Abstract
Induced pluripotent stem (iPS) cells are a type of undifferentiated cell that can be obtained from differentiated cells and have the pluripotent potential to differentiate into the musculoskeletal system, the myocardium, vascular endothelial cells, neurons, and hepatocytes. We therefore cultured mouse iPS cells in a DMEM containing 15% FBS, 10−7 M dexamethasone, 10 mM β-glycerophosphate, and 50 μg/ml ascorbic acid for 3 weeks, in order to induce bone differentiation, and studied the expression of the bone differentiation markers Runx2 and osteocalcin using RT-PCR in a time-dependent manner. Osteocalcin, a bone differentiation marker in bone formation, exhibited the highest expression in the third week. In addition, the deposition of calcium nodules was observed using Alizarin red S staining. iPS cells cultured for bone differentiation were transplanted into severe combined immunodeficiency (SCID) mice, and the osteogenic potential exhibited after 4 weeks was studied. When bone differentiation-induced iPS cells were transplanted into SCID mice, bone formation was confirmed in soft X-ray images and tissue specimens. However, teratoma formation was confirmed in 20% of the transplanted models. When mouse iPS cells were treated with irradiation of 2 Gray (Gy) prior to transplantation, teratoma formation was inhibited. When mouse iPS cells treated in a likewise manner were xenotransplanted into rats, bone formation was confirmed but teratoma formation was not observed. It is believed that irradiation before transplantation is an effective way to inhibit teratoma formation.
Introduction
Induced pluripotent stem (iPS) cells are undifferentiated cells induced from differentiated cells with genetic and morphological characteristics equal to those derived from embryonic stem (ES) cells (32). They have the potential to differentiate into various cells in vitro (13). Recent studies have demonstrated that ES cells cultured in an appropriate environment differentiate into osteogenic cells (5). As such, it is expected that the potential use of iPS cells will increase by differentiating them into various cells for regenerative purposes.
In the field of orthopedics, many materials (e.g., auto-bone graft, allobone graft, artificial bone, or sometimes bone cement) are used as bone fill in significant bone defect that results from aging, disease, or trauma. The purpose of bone fill is to augment bone intensity and to increase the regenerative potential around the bone defect. Autobone graft has the best property for bone fill (8), although there are downsides such as a limited bone harvest and invasion associated with bone harvest (3). Allobone graft has a risk of infection and its osteoinductive potential is limited because osteogenic cells inside it are killed by treatments before transplantation. Almost all artificial bones are ceramics, which have enough strength as bone fill. Therefore, they have no osteoinductive properties by themselves. In order to increase their bone-regenerative potentials, various cells and growth factors, such as mesenchymal stem cells (MSCs), osteoblasts, bone morphogenic protein, etc., were used with them. If creation were possible from autologous cells and clumps of cells that can facilitate bone formation or that can fill the bone void, it would surely expand the scope of regenerative medicine in the field of orthopedics.
We decided to study bone differentiation potential using mouse iPS cells. However, the problem with iPS cells with high differentiation potential is that they form teratomas after being transplanted in vitro (35). It is quite important to develop effective ways to induce bone differentiation and, at the same, develop techniques to prevent teratomas. This time, we studied the bone differentiation of mouse iPS cells in vitro and conducted an evaluation of bone regeneration and the frequency of occurrence of teratomas by transplanting bone differentiation-induced iPS cells into mouse models with calvarial defects. We also studied the effect of xenotransplantation of mouse iPS cells into Wistar rat models with calvarial defects.
Materials and Methods
iPS Cell Culture
We used mouse iPS cells of 10th passage supplied by Kyoto University (Cell No. APS0001, Cell name iPS-MEF-Ng-20D-17, Lot No. 006), which we purchased from RIKEN. They were cultured on mitomycin C (BIOMOL, PA, USA)-treated SNL cells (DS Pharma Biomedical, Osaka, Japan) used as feeder cells. Dulbecco's modified Eagle medium (DMEM; Nacalai Tesque, Kyoto, Japan) adding 15% fetal bovine serum (FBS) (Gibco), 50 U/ml penicillin and 50 mg/ml streptomycin (Invitrogen), 2 mM l-glutamine (Invitrogen), 1 × 10−4 nonessential amino acids (NEAA) (Invitrogen), and 1 × 10−4 M 2-mercaptoethanol (Invitrogen) was used. The medium was changed every 1 or 2 days. The passage of iPS cells was performed every 3 days. All subcultures were performed in a 37°C incubator with 5% CO2.
