Abstract
The recovery of all of the islets contained in a pancreas is the goal of islet isolation for transplantation. This study reveals an environment that injures the isolated islets during digestion and proposes a new model for optimal islet isolation. Islets were isolated from Wistar rat pancreases by stationary collagenase digestion while the digestion time was varied at 15, 30, 60, and 120 min. The digested pancreas and islets were analyzed histologically and adenosine nucleotides were measured. Overnight cultured islets (40 islets) were cocultured for 30 min with the supernatants obtained from pancreatic collagenase digestion at different digestion periods in order to assess the toxic environment. The peak yields of islets were obtained at 30 min of digestion. The histological study of digested pancreas showed that the exocrine cells lost their cellular integrity at 120 min of digestion, but the islet cells were left intact. Accordingly, the ATP levels of the pancreatic tissue decreased during the digestion period. The coculture experiment demonstrated that the islets cultured with the supernatants from the collagenase digestion showed digestion time-dependent disruption of the cellular integrity of islets in accordance with a rapid decrease of ATP levels in the islets. The addition of serine protease inhibitors into this coculture clearly showed protection of islets, which maintained high ATP levels in association with intact membrane integrity as assessed by AO/PI staining. Morphological deterioration of islets as well as a marked ATP decrease was evident in the entire digested pancreas as well as in islets cocultured in the supernatants from the collagenase digestion. Various factors toxic to the islets can therefore be analyzed in future experiments using this coculture model for obtaining a good yield of viable islets.
Introduction
Islet isolation is a crucial step for islet transplantation (1,15). Although there have been substantial advances in islet isolation technology during the past decades (11, 20), it is still difficult to recover all of the islets contained in a pancreas (13). In fact, even in the leading centers, a transplantable yield of isolated islets is obtained from less than 50% of the processed pancreases (18,19). A major obstacle to successful human islet isolation has thus been the variability of the collagenase digestion phase of islet isolation (10).
Islet isolation involves an enzymatic and mechanical digestion to release islets from the surrounding exocrine tissue, followed by a separation procedure to purify the islets. The collagenase digestion is critical to islet isolation, although it is potentially harmful to the islets (10). However, no monitoring system has yet been established.
The content of adenosines reflects the viability in various organ transplants (6–8). Assessment of the levels of adenosine triphosphate (ATP) or the adenosine diphosphate (ADP)/ATP ratio is useful for prediction of transplantation outcome in islet transplantation (3). A new method was recently established for determination of adenosine levels in isolated islets (6,7). This method is a bioluminescent enzymatic cycling assay, which allows quick measurement in samples as small as a single islet (6,7). This method allows the measurement of the levels of adenosines during the digestion phase not only in pancreatic tissue but also in isolated islets in experimental models using rats.
The collagenase digestion phase results in the release of endogenous pancreatic enzymes from the exocrine tissues (2,12,16,17,21–23). A strong increase of such enzyme activity, in particular serine proteases involved in the digestion process, is detrimental to the islet isolation outcome (2). However, the effect of the addition of serine protease inhibitors prior to digestion still remains controversial (12,16,17,21–23).
The aim of this study was to examine the changes in the energy status of islets during the digestion procedure, to elucidate the extracellular components of the digestive procedure as a supernatant to determine whether this supernatant is toxic to the islets and, if it is, whether a serine protease inhibitor could reduce the toxicity to the islets. This model provides a simple system to elucidate the mechanism of overdigestion and to select compounds for protecting islets during collagenase digestion.
Materials and Methods
Animals
Wistar rats (8 weeks, male, provided by CLEA Japan, Inc., Tokyo, Japan) were used for islet isolation under anesthesia with ether inhalation. The experimental protocol was approved by the Ethics Review Committee for Animal Experimentation of Fukushima Medical University, and all procedures in this experiment were performed according to the National Research Council's guidelines.
