Abstract
As oxygen carriers, perfluorocarbon emulsions might be useful to decrease hypoxia of pancreatic islets before transplantation. However, their hydrophobicity prevents their homogenisation in culture medium. To increase the surface of contact between islets and Perfluorooctyl bromide (PFOB), and consequently oxygen delivery, we tested effect of a PFOB emulsion in culture medium on β-cell lines and rat pancreatic islets. RINm5F β-cell line or pancreatic rat islets were incubated for 3 days in the presence of PFOB emulsion in media (3.5% w/v). Preoxygenation of the medium was performed before culture. Cell viability was assessed by apoptotic markers (Bax and Bcl-2) and by staining (fluoresceine diacetate and propidium iodide). β-Cell functionality was determined by insulin release during a glucose stimulation test and. Hypoxia markers, HIF-1α and VEGF, were studied at days 1 and 3 using RT-PCR, Western blotting, and ELISA. PFOB emulsions preserved viability and functionality of RINm5F cells with a decrease of HIF-1α and VEGF expression. Islets viability was preserved during 3 days of culture. Secretion of VEGF was higher in untreated control (0.09 ± 0.041 μg VEGF/mg total protein) than in PFOB emulsion incubated islets (0.02 ± 0.19 μg VEGF/mg total protein, n = 4, p < 0.05) at day 1. At day 3, VEGF secretion was increased as compared to day 1 in control (0.23 ± 0.04 μg VEGF/mg total protein) but it was imbalance by the presence of PFOB emulsion (0.09 ± 0.03 μg VEGF/mg total protein, n = 5, p < 0.05). While insulin secretion was maintained in response to a glucose stimulation test until day 3 when islets were incubated in the presence of PFOB emulsion preoxygenated (0.81 ± 0.16 at day 1 vs. 0.75 ± 0.24 at day 3), the ability to secrete insulin in the presence of high glucose concentration was lost in islets controls (0.51 ± 0.18 at day 1 vs. 0.21 ± 0.13 at day 3). Atmospheric oxygen delivery by PFOB emulsion might be sufficient to decrease islets hypoxia. However, to improve islets functionality, overoxygenation is needed. Finally, maintenance of islet viability and functionality for several days after isolation could improve the outcome of islets transplantation.
Introduction
The success of clinical islet transplantation for the cure of patients with type 1 diabetes is limited by the maintenance of a sufficient number of viable islets in vitro before transplantation. Indeed, islets in the pancreas are surrounded by an elaborate network of capillary vessels and connective tissue needed to satisfy their high metabolism. When islets are removed from their native environment, the islets' microvascular network is destroyed and their viability decreases dramatically (12). Although islets in vivo maintain a relatively constant O2 partial pressure throughout their diameter, those cultured in standard conditions are exposed to sharp oxygen partial pressure (pO2) gradients that may range from surface normoxia (142 mmHg) (11) to central anoxia (0 mmHg) (31). Thus, standard culture practice is not favorable for long-term islet survival and function (9,11,42). To minimize hypoxia and thereby reduce the number of required donors for each recipient, interest has been focused on the supply of oxygen during islets isolation and after transplantation using oxygen carriers (6,7,15,23,29). Perfluorocarbons (PFCs) are inert organic compounds in which the hydrogen atoms are replaced by fluorine atoms. This unique feature gives PFCs an unmatched ability to dissolve and transfer molecular oxygen (20,34,35). The solubility of O2 in PFCs is approximately 20 times higher than that of aqueous culture media (5). Furthermore, their O2 diffusivity is also 2.5-fold that in water or culture media (25,32). Because of physical dissolution, rather than chemical binding, and high diffusion rates related to low intermolecular cohesion forces in liquid PFCs, the partial pressure of O2 in PFC emulsions is in equilibrium with that in the surrounding media (21,34). Contrary to hemoglobin, the oxygen content of the emulsion and its partial pressure are linearly related.
Previous studies have already demonstrated the efficacy of the two-layer method that uses a neat oxygenated liquid PFC for pancreas preservation (1,2,26,29,30, 33,40,41,44). However, the use of PFC for islet preservation before transplantation is poorly described (4,41). Moreover, the use of the two-layer method for islet preservation in vitro seems to be deleterious for islet survival and function (4). This side effect of neat PFCs on pancreatic islets in vitro could be attributed to the hydrophobicity of PFC, which prevents mixture with aqueous media (20). Our interest has therefore been focused on the use of emulsified perfluorooctyl bromide (PFOB) in which submicronic droplets of perfluorocarbons are coated with a monolayer of phospholipids and further stabilized by a fluorocarbon/hydrocarbon diblock cosurfactant. Similar tailor-made, diblock-stabilized emulsions have recently been used in other studies where tissue oxygenation was needed (2,13). A PFOB emulsions has reached phase III clinical trials (16).
