Abstract
Porcine islets are considered to be a promising resource for xenotransplantation. However, it is difficult to isolate porcine islets because of the marked fragility and rapid dissociation. Endogenous trypsin is one of the main factors to damage islets during the isolation procedure. Recent studies have suggested that trypsin inhibitors during the preservation of pancreas or the collagenase digestion can improve the result of islet isolation. In this study, we examined whether a 1-antitrypsin (Aralast™), which inhibits several endogenous proteases and has immunomodulatory properties, can protect islets from the proteases and improve the results of porcine islet isolation. Twelve porcine pancreata were divided into three groups: without Aralast group (standard, n = 5), preserved with Aralast using the ductal injection (DI) method (DI, n = 3), and with Aralast using the DI method and in the collagenase solution (DI+C, n = 4). Efficacy of islet isolation was assessed by islet yields, purity, and viability. The trypsin activity of the preservation and the digestion solution during the isolation procedure was measured. During islet isolation, the trypsin activity in DI+C group was significantly inhibited compared to the standard group, whereas DI group showed less effect than DI+C group. The average of postpurification islet equivalents (IEQ) per pancreas weight in the DI+C group was significantly higher than the standard group (standard: 3516 ± 497 IEQ/g, DI: 4607 ± 1090 IEQ/g, DI+C: 7097 ± 995 IEQ/g; p = 0.017 between standard and DI+C). In the DI+C group, stimulation index was higher than in other groups, although there was no significant difference. The presence of Aralast in both DI solution and collagenase solution markedly inhibited trypsin activity during pancreas digestion procedure and improved the porcine islet isolation. Inhibition of trypsin activity by Aralast could improve porcine islet isolation.
Introduction
Islet transplantation has become a viable option for patients with type 1 diabetes (23,24). However, the worldwide donor shortage motivates the search for alternative donor strategies. Porcine islets are considered to be an unlimited source for xenotransplantation. However, a stable isolation method for porcine islet has not yet been established because the marked fragility and rapid dissociation of porcine islets is a basic problem in isolating functioning islets.
One factor that could potentially affect the recovery and viability of islets is the endogenous protease activity of the donor pancreas. Trypsin is considered to be one of the main proteases to damage islets during the isolation procedure (4,22). Moreover, trypsin produced by the exocrine pancreas could significantly hinder enzymatic digestion (28). Recent studies have shown that supplementation of trypsin inhibitors during collagenase digestion can improve the results of the porcine and human islet isolation (1–4,6,22). In addition, we and other groups previously reported that the ductal injection (DI) of preservation solution at the procurement site improved the islet isolation results and adding trypsin inhibitors into DI solution further improved the results for porcine and human islet isolation (14,17–19,27).
Human α1-antitrypsin (hAAT), a serine protease inhibitor, has been shown to be effective to suppress endogenous trypsin and to prevent β-cells from apoptosis (29). hAAT has also been reported to have immunomodulatory functions (5,7). Therefore, it is important to examine the effect of hAAT on islet isolation and islets themselves when hAAT will be used for islet transplantation. However, to our knowledge, the use of hAAT for islet isolation has not been reported. We have studied the effect of Aralast™, which is an α1-antitrypsin, both in the preservation solution using the DI method and in the collagenase solution for porcine islet isolation.
Materials and Methods
Porcine Islet Isolation
Pancreata from approximately 2-year-old female pigs were obtained at a local slaughterhouse. Twelve porcine pancreata were divided into three groups: without Aralast group (standard, n = 5), preserved with Aralast using the DI method (DI, n = 3), and with Aralast using the DI method and in collagenase solution (DI+C, n = 4). We started the procurement of pancreas about 30 min after the cessation of heart beating. After removing the duodenum from the donor pancreas, we immediately inserted a cannula into the main pancreatic duct and infused about 200–250 ml of extracellular-type trehalose-containing Kyoto (ET-KYOTO) solution (Otsuka Pharmaceutical Factory Inc., Naruto, Japan) for ductal protection. In the standard group, crude ET-KYOTO was infused whereas Aralast was added into the solution in DI and DI+C groups (1 g/L in DI group and 0.5 g/L in DI+C group, respectively). And then we removed fatty tissues from the pancreas and put the pancreas into the two-layer [ET-KYOTO/oxygenated perfluorochemical (PFC)] preservation container (13). Warm ischemic time (WIT) was defined as the time between the cessation of heart beating and the placement of the pancreas into the preservation solution. Cold ischemic time (CIT) was defined as the time between the placement of the pancreas into the preservation solution and the start of islet isolation.
