Abstract
Adipose tissue-derived stem/progenitor cells (ASCs) have attracted attention as a cell source that replaces marrow stromal cells (MSCs); ASCs may thus have applications in both regenerative medicine and cell transplantation. These medical treatments, however, require a high-quality supply of human ASCs. Therefore, the cryopreservation methods have been improved by changing a component of a cryopreservation medium. Sericin, a protein hydrolysate (with an average molecular weight of 30 kDa) is very rich in serine. The viability and the adipogenic/osteogenic potential of human ASCs were tested after freezing in a cryopreservation medium containing sericin. After thawing, the viability of the human ASCs frozen in the cryopreservation medium was found to be more than 95%. The proliferation rate of human ASCs frozen in CELLBANKER 2, and DMEM/Ham's F-12 medium (serum free) + 10% DMSO, 0.1 mol/L maltose, and 1% sericin was higher than that of the cells frozen in the maintenance medium + 10% DMSO. The adipogenic/osteogenic differentiation capabilities of frozen human ASCs were examined by Oil Red O staining/Von Kossa's method. The human ASCs were frozen using CELLBANKER 2, and DMEM/Ham's F-12 medium (serum free) + 10% DMSO, 0.1 mol/L maltose, and 1% sericin were positive. In conclusion, the cryopreservation medium containing sericin is therefore considered to have a beneficial effect on freezing human ASCs. This serum-free cryopreservation medium should be widely used in regenerative medicine, cell transplantation, and biological research.
Keywords
Introduction
Adipose tissue-derived stem cells (ASCs) are a promising cell source that replaces marrow stromal cells (MSCs). ASCs differentiate into various cells, such as osteoblasts, chondrocytes, myocytes, and adipocytes in vitro. The ASCs also have the potential to differentiate into endodermal cells such as hepatocytes and insulin-expressing cells, and ectodermal cells such as nerve cells. In clinical trials, human ASCs promote fat, vasculature, and bone regeneration (8,23,24). ASCs may thus have applications in regenerative medicine and cell transplantation (5). These medical treatments, however, require a high-quality supply of ASCs. Therefore, the technology of cryopreservation of ASCs is important (6,7), and we improved this technique by changing the components of the freezing medium.
Adipogenic/osteogenic differentiation of cryopreserved mouse ASCs has been examined using several commercial available cryopreservation mediums. CELLBANKER 2 yielded the optimal cell viability for mouse ASCs (17). In this study, continuously cultivated human ASCs were frozen using three cryopreservation mediums including 10% dimethyl sulfoxide (DMSO), maintenance medium + 10% DMSO, CELLBANKER 2, and DMEM/Ham's F-12 medium (serum free) + 10% DMSO, 0.1 mol/L maltose, and 1% sericin. The viability and the potential of adipogenesis/osteogenesis of human ASCs were compared after freezing in three kinds of cryopreservation media. Sericin, a protein hydrolysate (with an average molecular weight of 30 kDa) is very rich in serine and obtained from raw silk during the degumming process. Sericin was found to be useful for the cryopreservation of human primary hepatocytes (14), mammalian and insect cell lines (21). The current study examined whether the cryopreservation medium containing sericin could improve the adipogenic/osteogenic differentiation efficiency of frozen/thawed human ASCs.
Materials and Methods
Materials
Maintenance medium (#PM-1; preadipocyte medium), adipocyte differentiation medium (#DM-2), and osteoblast differentiation medium (#OB-1), were purchased from Zen-Bio, Inc. (Grand Island, NY). DMEM/Ham's F-12 (1:1, v/v; Dulbecco's modified Eagle medium: Nutrient Mixture F-12) was purchased from Gibco BRL, Life Technologies (Grand Island, NY). Dimethyl sulfoxide (DMSO, D-2650) was purchased from Sigma-Aldrich (St. Louis, MO). Maltose was purchased from Wako Pure Chemical Industries, Ltd. (Osaka, Japan). All other chemicals not specified above were of the highest available grade. Sericin hydrolysate with an average molecular mass of 30 k was generously donated by the Seiren Co. Ltd. (Fukui, Japan).
Cells
Cryopreserved human ASCs were obtained from Zenbio, Inc. (Research Triangle Park, NC). The cryopreserved cells (#ASC-F, Lot ASC062801; sex/age/BMI (average)/No. of patients: female/37/23.29/1 (single), and passage number 2) were purchased from Dainippon Sumitomo Pharma (Osaka, Japan).