Osteogenic Induction of iPS Cells
Our protocol of osteogenic differentiation of iPS cells was two staged (Fig. 1). The first stage was the formation of embryoid bodies (EB) and the second stage was the culturing in osteogenic medium.
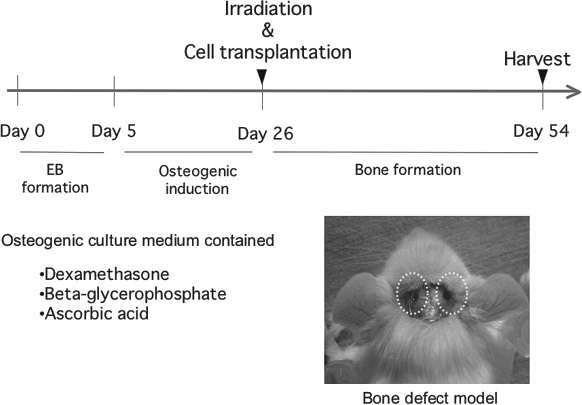
Specimens in the experiment to induce bone differentiation of induced pluripotent stem (iPS) cells. An experimental design of the present experiment is shown. EB, embryoid bodies.
EB Formation by iPS Cells
For effective induction, iPS cells were seeded on an ultralow attachment six-well plate at a density of 1 × 105 cells per well and were grown for 5 days in suspension cultures. Nodular cell clumps in suspension cultures were confirmed.
Osteogenic Induction by Culturing in Osteogenic Medium
After EB formation for 5 days, trypsinized EB cells were seeded on gelatine-coated six-well culture plates at a density of 1 × 105 cells/well and were started culturing in osteogenic medium. The osteogenic medium was DMEM supplemented with 10% fetal bovine serum, 10 nM dexamethasone, 50 mg/L ascorbic acid (Wako, Osaka, Japan), 10 mM β-glycerophosphate (Sigma-Aldrich, Tokyo, Japan), 50 U/ml penicillin, and 50 mg/ml streptomycin (Sigma-Aldrich) as previously described (31). The medium was changed every 3 or 4 days.
The Potential of Creating Calcium Nodules of iPS Cells In Vitro
On the 14th and 21st days of osteogenic induction, Alizarin red staining was performed. After 70% ethanol fixation for 10 min, they were washed in distilled water (DW) two times and absolute ethanol was added to remove water. They were stained with a 1% solution of pH 4.2 conditioned Alizarin Red S (Nacalai Tesque) for 2 min. Excess stain was washed off in tap water and the cultures were then air-dried. More than 10 photographs of each sample were taken using a phase-contrast microscope. The red staining area that contained calcium nodules was measured by Image-J.
The Expression of Bone-Related Genes of iPS Cells In Vitro
On the 7th, 14th, and 21st days, according to the procedure, total RNA was extracted using RNasey easy mini Kit (QIAGEN Science, Tokyo, Japan) and RT-PCR was performed with i-cyclor (Bio-Rad, Tokyo, Japan) to study the expression of runt-related transcription factor 2 (Runx2) and osteocalcin. One microgram of total RNA was transcripted with River-Tra Ace (Toyobo, Tokyo, Japan), oligo(dT)20 (Toyobo), RNase inhibitor (Toyobo), 5x RT buffer (Toyobo), and dNTP mixture (Toyobo). cDNA was amplified with Takara Taq (Mg2+ plus Buffer) (TaKaRa, Ootsu, Japan), 10x PCR buffer (TaKaRa), and dNTP mixture (TaKaRa). After a single incubation at 94°C for 5 min to activate the polymerase enzyme, samples were run at the following setting: 94°C for 30 s, 60°C for 30 s, followed by 72°C for 30 s for a total of 30 cycles; and final elongation at 72°C for 7 min. The PCR analysis was conducted with the following primers: Runx2, sense strands 5′-agaaggcacagacagaagc-3′ and antisense strands 5′-ctggaagggtccactctg-3′; osteocalcin, sense strands 5′-ttctgctcactctgctgacc-3′ and antisense strands 5′-accttattgccctcctgctt-3′; glyceraldehyde 3-phosphate dehydrogenase (GAPDH), sense strands 5′-tgcac caccaactgcttagc-3′ and antisense strands 5′-ggcatggactgt ggtcatgag-3′). Electrophoresis of PCR was done with ethidium bromide with added 2% agarose gel.