Histological Study and Energy Levels of the Pancreatic Tissue During Collagenase Digestion
Pancreatic grafts were taken from Wistar rats and were digested by the stationary method using 2 mg/ml collagenase (collagenase S-1, Nitta Gelatin Inc., Osaka, Japan). Pancreatic tissue samples for histological study and measurement of adenosine levels were taken at 0, 15, 30, 60, and 120 min after starting the digestion. Samples were fixed in paraformaldehyde for a histological study. Samples were taken and stored in deep freezer until the adenosine levels were measured.
Assessment of the Toxicity of Digested Supernatant During Collagenase Digestion on Overnight Cultured Islets
Islets were isolated and purified from Wistar rats by the stationary digestion method as previously reported (4), and cultured in RPMI in a 37°C incubator overnight. The 40 islets were cultured for 30 min at 37°C in Hanks balanced salt solution (HBSS), HBSS with 2 mg/ml collagenase, and in supernatants obtained from the collagenase digestion at 15, 30, 60, and 120 min after starting digestion. After 30 min of incubation, these islets were collected by gentle centrifugation (500 rpm for 1 min) and used for the histological analysis and measurement of adenosines.
Histological Analysis of Islets
The cultured islets and the treated islets were transferred into 1.5-ml Falcon microtubes, and collected by centrifugation at 500 rpm for 1 min. These islets were then fixed in 4% paraformaldehyde for 1 day, dehydrated, and embedded in paraffin. The sections were stained with hematoxylin and eosin (H&E) staining. For the immunohisotological staining of insulin, purified rabbit anti-insulin (H-86) (Santa Cruz Biotechnology, Inc., CA, USA) was used. To detect the insulin molecules the sections were stained using a peroxidaselabeled biotin-avidin detection system Histofine® Simple Stain Rat MAX PO(R) according to the instructions provided by the manufacturer (Nichirei Biosciences Inc. Tokyo, Japan).
Measurement of Adenosine Nucleotides (ANs)
Adenosine nucleotides were measured by the bioluminescence method using luciferase, as provided by Kikkoman Corp., Chiba, Japan (24) and the luminescence Mithras LB940 photon counter (Berthold Technologies GmbH & Co KG, Bad Wildbad, Germany). This method provides rapid results and has sufficient sensitivity to detect the ATP content even in one islet cell (6,7). ATP, ATP + adenosine monophosphate (AMP), and total adenosine nucleotide (TAN) content were measured by each reagent individually. Energy charge (EC) was calculated by obtaining the contents of ADP and AMP by subtraction of each of the measurable components. Twenty microliters of each sample was diluted with 80 μl of Tris buffer and 100 μl reagent was added immediately before the assay. It took 10 s to react with the ANs and the luminescence remained stable for 2 min. In the case of pancreatic tissue, samples were added with 0.5 N HClO4 and thoroughly homogenized for 30 s. Next, it was centrifuged for 5 min at 15,000 rpm and then the supernatant was taken and stored in liquid nitrogen. Samples were transferred and stored in a deep freezer until processed. It was diluted 1000 times and measured. Islets were placed in 500 μl of culture medium plus 55 μl of 0.5 N HClO4 for freezing at −80°C until processed. After centrifugation, the samples were assayed without dilution.
Measurement of Trypsin Activity
Aliquots of supernatant from the warm digested pancreatic tissue at 15, 30, 60, and 120 min after stationary digestion were collected and frozen until the measurement of trypsin activity. The trypsin activity was measured by absorption spectrophotometry (λ405 nm; Model 680 Series Microplate Readers Bio-Rad Laboratories, CA, USA) using the trypsin substrate kit containing carbobenzoxy-glycyl-d-alanine-p-nitroanilide-acetate, SPECTROZYME®TRY (American Diagnostica Inc., Stamford, CT, USA). The assay was calibrated using dilutions of a stock solution of trypsin from the bovine pancreas (Sigma-Aldrich Co., St. Louis, MO, USA) in 200 μl of 50 mM Tris buffer at 37°C to generate a standard curve. Absorbance was measured at 1, 5, and 10 min and the trypsin activity was calculated to compare it with the standard curve.
