Abstract
Currently, there is very limited large-scale real-world data on the use of total marrow irradiation (TMI) as conditioning for allogeneic hematopoietic stem cell transplantation (allo-HSCT). The primary objective of this study was to evaluate the real-world feasibility, efficacy, and safety of TMI-based conditioning in a large multicenter cohort. We retrospectively included consecutive patients undergoing allo-HSCT with TMI-based conditioning across four Chinese centers (2017–2024). The primary endpoint was overall survival (OS); secondary endpoints included disease-free survival (DFS), graft-versus-host disease–free/relapse-free survival (GRFS), nonrelapse mortality (NRM), graft-versus-host disease (GVHD), and safety. Among 205 patients, acute lymphoblastic leukemia (ALL) was the predominant diagnosis (81.5%). With a median follow-up of 19.3 months among survivors, 2-year OS, DFS, and GRFS were 71.7%, 60.0%, and 37.8%, respectively. One-year cumulative incidences of relapse and NRM were 20.2% and 13.8%. Day-100 grade II–IV acute GVHD occurred in 32.9%, and 1-year moderate-to-severe chronic GVHD in 19.1%. In multivariable analyses, acute myeloid leukemia (AML) diagnosis, haploidentical donor, and Eastern Cooperative Oncology Group (ECOG) performance status >1 were associated with inferior outcomes. Exploratory dose-stratified analyses were performed. Severe toxicities within 100 days were infrequent, predominantly infections (21.46%). In this large multicenter cohort, TMI-based conditioning demonstrated real-world feasibility with encouraging survival and manageable safety outcomes.
Keywords
Introduction
Total body irradiation (TBI) in combination with chemotherapy has long been used as a conditioning regimen for allogeneic hematopoietic stem cell transplantation (allo-HSCT) 1 . While effective in eradicating malignant cells and providing immunosuppression, TBI exposes normal tissues to radiation and carries risks of pulmonary, hepatic, and gastrointestinal toxicities 2 . These limitations have motivated efforts to develop alternative strategies that reduce toxicity without compromising disease control.
Total myeloid irradiation (TMI) uses conformal radiation therapy to focus the dose on the bone marrow tissue while sparing other vital organs3–5. Early studies have demonstrated its feasibility and potential efficacy, but most reports were from single institutions, involved small numbers of patients, and had short follow-up periods 6 . The lack of large-scale real-world data leaves uncertainty regarding outcomes across diverse diseases and conditioning settings.
To address these gaps, we conducted a multicenter retrospective study of patients with hematologic malignancies who received TMI-based conditioning regimens. Unlike previous small-sample studies limited to specific disease types or dose levels, this cohort study included patients with acute lymphoblastic leukemia (ALL), acute myeloid leukemia (AML), lymphoma, and other hematological malignancies, and evaluated in detail the real-world survival outcomes, toxicity, and dose-response patterns 7 . This work provides broader clinical insights into the outcomes of TMI-based conditioning regimens.
Methods
Study design and patient inclusion
This was a multicenter retrospective cohort study conducted at four transplant centers in China between March 2017 and September 2024. Consecutive patients with hematologic malignancies who received TMI-based conditioning followed by allo-HSCT were included. Eligible diagnoses comprised AML, T-cell acute lymphoblastic leukemia (T-ALL), B-cell acute lymphoblastic leukemia (B-ALL), lymphoma, and other hematologic malignancies. Patients undergoing autologous transplantation were excluded. Follow-up information was available for all, including patients. Risk stratification was determined according to established international criteria: ELN classification for AML, NCCN guidelines for ALL, and IPI for lymphoma. For other rare hematologic malignancies, risk status was assessed by the treating physician based on disease biology and response to prior therapy. This study was conducted in accordance with the Declaration of Helsinki and was approved by the Ethics Committee of Union Hospital, Tongji Medical College, Huazhong University of Science and Technology (approval No. 2025-1040). Given the retrospective design, the requirement for informed consent was waived.
Conditioning regimen
All patients received TMI-based conditioning. TMI was delivered using helical tomotherapy (TomoTherapy® platform) at all centers, with a total prescribed dose ranging from 4 to 12 Gy, most commonly 8 or 12 Gy delivered in 2–6 fractions. The specific dose and fractionation were determined by each center according to institutional protocols and patient characteristics. Additional irradiation, including craniospinal irradiation (CSI) for central nervous system involvement or high CNS relapse risk, total lymphoid irradiation (TLI), or selective boost fields (e.g. mediastinum, testes), was applied at the discretion of the treating physicians. Organs at risk (OARs) were contoured, and institutional dose constraints were applied to minimize exposure to critical organs. Chemotherapy backbones most commonly included etoposide (VP-16), cyclophosphamide (CY), and fludarabine (FLU), administered in combination with TMI. The choice of agents and schedules was determined by institutional practice. Conditioning regimens were categorized based on the presence of VP-16, CY, and FLU, and are summarized in Supplementary Table S1. In general, CY was administered at 60 mg/kg/day for 2 days, VP-16 at approximately 5 mg/kg every 12 h for 2 days, and FLU at 30 mg/m2/day for 4–5 days, with dose adjustments permitted according to institutional practice.