In a previous study, we examined the toxicity of PFOB emulsions on β-cells. Not only did PFOB emulsions exhibit an absolute inocuity, but they also proved capable of preventing the adhesion and enhance desorption of β-cells coming from the mouse and rat insulinoma cell lines—MIN-6 or RINm5F—from tissue culture petri dishes (36). Here, we have applied the concept of cell oxygenation using a PFC emulsion and evaluated its ability to decrease hypoxia in pseudoislets and in rat pancreatic islets.
Materials and Methods
PFOB Emulsions: Preparation and Characterization
A 60% w/v concentrated PFOB emulsion was prepared as previously described (36): egg yolk phospholipids (7.0 g, Lipoid E-80, Lipoid GmbH, Ludwigshafen, Germany) were dispersed in water (85.2 ml, deionized, Millipore, Molsheim, France) using a low-energy mixer (Ultra-Turrax T-25, Ika-Labortechnik, Staufen, Germany, 8000 rpm, 5 min). Then C6F13C10H21 (4.5 g) was added drop wise under stirring. Perfluorooctyl bromide (105 g, C8F17Br, Alliance Pharmaceuticals Corp. San Diego, CA, USA) was added gradually (8000 rpm, 8 min). After further homogenization at ~17,000 rpm during 30 min, the mixture was then submitted to the action of a microfluidizer (model 110T, Microfluidics Corp., Newton, MA, USA, 10,000 psi) for 40 min. The resulting emulsion was sampled into aliquots of 10 ml, sealed under nitrogen, and sterilized by static autoclaving (121°C, 15 min, 15 psi). The pH was 7.2 and the osmolarity 34 mOsm. The average particle size (80 nm + 5%) and particle size distribution histograms of the emulsion droplets were measured by a centrifugal sedimentation method coupled with light absorption measurements (Horiba Capa-700, Horiba Ltd., Kyoto, Japan). The oxygen dissolution capacity of this emulsion is ~16 vol.% in pure oxygen at atmospheric pressure.
The PFOB emulsion was diluted with an equivalent volume (1:1 dilution) of twofold concentrated cell culture medium (either RPMI-1640 or M199) containing 20% fetal calf serum (FCS, Eurobio, Les Ulis, France) and 2% of a combination of antibiotic and antimicotic agents (AB/AM, Invitrogen, Carlsbad, CA, USA): RPMI-1640 was used for RINm5F and M199 for islets (Lonza, Basel, Switzerland). These medium-diluted emulsions used for incubation were called PFOB working solutions.
Cell Culture
The rat β-cell line RINm5F (LGC-Promochem-ATCC, Molsheim, France) has been used for its capacity to form pseudoislets. RINm5F cells were maintained in culture with RPMI-1640 medium (Sigma, Steinheim, Allemagne) supplemented with 10% FCS and 1% AB/AM at 37°C in humidified 5% CO2/95% atmospheric air. RINm5F were cultured on six-well plates in 2 ml medium containing 10% of PFOB working solution for 1 and 3 days (PFOB emulsion). As controls, pseudoislets were formed in 2 ml medium on untreated plates (hydrophobic for cell suspension culture) (Greiner, Frickenhausen, Germany) in RPMI-1640, 10% FCS, 1% AB/AM. The atmosphere used for the comparison of RPMI-1640 with or without PFC contained 20% oxygen.
Assessment of RINm5F Number
Cells (30,000) were seeded in 96-well plates (Becton Dickinson-Falcon) in 200 μl of RPMI-1640 for 24 h. The medium was then removed and replaced by RPMI-1640 containing different concentrations of PFOB emulsion. The number of cells was assessed using Cell Titer® (Promega, Madison, WI, USA). After several rinses in medium, 20 μl of Cell Titer and 100 μl of RPMI-1640 were added to each well. Cells were incubated for 2 h at 37°C. The absorbance at 490 nm (OD 490) measures the quantity of formazan (products of the reduction of tetrazolium salts by dehydrogenases and reductases, produced by healthy cells) produced from the MTS-based assay and is directly proportional to the number of adherent cells. The relative number of adherent cells was calculated as the ratio between the optical density obtained in the presence of PFC emulsions and the optical density obtained in control conditions (i.e., with pure RPMI-1640) (ODPFOB/ODcontrol %).
Islet Isolation
Pancreatic islets were isolated from Wistar rats (250–300 g) (Depré, Saint Doulchard, France) by collagenase (Sigma) digestion as previously described (22). Two hundred isolated islets were cultured at 37°C in a humidified environment of 95% O2 and 5% CO2 for the experimentation. These studies were performed according to our institutional animal care policies.
Islet Culture
Islets were incubated in four different media. The control medium was composed of M199 supplemented with 10% FCS and 1% AB/AM. PFOB emulsion medium was composed of M199 supplemented with 10% of PFOB working solution, 10% FCS, and 1% AB/AM (atmosphere composition: 5% CO2 and 20% O2). Prior to setting up the culture, medium with or without PFOB was pregassed for 30 min with 99.5% medical oxygen.