Subculture of Human ASCs
For thawing, 2 × 105 human ASCs were seeded into a T-25 cell culture flask (NUNC) with maintenance medium at 37°C in 5% CO2. The cells attached and spread on the flask, and cultured until they were approximately 80–90% confluent. The cells were detached from the culture flask with trypsin, and repeatedly seeded and cultured on new culture flask. The passages were repeated from two to seven times, and the cells obtained after each passage were frozen and stored for 1–4 weeks.
Cryopreservation and Thawing of Human ASCs
The basal composition of the cryopreservation medium was maintenance medium and 10% DMSO. Maltose was added to DMEM/Ham's F-12 (1:1, v/v) medium containing 100 U/ml penicillin, 100 U/ml streptomycin at concentrations of 0.1 mol/L and Sericin at 1%. CELLBANKER 2 is serum-free freezing medium (Nippon Zenyaku Kogyo Co., Ltd, Japan). One milliliter of cell suspension containing 5 × 106 cells was quickly transferred to a 2.0-ml freezing tube and frozen at a cooling rate of 1°C/min. After cooling to −80°C, the cells were stored until use (typically from 1–4 weeks). Frozen tubes were placed in a 37°C water bath to thaw until the ice crystal had nearly melted. The cell suspension was then diluted 1:9 with ice-cold maintenance medium, and was centrifuged at 1200 rpm for 5 min. The supernatant was removed and the cells were resuspended in fresh medium. The cell viability was assessed using the trypan blue exclusion test. The final concentration of trypan blue (GIBCO BRL, Grand Island, NY) was 0.2% in the test.
Proliferation Rate of Human ASCs
Cryopreserved or refrozen human ASCs (1 × 104 cells) were seeded in 96-well plates (BD Biosciences) with 100 μl of maintenance medium. Viable cells were counted using a Cell Counting Kit-8 (CCK-8; DOJINDO Laboratories, Kumamoto, Japan). CCK-8 reagent (10 μ l) was added to each well and the reaction was allowed to proceed for up to 4 h. The absorbance of the sample at 450 nm was measured against a background control, using a microplate reader. The cell number was counted after 4, 7, and 11 days.
Differentiation of Human ASCs Into Adipocytes
Adipogenic differentiation was induced by culturing the cells for 3 days in an adipocyte differentiation medium. The cells were then further cultured in an adipocyte differentiation medium for 5 days. Differentiation was confirmed by microscopic observations of intracellular lipid droplets and Oil Red O staining as an indicator of intracellular lipid accumulation. Briefly, the cells were fixed in 10% formaldehyde in PBS for 10 min at room temperature, and then were washed with 60% isopropanol. The cells were then stained with 2% (w/v) Oil Red O reagent for 10 min at room temperature followed by repeated washing with distilled water and destaining in 100% isopropanol for 1 min. An assay of accumulated triglyceride by Triglyceride E-test™ (Wako Pure Chemical Industries Ltd.) in the cell samples was performed as described previously (17,19).
Differentiation of Human ASCs Into Osteoblasts
Osteogenic differentiation was induced by culturing the cells for 4 weeks in an osteoblast differentiation medium. The differentiation was confirmed by observation of extracellular matrix calcification using Von Kossa's method. The cells were washed twice with PBS and fixed in 10% formalin for 15 min at room temperature. They were washed and incubated with deionized water for 15 min. Thereafter, they were stained with a solution containing naphtol AS MX-PO4, N,N-dimethylformamide, Red Violet LB salt, and Tris-HCl (pH 8.3) for 45 min. Von Kossa staining was carried out with 2.5% silver nitrate solution for 30 min.
Results
Cell Viability of Refrozen Human ASCs
To investigate the function of refrozen human ASCs, the human ASCs are subcultured several times at passage numbers 2–7. The cell viabilities were estimated in the human ASCs that were cryopreserved in three cryopreservation solutions from 1 to 4 weeks. At first, the viability of cryopreserved ASCs (passage number 2) was over 95% soon after purchase. After the thawing of human ASCs refrozen in three cryopreservation solutions, the cell viability was shown to be more than 95% (Fig. 1). No difference in the cell viability from passage numbers 2–7 was observed.

