Abstract
Human embryonic stem (ES) cells and induced pluripotent stem (iPS) cells have an enormous potential; however, their potential clinical application is being arrested due to various limitations such as teratoma formation followed by tumorigenesis, emergent usage, and the quality control of cells, as well as safety issues regarding long-term culture are also delaying their clinical application. In addition, human ES cells have two crucial issues: immunogenicity and ethical issues associated with their clinical application. The efficient generation of human iPS cells requires gene transfer, yet the mechanism underlying pluripotent stem cell induction has not yet been fully elucidated. Otherwise, although human adult regenerative cells including mesenchymal stem cells have a limited capacity for differentiation, they are nevertheless promising candidates for tissue regeneration in a clinical setting. This review highlights the use of regenerative cells for transplantation in hepatic failure.
Introduction
Severe hepatic failure, such as hepatic cirrhosis and hepatocarcinoma, is not reversible. The only effective treatment to date is orthotropic liver transplantation, but alternative approaches are needed because of the limited number of donors and organ rejection. One such approach is regenerative medicine, which holds promise for the development of a cell-based therapy of the liver (113) and may allow the transplantation of hepatocyte-like cells generated from stem cells (4,8,94). The establishment of stem cell therapy requires multipotential, immunocompatible stem cells and a direct differentiation strategy that is not followed by posttransplantation complications or unwanted differentiation such as tumor formation. The cells are capable of supporting a host's liver function and thereby can open the way to further treatment options and liver regeneration.
Pluripotent Stem Cells
Human embryonic stem (ES) cells can be harvested from human blastocysts in human embryos (100). ES cells have been investigated for hepatic differentiation ability, using mostly mouse, but also monkey and human, ES cells (49). Mouse ES cells have been directly differentiated into functional hepatocytes in an adherent monoculture condition using a hepatic induction factor cocktail (HIFC) method (98,108). Growth factors directing hepatic fate specification have been identified on the basis of an in vivo transplantation of ES cells into liver-injured animals (106). Soto-Gutiérrez et al. demonstrated that approximately 70% of differentiated mouse ES cells express albumin following coculture with human liver nonparenchymal cell lines and that the albumin-positive cells can be recovered by albumin promoter-based cell sorting. The sorted cells produce albumin in culture and metabolize ammonia, lidocaine, and diazepam at approximately two thirds the rate of primary mouse hepatocytes (90). Chen et al. developed an efficient method for differentiating the human ES cell line KhES-1 into hepatocyte-like cells in vitro using 50 ng/ml of activin A and 500 ng/ml of a deleted variant of hepatocyte growth factor (dHGF) (21). Similarly, Soto-Gutiérrez et al. demonstrated the efficient differentiation of hES cells to functional hepatocytes using HGF and dpoly-amino-urethane-coated nonwoven polytetrafluoroethylene fabric (89). In addition, Basma et al. reported that human ES cells can be differentiated into cells with many characteristics of primary human hepatocytes. Hepatocyte-like cells can be enriched and recovered based on asialoglycoprotein receptor expression and potentially could be used in drug discovery research and developed as therapeutics (10). However, human ES cells have two crucial issues: immunogenicity and ethical issues in clinical usage.
Generating pluripotent stem cells from the postnatal tissue of an individual patient would greatly facilitate their therapeutic application by avoiding the ethical issues and the immune rejection by the recipient, which are associated with allograft transplants (38,109). Recently, it was shown that a specific set of genes can induce pluripotent stem cells from human fibroblasts (57,61,71,92,112). Human induced pluripotent stem (iPS) cells are similar to human embryonic stem cells in morphology, gene expression, cell surface antigens, long-term self-renewal ability, and teratoma formation and that human leukocyte antigen (HLA) genotypes of human iPS cell lines were identical to those of their respective parental somatic cells (61). However, at present, gene transfer is essential for the efficient generation of human iPS cells, and the mechanism underlying pluripotent stem cell induction has not yet been fully elucidated. Although human iPS cells have an enormous potential, many limitations such as teratoma formation followed by tumorigenesis, emergent usage, and quality control of cells and safety issues for long culture continue to delay their clinical application.
