Abstract
Understanding how bone marrow-derived cells (BMDCs) enter the central nervous system (CNS) is critical for the development of therapies for brain-related disorders using hematopoietic stem cells. We investigated the brain damages and blood–brain barrier (BBB) modification following either whole-body irradiation or a myeloablative chemotherapy regimen in mice, and the capacity for these treatments to induce the entry of BMDCs into the CNS. Neither treatment had a lasting effect on brain integrity and both were equally efficient at achieving myeloablation. Injection of bone marrow cells from green fluorescent protein (GFP) transgenic mice was able to completely repopulate the hematopoietic niche in the circulation and in hematopoietic organs (thymus and spleen). However, GFP+ cells only entered the brain following whole-body irradiation. We conclude that myeloablation, damages to the brain integrity, or the BBB and peripheral chimerism are not responsible for the entry of BMDCs into the CNS following irradiation.
Keywords
Introduction
Brain-specific macrophages called microglia are responsible for the immune defense of the brain through innate immunity processes (19). They are the only brain cell type deriving from bone marrow resident hematopoietic stem cells (18,22). The recruitment of bone marrow-derived cells (BMDCs) into the brain is highly active throughout the embryonic life during which microglia populate the central nervous system (CNS) (5). It appears to be marginal during the adult life, as self-renewal of microglia could be sufficient to maintain the microglial population (1), and more active in pathological conditions such as Alzheimer's disease (8). It was demonstrated that following whole-body irradiation, bone marrow transplantation was highly efficient to restrict amyloid plaque formation and resolve the cognitive declines of mouse models of Alzheimer's disease (23). More recently, a successful gene therapy of x-linked adrenoleukodystrophy (ALD) using lentivirus-transduced hematopoietic stem cells in humans was reported (2). A therapeutic potential of bone marrow-derived microglia was also suggested for the treatment of high-grade glioma, severe combined immunodeficiency, multiple sclerosis, and other CNS pathologies (6). Despite the high therapeutic potential of BMDCs for cerebral pathologies (13) and an inestimable tool to study microglial biology, the mechanisms by which they enter the brain are poorly understood.
Chimerism is the process by which injected bone marrow cells from a donor take over the hematopoietic system of a recipient. The use of the green fluorescent protein heterozygous (GFP+/-) transgenic mouse model has eased the study of chimerism by facilitating the tracking of injected GFP+ cells in a GFP- background. Numerous models have been designed to induce chimerism in mouse models such as parabiosis, immunosuppression of the recipient mice, and irradiation of the whole body, only the head, or the body with a protected head (24). It appears that only lethal doses of whole-body irradiation are able to create an efficient chimerism and to induce migrations of BMDCs to the brain. Concerns have arisen toward the use of irradiation in a clinical setting as the effects of this treatment on brain integrity and on the permeability of the blood–brain barrier (BBB) are poorly understood. A model of whole-body irradiation with the head protected from irradiation showed that BMDCs could not infiltrate the CNS and thus suggested that irradiation-induced damages to the brain were responsible for the entry of BMDCs into the CNS. However, such a model leaves intact many hematopoietic precursors in the vicinity of the brain that could compete with the injected cells for the niche. Furthermore, the lack of BMDC invasion following whole-body irradiation with a protected body demonstrated that peripheral myeloablation is also necessary for the entry of BMDCs into the brain.
In the present study, we sought to understand the effects of irradiation on the BBB integrity and to adapt a myeloablative chemotherapy regimen commonly used in hematopoietic stem cells transplants to determine if myeloablation alone is sufficient to allow the entry of BMDCs into the brain. We report that whole-body irradiation has no lasting effects on the integrity of the brain or BBB and that although the chemotherapy regimen is efficient to create a thorough myeloablation and peripheral chimerism, GFP+ BMDCs could not enter into the CNS. These results demonstrate that although irradiation does not alter significantly the integrity of the CNS, irradiation-induced myeloablation and peripheral chimerism alone are not sufficient to induce BMDC entry into the CNS.
Materials and Methods
Animal Treatments
All animals were used under the supervision of approved institutional protocols. Unless stated otherwise, 3-month-old C57/B16 mice were used. GFP+/- transgenic mice were also used in bone marrow transplants and as controls for fluorescence activated cell sorting (FACS) experiments. In both cases, initial strains were obtained from Charles River Laboratories.