GVHD prophylaxis
GVHD prophylaxis regimens were determined by each center according to routine clinical practice. Most patients received a calcineurin inhibitor (cyclosporine A or tacrolimus) in combination with short-course methotrexate (MTX), with or without mycophenolate mofetil (MMF). For patients undergoing haploidentical HSCT or matched unrelated donor (MUD) HSCT, additional agents such as anti-thymocyte globulin (ATG), antilymphocyte globulin (ALG), or interleukin-2 receptor antagonists (e.g. CD25 monoclonal antibodies) were administered as part of prophylaxis. No patient in this cohort received posttransplant cyclophosphamide (PTCy)–based GVHD prophylaxis.
Engraftment
Neutrophil engraftment was defined as the first of the three consecutive days with an absolute neutrophil count (ANC) ≥ 0.5 × 109/L. Platelet engraftment was defined as the first of the seven consecutive days with a platelet count ≥20 × 109/L in the absence of transfusion support. The first day of stem cell infusion was defined as day 0.
Supportive care
All patients received supportive care according to institutional guidelines. Antimicrobial prophylaxis regimens were determined by each center. CMV-DNA and EBV-DNA were detected by quantitative polymerase chain reaction (PCR) weekly during the first 100 days and monthly thereafter to monitor for reactivation of cytomegalovirus (CMV) and Epstein–Barr virus (EBV). Other supportive measures, including infection surveillance, transfusion thresholds, and nutritional or organ-specific management, were implemented in accordance with each center’s transplant protocols.
Endpoints
The primary endpoint of this study was overall survival (OS), defined as the time from transplantation to death from any cause or last follow-up. Secondary endpoints included disease-free survival (DFS), graft-versus-host disease–free/relapse-free survival (GRFS), cumulative incidence of relapse (CIR), nonrelapse mortality (NRM), incidence and grading of acute and chronic GVHD, and treatment-related toxicities. DFS was defined as the time from transplantation to relapse or death, whichever occurred first. CIR was defined as the first hematologic or extramedullary relapse, and NRM was defined as death without prior relapse. Acute Graft-versus-Host Disease (aGVHD) was diagnosed and graded according to the modified Glucksberg criteria, and chronic Graft-versus-Host Disease (cGVHD) was evaluated according to the 2014 National Institutes of Health (NIH) consensus criteria. GRFS was defined as the time from transplantation to the first occurrence of any of the following events: grade III–IV aGVHD, cGVHD requiring systemic immunosuppressive therapy, disease relapse, or death from any cause. Regimen-related toxicities within +30 days were graded by Bearman criteria. Other early organ-specific adverse events and complications within +100 days and late organ-specific adverse events and complications beyond +100 days were graded by CTCAE v5.0. The data cut-off for follow-up was April 9, 2025.
Statistical analysis
Descriptive statistics were used to summarize baseline characteristics. Continuous variables were expressed as medians with ranges, and categorical variables as counts and percentages. OS, DFS, and GRFS were estimated using the Kaplan–Meier method and compared with the log-rank test. The cumulative incidence of relapse (CIR), NRM, and acute and chronic GVHD were analyzed using the cumulative incidence function with competing risks, and compared with Gray’s test.
Univariate analyses were performed for all endpoints. Variables with a P value <0.10 in univariable analysis, together with clinically relevant covariates, were entered into multivariable models. Cox proportional hazards regression was used for OS, DFS, and GRFS, while Fine–Gray subdistribution hazard models were applied for competing-risk outcomes (CIR, NRM, and GVHD). Results were reported as hazard ratios (HRs) or subdistribution HRs with 95% confidence intervals (CIs). Multivariable analyses were performed on complete cases without imputation; missingness by variable is reported in Table 1. In sensitivity analyses, the transplant center was additionally included as a covariate in multivariable Cox and Fine–Gray models to evaluate the potential impact of center-level differences.
Baseline characteristics of the study cohort.

Percentages were calculated from available and applicable cases; denominators vary by variable. MRD by flow cytometry was not applicable in lymphoma, HLH, and PMF; extramedullary involvement was not applicable in HLH.
