Abstract
Hybrid artificial liver (HAL) is an extracorporeal circulation system comprised of a bioreactor containing immobilized functional liver cells. It is expected to not only serve as a temporary liver function support system, but also to accelerate liver regeneration in recovery from hepatic failure. One of the most difficult problems in developing a hybrid artificial liver is obtaining an adequate cell source. In this study, we attempt to differentiate embryonic stem (ES) cells by hepatic lineage using a polyurethane foam (PUF)/spheroid culture in which the cultured cells spontaneously form spherical multicellular aggregates (spheroids) in the pores of the PUF. We also demonstrate the feasibility of the PUF-HAL system by comparing ES cells to primary hepatocytes in in vitro and ex vivo experiments. Mouse ES cells formed multicellular spheroids in the pores of PUF. ES cells expressed liver-specific functions (ammonia removal and albumin secretion) after treatment with the differentiation-promoting agent, sodium butyrate (SB). We designed a PUF-HAL module comprised of a cylindrical PUF block with many medium-flow capillaries for hepatic differentiation of ES cells. The PUF-HAL module cells expressed ammonia removal and albumin secretion functions after 2 weeks of SB culture. Because of high proliferative activity of ES cells and high cell density, the maximum expression level of albumin secretion function per unit volume of module was comparable to that seen in primary mouse hepatocyte culture. In the animal experiments with rats, the PUF-HAL differentiating ES cells appeared to partially contribute to recovery from liver failure. This outcome indicates that the PUF module containing differentiating ES cells may be a useful biocomponent of a hybrid artificial liver support system.
Keywords
Introduction
The only effective therapy for liver failure is orthotropic liver transplantation, a treatment increasingly hampered by a shortage of donor organs. Hybrid artificial liver (HAL) might provide temporary support for liver function and also accelerate liver regeneration in recovery from hepatic failure. HAL is an extracorporeal circulation system comprised of a bioreactor containing immobilized functional liver cells. We previously developed a HAL using a polyurethane foam (PUF)/spheroid culture, in which the cultured cells spontaneously formed spherical multicellular aggregates (spheroids) in the pores of the PUF (11). Animal experiments in rats, dogs, and pigs have demonstrated the curative effect of the PUF-HAL (12,18,21).
However, a lack of hepatocytes is the most critical obstacle to the clinical application of HALs. Primary human hepatocytes would most likely be the best source of cells for HAL, but their availability is limited. The use of stem cells has recently received much attention as a novel source (24). Embryonic stem (ES) cells are pluripotent lines isolated from the inner cell mass of blastocysts (7,15). ES cells have unlimited proliferative capability and the potential to differentiate into a variety of cell lineages. Many groups have already reported the differentiation of ES cells into hepatocyte-like cells (1,4,5,9,14,20,22,26). In a prior study, we also reported that mouse ES cells in PUF/spheroid culture expressed spheroid formation and, with growth factor treatment, some liver-specific genes (16,17). In this study, we attempt to differentiate ES cells into hepatic lineage by using PUF/spheroid culture and treatment with sodium butyrate (SB), a compound known to be a differentiation-promoting agent (3,8). We also investigate the feasibility of PUF-HAL by comparing ES cells with primary hepatocytes in in vitro and ex vivo experiments.
Materials and Methods
PUF Stationary Culture for ES Cell Differentiation
The mouse ES cells (129SV line; Chemicon, Pitts-burgh, PA, USA) were maintained in an undifferentiated state on a mouse embryonic fibroblast (MEF). Undifferentiated ES cells were resuspended in Iscove's modified Dulbecco's medium (Sigma-Aldrich, St. Louis, MO, USA) containing 2 mM l-glutamine (Sigma), 100 U/ml penicillin (WAKO Pure Chemical Industries, Osaka, Japan), 100 μg/ml streptomycin (Meiji Seika Kaisha, Tokyo, Japan), 20% fetal bovine serum, and 300 μM monothioglycerol (Sigma). PUF was manufactured through a foaming process by blending isocyanate with polyol (Inoac Co. Ltd., Nagoya, Japan). We cut a PUF block into a flat plate for the PUF stationary culture. Mouse ES cells were inoculated at a density of 2.0 × 106 cells in a 35-mm petri dish containing the PUF plate in 2 ml of each culture medium. After 4 h of culture, the PUF plate with immobilized cells was transferred to a 60-mm petri dish containing 6 ml of each culture medium. The media were replaced daily. To induce hepatic differentiation, we added SB (Sigma) to the culture medium at a concentration of 1 mM. The SB treatment was started at 4, 9, and 15 days of culture (Fig. 1). We also compared the liver-specific functions in each culture condition.