Whole-Body Irradiation and Myeloablative Chemotherapy
A week before irradiation or chemotherapy, the mice were given water containing a commercially available mix of antibiotics (SEPTRA, GlaxoSmith Kline, Mississauga, ON, Canada). Mice were either exposed to 10 Gy whole-body irradiation using a cobalt-60 source (Theratron-780 model, MDS Nordion, Ottawa, ON, Canada) or received myeloablative chemotherapy. The chemotherapy regimen consisted of twice-daily (morning and evening) injections of 10 mg/kg of busulfan for 4 days (a total of 80 mg/kg) followed by injections of 100 mg/kg of cyclophosphamide for 2 days (a total of 200 mg/kg). The injections were performed in a total volume of 150 μl injected intraperitoneally, alternating sides between each injections. To counter chemotherapy-induced dehydration, mice received daily 1-ml injection of saline subcutaneously. Following either treatment, mice were transferred to sterile cages and given previously irradiated food. Antibiotic treatment continued for a week following treatment. Ten mice were included in each group (control, irradiated, and chemotherapy) and the experiments were performed three separate times.
Bone Marrow Transplants
Bone marrow cells were obtained by flushing the femurs of mice using PBS containing 5% fetal bovine serum (FBS, Sigma-Aldrich, St. Louis, MO). The cells were filtered on 35-μm nylon mesh (BD Bioscience, San Diego, CA), washed three times in FBS-free PBS (centrifugating at 300 × g for 5 min between washes) and counted with a hematocytometer. Cells (2 × 107) were then injected in the tail vein of recipient mice 6, 24, 48, 72, or 96 h following either irradiation or the end of the chemotherapy regimen. They were sacrificed 3 months later.
Fluorescence Assisted Cell Sorting (FACS)
Mice were anesthetized using a preparation of ketamine hydrochloride and xylazine. Endocardiac blood samples were taken with heparin-treated syringes and kept in ethylenediaminetetraacetic acid (EDTA)-coated vials (Sarstedt, Newton, NC) under constant agitation until used, no more than 3 h following sampling. Blood (100 μl) was transferred to 4.9 ml of magnesium- and calcium-free phosphate-buffered saline solution (mcPBS) containing 5% FBS. Cells were pelleted at 300 × g for 5 min and washed two more times in the same solution and finally suspended in 50 μl of 1% FBS-mcPBS. Nonspecific binding sites were blocked by adding 4 μl of CD16/CD32 at 4°C for 30 min. Antibodies were then added to the solution and incubated for 30 min at 4°C. The complete list of antibodies used in these experiments and their dilutions is summarized in Table 1. Following three more washes in mcPBS, cells were suspended in 250 μl mcPBS and brought for analysis on a FACS Aria analysis system (BD Biosciences). For every experiment, compensation controls were performed for each antibody on blood collected from untreated wild-type mice or GFP+ mice for the anti-GFP antibody. The results were analyzed using the FlowJo software (Treestand, Ashland, OR).
Nature and Dilutions of the Antibodies Used in the Experiments

The dilutions for antibodies used in fluorescence-activated cell sorting (FACS) are: 1 μl/200 μl of total blood (*), 1.5 μl/200 μl of total blood (**), and 2 μl/200 μl of total blood (***). GFP, green fluorescent protein; IBA-1, ionized calcium binding adaptor molecule 1; Gr-1, granulocyte differentiation antigen 1; Ly6-C, lymphocyte antigen 6 complex, locus C1; PE, phycoerythrin; APC, allophycocyanin; FITC, fluorescein isothiocyanate.