Stratified analyses were performed by diagnosis category (AML, T-ALL, B-ALL/lymphoma /others), extramedullary involvement (yes/no), and by TLI (yes/no), using the same definitions and modeling strategies as in the overall analysis.
Exploratory analyses assessed the association of TMI dose with outcomes, focusing on two comparisons: >9 Gy versus ≤9 Gy, and exactly 12 Gy versus <12 Gy. To address baseline imbalances, propensity score matching (PSM) was performed separately for each comparison. Because the two dose contrasts differed in group size and baseline distributions, matching parameters were determined independently based on standardized mean difference diagnostics. For the >9 Gy analysis, logistic regression models incorporating major clinical covariates were used to generate propensity scores, and 1:1 nearest-neighbor matching within a 0.2 caliper was applied. For the 12 Gy analysis, the same covariates were used with 1:3 nearest-neighbor matching within a 0.15 caliper. Adequate balance was confirmed (all standardized mean differences <0.1). Dose-specific analyses were restricted to patients with available TMI dose data; two patients receiving TMI doses <8 Gy were excluded to reduce heterogeneity related to nonstandard regimens. These analyses were considered hypothesis-generating.
A two-sided P value < 0.05 was considered statistically significant. The proportional-hazards assumption for Cox models was evaluated using Schoenfeld residuals. All statistical analyses were performed using R software (version 4.4.2; R Foundation for Statistical Computing, Vienna, Austria). Survival analyses were conducted using the survival package, and Kaplan–Meier curves were visualized using the survminer package. Competing risk analyses were performed using the cmprsk package. PSM was conducted using the MatchIt package, with balance assessed using the cobalt package.
Results
Baseline characteristics
A total of 205 patients who received TMI-based conditioning followed by allo-HSCT were included in this study. Detailed radiotherapy parameters were available for 169 patients (82.4%); the remaining 36 were confirmed to have received TMI but lacked specific dose records. Age ranged from 7 to 61 years, with a median age of 27 years at transplantation. Overall, 68.78% of patients were male. Among the cohort, 81.46% of patients had ALL, including B-ALL, 82 patients, 40.00%, and T-ALL, 85 patients, 41.46%. The Others group (n = 10) included rare entities such as hemophagocytic lymphohistiocytosis (HLH, n = 2), mixed phenotype acute leukemia (MPAL, n = 2), primary myelofibrosis (PMF, n = 1), myelodysplastic syndrome (MDS, n = 1), blastic plasmacytoid dendritic cell neoplasm (BPDCN, n = 1), unclassified dendritic cell tumor (n = 1), myeloid sarcoma (n = 1), and chronic myeloid leukemia in blast crisis (CML-BC, n = 1). A high-risk disease status was observed in 71.22% of the cohort, 18.52% had flow cytometry measurable residual disease (MRD) positivity, and 9.27% had not achieved complete remission (CR) at the time of transplantation. Extramedullary disease was observed in 24.63% of patients, including CNS, lymph node, and soft tissue infiltration. Most patients had an Eastern Cooperative Oncology Group performance score (ECOG-PS) of 0–1 (75.72%), a Karnofsky Performance Status (KPS) ≥90 (71.22%), and a Hematopoietic Cell Transplantation Comorbidity Index (HCT-CI) score <3 (87.80%). Donor types included haploidentical donors (HID, 68.66%), matched sibling donors (MSD, 23.88%), and MUD (7.46%). In haploidentical HSCT, 53.79% of patients received ATG/ALG-based GVHD prophylaxis, while 46.21% received CNI-based regimens without ATG (Supplementary Table S2). Peripheral blood stem cells (PBSC) were the predominant graft source (93.66%). The donor–recipient relationship included siblings (49.50%) and parent/child pairs (39.60%). Among gender combinations, female-to-male transplantation accounted for 22.45%. ABO blood group was mismatched in 42.16% of cases. Most patients received a TMI dose of 8 or 12 Gy. CSI and TLI were administered in 95.9 and 72.2% of patients, respectively (Table 1).
Engraftment
Neutrophil engraftment was achieved at a median of 11 days (range, 7–23), and platelet engraftment at a median of 12 days (range, 5–42).
Overall outcomes
The median follow-up of surviving patients was 19.3 months (range, 1.0–86.9 months). The 2-year OS, DFS, and GRFS rates were 71.7% (65.2–78.8), 60.0% (53.1–67.8), and 37.8% (31.2–45.9), respectively (Fig. 1). The 1-year CIR and NRM was 20.2% (14.5–25.9) and 13.8% (8.9–18.6). The cumulative incidence of grade II–IV acute GVHD at day 100 was 32.9% (26.4–39.4), and grades III–IV was 17.7% (12.4–22.9%). The 1-year cumulative incidence of overall chronic GVHD was 33.3% (26.6–40.1%), and the incidence of moderate-to-severe chronic GVHD was 19.1% (13.5–24.8). At 2 years, the cumulative incidence of overall and moderate-to-severe chronic GVHD were 36.1% (29.1–43.1%) and 21.3% (15.3–27.3), respectively (Fig. 2). Key transplantation outcomes at 180 days and 3 years are summarized in Table 2.
