Abstract
Recent advances in liver tissue engineering have encouraged further investigation into the evaluation of therapeutic benefits based on animal disease models. In the present study, liver tissues were engineered in coagulation factor IX knockout (FIX-KO) mice, a mouse model of hemophilia B, to determine if the tissue engineering approach would provide therapeutic benefits. Primary hepatocytes were isolated from the liver of wild-type mice and suspended in a mixture of culture medium and extracellular matrix components. The hepatocyte suspension was injected into the space under the bilateral kidney capsules of the FIX-KO mice to engineer liver tissues. The plasma FIX activities (FIX:C) of the untreated FIX-KO mice were undetectable at any time point. In contrast, the liver tissue engineered FIX-KO mice achieved 1.5–2.5% of plasma FIX activities (FIX:C) and this elevated FIX:C level persisted throughout the 90 day experimental period. Significant FIX mRNA expression levels were found in the engineered liver tissues at levels similar to the wild-type livers. The present study demonstrates that liver tissue engineering could provide therapeutic benefits in the treatment of hemophilia B.
Keywords
Introduction
Liver tissue engineering technology has evolved over the past several years, and spurred significant interest as potential new therapies for several types of liver disease, including genetic deficiencies (14, 18–21, 23). Several studies have succeeded in achieving functional persistency of the liver tissues engineered in subcutaneous sites, under the kidney capsule space, or the omentum (8, 9, 15–17, 19, 20, 28). Once the liver tissue engineering procedures established clinically applicable success, several types of liver diseases will be treated with the tissue engineering procedures. Because most blood clotting factors are produced by the liver (3, 11), liver tissue engineering could be a valuable approach in the treatment of inherited clotting factor deficiencies, including hemophilia A and B. As a proof-of-principle, therapeutic effects were achieved in a mouse model of hemophilia A engineered liver tissue using wild-type hepatocytes (19).
Hemophilia B is a recessive X-chromosome-linked congenital bleeding disorder, and its prevalence is approximately 1 in 30,000 males. The main clinical manifestations of hemophilia B are unpredictable, recurrent, spontaneous bleeding events in various sites including the major joints and internal organs. The bleeding diathesis of hemophilia B is due to a failure in the production of biologically active coagulation factor IX (FIX) (2). It has been well established that achieving plasma FIX activities (FIX:C) as low as 1% markedly changes the phenotype of hemophilia B patients from severe to moderate, and greatly improves the symptoms and quality of life of the individuals (2).
Provided that efficient engraftment of hepatocytes as well as functional maintenance of clotting factor productions can be achieved in individuals with hemophilia B, cell-based therapy could be a feasible and safe therapeutic option for this disease. Because hepatocytes are the primary cell type in the liver involved in FIX clotting factor production (3), hepatocyte-based therapy could be effective in establishing a novel therapy for hemophilia B. Recent experiments on hepatocyte transplantation into the liver of FIX knockout (FIX-KO) mice achieved 1–2% of FIX:C levels that persisted throughout the experimental period (26). These experiments have encouraged further investigation of the potential of a tissue engineering approach for hemophilia B.
The present study was conducted to engineer liver tissues using wild-type hepatocytes under the kidney capsule space of FIX-KO mice. The FIX-related functionality of the engineered liver tissues was assessed, including the plasma FIX activity levels, histological investigations, and FIX mRNA expression of the engineered tissues. The results show that hepatocytes in the engineered liver tissues retained the ability to produce FIX, thus resulting in an elevation of FIX:C at 1.0–2.5% of normal plasma levels. The results demonstrate that a liver tissue engineering approach could be a novel therapeutic option for the treatment of hemophilia B.
Materials and Methods
Animals
Factor IX knock-out (FIX-KO) mice and wild-type C57BL/6 mice, which were syngenic to the FIX-KO mice (originally purchased from The Jackson Laboratory, Bar Harbor, ME), were maintained at the Animal Care Center at Nara Medical University. FIX-KO mice, 10–15 weeks old, were used as the recipients of the liver tissue engineering procedure. Wild-type C57BL/6 mice, 10–12 weeks old, were used for hepatocyte donors. All animal studies were conducted under the protocol approved by the Animal Care Committees at Tokyo Women's Medical University and Nara Medical University. Mice were placed in cages within a temperature-controlled room with a 12-h light/dark cycle and ad libitum access to food and water.