Islet isolation was conducted by modified Ricordi method (9,11,12,20,23). In brief, after decontamination of the pancreas, the ducts were perfused with a cold Liberase HI (1 mg/ml; Roche Molecular Biochemicals) solution in a pressure-controlled manner. In the DI+C group, Aralast was added into the Liberase HI solution (1 g/L), whereas Aralast was not used in this step for the other two groups. After distention, the whole pancreas was then cut into about nine pieces, placed in a sterilized Ricordi chamber (1 L), and shaken gently. While the pancreas was digested by recirculating the enzyme solution through the Ricordi chamber, we monitored the extent of digestion. The digested tissue was collected into RPMI-1640 medium (Gibco) with 10% fetal bovine serum and washed with University of Wisconsin (UW) solution to remove the enzyme. The phase I period was defined as the time between the placement of the pancreas in the Ricordi chamber and the start of collecting the digested pancreas. The phase II period was defined as the time between the start and end of the collection.
Islets were purified with a continuous density gradient with iodixanol-ET-Kyoto solution as previously described (10,16). For the continuous density solution, low-density (density: 1.077) and high-density (density: 1.095–1.125) Iodixanol-ET-Kyoto solutions were produced by changing the volumetric ratio of iodixanol and ET-Kyoto solution.
Islet Evaluation
Islet preparations were evaluated for yield, purity, and morphology by using dithizone (3 mg/ml) (Sigma Chemical Co., St. Louis, MO) staining. The crude number of islets in each diameter class was determined by counting islets using an optical graticule. The crude number of islets was then converted to the standard number of islet equivalents (IEQ; diameter standardized to 150 μm) (21). Gross morphology was qualitatively assessed by scoring the islets for shape (flat vs. spherical), border (irregular vs. well rounded), integrity (fragmented vs. solid/compact), uniformity of staining (not uniform vs. perfectly uniform), and diameter (least desirable: all cells <100 μm/most desirable: more than 10% of the cells >200 μm). Each parameter was graded from zero (the worst) to 2 (the best), so that the worst islet preparations were given a cumulative score of zero and the best a score of 10. Spherical, well-rounded, solid/compact, uniformly stained, and large islets were characterized as the best islets (12). Islet viability after purification was assessed using trypan blue staining. Fifty islets were inspected and their individual viability was determined visually, followed by calculation of their average viability.
In vitro islet function was assessed by monitoring the insulin secretory response of the islets during glucose stimulation as described previously (23). Briefly, 5,000 IEQ were incubated with either 2.8 or 25 mM glucose in RPMI-1640 for 2 h at 37°C in a 5% CO2 atmosphere. The supernatant was collected and insulin levels were determined using an enzyme-linked immunosorbent assay kit (ALPCO Diagnostics, Windham, NH). The stimulation index was calculated by determining the ratio of insulin released from islets in the high glucose concentration to the low concentration. The data were normalized by total DNA.
For in vivo assessment of the islet function, nude mice (Harlan, Houston, TX) rendered diabetic by a single injection of streptozotocin (STZ) at a dose of 120 mg/kg were used. When the nonfasting blood glucose level exceeded 350 mg/dl for 2 consecutive days, the mice were considered to be diabetic. The 10,000 IEQ porcine islets obtained from DI and DI+C group were transplanted into the renal subcapsular space of the left kidney of a diabetic nude mouse immediately after isolation. During the 30-day posttransplantation period, the nonfasting blood glucose levels were recorded at three times per week. Normoglycemia was defined when two consecutive blood glucose level measurements showed less than 200 mg/dl. No statistical differences in either pretransplantation blood glucose levels or body weight were observed among the three groups. These experiments were approved by the Institutional Animal Care and Use Committee (IACUC) of Baylor Research Institute.