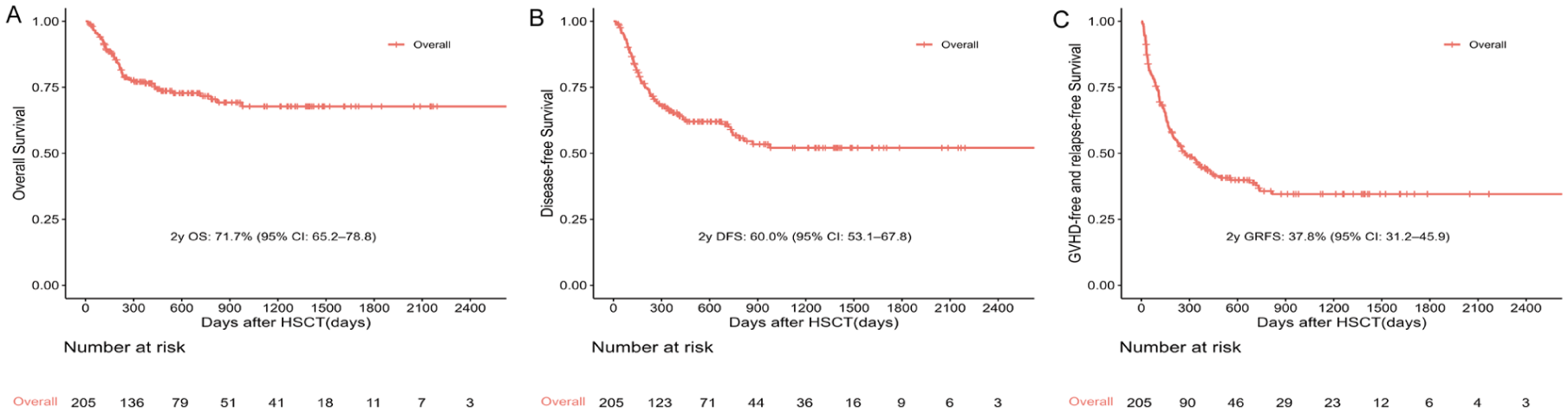
Survival outcomes after allo-HSCT. (a) Overall survival (OS). (b) Disease-free survival (DFS). (c) GVHD-free/relapse-free survival (GRFS).

Cumulative incidence outcomes after allo-HSCT. (a) Relapse (CIR). (b) Nonrelapse mortality (NRM). (c) Grade II–IV acute GVHD (aGVHD). (d) Moderate-to-severe cGVHD.
Patient transplantation outcomes.

Acute GVHD was evaluated at day 100, whereas OS/DFS/GRFS/CIR/NRM/chronic GVHD were evaluated at 3 years.
Stratified analyses
By diagnosis
When stratified by diagnosis, no statistically significant differences were observed among patients with B-ALL, T-ALL, AML, and other diagnoses. The 2-year OS was 73.8% (64.0–85.0), 71.3% (61.3–82.9), 62.9% (41.6–95.0), and 70.8% (53.4–93.9) (P = 0.580), respectively. Corresponding values for DFS were 57.3% (46.6–70.6), 63.4% (53.5–75.1), 62.9% (41.6–95.0), and 55.1% (36.8–82.3) (P = 0.955); for GRFS, 34.5% (25.1–47.3), 44.9% (35.0–57.6), 29.6% (13.1–66.8), and 34.4% (17.6–66.9) (P = 0.410); for CIR, 27.9% (17.1–38.7), 25.5% (15.7–35.3), 7.9% (0.0–23.4), and 25.2% (5.2–45.2) (P = 0.411); and for moderate-to-severe cGVHD, 20.8% (11.6–29.9), 21.0% (11.6–30.3),30.4% (3.4–57.3), and 19.2% (0.0–41.1) (P = 0.723) (Figure 3).

Survival and cumulative incidence outcomes stratified by diagnosis. (a) Overall survival (OS). (b) Disease-free survival (DFS). (c) Relapse (CIR). (d) Nonrelapse mortality (NRM).