Evaluation of the Effect of Serine Protease Inhibitors on Isolated Islets in a Toxic Environment During Collagenase Digestion
Overnight cultured islets were cultured for 30 min at 37°C in HBSS, HBSS with 2 mg/ml collagenase, and supernatants obtained from collagenase digestion at 15, 30, 60, and 120 min after starting of digestion. To assess the protective effect of serine protease inhibitor on islets in the presence of digested supernatant, Pefabloc (Pefabloc SC PLUS, Roche Diagnostics Co., Mannheim, Germany) or ulinastatin (MIRACLID Inc, Mochida Pharma., Tokyo, Japan) were added to the culture media. Pefabloc was used at 0.4 mM, which is the recommended concentration (21), and ulinastatin was added at a dose of 100 U/ml according to a previous report (17). After 30 min of incubation, these islets were collected by centrifugation and used for histological analysis and measurement of adenosines. The viability of islet cells was assessed by simultaneous use of the inclusion and exclusion dyes acridine orange (AO) and propidium iodide (PI), as described previously (5). Briefly, 40 islets were cultured with 0.67 μM AO and 75 μM PI for 30 min in HBSS or supernatants obtained after 15, 30, 60, and 120 of digestion. Next, the islets were examined by confocal microscopy (model FV300, Olympus, Japan). Fluorescent images were obtained under a fluorescent microscope (Eclipse E800, Nikon, Japan) and a confocal laser microscope (FV300, Olympus, Japan). A fluorescent illuminator consisting of a 100-W mercurial light source with a 490-nm excitation filter and a 510-nm barrier filter was used. This filter combination permits the simultaneous visualization of the green emission of AO and the red emission of PI.
Statistical Analysis
Data are expressed as the means ± SD. The ATP contents of pancreatic tissue and islets were compared using analysis of variance (ANOVA) and unpaired Student's t-test with Bonferroni correction. Values of p < 0.05 were considered to be statistically significant. All statistical analyses were performed using the Statview J 4.58 for Windows software package (Abacus Concepts Inc., Berkeley, CA, USA).
Results
Islet Yield and Adenosine Content in Relation to the Digestion Period
The islet yields per rodent after 30, 60, and 120 min of stationary digestion method using collagenase were 1304 ± 409, 534 ± 156, and 3.3 ± 0.9 islet equivalents (IEQ), respectively (n = 3), thus showing a significant difference between 30, 60, and 120 min of digestion period (p < 0.05). The ATP content of these islets, after 30, 60, and 120 min of stationary digestion, were 18.3 ± 0.2, 7.0 ± 0.5, and 3.5 ± 0.8 μM, respectively, also showing a significant difference between each digestion period (p < 0.05). Therefore, the islet yield and ATP content in the overdigestion period, 60 and 120 min of digestion, revealed a significantly low level in comparison to 30 min of digestion.
Histological Changes in Pancreas During Collagenase Digestion
The interstitial space of the pancreatic tissue after collagenase digestion was wider than that of the pancreatic tissue before distension, and the islets were separated from exocrine tissue after collagenase digestion. There were clearly many lobular structures in the pancreatic tissue after digestion. Half of the lobular structures were broken at 15 min of digestion and almost all disappeared after 30 min of digestion with the presence of islets free from the exocrine tissue. No lobular structures with destruction of exocrine tissue were obvious after 60 min of digestion. The densely stained nuclei and cytoplasmic swelling of exocrine cells were evident with significant damage to the surface of islets. Finally, the acinar cells were complete free from the lobar structures and islets were also disrupted after 120 min of digestion (Fig. 1).

Histological changes in the pancreas during collagenase digestion. Hematoxylin and eosin staining of paraffin-embedded pancreatic tissue of Wister rat. Pancreata were distended through pancreatic duct by collagenase solution and digested for 15, 30, 60, and 120 min (40x, 200x, and 400x magnification, respectively). The lobular structures disappeared after 30 min of digestion with the presence of islets free from the exocrine tissue. The acinar cells were complete free from the lobar structures and the islets remained relatively intact after 120 min of digestion. Scale bars: 500 μm (top row), 100 μm (middle row), 50μm (bottom row).