Regenerative Cells
Mesenchymal stem cells (MSCs) can be easily obtained from a patient's own tissues, isolated ex vivo, expanded (114), and then transplanted back into the patient as an autologous transplant. Such a possibility sidesteps the limits governing ethical issues and immunocompatibility problems. Importantly, human MSCs represent an advantageous cell type for allogenic transplantation as well, since they are immunoprivileged with low HLA-I and no HLA-II expression, thereby reducing the risk of allogenic transplant rejection and preventing graft-versus-host disease (GVHD) (1,9,27,50,51,102). MSCs act as “trophic mediators” (19,36,69,70,72,79,99), which by secretion of bioactive factors act either as immunosuppressors or promoters of regeneration (19). MSCs in animal models can engraft into the liver and ameliorate liver injury caused by carbon tetrachloride (CCl4) (39,68,82,85), allyl alcohol (74), or retrorsine (117) injection or combined radiation and CCl4 injection (116). The mechanisms of the effect of MSCs on hepatic regeneration, however, are still unclear and this point thus remains controversial (39,41,68,74,82,87,116,117). Among the proposed ideas is the hypothesis of the paracrine effects of MSCs, by which an amelioration of liver damage occurs through the secretion of active factors (69,70).
Therefore, human adult regenerative cells including multipotent stem cells are promising candidates for liver regeneration (39,68,72,82,85,99), and their usage might sidestep obstacles, such as ethical concerns and risks of rejection. The most promising regenerative cells are MSCs, found in human bone marrow (BM) (74), adipose tissue (116,117), scalp tissue (87), placenta (41), and umbilical cord blood (UCB) (13) as well as in various fetal tissues (17). These stem cells can differentiate in vitro into multiple types of lineages such as: chondrogenic, osteogenic, adipogenic (74), myogenic (30), neurogenic (81), and hepatogenic (39,68,82,85), depending on the microenvironment in which they reside. MSCs from BM and UCB have been differentiated into a hepatic lineage; however, the question of whether these are the best sources of stem cells for hepatic replacement and/or regeneration remains.
Bone Marrow-Derived Regenerative Cells
A well-characterized BM stromal cell population (74) emerged as a focus for regenerative therapy. There is a little confusion in terminology, with some authors suggesting (23) that subpopulations named colony forming units of fibroblasts (CFU-F), multipotent adult progenitor cells (MAPCs) (85), MSCs (74), or stromal cells are quite similar or closely related. Kucia et al. showed that human BM is composed of a heterogeneous nonhematopoietic (CXCR4+, CD34+, AC133+, lin-, CD45-) tissue-committed stem cell subpopulation (48). They postulate that BM contains stem cells with different levels of differentiation beginning from primitive pluripotent stem cells to tissue-committed stem cells (47,48,58). However, this subpopulation is different from MSCs (CXCR4-, CD34-). The predominant source for MSCs is adult BM, but they can also be obtained from various tissues of the human body, including compact bone, peripheral blood, adipose tissue, umbilical cord blood, amniotic fluid, and other fetal tissues. MSCs are capable of self-renewal and multilineage differentiation [adipogenic, osteogenic, chondrogenic (74), myogenic (30), neurogenic (105)] and can be expanded in vitro, thus making it possible to engineer transplantable tissue in association with appropriate scaffolds. Schwartz et al. showed that rat, mouse, and human BM-derived MAPCs, cultured with fibroblast growth factor (FGF)-4 and HGF on Matrigel, can differentiate into cells expressing several liver-specific markers (85). Sato et al. showed that human bone marrow mesenchymal stem cells (BM-MSCs) xenografted into rat liver differentiate into human hepatocytes, which express liver-specific markers, without fusion (82). Zhao et al. have demonstrated the protective effect of MSCs isolated from rat BM on fibrosis caused by CCl4 and dimethylnitrosamine (DMN) (115). We adapted the HIFC system to contribute to hepatic differentiation of human BM-derived MSCs (96). Hepatocytes derived from human MSCs reveal morphological, biological, functional, and therapeutic potential.
Therefore, MSCs represent a promising tool for cell transplantation. They can be obtained from the patient's own bone marrow, expanded in vitro, and then transplanted back into the liver as a native source for hepatic regeneration or, after differentiating ex vivo, maintained on a proper scaffold and transplanted back into a diseased liver. In the future, stem cell-derived hepatocytes might be used together with advanced tissue engineering technology for the development of the entire liver system.