By extramedullary involvement (EMI)
When stratified by extramedullary involvement (EMI), outcomes were broadly comparable between patients with and without EMI. The 2-year OS was 74.3% (62.1–88.8) versus 70.5% (62.9–79.1) (P = 0.491), DFS 58.6% (45.4–75.6) versus 61.0% (53.2–70.0) (P = 0.754), GRFS 43.6% (31.2–61.1) versus 37.2% (29.6–46.6) (P = 0.278), CIR 32.5% (18.0–47.0) versus 21.9% (15.0–28.9) (P = 0.253), and NRM 8.9% (0.4–17.4) versus 17.0% (10.6–23.5) (P = 0.106). Rates of aGVHD II–IV at day 100 (22.1% (10.4–33.8) versus 36.1% (28.5–43.8), P = 0.115) and moderate-to-severe cGVHD at 2 years (13.3% (3.2–23.4) versus 23.1% (16.0–30.2), P = 0.176) were also similar (Figure 4).

Survival and cumulative incidence outcomes stratified by extramedullary involvement. (a) Overall survival (OS). (b) Disease-free survival (DFS). (c) Relapse (CIR). (d) Nonrelapse mortality (NRM).
By TMI combining with TLI
In subgroup analyses by TLI exposure, survival and transplant-related outcomes did not differ significantly. Patients receiving TLI had a 2-year OS of 75.5% (67.4–84.5) compared with 61.5% (48.5–78.1) in those without TLI (P = 0.066). Corresponding estimates for DFS were 60.1% (51.3–70.4) versus 56.2% (43.3–72.9) (P = 0.431), GRFS 38.0% (29.7–48.7) versus 39.3% (27.3–56.6) (P = 0.621), CIR 28.1% (19.5–36.8) versus 26.5% (13.2–39.8) (P = 0.997), and NRM 11.8% (5.5–18.1) versus 17.3% (6.2–28.3) (P = 0.351). The incidence of aGVHD II–IV by day 100 (29.0% versus 34.0%, P = 0.541) and moderate-to-severe cGVHD at 2 years (21.5% versus 19.8%, P = 0.930) was likewise comparable between groups (Supplementary Figure S1).
Multivariable analysis
In multivariable analyses, several clinical factors were significantly associated with outcomes. Compared with B-ALL, AML diagnosis was associated with significantly inferior OS (HR 2.90, 95% CI 1.08–7.81, P = 0.035), higher NRM (sHR 6.68, 95% CI 2.03–21.98, P = 0.002), with a trend toward increased grade II–IV aGVHD (sHR 2.18, 95% CI 0.98–4.86, P = 0.056). T-ALL did not differ significantly from the reference across endpoints. HID transplantation was associated with worse OS (HR 2.49, P = 0.034) and DFS (HR 1.87, P = 0.044). Poor performance status (ECOG–PS >1) was independently associated with inferior OS (HR 2.14, 95% CI 1.15–4.01, P = 0.017) and markedly higher NRM (sHR 5.21, 95% CI 2.08–13.08, P < 0.001). Extramedullary involvement was associated with a lower risk of NRM (sHR 0.22, 95% CI 0.08–0.65, P = 0.006) and lower moderate-to-severe cGVHD (sHR 0.38, 95% CI 0.15–0.98, P = 0.045). Other variables, including risk category, HCT-CI, MRD, female-to-male transplantation, and MUD transplantation, were not significantly associated with outcomes after adjustment (Table 3, Supplementary Figure S2). In sensitivity analyses, additionally adjusting for transplant center, the magnitude and direction of associations remained largely consistent across endpoints (Supplementary Table S3).
Multivariate analysis of risk factors for survival and transplant outcomes.

Exploratory analysis by TMI dose
TMI >9 Gy versus TMI ≤9 Gy
Exploratory analyses suggested a potential benefit of a higher TMI dose (>9 Gy). At 2 years, OS was 76.9% (66.9–88.4) versus 65.9% (56.1–77.4) (P = 0.111), and DFS was 67.2% (56.5–80.0) versus 50.5% (40.4–63.0) (P = 0.033) for >9 Gy and ≤9 Gy, respectively(Supplementary Figure S3). Relapse (20.1% (10.6–29.7) versus 35.2% (24.3–46.1), P = 0.058) showed a trend toward lower incidence in the >9 Gy group, while NRM (12.6% (3.9–21.4) versus 14.4% (7.1–21.6), P = 0.551), GRFS 39.3% (29.0–53.3) versus 35.8% (26.5–48.4), P = 0.458), aGVHD 38.3% (27.0–49.6) versus 22.7% (14.1–31.3), P = 0.030) and cGVHD outcomes were comparable. PSM was performed using major pretransplant clinical variables; the matched cohort comprised 43 versus 43 patients with all postmatch standardized mean differences <0.10 (Supplementary Figure S4A). Similar trends were observed (2-year OS 72.2% (55.8-93.4) versus 64.8% (51.3–81.8); DFS 61.6% (44.2-85.7) versus 50.7% (37.1–69.2), P = 0.070), although differences did not reach statistical significance. In multivariable and PSM-adjusted models, higher TMI dose was consistently associated with HRs below 1 for OS, DFS, relapse, and NRM, supporting the robustness of these findings despite limited statistical power (Supplementary Table S4).