Hepatocyte Isolation and Purification
Hepatocytes were isolated from wild-type mice using a modified two-step collagenase perfusion method as previously described (8, 16, 19, 20). Briefly, the liver was primarily perfused with Hank's balanced salt solution (HBSS) (Sigma, St. Louis, MO) containing 0.09% EGTA followed by a second perfusion using HBSS containing 0.03% collagenase (Sigma) and 5 mM CaCl2. Isolated cells were filtered through a nylon mesh membrane and hepatocytes were then purified by centrifuging at 50 × g for 5 min followed by Percoll (GE Healthcare, Buckinghamshire, UK) isodensity centrifugation. Cells were resuspended in DMEM medium (Sigma), and the cell viability was determined by trypan blue exclusion test. In the present studies, experiments were conducted only when the hepatocyte viabilities exceeded 95%.
Liver Tissue Engineering Procedures
Hepatocytes were prepared for engineering liver tissue under the kidney capsule space as previously described (8, 15, 17, 19). In brief, isolated hepatocytes were resuspended with serum-free DMEM with an equal volume of Engelbreth-Holm-Swarm (EHS) matrix (Matrigel, BD Biosciences, Bedford, MA) to a final ratio of 1.5 × 106 hepatocytes/100 μl. A total of 2.4 × 106 hepatocytes were transplanted under the bilateral kidney capsule space of the FIX-KO mice. As an experimental control, several FIX-KO mice received only an abdominal incision and did not receive hepatocyte infusion (sham operation). All the surgical procedures were performed under inhalation anesthesia using isofluorane (Forane, Abbott Laboratories, Abott Park, IL). To avoid excessive surgical procedure-related bleeding, all FIX-KO mice including the sham operation group received an IP injection of 0.5 ml pooled normal mouse plasma twice, 30 min prior and 1 h after the tissue engineering surgical procedure (22).
Coagulation Assay for Plasma FIX Activity
Blood samples were periodically obtained from the retro-orbital plexus of the recipient FIX-KO mice before and after the tissue engineering procedures. The blood samples were immediately anticoagulated with 0.1 vol of 3.8% sodium citrate (Sysmex, Kobe, Japan). After centrifugation, plasma samples were stored at −80°C until being analyzed. The FIX activity (FIX:C) was quantified by one-stage clotting assay based on the activated partial thromboplastin time using human FIX-deficient plasma (Sysmex). Pooled mouse plasma obtained from wild-type C57BL/6 mice was used as FIX:C standard. Each measurement was reported after subtraction of the preoperational baseline FIX:C levels. We have previously confirmed that FIX activity levels as low as 0.25% of normal mouse plasma could be successfully measured using this assay (26). The development of neutralizing antibodies against FIX, called FIX inhibitor, was assessed by the Bethesda method using plasma obtained at sacrifice time (26). Briefly, residual FIX:C was determined after incubating equal volumes of sample plasma with normal mouse plasma at 37°C for 2 h, and was compared with residual FIX:C of a mixture of FIX knockout mouse and normal mouse plasma. The presence of a significant titer of FIX inhibitor was determined as >0.5 Bethesda unit/ml in this assay.
Histological Analyses
Naive liver specimens and engineered liver tissues containing the neighboring kidney of the recipient FIX-KO mice were harvested and fixed in 10% buffered formalin. The specimens were also embedded in Tissue-Tek O.C.T. compound (Sakura, Torrance, CA). Formalin-fixed specimens were paraffin embedded and sliced into 5-μm-thick sections, which were subsequently processed for hematoxylin & eosin (H&E) staining or histochemical staining. For the cellular glycogen detection, Periodic-Acid-Schiff (PAS) histochemical staining was performed as described previously (19). To confirm the staining specificity of the cellular glycogen, serial sections were pretreated with salivary amylase for 60 min followed by the same PAS staining procedures.