Tissue Engineering and Biomaterials
The microenvironment for long-term culture with a possibility to maintain stem cell-derived hepatocyte functions is important in the context of stem cell-based liver therapy and tissue engineering (37). The ideal scaffold is nonimmunogenic, nontoxic, biocompatible, bio-degradable, and easy to manufacture. Scaffolds should permit the easy diffusion of nutrients and cellular waste products and provide good mechanical support for cells during the repair process. Scaffolds currently used are natural or synthetic polymers. Liver tissue engineering started with maintaining a long-term culture of hepatocytes. Highly porous biocompatible polymers have been utilized, mainly poly(lactide-co-glycolide) (54), polyurethane, collagen, chitosan, alginate, and hydrogel. In the three-dimensional (3D) culture system, using poly-N-para-vinylbenzyl-lactonamide (PVLA)-coated reticulated polyurethane, Sato et al. maintained specific hepatocyte functions for up to 40 days in an in vitro culture. When transplanted into the peritoneal cavity of rats, the hepatocytes were able to survive and retain liver-specific functions for more than 1 month (83). Ohashi et al. successfully engrafted and maintained hepatocytes at extrahepatic sites, with extracellular matrix components for over 100 days (67). In a first-time demonstration, we show that ES cell-derived hepatocytes were capable of being maintained for a long time in a 3D culture with a porous hyaluronate (3D–HA) sponge (97). There is clear evidence that 3D–HA sponge formed hepatocyte spheroids within the pores and sustained cell viability and liver functions, such as albumin secretion, ammonia detoxification, and urea nitrogen synthesis for a prolonged time (more than 20 days), in comparison to a conventional monolayer culture and in collagen sponge cultures (17 days). Hepatocytes within an HA sponge secreted a constant level of albumin up to 32 days. Likewise, Navarro-Alvarez et al. compared the effect of a self-assembling peptide nanofiber (SAPNF) as an extracellular matrix (ECM) with collagen type I on hepatocyte metabolic and secretion activities following hepatocyte isolation. They clearly demonstrate the usefulness of SAPNF for maintaining the differentiated functions of porcine hepatocytes in culture (65). The demonstrated results of 3D cultures along with advances in stem cell technology might serve in the future as a basis for the development of an artificial liver. Soto-Gutiérrez et al. reported that treatment of 90% hepatectomized mice with a subcutaneously implanted bioartificial liver (BAL) seeded with ES cell-derived hepatocytes or primary hepatocytes improved the liver function and prolonged survival (88).
Adipose Tissue-Derived Regenerative Cells
The number of laboratories studying adipose tissue has increased dramatically since Zuk et al. reported on the identification of an adherent, multipotent cell from adipose tissue (117). Adipose tissue has been shown to be a source of MSCs for regenerative medicine (84, 116,117). A sufficient number of stem cells for cell-based therapy may be obtained from this tissue without invasiveness or damage to a patient's health. Adipose tissue mesenchymal stem cells (AT-MSCs), so-called processed lipoaspirate (PLA) cells (24,116,117), adipose-derived stromal cells (ADSCs) (91), adipose-derived adherent stromal cells/adipose-derived adult stem cells (ADASs) (16,34,43), and adipose tissue-derived stromal cells (ATSCs) (53) are considered to be the multipotent fraction of adherent cells, which, after the isolation of the adipose stromal vascular fraction (SVF), attach themselves to plastic culture dishes and then remain there as a heterogeneous population of fibroblast-like cells. AT-MSCs are very similar to BM-MSCs (24,104). Besides the fact that they are more heterogeneous (104), they have also been reported to demonstrate a surface antigen marker profile (33,43,44,104) and differentiation potential similar to BM-MSCs (18,26,40,80,86,91). AT-MSCs are characterized as CD45- CD34+/- CD105+ CD31-(14); however, there is confusion regarding CD34 marker. One of the markers defining MSC provenance is CD105 (endoglin) (35,56). CD105 is a component of the receptor complex of transforming growth factor-β (TGF-β), a pleiotropic cytokine involved in cellular proliferation, differentiation, and migration. CD105+ cells from bone marrow display more colony-forming unit-fibroblasts (CFU-Fs) and revealed a capacity to form bone in vivo (3) and differentiate into a chondrogenic lineage (59). The adipogenic and myogenic differentiation ratio of CD105+ BM-MSCs is not influenced by the age of the donor; however, the ratio usually decreased in older patients (6). The ratio of the number of CD105+ stem cells in adipose tissue to the age of the donor is still not clear, and little is known about the relationship between disease (e.g., cancer) and stem cell potential.
