in previously reported studies. This could also explain why we did not find statistically significant differences in terms of insulin independence in the follow-up between patients who were DSA positive and patients who were DSA negative.
Anti-HLA antibody determination has been strongly modified by the development of the single-antigen bead assay [or solid-phase assay (SPA)]. This technology is very sensitive, and a significant number of DSAs detected by SPA are not clinically relevant. It is still difficult to discriminate between clinically relevant and irrelevant DSAs. Factors such as MFI, a semiquantitative measure of the anti-HLA antibody titer, complement-binding capability of DSAs, or the DSA specificity against distinct HLA classes and loci have been considered to stratify the risk of sensitization and clinical events. Several studies still determine immunization with the CDC technology (i.e., PRAs alone) (13). Analysis based on PRA alone could lead to different interpretation, and as this technique is less sensitive, any positive PRA is associated with high anti-HLA antibody titers.
With the SPA assay, between 20% and 30% of patients develop de novo DSAs posttransplant (7). In the present study, the rate of de novo DSAs in both islet and pancreas transplantation is slightly higher than in previously reported studies. This difference is mainly due to the fact that the level of clinically relevant DSAs in our center is at MFI 1,000, which is lower than those used in previously reported studies. As displayed in Table 2, most DSAs have an MFI below 2,500 (19/24) for HLA class I and below 5,000 for HLA class II (15/17). Only one patient had a DSA (anti-B8) above MFI 10,000 in the islet cohort without clinical problems until now. The role of anti-HLA class II remains controversial: pancreas and islet endocrine tissues do not express HLA class II molecules (9), but cytokines such as interferon-γ (IFN-γ) could have induced major histocompatibility complex (MHC) class II expression in any tissue. Any clinical events such as rejection or infection could induce the expression of MHC class II in the transplant tissue and favored the binding of DSAs against MHC class II. In our cohort, the rate of rejection was low (5.6% in the islets group and 11% in the pancreas group), and the number of infections was limited and treated promptly.
Finally, this study highlights that the number of PIRCHE-II is related to de novo DSAs in islet and pancreas transplantation. The number of PIRCHE-II has already been associated with de novo DSA development after kidney transplantation (15) and pregnancy (5), but it is the first time that it is reported in pancreas and islet transplantation. Interestingly, only HLA class II antibodies were found to be related to the number of PIRCHE-II, and the number of PIRCHE-II for nonimmunogenic HLA class I antibodies was higher for islet transplantation. The latter observation suggests different immune tolerance mechanisms between islet and pancreas transplantation settings. Such differences could be partly explained by the different environment endured by the grafts after transplantation due to different graft localizations.
In conclusion, we believe that evaluation for the presence of anti-HLA antibodies before transplantation by SPA is critical, and, depending on the level of DSA, islet or pancreas transplantation across DSAs seems to be possible in some situations with an appropriate immunosuppression protocol. Before transplantation, evaluation of the number of PIRCHE-II could be considered to assess the risk for de novo anti-HLA class II DSA development. After transplantation, regular anti-HLA antibody monitoring should be part of the routine follow-up. Detection of de novo DSAs is critical after pancreas or islet transplantation for patients to take adequate decisions with regard to IS modification.
Footnotes
Acknowledgment