TMI=12Gy versus TMI<12Gy
Exploratory analyses also evaluated the effect of receiving exactly 12 Gy versus lower TMI doses. Before matching, 2-year OS and DFS were higher with 12 Gy (78.2% (66.6–91.9) and 66.9% (54.1–82.7)) compared with <12 Gy (69.0% (60.6–78.5) and 55.5% (46.8–65.9)), although differences were not significant (P = 0.409 and P = 0.572) (Supplementary Figure S5). GRFS, CIR, and NRM were largely similar, but the 12 Gy group experienced a higher incidence of grade II–IV aGVHD at day 100 (45.4% vs. 23.9%, P = 0.008). After PSM (35 vs. 77 patients, all standardized mean differences <0.10) (Supplementary Figure S4B), similar trends were observed (2-year OS 81.6% vs. 73.1%, DFS 70.0% vs. 60.3%), though none reached statistical significance, and differences in GRFS, CIR, NRM, and GVHD outcomes remained nonsignificant. In multivariable models, TMI =12 Gy was associated with improved OS (HR 0.39, 95% CI 0.16–0.95, P = 0.038) and reduced NRM (HR 0.16, 95% CI 0.04–0.63, P = 0.009), albeit at the cost of higher grade II–IV aGVHD (HR 2.07, 95% CI 1.06–4.04, P = 0.034). These associations attenuated after PSM adjustment (OS HR 0.73, NRM HR 0.53, aGVHD HR 1.60; all P > 0.2). (Supplementary Table S5)
Adverse events
Severe toxicities within 100 days posttransplantation were relatively infrequent. The most common grade ≥3 adverse events were infections (21.46%), followed by vascular disorders(6.83%), gastrointestinal disorders (2.93%), nervous system disorders (2.44%), and renal/urinary disorders (2.44%). Other categories, including cardiac, hepatobiliary, respiratory, metabolic, and vascular disorders, each accounted for <2% of cases. Beyond severe events, early complications were common. Viral reactivation was frequent, with CMV activation in 44.88% and EBV activation in 23.90%; Epstein–Barr virus related posttransplant lymphoproliferative disorder (EBV-PTLD) developed in 3.90% and CMV disease in 2.93%. According to Bearman’s criteria, gastrointestinal (14.63%) and oral mucosal (16.10%) complications were notable. Late complications included endocrine and metabolic disorders (e.g. hypothyroidism 2.44%, lipid disorders 2.44%, osteoporosis 6.34%), and infertility (2.93%). Late-onset infections were observed in 15.12%, while neurologic and gastrointestinal late toxicities occurred in 5.85% and 3.41%, respectively. Most late effects were grades I–II (Supplementary Table S6). Among the 54 deaths recorded, the leading causes were primary disease relapses (27.78%), infections (25.93%), and GVHD (12.96%), followed by organ failure, hemorrhage, and subsequent malignancies (Supplementary Table S7).
Discussion
In this multicenter retrospective cohort, the 2-year OS, DFS, and GRFS were 71.7%, 60.0%, and 37.8%, respectively, with 1-year CIR and NRM of 20.2 and 13.8%. These outcomes indicate that TMI-based conditioning is clinically feasible within heterogeneous real-world transplant settings. When interpreted in the context of previously published international series, our results fall within the range of reported contemporary outcomes. For example, Haraldsson et al. 7 reported similar engraftment and survival in a prospective cohort evaluating TMI- and TBI-based approaches. In a haploidentical cohort, Sarina et al. 8 reported 2-year OS and PFS of 63% and 54% using low-dose TBI or TMLI, with no major differences in engraftment or GVHD endpoints. A Chinese single-center study reported a 2-year OS of 74.7% using hypo-fractionated TMLI, further supporting feasibility in acute leukemia and lymphoma 9 . In this context, the present multicenter cohort adds real-world data from a relatively large Chinese population, complementing prior single-center observations across diverse hematologic malignancies. In this multicenter cohort, TMI should be understood as a radiotherapy platform integrated within diverse conditioning backbones rather than a uniform, standalone intervention. The observed outcomes, therefore, reflect the composite effect of TMI delivery in combination with chemotherapy intensity, donor type, and GVHD prophylaxis strategies. As no contemporaneous TBI control group was included, these cross-study comparisons are descriptive and should not be interpreted as evidence of equivalence or superiority.