Laser Capture Microdissection (LCM)
The specimens embedded in the Tissue-Tek O.C.T. compound were sliced into 8-μm-thick sections. The sections were then stained with HistoGene LCM Frozen Section Staining Kit (Arcturus, Mountain View, CA) to visualize the engineered liver tissues under the kidney capsule followed by LCM using a PixCell II LCM System (Arcturus). The microdissected tissues were harvested and subjected to total RNA extraction using the PicoPure RNA Isolation Kit (Arcturus). Isolated RNA was amplified using the RiboAmp RNA Amplification Kit (Arcturus) as described previously (28).
RT-PCR
One microgram of total RNA was reverse transcribed using Omniscript RT Kit (Qiagen, Hilden, Germany), and the first-strand cDNA samples were subjected to PCR amplification in the StepOnePlus Real-Time PCR Systems (Applied Biosystems, Tokyo, Japan) followed by 2% agarose gel electrophoresis. In addition to the FIX gene expression, albumin (Alb), ornithine transcarbamylase (OTC), and factor VII (FVII) were also analyzed as other gene expression specific to hepatocytes. Housekeeping gene, β-actin (ACTB) was used as reference gene. PCR primers for the mouse sequences (FIX, FVII, Alb, OTC, and ACTB) were purchased from TaqMan Gene Expression Assay (Applied Biosystems, Assay ID: Mm01308427_m1, Mm00487329_m1, Mm 00802090_m1, Mm00493267_m1, and Mm00607939_s1, respectively).
Statistical Analysis
Significant differences were tested by repeated measures ANOVA with the use of the ystat2006 software program (Igakutoshoshuppan, Tokyo, Japan). If a probability value of p < 0.05 was obtained, the Dunnett's test was used for comparison for each individual group. A value of p < 0.05 was considered to be significant. Data were presented as the mean ± SD.
Results
Liver Tissue Engineering Procedure Used for the FIX-KO Mice
In order to avoid surgical procedure-related bleeding, all the FIX-KO mice including the sham operation group received IP injections of 0.5 ml pooled normal mouse plasma 30 min before and 30 min after the surgical procedure. Liver tissue engineering procedures based on transplantation of hepatocytes suspended with EHS-matrix or sham operation were successfully carried out using FIX-KO mice without any issues related to bleeding and all the recipient mice survived throughout the experimental period. At day 90, some of the recipient FIX-KO mice were sacrificed for histological analyses and mRNA analyses of the engineered liver tissues.
Effect of Liver Tissue Engineering on Coagulation FIX Activity of Hemophilia B
The FIX activity (FIX:C) in plasma obtained from FIX-KO mice received liver tissue engineering or sham operation was quantified by one-stage clotting assay (Fig. 1). The FIX:C of the FIX-KO mice prior to the tissue engineering procedures and the sham operation groups were consistently below the detection limit (0.25% of normal) and thus the values were expressed as 0. In contrast, the FIX:C of FIX-KO mice that received liver tissue engineering procedures increased to 1.60 ± 0.55 at week 3, and this elevated FIX:C was stably maintained throughout the experimental period. On the other hand, no elevation in the FIX:C levels were detected in the sham-operated mice. Overall, the FIX:C levels were significantly higher in the recipient mice in comparison to the levels found in the sham-operated mice at every time point examined (p < 0.05). These results indicated that engineering liver tissues by transferring nonhemophilic hepatocytes into the individuals with hemophilia B could provide a therapeutic effect by de novo production of coagulation factor IX.

The plasma FIX activity (FIX:C) levels of FIX-KO mice following liver tissue engineering. FIX-KO mice received an injection of hepatocytes resuspended with EHS-matrix into the bilateral under the kidney capsule spaces. Each line with circles (n = 7) represents an individual FIX-KO mouse that received hepatocyte injection and the line with triangles (n = 7) represents FIX-KO mouse with sham operation.
Morphology and Functions of the Engineered Liver Tissues
The kidney tissues were harvested at day 90 to determine if the hepatocytes within the engineered liver tissues maintained healthy morphology and functions. H&E staining revealed hepatocytes with large cuboidal shapes and eosinophilic cytoplasm next to the renal tubulus (Fig. 2A). Glycogen synthesis, an important liver function, was confirmed by positive PAS staining in the hepatocytes of the engineered liver tissues (Fig. 2B). Staining specificity for cellular glycogen in the PAS staining was confirmed by the finding that pretreatment with salivary amylase in the tissue sections diminished the PAS staining (Fig. 2C).