Irradiation of iPS Cells
In order to inhibit teratoma formation by iPS cells transplanted into models with calvarial defects, iPS cells were irradiated after being bone differentiated. Irradiation was performed at the irradiation facility attached to the animal control center using MBR-1520R (Hitachi Medico, Tokyo, Japan). iPS cells, plated on a gelatin-coated six-well culture plate at a density of 5 × 105 cells/well to induce bone differentiation for 3 weeks, were irradiated with doses of 1, 2, and 4 Gy, in order to study cell viability by thiazolyl blue tetrazolium bromide (MTT) assay. The expression of bone-related genes was assessed by RT-PCR 1 week after radiation treatment.
In Vivo Transplantation Experiments
All experiments were performed in accordance with Okayama University ethical committee and were conducted at Okayama University Animal Control Center in compliance with the Guidelines for Proper Conduct of Animal Experiments.
Two Bone Defect Models
In order to examine whether iPS cells induce bone formation, two different bone defect models were prepared. Eight-week-old adult SCID mice (Nippon CLEA, Tokyo, Japan) and 10-week-old Wister rats (Nippon CLEA, Tokyo, Japan) were used. A critical calvarial bone defect model was used as previously described in detail (7). Under general anesthesia, the scalp was opened sharply transversely to midline to expose the calvaria. Periosteal tissue over the parietal bone was removed. Two calvarial defects of 3 mm diameter for mouse and 5 mm diameter for rat were made in both parietal bones with diamond-bit H-282 (Sunflex, Osaka, Japan) revolving by Joy-Robo Pro H-027 (Sunflex, Osaka, Japan), as we conducted previously (26). The bone defects were washed with saline to remove bone powders and blood, and then transplanted samples. After the cell transplantation, the skin was sutured with 4–0 nylon sutures.
Cell Transplantation to Bone Defects
After EB formation, mouse iPS cells were divided into the following three groups and cultured in the different conditions for the transplantation experiment. Group 1 (G1): iPS cells cultured in regular medium. iPS cells were plated on a gelatin-coated six-well culture plate at a density of 1 × 105 cells per well to create a further 21-day culture in the regular medium without growth factors, DMEM with added 10% FBS, 50 U/ml penicillin, and 50 mg/ml streptomycin. Group 2 (G2): iPS cells cultured in osteogenic medium. iPS cells were plated on a gelatin-coated six-well culture plate at a density of 1 × 105 cells/well and cultured for 21 days in an osteoinductive medium. Group 3 (G3): irradiated iPS cells after osteogenic differentiation. Induction of bone differentiation was carried out as with group 2, followed by irradiation with doses of 2 Gy.
Examination of the Osteogenic Potential of iPS Cells Using Mouse Models with Calvarial Defects
In order to make it easier to compare the bone formation between groups, two different groups of cells were transplanted to each bone defect (G1 vs. G2, G1 vs. G3, and G2 vs. G3). The number of transplanted cells was 1 × 106 cells per defect. Five models for each comparison were made and therefore 10 specimens for each group were analyzed.