Immunohistochemistry
To collect the brain tissues, mice were deeply anesthetized via an IP injection of a mixture of ketamine hydrochloride and xylazine, and then rapidly perfused transcardially with 0.9% saline, followed by 4% paraformaldehyde/3.8% borax in sodium phosphate buffer (pH 9 at 4°C). Brains were rapidly removed from the skulls, postfixed overnight, and then placed in a solution containing 10% sucrose diluted in 4% paraformaldehyde/3.8% borax buffer (pH 9) overnight at 4°C. The frozen brains were mounted on a microtome (Reichert-Jung, Cambridge Instruments Company, Deerfield, IL), frozen with dry ice, and cut into 25-μm coronal sections from the olfactory bulb to the end of the medulla. The slices were collected in a cold cryoprotectant solution (0.05 M sodium phosphate buffer, pH 7.3, 30% ethylene glycol, 20% glycerol) and stored at −20°C. Free-floating sections were incubated for 30 min in potassium phosphate-buffered saline (KPBS) containing 4% goat serum, 1% bovine serum albumin (BSA), and 0.4% Triton X-100. Using the same buffer solution, the sections were then incubated for 90 min in primary antibody at room temperature. The sections were then rinsed four times for 5 min in KPBS, followed by a 90-min incubation in fluorochrome- or biotin-conjugated goat secondary antibody. Fluorochromes were fluorescein isothiocyanate (FITC) and tetramethyl rhodamine isothiocyanate (TRITC). For nonfluorescent staining, biotin-conjugated secondary antibodies were detected using the peroxidase-based Vectastain ABC kit (Vector Labs, Burlingame, CA) following the manufacturer's instructions. The complete list of antibodies used in these experiments and their dilutions is summarized in Table 1. Sections were then rinsed four times for 5 min in KPBS, mounted onto SuperFrost slides (Fisher Scientific, Nepean, Ontario, Canada), stained with 4′,6-diamidino-2-phenylindole (DAPI) if needed (0.0002% dilution for 10 min; Molecular Probes), and coverslipped with antifade medium composed of 96 mM Tris-Hcl, pH 8.0, 24% glycerol, 9.6% polyvinylalcohol, and 2.5% diazabicyclooctane (Sigma). Confocal laser scanning microscopy was performed with a BX-61 microscope equipped with the Fluoview SV500 imaging software 4.3 (Olympus America Inc, Melville, NY). Confocal images were acquired by sequential scanning using a two-frame Kalman filter and a z-separation of 1 μm.
In Situ Hybridization
In situ hybridization was performed on every 12th section of the brain, starting from the end of the olfactory bulb to the end of the cortex, using 35S-labeled cRNA probes as described previously (9,10,15). The mRNA probes were generated with the following primers: GGCTCTTCTGGATCTTGGTGGCC (forward) and GGGCCACTCCAGGTAGGTCTTGG (reverse) for toll-like receptor 2 (TLR2) and GCCTGTAGCCCACG TCGTAGC (forward) and AAAGTAGACCTGCCCG GACTC (reverse) for tumor necrosis factor-α (TNF-α). The probes for interleukin (IL)-1β and monocyte chemoattractant protein-1 (MCP-1) were both kind gifts from Dr. Pennica (7) and Dr. Stiles (20), respectively. These probes are clones of the full-length cDNAs.
Fluorojade Staining
Neuronal death was labeled with the fluoro-jade b (FJB) method. Briefly, every sixth section of the whole rostrocaudal extent of each brain was mounted onto poly-l-lysine-coated slides, dried under vacuum for 2 h, dehydrated through graded concentrations of alcohol (50%, 70%, and 100%, 1 min each), and rehydrated through graded concentrations of alcohol (100%, 70%, and 50%, 1 min each) and 1 min in distillated water. They were then dipped into and shaken in potassium permanganate (0.06%) for 10 min, rinsed 1 min in distillated water, and dipped into and shaken in a solution containing FJB [FJB 0.0004% (Histochem, Jefferson, AR) + acetic acid 0.1% (Sigma-Aldrich) + DAPI 0.0002% (Molecular Probes Eugene, OR)] for 20 min. The slides were thereafter rinsed three times in distillated water (1 min each), dried, dipped in xylene three times (2 min each), and coverslipped with a mounting medium (DPX, Sigma-Aldrich).