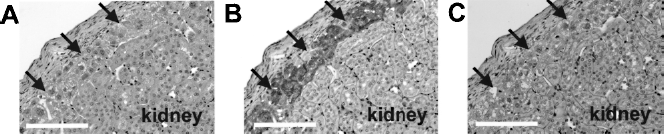
Histological analyses of the engineered liver tissues in the FIX-KO mice. Sections were obtained from FIX-KO mice at day 90 of the experiment. (A) H&E staining and (B, C) PAS staining. PAS staining was performed to confirm the cellular glycogen in the engineered liver tissues that were either nontreated (B) or pretreated with salivary amylase (C). Arrows indicate engineered liver tissues under the kidney capsule. Scale bars: 100 μm.
The liver-specific mRNA expression levels were assessed in the engineered liver tissue specimens. To minimize contamination from the surrounding renal tissues, the engineered liver tissues were selectively dissected from the surrounding kidney tissues by LCM. As shown in Figure 3, the engineered tissue specimens showed significant expression levels of FIX, FVII, albumin, and OTC at levels to those of naive healthy livers. Negative FIX signals for samples from the liver and kidney of FIX-KO mice and positive FIX signal from the liver of wild-type mouse confirmed the detection specificity for mouse FIX mRNA in the present assay. No detectable signals were observed for the hepatocyte-specific mRNAs in the kidney samples.
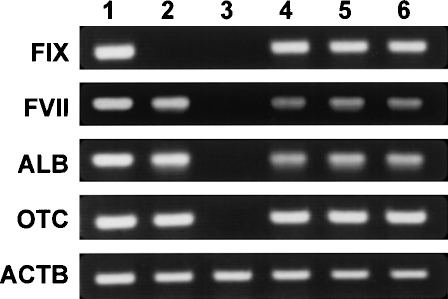
Hepatocyte-specific mRNA expression in the livers, kidney, and engineered liver tissues. The target tissues were selectively dissected under LCM from cryosections obtained at day 90 of the experiment. mRNA expression levels were assessed for coagulation factor IX (FIX), coagulation factor VII (FVII), albumin (ALB), ornithine transcarbamylase (OTC), and β-actin (ACTB). Lane 1: native liver of wild-type C57BL/6 mouse; lane 2: native liver of FIX-KO mouse; lane 3: native kidney tissue of FIX-KO mouse; lanes 4–6: liver tissues engineered under the kidney capsule space composing hepatocytes isolated from wild-type C57BL/6 mouse. Each lane demonstrates samples from three different FIX-KO recipient mice.
Discussion
The present study demonstrates a relatively simple procedure to engineer liver tissue in hemophilic animals. After the infusion of wild-type hepatocytes under the kidney capsule space of FIX-KO mice, engrafted hepatocytes functionally survived and small liver tissues were engineered. The functional performance for the FIX production of the engineered liver tissues yielded therapeutic levels of plasma FIX:C. The FIX:C plasma levels persisted for 90 days, the length of the present study. The present report is the first conclusive therapeutic evidence of a liver tissue engineering approach to hemophilia B.
The FIX-KO mouse is a valuable mouse model for hemophilia B. FIX-KO mice fail to produce any hepatic FIX mRNA, have no circulating FIX protein, bleed excessively with hemostatic events, and display changes of bleeding-induced poor wound healing (11). In order to avoid any surgical process related to bleeding events, normal mouse plasma was injected twice prior and after the tissue engineering procedure. With these plasma replenishments, all the recipient mice tolerated the tissue engineering surgical procedures. This uneventful process may highlight the simplicity of the tissue engineering procedures, and they could be reasonably clinically applied to individuals with hemophilia.