Xenotransplantation of iPS Cells Using Rat Models with Calvarial Defects
The main purpose of this experiment was to study the prevention of teratoma formation of mouse iPS cells by radiation treatment and subsequent xenotransplantation. Irradiated iPS cells (3 × 106) after osteogenic differentiation (G3 cells, as mentioned above) were transplanted into the left side of calvarial defects (5 mm diameter) in rats. The defect in the right side was irrigated by PBS containing no cells and served as a control.
Radiological and Histological Examination
At 4 weeks following transplantation, in order to evaluate bone defects, models were given an overdose of anesthesia before their skulls were extracted for soft X-ray radiographs using Softex type CMB (Softex, Tokyo, Japan). Having been fixed in 4% formaldehyde for 24 h, the extracted skulls were placed in ethylenediamine tetraacetic acid (EDTA) for 7 days for demineralization, then wrapped in paraffin, and thinly sectioned at 6 μm for hematoxylin and eosin (H&E) staining. Bone density inside bone defect was measured using Image-J. First, we measured the density of normal cortical bones as a control. Then the density inside of defects was compared with control. Teratoma formation was confirmed by histological findings.
Statistical Analysis
The t-test was used to statistically process the mean value. Multiple comparisons were performed by Bonferroni test. A value of p < 0.05 was set as an indication of significant difference.
Results
In Vitro Potential of iPS Cells to Induce Bone Differentiation
Bone differentiation was induced for iPS cells for 3 weeks and then the cells were irradiated with doses of 1, 2, and 4 Gy. The MTT assay was performed for iPS cell viability 3 days after irradiation. The MTT values were 0.63 ± 0.11 for 1 Gy, 0.52 ± 0.03 for 2 Gy, and 0.31 ± 0.06 for 4 Gy. Irradiation of 4 Gy markedly induced cell injury.
The extent of calcium nodule formation by iPS cells during bone differentiation induction was studied using Alizarin red staining. The rate of staining in calcium nodules was 10 ± 3% in the first week of bone differentiation induction, 40 ± 3% in the second week, and 80 ± 3% in the third week, thereby demonstrating an increase in the number of calcified nodules in a time-dependent manner. The rate of staining in calcium nodules was 85 ± 5% in cases in which irradiation was performed after the third week of bone differentiation induction (Fig. 2). This was comparable to the results of the nonirradiation cases. Gene expression of the bone differentiation induction markers Runx2 and osteocalcin, shown by RT-PCR, confirmed the expression of Runx2, an initial stage marker of osteoblast differentiation, and the expression of osteocalcin, late stage markers of osteoblast differentiation. Following confirmation of the enforced expression of an initial stage marker Runx2, the expression of osteocalcin, which demonstrates differentiation into osteoblasts, increased in a time-dependent manner (Fig. 3).

Examination of the extent of calcified nodules by Alizarin red staining. Bone differentiation of iPS cells was performed for 3 weeks. The rate of staining in calcified nodules was 10 ± 3% in the first week of bone differentiation induction, 40 ± 3% in the second week, and 80 ± 3% in the third week, thereby demonstrating an increase in the number of calcified nodules in a time-dependent manner. The rate of staining in calcified nodules of irradiated cells was 85 ± 5%, compared to nonirradiated cells (A: first week of bone differentiation induction; B: second week of bone differentiation induction; C: third week of bone differentiation induction; D: fourth week of bone differentiation induction for 3 weeks, irradiation, and culture for a week). Scale bars: 200 μm.

Gene expression of bone differentiation markers during bone differentiation of iPS cells. The gene expression of runt-related transcription factor 2 (Runx2), an initial stage marker of bone differentiation, was confirmed. The expression of osteocalcin, an osteoblast expression marker, increased in a time-dependent manner. The value of Runx2, an initial stage marker of bone differentiation, was highest in the first week, and gradually decreased thereafter. Comparison between the irradiation group and the nonirradiation group found no differences after the third week of bone differentiation. Glyceraldehyde 3-phospahte dehydrogenase (GAPDH) was used as a control.