A schematic diagram of the experimental protocol for embryonic stem (ES) cell differentiation in polyurethane foam (PUF) stationary culture. Mouse ES cells were inoculated in a PUF plate and cultured. To induce hepatic differentiation, sodium butyrate (SB) was added to the culture medium at a concentration of 1 mM. The effect of SB treatment was compared under three different conditions (SB treatment 4d, 9d, and 15d, respectively).
PUF-HAL Module
The PUF-HAL module is a cylindrical PUF block with an 11-mm diameter and a height of 50 mm. It has many capillaries of 1.5 mm diameter for medium flow (Fig. 2). Undifferentiated ES cells (4.0 × 106) were immobilized in the PUF-HAL module using centrifugal force as previously reported (13). The module was connected to a simple culture circuit and cultured in the medium with a flow rate of 12 ml/min. After 9 days of culture, 1 mM SB was added to the culture medium for hepatic differentiation. The medium was replaced daily. Liver-specific functions were evaluated from 12 days of culture.
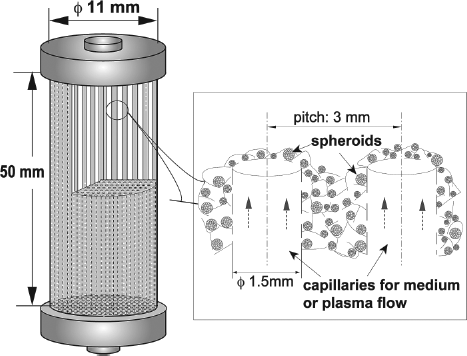
A schematic diagram of the PUF-HAL module. This is the multicapillary polyurethane foam packed-bed type of artificial liver module. This module is comprised of a cylindrical PUF block with many capillaries for medium or plasma flow. ES cells spontaneously formed spheroids in the pores of PUF between each capillary. HAL, hybrid artificial liver.
Evaluation of Liver-Specific Functions
We measured ammonia removal activity by replacing the culture medium with fresh culture medium with 1 mM NH4Cl at each evaluation. The decrease in ammonia concentration during a 6-h period was measured using the Wako ammonia test (WAKO). We measured the albumin concentration in the medium with an enzyme-linked immunosorbent assay, the ELISA starter accessory package (Bethyl Laboratories, Montgomery, TX, USA), and the mouse albumin ELISA Quantitation Kit (Bethyl Laboratories). The control for liver-specific function was primary hepatocytes isolated from C57BL/6 mice (6–7 weeks old, male, 20–23 g) (CLEIA Japan Inc., Tokyo, Japan) by the liver perfusion method using 0.05% collagenase. These were cultured and evaluated in an identical manner to the differentiating ES cells.
Ex Vivo Experimental Design
The extracorporeal circulation system for rats and the experimental procedure have been previously described (21), but they were slightly modified for the present study. Our system is comprised of an extracorporeal blood circulation tube connected to a plasma separator, and a plasma circulation tube connected to an artificial liver module (PUF-HAL module) (Fig. 3). The blood circulation, plasma circulation, and plasma separation flow rates were set to 1.0, 12.0, and 0.1 ml/min, respectively. Before the ex vivo experiment, all circulation was heparinized.

A schematic diagram of the extracorporeal circulation system containing the PUF-HAL module. Rats with a partial hepatectomy were treated with a PUF-HAL module with immobilized differentiating mouse ES cells (ES-HAL, n = 3) or primary mouse hepatocytes (Hep-HAL, n = 3). The same system without cells in the PUF-HAL module was used as a control (Control, n = 4).