Therapeutic Potential of Differentiated Cells
The possibility for future application of AT-MSCs in the therapy of liver diseases is very promising. A previous study evaluated the hepatogenic potential of AT-MSCs. AT-MSCs from nonobese cancer patients undergoing a gastrectomy were used because therapy concerns patients, not healthy donors. Magnetically activated cell-sorting (MACS) of the CD105 fraction yielded a multipotent and homogeneous subpopulation of cells. CD105+ AT-MSCs were highly inducible into the hepatic lineage, and derived hepatocyte-like cells expressed the liver markers, proteins, enzymes, and functions of human primary hepatocytes. The differentiation potential of AT-MSCs was demonstrated by in vivo transplantation into immunodeficient mice, which resulted in the incorporation of AT-MSC-derived hepatocytes into the CCl4-injured liver.
Therefore, human AT-MSCs, which might be obtained from a large number of cells, represent a very attractive tool for future stem cell therapy of liver diseases. Transplantation of hepatocytes might become easier, more efficient, and safer than whole organ transplantation to cure patients suffering from end-stage liver dysfunction. The maintenance of hepatocyte function in vitro is difficult if not nearly impossible, thus the generation of stem cell-derived hepatocytes holds considerable promise for future clinical applications. The hepatogenic potential of MSCs from different sources has been previously described (39,68,82,85). There have been two reports so far indicating the endoderm differentiation capacity of AT-MSCs: insulin-, somatostatin-, and glucagon-expressing cells (101) as well as albumin-and α-fetoprotein-expressing hepatocyte-like cells with the ability to synthesize urea and uptake low-density lipoproteins (LDL) (86); however, the second reported system includes the use of dimethyl sulfoxide (DMSO), which has been shown to maintain hepatic morphology. In contrast, a preferable system simulates in vivo endoderm development of the liver, as previously reported (35,56). The isolation ratio and minimal decline in donor health associated with isolation of these cells suggests that AT-MSCs represent an attractive tool for studies on stem cell therapy for the liver. MSCs from a predominant source, such as bone marrow, reveal a lower proliferation capacity and frequency than AT-MSCs (76), and their frequency is influenced by age, gender, the presence of osteoporosis, and prior exposure to high-dose chemotherapy or radiation. Importantly, 200 ml of lipoaspirate yields approximately 40-fold more stem cells than from 40 ml of marrow. Even though adipose tissue provides a more heterogeneous population of stem cells, the potential number of AT-MSCs is still sufficiently large to allow the selection/sorting of the multipotential fraction of AT-MSCs. The CD105+ fraction from BM-MSCs has more homogeneity and a greater ability to form CFU-Fs (3,59,77). Some scientists have used other isolation procedures to select multipotent AT-MSCs (76); however, the study described above used a CD105+ fraction using magnetic beads. The CD105+ fraction of BM-MSCs is age resistant (77), and therefore sorting might be very useful because many patients with liver failure or hepatocarcinoma tend to be elderly. Large amounts of fat are discarded during liposuction, and, in the future, this valuable resource could be used for the isolation of AT-MSCs, which might then be stored in each patient's cell bank or cell products.
The hepatic differentiation of CD105+ AT-MSCs results in the generation of functional and transplantable hepatocytes (60–85%). Unfractionated AT-MSCs reveal hepatogenic potential as well; however, the ratio of differentiation is relatively lower (20–60%). The hepatogenic potentiality of AT-MSCs is confirmed by the detection of hepatic-specific markers and biochemical functions. The major protein produced by the hepatocytes, albumin, is synthesized and secreted into a medium at days 30 and 50 (subsequently: 5 and 14 mg/dl/4 days/106 cells). The expression and activity of the microsome cytochrome P450 (CYP) enzymes involved in drug, xenobiotic metabolism, and sterol and bile acid synthesis indicates hepatocyte specificity. Hepatocyte-like cells generated from CD105+ AT-MSCs express CYP7A1, CYP1A1, CYP2C9, CYP3A4, and nicotinamide adenine dinucleotide phosphate (NADPH)-CYP reductase. Therefore, these findings suggest that there might be some value in in vitro preclinical drug investigations.
Transplantation experiments of AT-MSC-generated hepatocytes into CCl4-injured nude mice revealed direct incorporation into the liver, which was confirmed by human albumin immunostaining. This induction system does not require coculturing, and therefore the undesired cell–cell interactions can thus be reduced. Nevertheless, further studies examining the in vivo mechanism of homing, engraftment, and liver regeneration need to be performed in order to eliminate posttransplantation complications. Some in vivo functions, such as the serum ammonia level and alanine aminotransferase (ALT), decreased after the transplantation of undifferentiated AT-MSCs as well. These findings indicate the possibility of the in vivo differentiation of AT-MSCs caused by a regeneration microenvironment. The subsequent interaction of transplanted undifferentiated MSCs with liver parenchyma cells also needs to be evaluated in the context of the promotion of fibrosis (78), cancer, or liver dysfunction. Another topic of interest deserving evaluation is the correlation between such factors as donor age, cancer history, and the differentiation potentiality of stem cells. It should be emphasized that three independent donor-derived CD105+ AT-MSCs show similar hepatic differentiation ability. In the context of clinical application, the use of AT-MSCs, which can be obtained from a patient's own tissue in contrast to highly potent ES cells, would eliminate many obstacles such as ethical issues, rejection, and the risk of teratoma and tumor formation.