Changes of ATP, TAN Content, and EC in the Pancreas During Collagenase Digestion
The ATP contents of the pancreatic tissue decreased during collagenase digestion, showing significant differences before and after digestion [ATP content before digestion: 1599 ± 431, 15 min of warm digested tissue (WDT): 238 ± 114, 30 min of WDT: 109 ± 27, 60 min of WDT: 72 ± 22, 120 min of WDT: 38 ± 23 μM, p < 0.01]. During digestion, ATP content after 120 min of WDT was significantly lower than that of 15 min and 30 min of WDT (p < 0.05) (Fig. 2a).
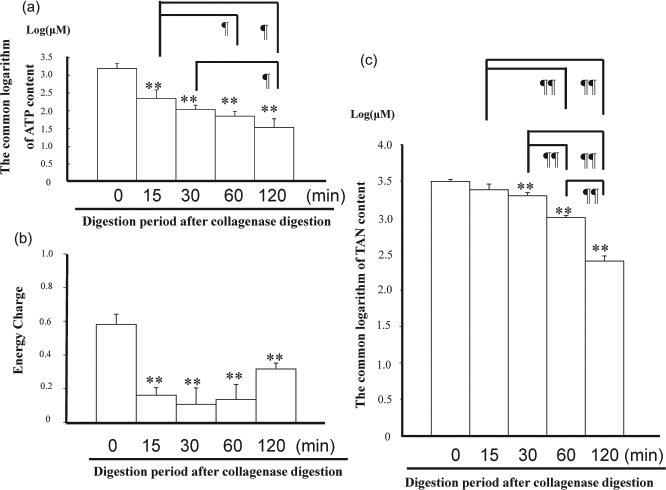
(a) Changes of adenosine triphosphate (ATP) content in pancreas during collagenase digestion (n = 3). ATP contents were adjusted by a common logarithm. The ATP contents of the pancreatic tissue decreased during collagenase digestion showing significant differences before and after digestion. **Significant difference in comparison to 0 min, p < 0.01. ¶Significant difference between digestion periods, p < 0.05. (b) Changes of total adenine nucleotide (TAN) content in the pancreas during collagenase digestion (n = 3). TAN contents were adjusted by the common logarithm. The TAN contents of pancreatic tissue revealed significant decrease during digestion periods. **Significant difference in comparison to 0 min, p < 0.01. ¶¶ Significant difference between digestion periods, p < 0.01. (c) Changes of energy charge (EC) in the pancreas during collagenase digestion (n = 3). Time means digestion period after distension. The EC of the pancreatic tissue decreased during collagenase digestion. **Significant difference in comparison to 0 min, p < 0.01.
The TAN content failed to show a significant difference between pancreatic tissue before digestion and 15 min of WDT, but TAN content after 30, 60, and 120 min of WDT was significantly lower than that before digestion (TAN content before digestion: 3106± 194, 15 min of WDT: 2411 ± 419, 30 min of WDT: 1978 ± 199, 60 min of WDT: 990 ± 67, 120 min of WDT: 253 ± 42 μM, p < 0.01). The TAN contents revealed significant differences during digestion between 30, 60, and 120 min of WDT (p < 0.01), respectively (Fig. 2b).
The EC of the pancreatic tissue decreased during collagenase digestion showing a significant difference between before and after digestion (EC before digestion: 0.58 ± 0.06, 15 min of WDT: 0.16 ± 0.04, 30 min of WDT: 0.11 ± 0.10, 60 min of WDT: 0.14 ± 0.09, 120 min of WDT: 0.32 ± 0.04, p < 0.01) (Fig. 2c).