Stereological Analysis
Immunostained brain sections were analyzed using a stereological apparatus (n = 4 for each group). Slices were stained for microglia [anti-IBA-1 (ionized calcium binding adaptor molecule 1)[(see Table 1), GFP, and nuclei (DAPI, 2e–4%). A total of 10 animals per group were analyzed. Two sections per animal were selected that contained the hypoglossal nucleus and the loci of migration. Real-time images (1600 × 1200 pixels) were obtained using a Nikon C80i microscope equipped with both a motorized stage (Ludl) and a Microfire charge couple device (CCD) color camera (Optronics). Such an apparatus was operated using the Stereo Investigator software designed by Microbrightfield. The sampling method was previously tested in a pilot study to ensure correct estimation in the number of quantified elements. First, the Stereo Investigator software was used to delimit counting frames of 670 × 500 μm, and located at each 2000 μm in the x-axis and at every 1000 μm in the y-axis of the previously selected cortex and hippocampus regions. Microglia were counted for each frame using the pen display, a 40× Plan Apochromat objective (NA 0.95), and a triple-band filter (DAPI/FITC/TRITC, Chroma Technology). IBA-1-immunoreactive cells were counted only when their nuclei (DAPI labeled) were in the dissector area (20 mm) and not in contact with the two forbidden contours of the counting frame. A similar method was used for GFP+ cells. Results are expressed as mean ± SEM of the number of cells positive for both GFP and IBA-1.
Statistical Analysis
A two-tailed Student's t-test was performed to determine the statistical significance of the differences observed in quantitative experiments. A value of p < 0.05 was used as a cutoff to reach significance.
Results
Adaptation of a Myeloablative Chemotherapy Regimen for the Transplantation of Bone Marrow Cells
In order to understand whether myeloablation is sufficient to allow for the entry of BMDCs into the brain, a myeloablative chemotherapy regimen was adapted from those used in bone marrow transplants in humans. It consisted of 4 days of twice daily injections of 10 mg/kg of busulfan followed by 2 days of 100 mg/kg of cyclophosphamide. The advantage of this treatment is that it circumvents the need for irradiation and also completely depletes the myeloid cells, including those that survive in a protected head irradiation protocol. In our setting, chemotherapy led to a complete depletion of monocytes in the circulation as seen in the scatter plot of CD45+ cells of treated mice (Fig. 1). We found that chemotherapy depleted the monocyte population (CD115+, CD11b+) by 90.1%, comparable to the 88.6% depletion induced by irradiation (Fig. 1B).

A busulfan-cyclophosphamide chemotherapy regimen is as efficient as whole-body irradiation to induce myeloablation, which was confirmed by fluorescence activated cell sorting (FACS) analysis of the blood of treated and untreated mice. Displayed are the scatter plots of hematopoietic cells isolated by staining for the CD45 antigen (A). Monocytes were also counted by isolating the CD11b+ CD115+ cells (B). Represented are the means of the levels observed in three replicates of experiments implicating five mice for each conditions. *p < 0.001 versus wild-type (WT).
Limited Effects of Irradiation and Chemotherapy on Brain Integrity
In order to understand the effects of irradiation on the brain, 3-month-old wild-type C57Bl/6 mice received lethal doses of whole-body irradiation (10 Gy) (Fig. 2). Rapid and transient damages to neurons were observed with fluorojade staining of brain sections of these mice (Fig. 2A, C, E). Damage was observed in the olfactory limb, in cells of the caudoputamen bordering the lateral ventricles, and in the granular layer of the dentate gyrus. The damages were also found by staining for the apoptosis marker cleaved caspase 3 (Fig. 2B, D, F). In all cases, the damage could not be detected 48 h following irradiation and no long-term damage was found 7 days after irradiation. Another concern about irradiation is that it induces strong, sustained neuroinflammation. However, in situ hybridization for TLR2, MCP-1, IL-1, and TNF-α, all classical markers of neuroinflammation, showed that limited inflammation occurred 6 h after irradiation but it did not persist (Fig. 3). Neither cell death nor inflammatory markers could be detected in animals that had received chemotherapy (Figs. 2, 3). Moreover, injecting bone marrow cells from GFP+ transgenic mice 6, 24, 48, 72 h, or 7 days after irradiation did not influence the blood chimerism and brain infiltration of GFP+ cells (Fig. 4), showing that the presence of the damage is not necessary to induce the entry of BMDCs into the brain.