The Transcriptome and Signal Pathways in Hepatic Differentiation
The cells generated from AT-MSCs are transplantable hepatocyte-like cells with functional and morphological similarities to hepatocytes. AT-MSC-derived hepatocytes (AT-MSC-Hepa) demonstrate several liver-specific markers and functions, such as albumin production, LDL uptake, and ammonia detoxification. This system induces transplantable cells with morphological and functional characteristics of hepatocytes (7). Other groups have also provided evidence of hepatic differentiation from human AT-MSCs (86,93). However, the molecular mechanisms underlying the differentiation of AT-MSCs are still largely unknown. None of the reports provide a comprehensive analysis of the process underlying the differentiation of AT-MSCs into hepatocytes. Therefore, the molecular events involved in controlling the plasticity of AT-MSCs that give rise to hepatocytes were investigated (7). A microarray analysis showed that the gene expression pattern of AT-MSC-Hepa is similar to that of adult human hepatocytes and liver. Moreover, the enriched categories of genes and the signaling pathways in the AT-MSC-Hepa were relevant to liver-specific functions. An analysis of the gene ontology (GO) groups indicated that many of the 1639 up- or downregulated genes belonged to GO categories relevant to hepatic function, including the steroid and lipid metabolisms. In addition, a gene signaling pathway analysis has identified gene signals that are remarkably activated in AT-MSC-Hepa, and these signals are also upregulated in the whole liver. Therefore, a microarray analysis provides a potentially valuable resource for determining the key molecules involved in hepatocyte differentiation and function. These integrative perspectives on the gene expression profile might be useful for revealing the control of plasticity of AT-MSCs that give rise to hepatocytes. The expression of liver-selective transcription factors, such as hepatocyte nuclear factors (HNFs), CCAAT/enhancer-binding proteins, and GATA-binding proteins, is essential for the induction of liver development and its progression. These transcription factors exhibit temporal- and site-specific expression patterns during organogenesis, with a distinct narrow time interval of transcription initiation (107), and thus regulate the transactivation of several endoderm and hepatocyte-specific factors, including transthyretin, albumin, and tyrosine aminotransferase (22,28). HNF3b/for-khead box A2 (FOXA2) plays an important role in endoderm specification and subsequent hepatocyte differentiation both in vivo and in vitro (15,52). In this study, the induction of HNF3b/FOXA2 expression was clearly seen in AT-MSC-Hepa by a microarray analysis. Furthermore, the expression of other hepatic transcription factors, including HNF3a/FOXA1, GATA4, HNF6/ onecut homeobox 1 (ONECUT1), and HNF1 were observed to be 10-fold upregulated in AT-MSC-Hepa in comparison to undifferentiated AT-MSCs. These results suggest that the transcription factor networks are precisely regulated in the hepatic differentiation system, and that the AT-MSCs differentiate into mature hepatocytes.
Rapid Hepatic Fate Specification
Clinical applications in the future would require a special approach, such as shortening the ex vivo manipulations as much as possible, including cultivation and direct hepatic fate. Therefore, an attempt was made to modify the hepatocyte differentiation strategy, based on the current knowledge on in vivo mouse fetal liver development (5). A period as short as 13 days is required and this strategy is enriched by pretreatment with activin A and FGF-4. In addition, the content of the growth factor cocktail was reorganized and enriched with the addition of DMSO, nicotinamide, and insulin-transferrin-selenium. This protocol yielded functional hepatocyte-like cells in a much shorter period of time. Consequently, hepatocyte-like cells differentiated from adipose-derived stem cells (ASCs) were transplanted into immunodeficient mice with nonsevere acute liver injury. These results showed a significant decrease of ammonia, aspartate aminotransferase (AST), ALT, and uric acid (UA) in the blood plasma of mice after the transplantation of hepatocyte-like cells from ASCs. These results therefore show very important progress towards the further establishment of an alternative and successful therapy for liver disease. The functional benefits of transplantation of the hepatocyte-like cells from ASCs may be because of the functional support of the transplanted cells. The mechanisms by which the transplanted cells improve the functioning of the liver remain unclear. The support and activation of endogenous progenitors are possible factors. Further studies examining the in vivo mechanism of homing, engraftment, liver regeneration, and the trophic effects need to be conducted.