Adult Wistar rats (7–8 weeks of age, male, 230–280 g) (Kyudo Co., Ltd, Saga, Japan) were given food and distilled water in a room at a constant temperature. They were maintained in accordance with the guidelines for animal experiments at Kyushu University, and the experimental protocols were approved by the Ethics Committee on Animal Experiments at Kyushu University. All surgical procedures were done under general anesthesia using isoflurane. The conditions were clean but not sterile. The two anterior liver lobes (two thirds of the liver) were removed using the standard Higgins and Anderson technique (10). The incision in the abdomen was sutured after surgery. We accessed extracorporeal circulation of blood through the carotid arteries and jugular veins of the rats.
We performed the ex vivo experiments using the same previously described procedure (21). The rats were treated with the PUF-HAL module containing differentiating mouse ES cells (ES-HAL) and primary mouse hepatocytes (5 × 107 cells, Hep-HAL). The control was the same system without cells in the module. We compared the levels of biochemicals used during treatment and posttreatment survival periods.
Results
Spheroid Formation of Mouse ES Cells in the Pores of PUF
Mouse ES cells formed multicellular spheroids in the pores of PUF (Fig. 4). The spheroids grew larger with cell proliferation. Cell proliferation was suppressed after SB treatment in each culture condition, and cell numbers gradually decreased (Fig. 5). Figure 6 shows the ammonia removal and albumin secretion rates of differentiating mouse ES cells per unit volume of PUF plate among different SB treatment conditions. Under all conditions, mouse ES cells expressed ammonia removal and albumin secretion functions after 21 days of culture. Starting SB treatment from 9 days of produced the strongest response.

Morphology of the mouse ES cells in PUF stationary culture at 3 days of culture. Mouse ES cells spontaneously formed spheroids in the pores of PUF. The spheroids grew larger with cell proliferation.
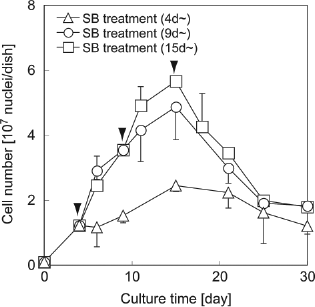
The changes in the cell numbers in the PUF stationary culture. The arrowheads show the start time of the SB treatment under each treatment condition. Cell proliferation was suppressed by SB treatment. The total cell number decreased after 15 days of culture under all conditions. The values show the mean of two experiments. Error bars represent SD.

Evaluation of the liver-specific functions of differentiating ES cells in PUF stationary culture. The 1 mM SB treatment was performed for three different terms, and the activities of liver-specific functions were compared in each condition. (A) The ammonia removal rate per unit volume of PUF plate. (B) The albumin secretion rate per unit volume of PUF plate. The values show the mean of two experiments. Error bars represent SD.
Evaluation of Liver-Specific Functions of the PUF-HAL Module in In Vitro Perfusion Culture
Figure 7 shows the comparison of ammonia removal and albumin secretion rates of PUF-HAL using different cell sources. In the ES cell differentiation culture, we started SB treatment from 9 days of culture. In the primary mouse hepatocyte culture, we saw the highest activity of ammonia removal at 1 day of culture, and then the activity decreased rapidly by the first week of culture. In ES cell differentiation culture, we detected ammonia removal activity at 21 days of culture. Although expression was observed for at least 1 month of culture, the level was much lower than that of primary hepatocyte culture. The changes in albumin secretion activity in primary hepatocyte culture showed the same tendency observed in the ammonia removal function. In ES cell differentiation culture, albumin secretion activity was detected from 15 days of culture and maintained for at least 1 month. Unlike expression of ammonia removal, the levels of albumin secretion activity by differentiating ES cells was comparable to that of primary mouse hepatocytes.

The changes in the activity of liver-specific functions of the PUF-HAL module. The activities of PUF-HAL with differentiating mouse ES cells (ES-HAL) and with primary mouse hepatocytes (Hep-HAL) were compared. (A) The ammonia removal rate per unit volume of module. (B) The albumin secretion rate per unit volume of module. The values show the mean of six (ES-HAL) or five (Hep-HAL) experiments. Error bars represent SD.
Evaluation of the Curative Effects of PUF-HAL in Animal Experiments
Figure 8 shows the time course of blood ammonia in rats with a partial hepatectomy during HAL treatment. In the control group, the blood ammonia increased with circulation time. In the Hep-HAL group, blood ammonia concentration was at a lower level compared with the control group. However, the initial blood ammonia concentration in the ES-HAL group was higher than that in the other two groups. Blood ammonia concentration was maintained at the initial level during treatment. In the control group (n = 4), all rats with a partial hepatectomy died within 5 h after treatment. In the HEP-HAL and ES-HAL groups (n = 3, respectively) two rats died within 6 h after treatment, and one rat recovered.