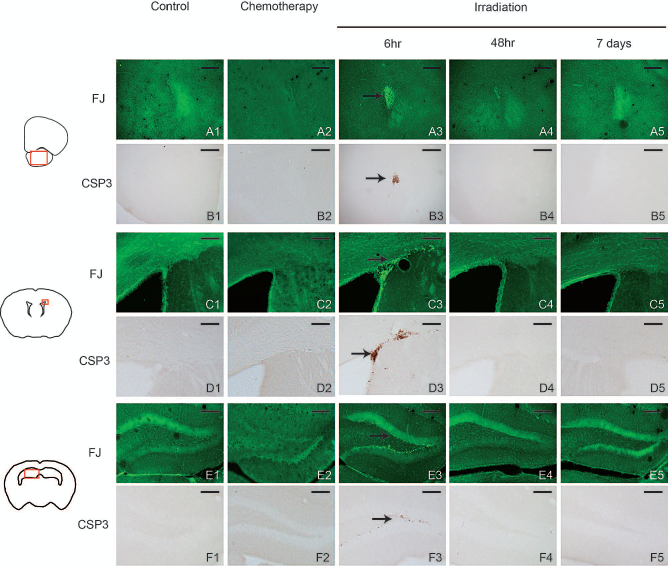
Irradiation and chemotherapy have no lasting effect on brain integrity. C57Bl6 mice received either a 10-Gy dose of irradiation and were sacrificed 6 or 48 h and 7 days later or a course of myeloablative chemotherapy and sacrificed 24 h after the end of the protocol. Fluorojade-B (FJ) staining revealed neuronal death (A, C, E) and diaminobenzidine (DAB) staining for cleaved caspase 3 (CSP3) revealed cellular apoptosis (B, D, F). These regions are indicated by a black arrow. Represented regions are located by a red square to the left and include the olfactory limb (A, B), the caudoputamen (C, D), and the dentate gyrus (E, F). In all images, the black bars represent 100 μm except in slides (C) and (D) where it represents 50 μm.

Irradiation does not induce a strong, sustained neuroinflammation profile. Expression levels of neuroinflammatory markers of following irradiation were determined by in situ hybridization. A wild-type untreated control served as negative control. Mice that received a single IP lipopolysaccharide (LPS) injection were included as positive controls. In all images, the white bars represent 50 μm except for the TLR2 images in which the bars represent 100 μm. TLR2, toll-like receptor 2; MCP1, monocyte chemoattractant protein-1; IL-1, interleukin-1; TNF-α, tumor necrosis factor-α
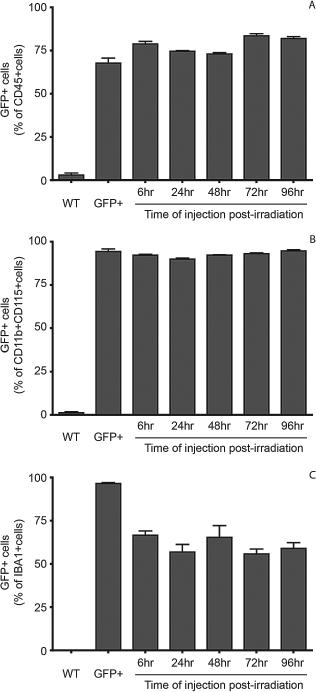
The expression of cell death markers is not needed to allow the migration of GFP+ cells into the CNS. Green fluorescent protein positive (GFP+) bone marrow cells were injected at various times following irradiation. Levels of chimerism were determined in the circulation (A, B) and in the brain (C) as determined by FACS analysis of CD45 cells (A) and CD11b+CD115+ cells (monocytes, B) or by stereological analysis of ionized calcium binding adaptor molecule 1 positive (IBA-1+cells in the brain (C). Also included are a negative control (untreated wild-type mice) and a positive control (GFP+ transgenic mice). In every case, the amount of GFP+ cells in GFP+ mice and irradiated mice were significantly different from WT mice (p < 0.0001).
Whole-Body Irradiation and Chemotherapy Does Not Affect the Integrity of the BBB
Damage to the BBB has been assumed to be responsible for the entry of BMDCs into the brain (17). We used classic immunostaining for BBB damage by staining for mouse immunoglobulin-g (IgG) and albumin (Fig. 5). These proteins are only found in the circulation and their presence in the brain is a clear sign of BBB disruption (16). This technique also alleviates the need for complex surgeries that require the installation of stereotaxic dialysis probes and skull screws, themselves disrupting the BBB integrity. Normal staining for IgG and albumin was observed in the dentate gyrus, the fimbria, and periventricular hypothalamic nucleus of irradiated mice (Fig. 5D, E, I, J). This staining pattern was similar to that observed in untreated controls (Fig. 5A, F). No sign of immunostaining for IgG or albumin was found in the cortex or any other part of the brain as compared to a positive control of a BBB mechanically disrupted with a micro syringe (Fig 5B, G). These results clearly show that whole-body irradiation as a means to permit the entry of BMDCs into the brain has no effect on the integrity of the BBB and its permeability to components of the circulation.