In our cohort, day-100 grade II–IV and III–IV aGVHD were 32.9 and 17.7%, and 1-year overall and moderate-to-severe cGVHD were 33.3 and 19.1%, respectively. Previous reports of TMI-based approaches using modern irradiation platforms have described early toxicity profiles and organ dose distributions in transplant populations7,10–12. The most common serious nonhematologic adverse event within 100 days was infection (21.5%), while gastrointestinal, neurologic, and renal toxicities each accounted for <3%. The observed CMV and EBV reactivation rates may partly reflect the high proportion of haploidentical transplantation and ATG use, both recognized risk contexts for delayed immune reconstitution13,14. However, clinically significant CMV disease and EBV-PTLD were infrequently observed. Prior studies have reported grade III–IV nonhematologic toxicities in TMI-treated cohorts. Potential biological differences between marrow-targeted irradiation and conventional TBI have been proposed, although such mechanisms were not evaluated in this study15–17. Long-term safety and rates of subsequent malignant neoplasms have been examined in prior reports, although follow-up durations remain limited 18 . Importantly, the risk of subsequent malignant neoplasms (SMN) appears comparable between TMI and TBI, despite higher target doses in TMI cohorts 19 . In our study, no SMNs were detected; however, the follow-up duration was limited. Overall, these findings describe short-term toxicity patterns in this cohort and highlight the need for longer follow-up to characterize chronic and delayed toxicities. In stratified analyses, outcomes varied across diagnostic subgroups. Patients with B-ALL in our cohort showed intermediate survival with lower NRM compared with AML. Prior studies have evaluated TMI/TMLI in ALL populations, including cohorts largely composed of B-ALL patients, reporting 2-year OS and DFS in the range of 70% to 80%20,21. However, differences in disease status, donor composition, and conditioning strategies limit direct comparison across studies. In our cohort, T-ALL patients demonstrated survival outcomes comparable to those observed in other ALL subtypes, without statistically significant differences 22 . In our cohort, AML patients (n = 15) showed a descriptive pattern of inferior outcomes compared with other diagnoses, with 2-year OS and DFS around 63% and higher NRM (29.3%). Given the small sample size and wide confidence intervals, AML-specific findings should be interpreted cautiously. Prior studies of TMI/TMLI in AML and other myeloid malignancies have reported heterogeneous outcomes across different conditioning intensities and transplant strategies23–25. Differences between series may be related to variation in remission status, donor type, and GVHD prophylaxis strategies rather than radiation modality alone. Accordingly, the AML-related observations in this study are descriptive and not powered for formal inference. Prior studies have suggested that pretransplant EMI is a predictor of post-HSCT extramedullary relapse independent of radiation modality 26 . Interestingly, EMI was not significantly associated with inferior survival in our cohort. EMI-positive patients showed a trend toward higher relapse but lower NRM, and in multivariable analysis, EMI was independently associated with reduced NRM. Given the limited sample size and potential residual confounding, these findings should be interpreted cautiously and warrant validation in larger cohorts. In multivariable analyses, donor type and performance status remained critical prognostic determinants. Haploidentical transplantation was associated with inferior OS compared with MSD, although this association should be interpreted cautiously given differences in GVHD prophylaxis strategies. Some registry studies in older AML/MDS populations have reported comparable findings 27 . Poor performance status (ECOG-PS >1) strongly predicted higher NRM and inferior OS, in line with established evidence on ECOG-PS and HCT-CI28,29. Collectively, these findings suggest that baseline disease biology, donor source, and patient fitness continue to exert dominant influence on transplant outcomes.
In our cohort, patients receiving TMI >9 Gy had higher 2-year DFS compared with those receiving ≤9 Gy (67.2% vs. 50.5%, P = 0.033), whereas OS did not differ significantly. In the 12 Gy subgroup, no statistically significant differences in 2-year OS or DFS were observed compared with <12 Gy, whereas grade II–IV aGVHD at day 100 occurred more frequently in the 12 Gy group (45.4% vs. 23.9%, P = 0.008). Multivariable models showed associations with lower mortality risk at 12 Gy, though these effects were attenuated after propensity matching. Taken together, these dose-stratified analyses are exploratory in nature. Given the nonrandomized design and the correlation between conditioning intensity, donor type, and GVHD prophylaxis strategy, residual confounding by indication cannot be excluded. Patients selected to receive higher TMI doses may have differed in baseline characteristics or treatment context. Therefore, these findings do not establish an optimal TMI dose or support dose escalation in routine practice. Biologically, higher TMI doses may plausibly enhance systemic inflammatory signaling and antigen-presenting cell activation, thereby intensifying donor T-cell responses. While such mechanisms are consistent with established models of radiation-induced immune modulation. The biological basis underlying this observation was not evaluated and remains speculative 30 . Prior studies have examined conditioning intensity and TMI/TMLI dose strategies18,20,31–33. However, differences in patient selection and transplant strategies limit direct comparison with our real-world cohort. Taken together, these data do not establish an optimal TMI dose, and prospective studies are required before any conclusions regarding dose escalation can be drawn. Prospective studies are required to more clearly define the role of dose intensity in marrow-targeted irradiation.