Therapeutic Potential of Undifferentiated Cells
A recent study reported the therapeutic potential of undifferentiated human AT-MSCs with respect to their future clinical usage for the treatment of hepatic failure (6). The usage of such AT-MSCs, which are minimally manipulated ex vivo and easily obtained within a short period of time, is crucial for future clinical emergency usage, quality control of cells, and safety issues. AT-MSCs were transplanted into immunodeficient mice with acute liver failure caused by CCl4 injection. The results showed a significant decrease of ammonia, AST, UA, and ALT concentration 24 h after transplantation of AT-MSCs, and also indicate that AT-MSCs are involved in liver regeneration in vivo. HLA-1 immunostaining revealed human AT-MSCs within the liver of immunodeficient mice 24 h after transplantation. Few positive cells were observed in different areas of the liver such as parenchyma, vessel, and bile duct. Monitoring AT-MSC migration, mobilization, and engraftment for a longer time is essential and very informative. Evaluating the effect of AT-MSCs on endogenous progenitor activation has proven to be interesting (60). There may be a different response of oval cells in vivo between mice and rats as well as in the injury model (42). The possible actions of AT-MSCs in vivo are that they act directly by causing intracellular signaling and/or indirectly by secreting or making other cells in the vicinity secrete functionally active agents. However, involvement may be in part due to the pleiotrophic contribution through the direct and/or indirect activity of AT-MSCs. Actually, according to several recent noteworthy reports, generalized administration of the culture supernatant from MSCs alone is successful in effectively prolonging the lives of model animals with life-threatening liver damage (70,103). The in vitro production of cytokines/growth factors by AT-MSCs was evaluated and compared with undifferentiated BM-MSCs and normal human dermal fibroblasts (NHDFs). This revealed a higher secretion of interleukin (IL)-1 receptor antagonist (IL-1RA), IL-6, IL-8, granulocyte colony-stimulating factor (G-CSF), granulocyte macrophage colony-stimulating factor (GM-CSF), monocyte chemotactic protein-1 (MCP-1), nerve growth factor (NGF), and HGF by AT-MSCs, as opposed to BM-MSCs and NHDFs. However, vascular endothelial growth factor (VEGF) was secreted similarly by both types of MSCs higher than by NHDFs. This in vitro study shows a higher production of bioactive factors in AT-MSCs than in BM-MSCs. Moreover, this in vivo study shows the ability of AT-MSCs to incorporate into the liver and improve its function. The regenerative ability of AT-MSCs may be possibly due to trophic activity of AT-MSCs.
Therefore, undifferentiated AT-MSCs have the ability to restore and improve liver functions and promote liver recovery in mice with liver injury. Parameters such as AST, ALT, ALP, UA, and ammonia decrease in injured mice after AT-MSC transplantation, suggesting that AT-MSCs have the ability to regenerate the liver. However, no morphological improvement of the liver was observed after AT-MSC transplantation. A long-term course experiment with histological analyses as well as safety issues should be carefully evaluated, because the effect of transplantation of AT-MSCs is not transient. Little is known regarding posttransplantation modifications and the risk of carcinogenesis. AT-MSCs may undergo spontaneous transformation in vitro; however, that was observed in cells at the senescent phase after a prolonged period of culturing.