Irradiation and chemotherapy do not alter the blood–brain barrier (BBB). Immunoglobulin-g (A–E) and albumin (F–J) staining was used to test the integrity of the blood-brain barrier (BBB). Displayed are a positive control (B, G: a BBB mechanically disrupted by a stereotaxic needle) and a representation of the staining we found in untreated wild-type controls (negative control; A, F), irradiated mice (D, E, I, J), and mice that received chemotherapy (C, H). In all images, the black bars represent 100 μm except in slides (B) and (G) where it represents 50 μm.
Myeloablation Is Sufficient to Induce Peripheral Chimerism in Bone Marrow Transplantations
Bone marrow cells from GFP+ transgenic mice were injected into wild-type littermates 24 h following either irradiation or the end of the chemotherapy regimen. Three months later, GFP+ cells were found as abundant in chemotherapy-treated mice as in irradiated mice, representing 75.5% of CD45+ cells and 94.2% of CD115+/CD11b+ cells (Fig. 6A, B). Most importantly, GFP+ cells effectively infiltrated the peripheral organs (Fig. 6C–H). In the spleen, GFP+ cells were found both in the red pulp and in lymphatic nodes (Fig. 6C–E), implicated respectively in granulocyte and macrophage maturation and in T-lymphocyte maturation. In the thymus, GFP+ cells were restricted to lobules' medulla (Fig. 6F–H) where T-lymphocytes undergo their final maturation process. Thus, chemotherapy was as efficient as irradiation both to induce myeloablation and to allow for a sustained and robust chimerism, both in the circulation and in peripheral organs.

Chemotherapy and irradiation are both efficient to induce a strong chimerism in the periphery. Following either chemotherapy or irradiation, bone marrow cells from GFP+ transgenic mice were injected in the tail vein of recipient mice. Three months later, they were sacrificed and their blood was analyzed by FACS. Displayed is a representative cell count for the number of CD45+ cells in relation to their intensity of GFP fluorescence (A). The chimerism was observed in all subpopulations of the circulation (B). Displayed are the means and standard deviation of the percentage of GFP+ cells from five replicates of experiments performed twice in every case, the amount of GFP+ cells in GFP+ mice, irradiated and chemotherapy-treated mice were significantly higher than in control mice (p < 0.0001). The difference between the treatment groups did not reach statistical significance. GFP+ cells were also identified in the major hematopoietic peripheral organs, spleen (C–E), and thymus (F–H), by immunofluorescence. Displayed are the merges of DAPI staining (nuclei) in blue and GFP staining in green. In panels (C)–(H), the white bars represent 50 μm.
Myeloablation and Peripheral Chimerism Is Not Sufficient to Induce the Entry of BMDCs Into the Brain
Is myeloablation and peripheral chimerism sufficient to allow for the entry of BMDCs into the CNS? We investigated the presence of GFP+ cells in the brain using the hypoglossal nerve axotomy model that induces a strong migration of microglial cells in the ipsilateral side of the hypoglossal nucleus (14) (Fig. 7A–C). Microglial cells were revealed by staining for IBA-1. As expected, most IBA-1+ cells that migrated into the hypoglossal nucleus in mice that had been irradiated were also GFP positive (Fig. 7D, E), indicating a robust infiltration of BMDCs. GFP+ IBA-1+ cells were also found throughout the cortex of irradiated mice (Fig. 7F). In mice that received chemotherapy, GFP+ cells could not be found in the hypoglossal nerve region (Fig. 7G, H) or anywhere else in the cerebral cortex. GFP+ cells were only found in the choroid plexus and the circumventricular organs (CVOs, Fig.7H, I). As such, it appears that grafted cells can migrate to regions of the brain not protected by the BBB or the blood–cerebrospinal fluid barrier (BCSFB) but are not able to cross through to populate the CNS.