Cell viability of refrozen human ASCs with different cryopreservation media and different subculture numbers. 1: cryopreserved adipose tissue-derived stem cells (ASCs), passage number 2. 2: Maintenance medium + 10% dimethyl sulfoxide (DMSO), passage number 5. 3: DMEM/Ham's F-12 medium + 10% DMSO, 0.1 mol/L maltose, 1% sericin, passage number 5. 4: CELLBANKER 2, passage number 5. 5: Maintenance medium + 10% DMSO, passage number 7. 6: DMEM/Ham's F-12 medium + 10% DMSO, 0.1 mol/L maltose, 1% sericin, passage number 7. 7: CELLBANKER 2, passage number 7. The data are presented as the mean ± SD of three independent experiments.
Proliferation Rate of Refrozen Human ASCs
The proliferation rate of refrozen human ASCs was examined at 0, 4, 7, and 11 days using a Cell Counting kit-8 (Fig. 2). The cryopreserved human ASCs (passage number 2) showed a higher proliferation rate in comparison to refrozen human ASCs (passage number 5 and 7). On day 11, the thawed human ASCs that had been refrozen in CELLBANKER 2, and DMEM/Ham's F-12 medium (serum free) + 10% DMSO, 0.1 mol/L maltose, and 1% sericin had a higher rate of cell proliferation in comparison to those refrozen in the maintenance medium + 10% DMSO.
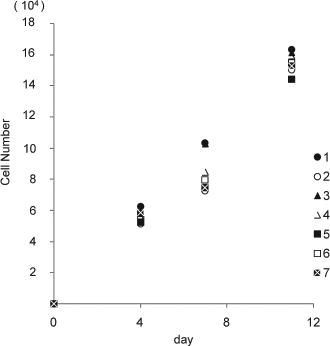
Proliferation rate of refrozen human ASCs. 1: Cryopreserved ASCs, passage number 2. 2: Maintenance medium + 10% DMSO, passage number 5. 3: DMEM/Ham's F-12 medium + 10% DMSO, 0.1 mol/L maltose, 1% sericin, passage number 5. 4: CELLBANKER 2, passage number 5. 5: Maintenance medium + 10% DMSO, passage number 7. 6: DMEM/Ham's F-12 medium + 10% DMSO, 0.1 mol/L maltose, 1% sericin, passage number 7. 7: CELLBANKER 2, passage number 7. The number of cells was evaluated by Cell Counting Kit-8 after 0, 4, 7, and 11 days. The data are the means and SD of three independent experiments.
Differentiation of Refrozen Human ASCs Into Adipocytes
The differentiation from refrozen/thawed human ASCs (passage number 5) to adipocytes was examined by the cell shape. The human ASCs were initially cultured on 35-mm culture dishes in adipocyte differentiation medium. After 5 days, the human ASCs showed round morphologies with lipid vacuoles in the cytoplasm. The human ASCs were positive for Oil Red O staining, thus suggesting that the cells had adipogenic differentiation capabilities (Fig. 3A). The human ASCs refrozen in each cryopreservation medium showed no difference in the shape of adipocytes (data not shown).
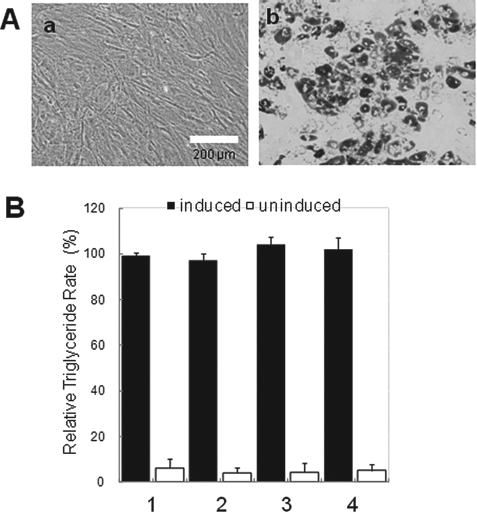
The capabilities of adipogenic differentiation of refrozen human ASCs. (A) Adipogenic differentiation from refrozen human ASCs (passage number 5). Phase-contrast photomicrographs of refrozen human ASCs before (a) and after adipogenic differentiation (b). Scale bar: 200 μm. (B) Accumulation of triglyceride in refrozen human ASCs. The capabilities of adipogenic differentiation of refrozen human ASCs with three cryopreservation mediums were examined using Triglyceride E-test™. 1: Cryopreserved ASCs, passage number 2. 2: Maintenance medium+10% DMSO, passage number 5. 3: DMEM/Ham's F-12 medium + 10% DMSO, 0.1 mol/L maltose, 1% sericin, passage number 5. 4: CELLBANKER 2, passage number 5. The data are presented as the means ± SD of three independent experiments.