Transplantation of Bone Differentiation-Induced iPS Cells Facilitates Regeneration in Mouse Models of Calvarial Defects
Soft X-ray images demonstrated new bone formation in bone defects in the group of models with transplantation of bone differentiation induced iPS cells (G2 and G3) (Fig. 4A). Recovery of bone defects was significantly better in this group than in the group of models with transplantation of iPS cells that had not undergone bone differentiation induction (G1) (G1 vs. G2, p = 0.046; G1 vs. G3, p = 0.040; G2 vs. G3, p = 0.839) (Fig. 4B). H&E staining also showed, in the group of models with transplantation of bone differentiation-induced iPS cells, new bone formation not only in the periphery of the bone defects but also in the center (Fig. 5). On the other hand, in the group of models with transplantation iPS cells that had not undergone bone differentiation induction (G1), although bone formation was observed in the periphery of the bone defects, bone formation in the centre was found to be insufficient (Fig. 5). Further, the expression of Runx2 and osteocalcin shown by RT-PCR was significantly higher in the group of models with transplantation of bone differentiation induced iPS cells. However, tumor formation was visually observed in both G1 and G2 (Fig. 6). The results from H&E staining demonstrated that these tumors consisted of pseudo-glandular, dermoid, and chondroid structures; in other words, teratomas differentiated into triploblastic cells.

Soft X-ray evaluation of new bones after cell transplantation. (A) Bars indicate 10 mm in soft X-ray photographs. (B) The bone density relative to the normal bone area is shown. Compared with G1, new bone formation was significantly better in G2 and G3. In G3 and G2, there was no difference in the extent of new bone formation (G1: iPS cells that were not bone differentiation induced, G2: bone differentiation induced iPS cells, G3: iPS cells that were bone differentiation induced and irradiated).

Histological evaluation of new bones by way of transplantation of iPS cells into mouse models with calvarial defectsIn G1, bone differentiation was not performed, and new bone formation was not observed following cell transplantation. Massive tumor was observed and this tumor confirmed to be teratoma histologically. In this series, teratoma formation at a rate of 40% was observed. After the transplantation of bone differentiation induced iPS cells (G2), new bone formation was observed, but at the same time teratoma formation was also observed. In G3 where bone differentiation induced and irradiated iPS cells were transplanted, new bone formation comparative to that in G2 was observed, without teratoma formation being detected (G1: iPS cells that were not bone differentiation induced; G2: bone differentiation induced iPS cells; G3: iPS cells that were bone differentiation induced and irradiated). The arrow indicates cells and the range of calvarial defects. Scale bars: 1 mm.
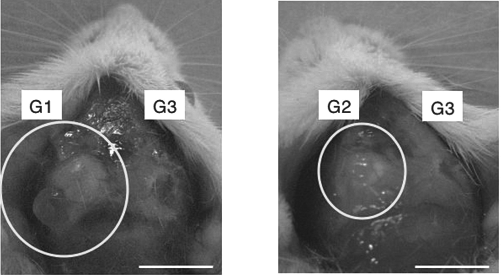
Potential of iPS cells to form teratomas. In the nonirradiated groups (G1 and G2; n = 10) as both groups show tumors. On the other hand, in the irradiation group (G3, n = 10), no teratoma formation was observed in any of the models. A typical example of these findings is shown (G1: iPS cells that were not bone differentiation induced; G2: bone differentiation induced iPS cells; G3: iPS cells that were bone differentiation induced and irradiated). Scale bars: 10 mm.
Xenotransplantation Using Bone Differentiation-Induced Mouse iPS Cells
Bone differentiation-induced mouse iPS cells were transplanted into 12-week-old Wistar rats (n = 3), at 1 × 106 cells each, that were then observed for 4 weeks. In the group of models with transplantation of iPS cells that had undergone bone differentiation induction and were irradiated with dose of 2 Gy, bone formation was induced without teratoma formation (Fig. 7).

Histological evaluation of xenotransplantation of iPS cells into rat models with calvarial defects. (A) Photograph showed two bone defects, no cells (PBS only) were transplanted in the left side and osteogenic inducted and irradiated iPS cells in the right side. (B) Soft X-ray images demonstrated new bone formation in the right side of the bone defect. Scale bars: 10 mm. (C) H&E staining also revealed a bone bridge that was induced by new bone formation. The area between arrows indicates newly formed bones. No teratoma formation was observed in the 4-week follow-up observation following transplantation.