Changes in the Trypsin Activity in Supernatants of Digested Fluid During Collagenase Digestion
Endogenous enzymes were thought to induce a toxic environment during the digestion phase; thus, trypsin was selected as one of the factors that damaged the pancreatic tissue. Trypsin activity of digestion supernatant increased time dependently with the progression of digestion. The trypsin activities at 15, 30, 60, and 120 min were 339 ± 66.8, 795 ± 371.8, 1504 ± 257.4, and 1545 ± 800.2 U/ml, respectively, thereby showing significant differences between 15 and 60 min, and between 15 min and 120 min (Fig. 3).
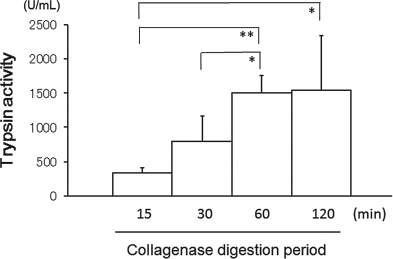
The trypsin activity in the supernatant of warm digested pancreatic tissue (n = 4). The trypsin activity of the digestion supernatant increased time dependently with the progression of digestion. Significant difference between the digestion periods: *p < 0.05, **p < 0.01.
Evaluation of Effect of the Serine Protease Inhibitors on the Morophological Changes of Isolated Islets in a Toxic Environment During Collagenase Digestion
The islets were used after an overnight culture in RPMI. Forty islets were cultured in HBSS or the digestion supernatants after 15, 30, 60, and 120 min of digestion for 30 min, with or without serine protease inhibitors to assess the effect of serine protease inhibitors. The islets cultured without serine protease inhibitors served as the control group.
Histologically, the islets in the control group showed limited changes in cultured with 15 and 30 min of digestion supernatant, but evident in cultured with 60 and 120 min of digestion supernatant, as described above (Fig. 4a). In contrast, the islets cultured with serine proteases (both Pefabloc and ulinastatin) showed very limited damage to their surface when cultured with 15, 30, and 60 min of digestion supernatant. Moreover, only limited damage to the islets with serine proteases was observed and they maintained their integrity even when cultured with a digestion supernatant for 120 min. An immunohistological study using anti-insulin antibody revealed diffusely positive cells for anti-insulin antibody within the islets, and almost the same staining level of insulin was observed regardless of whether or not serine proteases were added when cultured for 15, 30, and 60 min in the digestion supernatant. Only limited damage to the islets with serine proteases was also observed when cultured with a digestion supernatant for 120 min, and the insulin positive cells in the islets with serine proteases also maintained their integrity (Fig. 4b).

(a, b) Histological finding of isolated islets cultured with supernatants of digestion supernatant during collagenase digestion for 30 min, with or without serine protease inhibitors. H/E staining of paraffin-embedded pancreatic islets of Wister rat (a). Islets were cultured overnight and cultured in digestion supernatant for 30 min at 37°C, with or without serine proteases, Pefabloc or ulinastatin. The islets cultured with pancreatic digestion supernatant showed prominent damage leading to disruption of the cellular integrity of the islets in the control group. The islets cultured with serine proteases (both Pefabloc and ulinastatin) showed very limited damage to their surface compared to the control group. The immunohistological findings of the pancreatic islets of Wister rat using anti-insulin antibody (b) were similar to those of H/E staining. Scale bars: 100 μm.
Changes of ATP Content in Isolated Islets Cultured with Digestion Supernatant During Collagenase Digestion
The ATP levels of 40 islets cultured with collagenase decreased, but failed to show a significant difference in comparison to HBSS alone. In contrast, the ATP content of the islets decreased in accordance with the period of digestion showing a significant decrease when cultured with the pancreas digestion supernatant taken from 15, 30, 60, and 120 min of digestion in comparison to either HBSS or collagenase (ATP content in HBSS: 0.32 ± 0.05 μM, in collagenase: 0.24 ± 0.04 μM, in 15-min digested fluid: 0.17 ± 0.03 μM, 30-min digested fluid: 0.13 ± 0.02 μM, 60-min digested fluid: 0.07 ± 0.02 μM, and 120-min digested fluid: 0.03 ± 0.02, p < 0.01) (Fig. 5).