In addition, the capabilities of adipogenic differentiation between cryopreserved (passage number 2) and refrozen human ASCs (passage number 5) were examined using Triglyceride E-test™. The refrozen human ASCs were compared after adipogenic differentiation and no difference was seen in the accumulation of triglyceride (Fig. 3B). In addition, no difference was seen in the capabilities of adipogenic differentiation when the cells were refrozen after subculture (passage number 7).
Differentiation of Refrozen Human ASCs Into Osteoblasts
Human ASCs were cultured for 4 weeks to observe the osteogenic differentiation. The human ASCs (passage number 5) refrozen in CELLBANKER 2, and DMEM/Ham's F-12 medium (serum-free) + 10% DMSO, 0.1 mol/L maltose, and 1% sericin were positive for von Kossa staining, thus suggesting that the cells had osteogenic differentiation capabilities (Fig. 4). In addition, no differentiation capabilities into osteoblasts were observed if the cells were refrozen after the subculture (passage numbers 7 and 9; data not shown).

Osteogenic differentiation from refrozen human ASCs. Morphology of refrozen human ASCs (passage number 5) before (A) and after osteogenic differentiation (B). Scale bar: 200 μm.
Discussion
Research has identified numerous cell types that can be used for regenerative medicine and cell transplantation, such as induced pluripotent stem cells (iPS cells) (15,18,22), embryonic stem cells (ES cells) (15,18), and somatic stem cells. It is difficult to secure a sufficient number of cells for clinical applications with bone marrow mesenchymal stem cell in somatic stem cells. To solve these problems, ASCs, with multilineage potential, have been investigated as a new cell source (5,18,23,24). In comparison to human hepatocytes and other cells, human ASCs can be obtained with comparative ease. Moreover, the separation, establishment of the obtaining route, and large numbers of the necessary cells can therefore be prepared for human ASCs.
Therefore, ensuring the quality, amount, and the safety of the human ASCs is extremely important for further promoting the clinical application of this treatment modality (23,24). Continual improvement in the preservation technology (11,12) is therefore the key to resolving these problems. The current study focused on the effect of the composition of cryopreservation medium of various factors related to the preservation of the cells (9,10,16,20). When trehalose and anti-freeze protein (AFP), etc., are added to cryopreservation medium as a protective material, it thereby controls the damage to the cells induced by ice crystals (1–4).
Cryopreservation of mouse ASCs was evaluated using commercial cryopreservation solutions, and CELLBANKER 2 was the most effective (17). Moreover, using sericin (14) and oligosaccharides (13) was effective for human hepatocytes that are difficult to do cryopreserve. The present study showed that these cryopreservation mediums were effective for human ASCs. Human ASCs frozen after the subculture (passage number 5) using CELLBANKER 2 and DMEM/Ham's F-12 medium (serum free) + 10% DMSO, 0.1mol/L maltose, and 1% sericin maintained their cell proliferation, and adipogenic/osteogenic differentiation capabilities (Figs. 2, 3, and 4). Although the induction of differentiation from human ASCs (passage number 5) that had been frozen in the maintenance medium + 10% DMSO to the osteoblasts was performed, the differentiation into osteoblasts could not be confirmed after 4 weeks (data not shown). This findings suggests that controlling the decrease in the multilineage potential by cryopreservation of the human ASCs required not only 10% DMSO but also sericin and oligosaccharides as protective materials.
In conclusion, this serum-free cryopreservation medium should therefore be widely used in regenerative medicine, cell transplantation, and biological research. However, the functional maintenance of human ASCs with multilineage potential requires further study, not only regarding the influence of cell cryopreservation, but also to elucidate the influence of cell subcultures.
Footnotes
Acknowledgments
We thank Ms. Rina Yokota (Nagoya University) for assistance. This work was supported in part by Health and Labor Sciences Research Grants from the Ministry of Health, Labour and Welfare (Tokyo, Japan); Grant-in-Aid for Scientific Research from Japan Society for the Promotion of Science; Sasakawa Scientific Research Grant from The Japan Science Society. The authors declare no conflicts of interest.