Measurement of Trypsin Activity During Islet Isolation
To measure the trypsin activity released from the pancreas during the preservation and digestion, samples were taken from solutions four times (1: preservation solution after arriving at the isolation facility; 2: the collagenase solution after the distention step; 3: the collagenase solution when the first free islet was detected in the solution, named the first collection; 4: the collagenase solution at the end of phase I). Three milliters of 0.3 mM N-bensoyl-l-arginine ethylester reagent (BAEE; Sigma) were incubated for 5 min and then 50 μl of the solution sample was added. The trypsin activity was measured by absorption spectrophotometry (λ253 nm) using BAEE for the trypsin substrate, as previously reported (19).
Statistical Analysis
All results were expressed as the mean ± SE. Differences among the three groups were analyzed by ANOVA followed by Student's t-test with Bonferroni correction. Values of p < 0.05 were considered significant in ANOVA and in Student's t-test with Bonferroni correction. Differences of ratio among the three groups were analyzed by chi-squared test with Ryan method.
Results
Porcine Islet Isolation Characteristics
The islet isolation variables are shown in Table 1. There were no significant differences in WIT, CIT, pancreas weight, phase I time, phase II time, and undigested tissue volume among the three groups. The average of WIT was more than 40 min; therefore, these pancreata were in marginal condition.
Porcine Islet isolation Variables
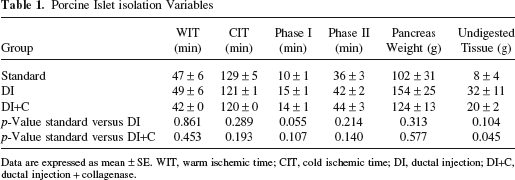
Data are expressed as mean ± SE. WIT, warm ischemic time; CIT, cold ischemic time; DI, ductal injection; DI+C, ductal injection + collagenase.
Postpurification variables are shown in Table 2. There were no significant differences in purity, viability, final tissue volume, and the rate of postpurification recovery among the three groups. Islet yield (IEQ) per pancreas weight (IEQ/g) before purification was higher in the DI+C group than the standard and DI groups (standard: 6780± 1118 IEQ/g, DI: 5951 ± 1379 IEQ/g; DI+C: 10360 ± 903 IEQ/g, respectively) (Fig. 1A, although there was no significant difference. IEQ/g after purification was significantly higher in the DI+C group than the standard group (standard: 3516 ± 497 IEQ/g; DI: 4607 ± 1090 IEQ/g; DI+C: 7097 ± 995 IEQ/g, respectively, p = 0.017 in standard vs. DI+C group) (Fig. 1B).
Postpurification Variables

Data are expressed as mean ± SE. Recovery rate (%): islet equivalents (IE) after purification/IE before purification × 100.

Islet yield before and after purification. (A) Prepurification islet equivalents (IEQ)/g in three groups [standard, ductal injection (DI), ductal injection + collagenase (DI+C)]. (B) Postpurification IEQ/g in the three groups. Data are expressed as mean ± SE. *p = 0.017.
Trypsin Activity
We measured the trypsin activity of the solution during the preservation and the digestion steps, and examined whether Aralast in DI and the collagenase solution could inhibit the trypsin activity. In all groups, the trypsin activity in the preservation solution was very low (Fig. 2). On the other hand, the trypsin activity rose from the distention step and it increased along with the proceeding of the digestion step (Fig. 2). At the end of phase I, the DI group showed lower, but not significant, trypsin activity than the standard group, whereas the DI+C group showed further low value and it was significantly lower than the standard groups (BAEE unit: standard: 58±14; DI: 11 ± 3; DI+C: 6 ± 2, respectively; p = 0.012 in standard vs. DI+C groups). These findings indicated that Aralast in both DI and collagenase solution could inhibit trypsin activity most effectively.

Impact of human α1-antitrypsin (hAAT) on trypsin activity in the preservation and the digestion solution in the three groups (as above). The method of measuring trypsin activity is described in Materials and Methods. A N-bensoyl-l-arginine ethylester (BAEE) unit was defined as a change in optical density of 0.001 per minute. Data are expressed as mean ± SE. *p = 0.012.
Evaluation of Islet Function In Vitro and In Vivo
The average of the stimulation index in the DI+C group was higher than the other two groups, although it did not reach the statistical significance (standard: 1.4 ± 0.3; DI: 3.3± 1.9; DI+C: 7.1 ± 3.1, respectively) (Fig. 3).

Assessment of islet function after isolation in vitro and in vivo. (A) Stimulation index in the three groups. There was no significant difference among the three groups. (B) In vivo transplantation success rate according to the three groups. There was no significant difference among the three groups. Data are expressed as mean ± SE (A) or mean (B).