ATP content of 40 islets cultured with digestion supernatants during collagenase digestion for 30 min (n = 3). The ATP levels of 40 islets cultured with pancreas digestion supernatant decreased in a time-dependent fashion. Significant difference between digestion periods: *p < 0.05, **p < 0.01.
Evaluation of Effect of the Serine Protease Inhibitors on the ATP Content, AO/PI Staining of Isolated Sslets in a Toxic Environment During Collagenase Digestion
The ATP levels of these islets were measured, and expressed as the “ATP content ratio,” which was calculated by the formula: ATP contents ratio = measured ATP content of islets/ATP content of islets cultured in HBSS for 30 min (Fig. 6). The ATP content ratio of islets in the control group decreased significantly after culture with 15-, 30-, 60-, and 120-min digestion supernatant. In contrast, the ATP content ratio in the presence of Pefabloc or ulinastatin maintained the same level as that before digestion in the islets cultured for 15 min. Moreover, the ratio of islets with Pefabloc maintained the level even in islets cultured with 30 min digestion supernatant, thus showing a significant difference in comparison to the control islets cultured for the same period.
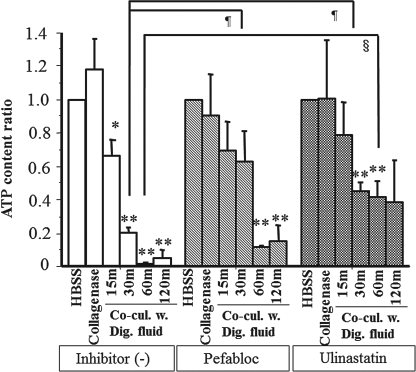
The ATP content of 40 islets cultured in digestion supernatants during collagenase digestion for 30 min, with or without serine protease inhibitors. Significant difference between digestion periods: *p < 0.05, **p < 0.01. ¶Significant difference in comparison to islets cultured with 30-min digestion supernatants in the control group, p < 0.05. §Significant difference in comparison to the islets cultured with 60-min digestion supernatants in the control group, p < 0.05. The ATP content ratio in the presence of Pefabloc or ulinastatin was maintained at relatively high levels in comparison to the control group.
The observation of the islets using confocal microscopy with AO/PI revealed that almost all of the islets in the control group stained with PI at the end of 30-min culture (Fig. 7). In contrast, the majority of islets cultured in the presence of Pefabloc or ulinastatin were stained with AO and showed a “green color,” but they were not stained with PI, thus suggesting the protective effects of Pefabloc and ulinastatin against lethal damage.

Histological findings of AO/PI staining observed by confocal microscopy for isolated islets cultured with digestion supernatants during collagenase digestion for 30 min, with or without serine protease inhibitors. The islets were cultured overnight and cultured in the supernatant for 30 min at 37°C in a culture dish. A green color after acridine orange (AO) staining means viable cells, while a red color of propidium iodide (PI) staining means dead cells. A yellow color after AO/PI indicates the presence of damaged and dead cells. The observation of the islets using confocal microscopy with AO/PI revealed the protective effects of Pefabloc and ulinastatin against lethal damage. Scale bars: 200 μm.
The islet yields after 30 min of stationary digestion without serine protease, with Pefabloc or ulinastatin were 1304 ± 409, 1019 ± 259, and 851 ± 686 IEQ, respectively, thus showing no significant difference. However, the ATP content of these islets digested without serine protease, with Pefabloc or Ulinastatin was 1.8 ± 0.1, 2.3 ± 0.2, and 2.4 ± 0.4 μM, respectively, thus showing a significant difference between those islets without serine protease and those with Pefabloc (p < 0.05).