One of the approaches toward engineering a functional liver tissue has been the infusion of a hepatocyte mixture with extracellular matrix components under the kidney capsule space (15, 17, 19). The extracellular matrix rich in laminin and type IV collagen contributes to the stable persistence of the hepatic functions including FIX expression (15). The ability to express clotting factor VIII was documented in previous studies (17, 19). The present study demonstrated that elevated FIX:C in the recipient FIX-KO mice persisted throughout the 90-day experimental period. Life-long functional persistence was confirmed in a recent study where the tissue-engineered mice were followed for over 450 days, which is nearly the life span of a normal mouse (unpublished data).
Another important determinant in the liver tissue engineering is an ability to undergo regenerative growth, an important feature of the liver (10). Although the present study did not address to this point, previous investigations established that engineered liver tissues established by the current approach are able to proliferate at similar levels to the naive livers in face of liver regenerative stimuli (16, 19, 20). Liver regeneration occurs through two distinct pathways: compensatory regeneration as well as the direct hyperplasia mode of regeneration (10). Previous studies have demonstrated engineered liver tissue specimens are also able to undergo both regeneration modes. If it is possible to regulate the in vivo liver growth process, engineered liver tissue could be induced to form a larger tissue. Functional control of the liver volume will allow liver tissue engineering approach to provide the next generation therapy for hemophilia as well as other forms of liver diseases.
The kidney capsule space offers favorable hepatocyte engraftment followed by persistent survival (8, 15–17, 19). The kidney site offered higher cell engraftment rate in comparison to intrahepatic transplantation when human hepatocytes were transplanted into NOD/SCID mice (17). Successful phenotypic corrections of the disease phenotype of hemophilia A have been described in experiments where wild-type hepatocytes or coagulation factor-producing cells were transplanted into the kidney capsule (13, 19). The advantage of the kidney site over the conventional liver site was also documented by Horne et al. (6) describing that humoral and cellular responses to the allogenic hepatocytes engrafted into the kidney site were blunted in comparison to those responses to the intrahepatic engrafted hepatocytes. Tissue factor-dependent coagulation around the transplanted hepatocytes has also been shown to be one of the mechanisms for poor cell engraftment when hepatocytes are transplanted intrahepatically (24). We speculate that transplanting these cells into a closed space (i.e., under the kidney capsule space) may therefore prevent the generation of such coagulation, thus resulting in a higher hepatocyte engraftment rate for the kidney capsule site in comparison to the intrahepatic site.
It is important to note that the hepatocytes within the engineered tissues showed stable functionality for the FIX production throughout the 90 days experimental period. Hepatocytes are known to possess cellular machineries (i.e., posttranslational modification) to produce biologically active forms of clotting factors (1) and thus are considered as a suitable candidate for the cell-based therapy for hemophilia (4, 12, 21). However, given that clotting factor-producing cells could be successfully and stably engrafted in vivo, the cell source may not be limited to wild-type hepatocytes. The tissue engineering approach could also be highlighted by the recently developed cellular technologies to generate coagulation factor-producing cells (5, 7, 13, 25, 27). Considerable progress has recently been made using gene therapy vector systems. The addition of the FIX expression cassette to the naive FIX expression system of the hepatocytes could theoretically enhance the capacity for the FIX production. It would be reasonable to propose that combining tissue engineering and gene therapy technologies will bring higher levels of therapeutic efficacy.
In summary, this study demonstrated that hepatocytes isolated from wild-type C57BL/6 mice can be effectively engrafted under the kidney capsule space of FIX-KO mice and thus form liver tissue that sufficiently and stably provides therapeutic levels of FIX in the recipients. The therapeutic effect might be enhanced by genetically modifying the hepatocytes to induce the cells to produce biologically active FIX at higher levels. In all, the current work thus can serve as the basis to create a novel cell-based therapy for hemophilia B.
Footnotes
Acknowledgments
The authors would like to thank Ms. S. Taminishi (Department of Pediatrics, Nara Medical University) for technical assistance and C. Kohno (Tokyo Women's Medical University) for histological assistance. This work was supported in part by Grant-in-Aid (K.O. No. 21300180) and Special Coordination Funds for Promoting Science and Technology (K.O. and T.O.) from the Ministry of Education, Culture, Sports, Science and Technology (MEXT) Japan, Bayer Hemophilia Award Program (K.O.), Novartis Foundation for the Promotion of Science (K.O.), and Public Trust Surgery Research Fund (K.O.).