The changes in the blood ammonia concentration of rats during HAL treatment. In the ES-HAL group (n = 3), the PUF-HAL immobilized differentiating mouse ES cells were applied to rats with partial hepatectomy. In the Hep-HAL group (n = 3), the PUF-HAL immobilized primary mouse hepatocytes were applied to rats. Rats in the control group (n = 4) were treated by PUF-HAL without cells. We observed an increase of blood ammonia with circulation time in the control group. This result was not seen in the Hep-HAL and the ES-HAL groups.
Discussion
Various protocols for hepatic differentiation of ES cells have been reported by many researchers, but studies that apply such protocols to an artificial liver device (6,23) are scarce. There are two ways to apply ES cells to a HAL. In one, ES cell differentiation is performed in a HAL module. The other way purifies hepatic cells induced from differentiation culture and then immobilizes them in a HAL module. In this study, we differentiated ES cells into the PUF-HAL module directory; we have yet to develop an effective method for purification of a hepatic population in a differentiation culture. Development of a HAL module using a homogenous population of hepatocyte-like cells obtained from ES cell differentiation culture will be needed in the near future.
Mouse ES cells formed spheroids in the pores of the PUF and proliferated with culture time. SB treatment then inhibited the proliferative activity. Total cell number decreased after 15 days of culture in all conditions (Fig. 5). SB is known to suppress proliferation and induce apoptosis following the expression of tumor suppressor genes, such as p21 (27,28). In a preliminary study, ES cells in PUF/spheroid culture nearly disappeared when treated with 3 mM SB (data not shown).
We evaluated the changes in gene expression of α-fetoprotein (AFP) and albumin. The AFP gene expression started to increase at 9 days of culture and the albumin gene expression started to increase at 12 days of culture (data not shown). These results indicated that SB treatment at an early stage of culture (~9 days) might not be effective for hepatic differentiation. However, spontaneous differentiation and proliferation with spheroid formation for approximately 9 days before SB treatment may effectively induce a high expression of liver-specific functions.
The expression levels of liver-specific functions of ES-HAL per unit volume of module were compared to those of Hep-HAL. The albumin secretion rate was comparable to that of Hep-HAL. This resulted in a high cell density of 2.09 × 107 cells/cm3 of proliferating ES cells in the ES-HAL module. This figure was two times higher than initial cell density of nonproliferative primary hepatocytes immobilized in the Hep-HAL module (0.89 × 107 cells/cm3). Conversely, the ammonia removal activity of ES-HAL was very low compared with that of Hep-HAL. Not all cells in the ES-HAL differentiated into hepatic lineage, and the cell population was heterogeneous. The apparent decrease in ammonia was due to production of ammonia as metabolic waste by nonhepatic cells. More effective differentiation protocols must be established by investigating other differentiating agents, such as growth factors.
Although the device scale and immobilized cell mass in the animal experiments was smaller than those in a prior study on the use of primary rat hepatocytes (21), ES-HAL seemed to contribute, in part, to rat recovery from liver failure. The effective differentiation method for hepatic lineage in the HAL device and its scale-up will lead to a greater curative effect in animal experiments. The development of a HAL immobilized homogenous population purified by a specific cell marker, such as asialoglycoprotein receptor, is a priority (2). Similarly, induced pluripotent stem cells (iPS cells) (19,25) are needed to bring clinical use of HAL to fruition.
In conclusion, the PUF-HAL module containing differentiating ES cells expressed liver-specific functions. In the animal experiments, this module showed a good potential to support the liver functions. Our outcomes indicate that the PUF-HAL module containing differentiating ES cells might therefore be a useful biocomponent of a hybrid artificial liver support system.
Footnotes
Acknowledgments
This study was supported, in part, by a Grant-in-Aid for Scientific Research (A)(2): 14205119, Grant-in-Aid for Scientific Research (B): 19360375, and Grant-in-Aid for Scientific Research (B): 21360407 from the Japan Society for the Promotion of Science. The authors declare no conflicts of interest associated with this study.