Hepatic disease is accompanied by inflammation and fibrosis in a heterogeneous condition, and so far no effective treatment strategy has yet been established. Kupffer cells and liver macrophages are major sources of IL-1β and tumor necrosis factor-α (TNF-α) production. These cytokines enhance the expression of adhesion molecules in endothelial cells, which mediate neutrophil adhesion to endothelial cells causing cell dysfunction and destruction, leading to hepatocyte necrosis (31). MSCs secrete a broad spectrum of active cytokines, chemokines, and growth factors. AT-MSCs produce significantly more bioactive factors than BM-MSCs, and, therefore, they may have either an equal or even stronger regenerative effect than BM-MSCs. This issue is highly interesting and needs precise evaluation by using MSCs isolated from adipose tissue and bone marrow from the same donor. AT-MSCs secrete many active factors in vitro, among them immunosuppressive IL-1RA, hepatocyte growth promoting IL-6, IL-8, HGF, NGF, MCP-1, and hematopoiesis promoting G-CSF, GM-CSF, and that they are secreted in a larger volume than both BM-MSCs and NHDFs. IL-1RA is a strong anti-inflammatory cytokine that binds to IL-1 receptors and inhibits the binding of IL-1α and IL-1β. Therefore, the immune activity of IL-1α and 1β is neutralized. Inflammatory cytokines such as IL-1 and TNF-α are not produced by AT-MSCs or by BM-MSCs or NHDFs. On the other hand, IL-6, identical to hepatocyte-stimulatory factor (HSF), elicits an acute phase response in liver cells; IL-8 and MCP-1 are inflammatory-related factors, although it is difficult to predict their actual in vivo activity. In fact, IL-6 together with TNF-α is responsible for a hepatocyte entering to the state of replication competence (95). IL-6, IL-8, and MCP-1 are produced by human intrahepatic biliary epithelial cells through the TLR (toll-like receptor), 4-NF (nuclear factor)-KB, and MAPK (mitogen activated protein kinase) signaling pathways, and that they are possibly mediating an innate immune-system function and modulating hepatic regeneration in vivo (46,110). HGF and TGF-α stimulate proliferation of hepatocytes in the proliferation phase. HGF, which is proapoptotic for liver stellate cells as well as for myofibroblasts (45), acts as an anticirrhotic agent. Stellate cells are a major source of collagens and other extracellular matrix (ECM) proteins in liver fibrosis. In addition, they regulate matrix metalloproteinases (MMP). Other highly secreted factors by AT-MSCs were G-CSF, GM-CSF, and IL-6, considered as a supporting process of hematopoiesis. There is evidence that many tissues contain their own multipotential stem cells (11,12); thus endogenous stem cells (oval cells, liver epithelial progenitors, etc.) may be the target of MSC-derived direct or indirect actions. In addition, bioactive factors may act on oval cells and therefore provoke hepatocyte generation in vivo. G-CSF promotes rat liver repair and induces oval cell migration and proliferation (25,73); however, there are controversial opinions regarding the G-CSF effect (20). Recent evidence suggests that neurotrophins, such as NGF, may have a role in hepatic regeneration (2,29,32,66,75), and their mRNA and protein levels become elevated in association with hepatocyte proliferation induced by, for example, administration of CCl4 (66). NGF, like HGF, induces the apoptosis of stellate cells (2,29,66). NGF is secreted by cholangiocytes and also induces their proliferation (32). In addition, MSCs can inhibit the proliferative and fibrogenic function of activated stellate cells in a paracrine manner. This inhibition is caused by MSC-derived IL-10 and TNF-α, which act synergistically. The secretion of IL-10 by MSCs is a response to IL-6 secretion by activated stellate cells. These events are not detected while stellate cells are cultured with fibroblasts. The production of trophic agents by AT-MSCs exhibit donor to donor variations. In fact, the secretion of IL-10 was detected in AT-MSCs of one donor only. Recently, Akt-modified MSCs are proposed to be candidate cells for paracrine-mediated actions (64). Akt-modified MSCs can upregulate the expression of several candidate mediators such as VEGF, FGF2, HGF, and insulin-like growth factor-1 (IGF-1), secreted frizzled related protein 2 (SFRP2), which is the key Akt-modified-MSC released paracrine factor. Of note, these data show only the in vitro production of bioactive factors. In vivo MSCs may act through multiple yet unknown mechanisms to coordinate a dynamic integrated response to fibrosis (69). It is also likely that similar immunomodulatory mechanisms may influence the phenotype of resident hepatocytes, stellate cells, Kupffer cells, sinusoidal endothelial cells, and immune cells that infiltrate the liver during inflammation.