Discussion
The procedure of human islet isolation is complex and it requires a great deal of experience to yield sufficient islets for transplantation (1,15). The technique of islet isolation has advanced (10,11,13,18–20). A tissue dissociation chamber and recirculation of enzyme solution established by Ricordi et al., and purified enzyme, Liberase HI, has been used worldwide (11,13,18–20). Although substantial advances in islet isolation technology have been achieved, it is still difficult to recover all of the islets contained in a pancreas. Many factors influencing the collagenase digestion phase have been reported (10), but little is known about the morphological changes and energy levels of pancreatic grafts or islet cells during digestion. This study focused on the digestion phase of islet isolation and demonstrated a reduction of ATP and EC levels of pancreatic graft during the digestion phase, and established an evaluation system of the toxicity of digestion supernatant involving endogenous pancreatic enzymes against isolated islets. Moreover, the effect of serine protease inhibitors has also been clarified using this newly established system.
The current study confirmed the decrease of islet yield after over digestion of the pancreatic graft, which was consistent with previously reports (9,25). Morphological changes in the islets were commonly observed in the peripheral cells of the isolated islets during isolation, including breakage of the plasma membrane and loss of cells (14). Secondly, islet fragmentation and disintegration was observed after 60 and 120 min of collagenase digestion. These changes are caused by the toxic environment during digestion phase, including elevation of endogenous pancreatic enzymes (2,10,12,17,21–23). In fact, the trypsin activity was observed to increase with the progression of digestion.
The ATP contents of pancreatic grafts or islets are used to evaluate the viability of the graft or isolated islets (3,6–8). However, little is known about the ATP or adenosine levels during the digestion phase. This study showed that the ATP and TAN contents of pancreatic tissue significantly decreased during collagenase digestion from 15 min after starting the stationary digestion, when histological damage to the exocrine tissue was evident, but such damage to the islets was limited.
Histological damage to the exocrine tissue and islets was evident at 60 min after starting digestion. Therefore, ATP and TAN contents in digesting pancreatic grafts could be more sensitive and quantitative markers than the histological findings.
Isolated islets were cultured with supernatants obtained from various digestion periods. The histology of islets cultured with these fluids clearly showed a time-dependent disruption of the cellular integrity of islets. Furthermore, the levels of ATP of islets cultured for 30 min in the digestion supernatants were decreased with the length of digestion periods, indicating the detrimental effects of endogenous enzymes to the isolated islets.
The trypsin activity of the supernatants obtained from collagenase digested tissue was observed to increase with the progression of digestion, thus indicating the leakage of various endogenous enzymes including serine proteases from the digested exocrine tissue. An increase of such enzyme activity is harmful for islet isolation (2,10,12,17,21–23). However, the effect of inhibition of serine proteases on islet isolation still remains controversial (12,17,21–23).
The protease inhibitors, Pefabloc and ulinastatin, were added to islets cultured with supernatants obtained from collagenase digested tissue at various digestion periods, and these inhibitors clearly prevented the occurrence of histological damage to the islets. Moreover, these inhibitors maintained the energy levels of islets under such conditions. Finally, the assessment of the viability using confocal microscopy and staining of AO/PI indicated the maintenance of membrane integrities in the presence of these inhibitors. Therefore, the protective effect of Pefabloc and ulinastatin during the digestion phase was evident. This islet culture system might therefore be useful for evaluating the direct effect of drugs on islets during the digestion phase in the isolation of islet cells.
Footnotes
Acknowledgments
This work was supported in part by grants from the Japanese Ministry of Education, Culture, Sports, Science and Technology and in part by a Grant-in-Aid for Research on Human Genome, Tissue Engineering Food Biotechnology, Health Sciences Research Grants, Ministry of Health, Labor and Welfare of Japan and in part by a research grant from Fukushima Medical University. The reagents of adenosine nucleotides luciferase were provided by Kikkoman Corp. The authors also wish to thank Dr. Yuichi Endo (Immunology), and Dr. Masaya Yamamoto and Dr. Satoshi Waguri (Histology) for their valuable technical advice. The authors declare no conflict of interest.