Migration of microglial cells into the hypoglossal nucleus region following hypoglossal nerve axotomy. Slides were stained in red for IBA-1, a microglial cell marker (A, D, G), and for the presence of GFP in green (B, E, H). Represented are classical staining patterns observed in untreated mice (A, B), after whole-body irradiation (D–F), and after chemotherapy (G, I). Panels (A), (B), (D), (E), (G), (H) represent the region encompassing the hypoglossal nucleus and the area postrema, as depicted by a red square in (C). The high number of IBA-1+ cells migrating to the hypoglossal nucleus is a result of the hypoglossal nerve axotomy performed in these mice a week prior to sacrifice. Also shown is the presence of GFP+ IBA-1+ cells throughout the cortex of irradiated mice (F) and in the choroid plexus of mice that received chemotherapy (I). Both images are merges of IBA-1 staining in red and GFP in green. In all images, the white bars represent a length of 100 μm except in (F) where it represents 25 μm.
Discussion
We report that a lethal dose of whole-body irradiation has limited effects on cell and neuron death in the brain. The limited damages found were all in regions with active neurogenesis (11), which could explain why they display cell death, as irradiation is known to have more effects on dividing cells (3). These damages could not be detected 24 h after irradiation. The transient nature of the damages in these few regions could also be attributed to the ability of these cells to regenerate. We could not detect any changes in the integrity of the BBB. Previous reports on the effects of irradiation on the BBB indicated the contrary. However, most models studying irradiation-induced BBB damages usually use either high doses of irradiation (20–50 Gy) (12), deliver the irradiation directly to the denuded brain or nerve region (12), and use radiolabeled tracer compounds (4). The irradiation protocol used in our study was less aggressive, delivering 10 Gy to the whole body, but sufficient to induce BMDC entry into the brain, and the method used to detect BBB leakage was much less invasive. Taken together, our results show that brain entry permitting irradiation doses do not induce lasting defects to the brain integrity or to the BBB, which warrants the use of irradiation in a clinical setting, as is already the case for the treatment of different types of leukemia.
In our setting, myeloablative chemotherapy was sufficient to induce a strong chimerism in the periphery, both in the circulation and in hematopoietic organs such as thymus and spleen, but these cells did not invade the CNS. These results confirm those observed in the models of irradiated mice with a protected head in that it appears that irradiation has an effect on brain cells that allows for the entry of BMDCs into the brain. Grafted cells were found in the vicinity of the brain, in regions unprotected by the BBB such as circumventricular organs or the choroid plexus. However, these cells seem unable to cross the BBB or the BCSFB to populate the CNS parenchyma. The BCSFB's involvement has previously been suggested in BMDC entry into the CNS (21). It is already implicated in the control of the migration of leukocytes into the CNS in conditions like amyotrophic lateral sclerosis (ALS) and experimental autoimmune encephalitis (EAE). Possible effects of irradiation on the BBB or BCSFB could include weakening of the junctions between cells that are the basis of the permeability of these barriers or the release of chemoattractant factors such as vascular endothelial growth factor (VEGF) that could induce the entry of the cells. Clearly, further work is needed to understand the effect of irradiation on the choroid plexus and the permeability of the BCSFB.
On the other hand, the chemotherapy protocol we applied was the same that was used to prepare the patients in the gene therapy study of X-linked ALD (2). Since the results of the therapy are indisputable when comparing the computed tomography (CT) scans of the treated brains to controls, we can assume that in certain pathological conditions such as ALD, stroke, direct brain injury, or even Alzheimer's disease, the permeability of the brain to BMDCs is increased and chemotherapy alone could prove a sufficient pretreatment.
Three months after the injection of bone marrow cells, myeloid cells in the circulation and in the brain of irradiated recipient mice were predominantly grafted cells. This demonstrates that grafted bone marrow cells are not transient in the CNS and could thus be used for the long-term treatment of various pathologies as a onetime treatment option.
Footnotes
Acknowledgments
The presented work was supported by grant number 219561 from the Canadian Institutes for Health Research. S.R. holds a Canadian Research Chair in Neuroimmunology. The authors would like to thank Dr. Stiles and Dr. Pennica for graciously providing us with the plasmids coding for the cRNA probes for MCP-1 and IL-1β. The authors declare no conflicts of interest.