Therapeutic Potential of Uncultured Cells
The most promising perspectives for cell transplantation would be to use uncultured (freshly isolated) regenerative cells in clinical application. Adipose tissue is an abundant source of regenerative cells that is easily harvested. Such adipose tissue-derived regenerative cells (ADRCs) include adipose-derived mesenchymal stem or progenitor cells and mesenchymal cells such as vascular cells and hematopoietic cells (Fig. 1). Similar to BM-MSCs, AT-MSCs can differentiate into multiple functional phenotypes with potential regenerative applications. Cytori Therapeutics, Inc., has developed a closed system called Celution™ System to isolate and concentrate ADRCs including mesenchymal stem and progenitor cells automatically from adipose tissue (55). ADRCs can be processed by Celution™ System within the time frame of a single surgical procedure and are equivalent to those isolated manually. A therapeutic dose of ADRCs can be prepared aseptically with the Celution™ System in nonspecified areas such as medical clinics and hospitals; thus. this system could provide a “real-time treatment setting at the bed side” that is cost-effective and safe (Fig. 2). Regenerative cells from the Celution™ System are composed of heterogeneous cell populations including putative AT-MSCs (CD31-, CD34+/-, CD45-, CD90+, CD105-, CD146-), endothelial (progenitor) cells (CD31+, CD34+, CD45-, CD90+, CD105-, CD146+), vascular smooth muscle cells or pericytes (CD31-, CD34+/-, CD45-, CD90-, CD105-, CD146+), and hematopoietic cells (CD45+) in an uncultured condition. Adipose tissue could be one of the most suitable cell sources for cell therapy because of its easy accessibility, minimal morbidity, and abundance of stem cells. The large numbers of stem cells in adipose tissue mean that clinically relevant stem cell numbers could be extracted from the tissue, potentially eliminating the need for in vitro expansion. While up to 2 L of adipose tissue can be safely extracted under local anesthesia with minimal morbidity in an outpatient setting, this is partially BMI dependent. Therefore, for the sake of this comparison, tissue volumes of approximately equivalent donor site morbidities typically yield 40 ml of marrow and 800 ml of adipose. Fibroblast clonogenic assays demonstrated the colony forming potential, for bone marrow, total mononuclear cell counts are ~3 × 107 cells/ml bone marrow, and at a stem cell frequency of ~1:100,000; thus, a bone marrow aspirate of 40 ml yields 1.2 × 104 BM-MSCs. In comparison, ~2 × 106 nonbuoyant mononuclear cells can be harvested per milliliter of adipose tissue and at a frequency of ~1:100, and 1.6 × 107 ADRCs can be isolated (62,63,111). Therefore, an approximately 1,300-fold increase in adherent stem cells from adipose tissue is observed in comparison to bone marrow with equal donor site morbidity.

Adipose tissue is the most abundant source of regenerative cells that is easily harvested by liposuction. The resulting lipoaspirate is rich in fibroblast-like cells including adipose progenitor, endothelial progenitor, smooth muscle cells, mesenchymal stem cells, and fibroblast. These cells proliferate and differentiate functional mature cells, and they possess trophic effects to inhibit fibrosis and apoptosis, promote angiogenesis, while also stimulating host progenitors to proliferate and differentiate into somatic resident cells.
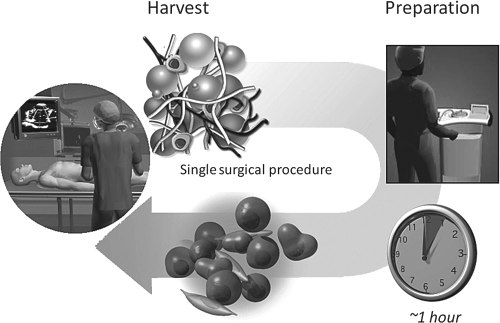
Flow of autologous cell transplantation is shown. Adipose tissue derived regenerative cells (ADRCs) such as mesenchymal stem cells and progenitor cells are harvested by liposuction and prepared from lipoaspirate automatically within 1 h or manually. ADRCs are fibroblast-like cells that can be processed and transplanted within the time frame of a single surgical procedure.
Concluding Remarks
The transplantation of human undifferentiated AT-MSCs has functional benefits, at least in part because of their ability to produce a large number and volume of bioactive factors such as growth factors and cytokines. This trophic activity may result in future therapeutic usage of uncultured regenerative cells for liver failure. The trophic activity would be one of the most promising actions in vivo of uncultured and/or undifferentiated regenerative cells. Nevertheless, the efficacy needs to be precisely confirmed and analyzed with a long-term course of experiments, and other mechanisms have not been excluded. Further characterization as to whether regenerative cells can produce bioactive factors that inhibit fibrosis and apoptosis, promote angiogenesis, and stimulate host progenitors to proliferate and differentiate into functional cells will be required in order to establish an effective therapy for hepatic failure.
Footnotes
Acknowledgments
This work was supported in part by a Grant-in-Aid for the Third-Term Comprehensive 10-Year Strategy for Cancer Control; Health Science Research Grants for Research on the Human Genome and Regenerative Medicine from the Ministry of Health, Labor, and Welfare of Japan; and a Grant from the Program for Promotion of Fundamental Studies in Health Sciences of the National Institute of Biomedical Innovation (NiBio). The authors declare no competing financial interests.