To assess the islet graft function in vivo, 10,000 IEQ of DI and DI+C group were transplanted below the kidney capsule of STZ-induced diabetic nude mice. The blood glucose levels in 5 of 12 mice (41.7%) receiving islets from the DI group and 6 of 11 mice (54.5%) in the DI+C group decreased and reached normoglycemia. The success rate in the DI+C group was higher than the DI group, although there was no statistical significance.
Discussion
Porcine islet cells are potentially excellent substitute for human islet cells. However, porcine islets are known to be particularly difficult to isolate because of a weak islet capsule (15) and the fragility to be easily fragmented (25) during isolation. The endogenous proteases from the exocrine pancreas could aggravate the disintegration of the islets.
We showed an increase in trypsin activity during the porcine islet isolation procedure (Fig. 1), which is well consistent with other reports (4,22). Trypsin disintegrates a large number of proteins and accelerates the digestion process. This may influence the islet isolation in two aspects: it damages islets directly and decreases the exogenous enzymatic activity. The combination of exogenous collagenase and endogenous trypsin may give rise to a nonspecific, fast, and uncontrollable tissue disintegration process. Heiser et al. reported that low trypsin activity during the isolation procedure guaranteed high islet yields (4). It has been reported that using protease inhibitors during preservation or digestion step improved the islet isolation results in both animal and human isolation (1–4,6,14,17–19,22,27). We previously demonstrated that a trypsin inhibitor, ulinastatin, was extremely useful for human pancreas preservation from marginal donors (9,10). Indeed, ulinastatin is a potential trypsin inhibitor; however, its availability is limited in many countries such as the United States. Therefore, we have sought an alternative which can be used worldwide.
hAAT is a serine trypsin inhibitor, which is used for treatment of α-antitrypsin deficiency (8). It can inhibit several endogenous proteases. Moreover, it can prevent diabetes onset in non-obese diabetic (NOD) mice (5) and prolong islet allograft survival in a non-autoimmune murine model (7) and inhibit pancreatic B-cell apoptosis in vivo (29). Therefore, hAAT could apply for immunomodulation and/or islet projection for islet transplantation. Hence, it is important to clarify the effect of hAAT on islet isolation.
In this study, we evaluated the effects of hAAT in the preservation and the digestion solution on the porcine islet isolation and the function of isolated islets. Our data clearly showed for the first time that hAAT inhibited trypsin activity during the islet digestion step and improved the islet yield from porcine pancreas. The islet function evaluated by in vitro and in vivo analysis was better in the DI+C group than the other two groups, although there was no significant difference.
There are several considerable points in the present study. First, the used pancreata were in a marginal condition. The average of the warm ischemic time was more than 40 min, due to necessary processes for slaughtering pigs at a local slaughterhouse. Even 30 min of warm ischemic condition can cause severe apoptosis in porcine islets (26); therefore, islets in this study are considered to be strongly damaged. Indeed, for the in vivo functional analysis, 10,000 IEQ were required to cure diabetic nude mice. In other words, however, our method with hAAT enabled us to obtain functional islets from porcine pancreata even under such a severely damaged condition.
Second, the interaction between hAAT and the digestive enzymes was our concern. We examined the effect of collagenase on the hAAT activity for trypsin inhibition. Actually, collagenase inhibited hAAT activity for trypsin inhibition after 30-min exposure (data not shown). In other words, the effect of hAAT can maintain for at least 30 min; therefore, hAAT should work efficiently during the digestion step. On the other hand, we did not examine whether the collagenase activity was inhibited by hAAT directly. However, our isolation results did not show any tendency that the collagenase activity was suppressed when hAAT was added (there were no differences in the digestion time, the dilution time, and the undigested tissue volume among the three groups).
In conclusion, addition of hAAT into ductal injection solution and collagenase solution effectively suppressed trypsin activity and substantially increased islet yields from marginal porcine pancreata.
Footnotes
Acknowledgments
The authors would like to thank Ms. Yoshiko Tamura and Mr. Greg Olsen (Baylor University Medical Center) for their technical supports. This research is partially supported by All Saints Health Foundation and Otsuka Pharmaceutical Factory Inc. The authors declare no conflicts of interest.