Nonetheless, several limitations should be acknowledged. First, as a retrospective single-arm study, inherent selection bias and residual confounding cannot be excluded, and causal inference remains limited. Although we applied PSM to mitigate baseline imbalances, this approach only accounts for observed covariates and is subject to loss of sample size and residual unmeasured confounding. Second, heterogeneity across centers existed in radiotherapy platforms, conditioning details, GVHD prophylaxis strategies, and supportive care, which may have influenced outcomes. Given the limited sample size per center, models were not stratified by center nor clustered by center, thus residual inter-center heterogeneity may have affected variance estimates. Third, the lack of a contemporaneous TBI- or chemotherapy-based control group prevents direct comparison of relative efficacy. Fourth, although the total sample exceeded 200 patients, some subgroups (e.g. AML, extramedullary involvement, MUD transplantation) were small, resulting in wide confidence intervals and limited robustness of subgroup findings. Fifth, the median follow-up of 19.3 months is insufficient to fully capture late toxicities such as endocrine, metabolic, pulmonary complications, and secondary malignancies. Detailed patient-level dosimetric parameters for organs at risk were not uniformly available in this retrospective multicenter dataset and were therefore not analyzed. Finally, mechanistic endpoints such as immune reconstitution and biomarker profiling were not systematically assessed, limiting biological interpretation. Prospective studies with longer follow-up are required to validate these findings and more clearly define the role of TMI within evolving conditioning strategies.
Conclusion
In this multicenter real-world cohort, TMI-based conditioning for allogeneic hematopoietic stem cell transplantation was feasible across heterogeneous clinical settings and achieved survival outcomes consistent with contemporary practice. Baseline disease characteristics, donor type, and patient performance status remained the primary determinants of outcome. Exploratory dose analyses suggested potential associations between higher TMI dose and survival outcomes, but did not define an optimal dose. Prospective studies with standardized protocols and longer follow-up are warranted to further clarify the role of TMI in conditioning strategies.
Supplemental Material
sj-docx-1-cll-10.1177_09636897261440297 – Supplemental material for Total marrow irradiation-based conditioning for allogeneic hematopoietic stem cell transplantation in patients with hematologic malignancies: A multicenter real-world study
Supplemental material, sj-docx-1-cll-10.1177_09636897261440297 for Total marrow irradiation-based conditioning for allogeneic hematopoietic stem cell transplantation in patients with hematologic malignancies: A multicenter real-world study by Yi Zhang, Ran Zhang, Xiena Cao, Yang Yang, Weiwei Jin, Yu Chen, Liling Zhang, Hui Zeng, Ruowen Wei, Ao Zhang, Hongru Chen, Li Wang, Yang Cao, Weijie Cao, Linghui Xia and Wei Shi in Cell Transplantation
Footnotes
Acknowledgements
Not applicable.
Ethical Considerations
This study was conducted in accordance with the Declaration of Helsinki and was approved by the Ethics Committee of Union Hospital, Tongji Medical College, Huazhong University of Science and Technology (approval No. 2025-1040, approved on October 13, 2025). Given the retrospective design, the requirement for informed consent was waived.
Author Contributions
Yi Zhang designed the study, performed statistical analyses, interpreted results, and drafted the manuscript. Ran Zhang coordinated multicenter data collection and contributed to study design and manuscript revision. Xiena Cao and Yang Yang collected and verified data. Weiwei Jin, Yu Chen, and Liling Zhang interpreted data and assisted with manuscript editing. Hui Zeng and Ruowen Wei supervised data acquisition and management. Ao Zhang and Hongru Chen conducted the literature review, statistical verification, and figure preparation. Li Wang provided clinical expertise and supervised the study. Yang Cao and Weijie Cao contributed to the study conception, supervised analyses, and interpreted results. Linghui Xia and Wei Shi supervised the overall project, critically revised the manuscript, and approved the final version.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China [82370220]. Hubei Provincial Natural Science Foundation of China [2025AFD499, 2025AFB519]. Medical Youth Top Talent Program of Hubei Province of China [Chen Yu]. Research Grant of Key Laboratory of Molecular Biological Targeted Therapies of the Ministry of Education (Huazhong University of Science and Technology) [2024SWBS002].
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statements
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