Discussion
Tissues are formed from mature stem cells and pre-cursor cells. In recent years, studies have been actively conducted in order to make use of pluripotent stem cells for bone regeneration and, with regard to bone regeneration, studies have been conducted using ES cells (16) and bone marrow MSCs (29). In particular, differentiation of MSCs is comparatively easy to regulate and these cells are thus being applied clinically. In this context, the study and application of these cells may be more advanced in the field of bone regeneration than other types of cells (25). However, one of the major problems associated with mesenchymal stem cells is the fact that there is a limit to the proliferative capacity of its populations and that they become senescent (4). We therefore tried to create an in vitro a cell line that could proliferate indefinitely by introducing telomerase genes into MSCs (26). Progress in cell biology has made it possible to initialize mature cells. It was reported in 2006 that iPS cells could be created by introducing four transcription factors into mouse mature fibroblasts (33). In 2007, it was demonstrated that iPS cells could also be created using human cells, thereby substantially raising expectations for future clinical use in regenerative medicine (32).
iPS cells, with high proliferative capacity of its populations as ES cells, can be created from the distal cells of each individual and thus offer an advantage of enabling cell transplantation that does not require administration of immunosuppressant (10). As iPS can be created from the patient's own cells, once a method to differentiate them into envisaged organs has been established, it becomes possible to avoid rejection of the transplant. This would be a major breakthrough for its application in both transplantation and regenerative medicine.
The actual application of regenerative medicine in the field of orthopedics has taken a form of a tissue-engineered ceramic artificial bone (19). The artificial bone has no osteogenetic potentials in itself. Many growth factors like bone morphogenetic protein-2 (BMP-2) (17) and connective tissue growth factor (CTGF) (15) and cells like osteoblasts and MSCs (20) are used with them to enhance a bone regenerative capacity. However, there have been few studies reported on bone regeneration using iPS cells (6). Against this background, this time we studied bone regeneration using mouse iPS cells.
As iPS cells have biological features similar to that of ES cells, it was decided that induction of bone differentiation of ES cells should be applied in this study. The cells themselves were not genetically engineered and their differentiation was inducted using the addition of liquidity factors including dexamethasone, ascorbic acid, and β-glycerophosphate (31). This method only requires that these liquidity factors be added into cell culture fluids, and is thus a simple procedure from which results with high reproducibility can be obtained. This method will also enable automated cell cultures in the future. As a result, Alizarin red staining revealed a number of calcified nodules and demonstrated that bone minerals had been induced (23). Furthermore, the expression of Runx2, an initial stage marker of osteoblast differentiation (11), and the expression of osteocalcin, late stage markers of bone differentiation (22), were observed. However, in the group of models for transplantation of iPS-derived osteoblasts obtained in this method, good bone regeneration was obtained, but at the same time, teratoma formation was confirmed.
Important issues in clinical cell transplantation that need to be resolved are the control of rejection and the prevention of tumor formation, including formation of teratomas (35). Tumor formation associated with transplantation must always be avoided when the procedure is used in humans. With regard to the occurrence of malignant tumors, the involvement of c-Myc introduced at the time of the generation of iPS cells has been reported, but the involvement of c-Myc has not been reported in relation to teratomas (12). Also, no report has been made that other genes are directly involved (24). Although the incidence of teratomas after the subcutaneous transplantation of ES cells for the purposes of examining the undifferentiation potential of ES cells to SCID mice is said to be 25–100% (28), long-term follow-ups may find the incidence of teratomas even higher. In regard to the number of cells to be transplanted, it has been reported that 5 × 105 or more cells would be necessary for ES cell teratoma formation (18). The incidence of teratoma formation was reported higher in iPS cells than ES cells (9).
Inevitably, in this study, a high incidence of teratoma formation was expected after the transplantation of iPS-derived osteoblasts into SCID mice without self-immunity. In this examination, teratoma formation was seen at a rate of 40% during the 4-week follow-up observation in the group of models with transplantation of nonirradiated iPS cells that underwent bone differentiation. On the other hand, in the group of models with transplantation of iPS cells that underwent bone-differentiating induction and irradiated with 2 Gy, none of 10 mice induced teratoma formation during the 4-week follow-up observation after transplantation. With regard to irradiation of 2 Gy, the expression of genes closely related to osteoblasts was maintained, and after the transplantation, efficient bone regeneration was histologically confirmed. In terms of irradiation doses, the irradiation of 4 Gy substantially decreased the bone differentiation induction activity of iPS cells. In this examination, it was considered that irradiation of 2 Gy may be an effective method. In the examination of xenogenic cell transplantation in rats performed in our study, no teratoma formation of mouse iPS cells was observed, suggesting its potential efficacy in preventing teratoma formation. This time, it is likely that our report is the first report demonstrating that no teratoma formation was observed in allogeneic transplantation and xenotransplantation of iPS cells that are bone differentiated and irradiated. The molecular biological mechanism action of differentiation-induced iPS cells is currently being examined. In general, as an effect of the radiation itself, various signals inside the cell are transmitted as a result of the radiation stress response inside the cell (30). The well-known pathway is that with emphasis on the role of p53 (21). As a result, cells die when the cell cycle is blocked. It is believed that, in an undifferentiated state, in other words when cells are very active, teratoma formation tends to take place. It is believed that, treated with irradiation, and due to the DNA damage as a result thereof, phosphorylation of specific proteins occurs, which in turn initiates a series of intracellular signal transduction pathways, thereby inhibiting the expression of genes necessary for cell cycle progression. Specifically, it is known that a mechanism exists to block progression from the G1 phase to the S phase and from the G2 phase to the M phase (27). This time, in our study, iPS cells were irradiated, and although the duration of follow-up observation after the transplantation was up to 4 weeks, no teratoma formation was observed. It has been reported that no teratoma formation was observed of iPS cells created from mice that had received resveratrol (14). The possibility has been demonstrated that resveratrol would lower the level of undifferentiation of iPS cells, thereby inhibiting teratoma formation.
With regard to the inhibition of teratoma formation after cell transplantation, it has been reported that, for cases in which iPS cells had been created from hepatocytes and gastric parietal cells at the cell creation stage (1), there was less tumor formation by these cells with the decreased level of reactivation of retrovirally introduced c-Myc (2). The usage of these cells and resveratrol may reduce the potential to form teratomas from the initial stage of differentiation induction. It has also been reported that the introduction of genes, such as Runx2 and peroxisome proliferator-activated receptor-γ (PPAR-γ) that are involved in cell differentiation, into iPS cells improves differentiation efficiency (34), and we would like to conduct a study on this in the future.
In this study, we examined bone regeneration using single iPS cells, and it seems to be necessary to consider the efficacy of this bone differentiation induction method using other types of iPS cells. It will inevitably be necessary to further study the possibility of teratoma formation in longer term follow-up observation. If these problems are all resolved, then the actual application of iPS cells will no longer be a dream. In fact, as an impact of an aging society with fewer children, the number of chronically ill patients with compression fractures due to osteoporosis and with pseudarthrosis as a result thereof, is surging in Japan. The actual application of cell transplantation procedures for osteoporosis, using bone cells differentiated and induced from iPS cells derived from the patient, would also be desirable in terms of health services.
Conclusions
In the present study, we conducted an elementary study on the bone differentiation of mouse iPS cells. It was suggested that bone-differentiated mouse iPS cells have an osteoinductive potential. They also facilitated bone formation in mouse models with bone defects. After transplanting bone-differentiated iPS cells, teratoma formation was observed, but it was suggested that cell irradiation before transplantation inhibits teratoma formation. The xenotransplantation of cells into rat models with bone defects facilitated bone formation.
Footnotes
Acknowledgment
The authors declare no conflicts of interest.