The pO2, pCO2, and pH in cell-free media were measured prior to experiments using a blood gas analyzer (Radiometer ABL 700 Series, Copenhagen, Denmark) in order to determine baseline parameters.
Assessment of Islet Viability Using FDA/PI
Cell viability was determined using fluorescein diacetate (FDA, Sigma) and propidium iodide (PI, Sigma). Ten islets of each condition were randomly selected by two independent investigators. The ratio between green cells and red cells gives a percentage of viability.
Stimulation Test and Insulin Quantification
Insulin secretion was evaluated by static incubation of RINm5F pseudoislets and rat pancreatic islets. Briefly, pseudoislets and islets were incubated in Krebs buffer containing 3 mmol/L glucose for 1 h (basal secretion) and they were incubated in 27.5 mmol/L glucose Krebs solution for 1 h (stimulated secretion). Insulin concentration was measured using a rat insulin ELISA kit (Mercodia, Uppsala, Sweden). Concerning the rat islets, a stimulation index was calculated as the ratio of stimulated insulin secretion to basal insulin secretion.
Reverse Transcriptase-Coupled Polymerase Chain Reaction
By days 1 and 3, RNA isolation from pseudoislets or islets was performed using a Trizol kit (Invitrogen). Reverse transcriptase-coupled polymerase chain reaction (RT-PCR) was used to evaluate the mRNA expression of hypoxia inducible factor (HIF-1α; 5′-tcaagtcagcaacgt ggaag-3′, 3-‘atcgaggctgtgtcgactg-5′), vascular endothelial growth factor (VEGF; 5′-caatgatgaagccctggagt-3′, 3′-tttcttgcgctttcgttttt-5′), B cell lymphoma-2 (Bcl-2; 5′-atacctgggccacaagtgag-3′, 3′-tgatttgaccatttgcctga-5′), and Bcl-2 associated X protein (Bax; 5′-tgcagaggatgattgct gac-3′, 3′-gatcagctcgggcactttag-5′) in pseudoislets, and Bax and Bcl-2 in pancreatic islets. The RT-PCR amplifications were performed according to the manufacturer's instructions (Invitrogen). RT-PCR products were analyzed by gel electrophoresis in 1.5% agarose-1000 (Euromedex, Mundolsheim, France). The amount of cDNA was normalized on the basis of the signal obtained from constitutively expressed β-actin mRNA (5′-aacactggcattgtcatgga-3′, 3′-tacatggcggtaccaacaga-5′). Gel images were captured with a BioRad Gel Doc 1000 system and analyzed by ImageJ.
Vascular Endothelial Growth Factor (VEGF) Quantification
Five islets were incubated in the presence of 10% PFOB emulsion for 1 and 3 days. VEGF was quantified in conditioned media using the RayBiotech Rat VEGF ELISA kit (Norcross, GA, USA), according to the manufacturer's protocol. Islets were lysed in cell lysis buffer [20 mmol/L Tris, pH 8, 137 mmol/L NaCl (Sigma), 31 mmol/L phenylmethylsulfonyl fluoride (Sigma), 10% glycerol, 1% IgPal (Sigma)]. Lysate protein concentration was measured by the Biorad DC protein assay. Results were expressed as the ratio between VEGF secreted (pg) and total protein (μg).
Immunohistochemistry
After 1 and 3 days of culture, rat islets were fixed in formalin 4% (Sigma) for 30 min and embedded in paraffin. Sections were incubated with anti-VEGF 1/200 (Abcam, Paris, France) for 1 h. After washes in PBS, sections were incubated with biotinylated anti-mouse IgG (1/100) for 30 min following by incubation with Extravidin-peroxidase® for 30 min. Detection was performed with 3-amino-9-ethyl-carbazole (AEC) chromogen substrate solution for horseradish peroxydase. The section were counterstained with Mayer's hematoxylin (Surgipath®, Cambridgeshire, UK) and mounted on glass slides. Control sections were treated with PBS + 1% BSA to determine the specificity of the staining.
Statistical Analysis
Statistical analysis was performed using SigmaStat 3.10 (Systat Software, San Jose, CA, USA). All values were expressed as means + SD. Statistical comparison was performed by ANOVA (comparison between basal/stimulated, control/PFOB emulsion, oxygenated control/oxygenated PFOB emulsion) with a post hoc Bonferroni correction in different groups. Significance was expressed as p < 0.05.
Results
Physical Parameters of Culture Media
All manipulations were performed at room temperature in normal atmospheric air. We felt it was important to measure physical parameters of the culture media in addition to oxygenation; saturation of the medium with O2 displaces CO2, a principal component of the buffering system in the media used in our experiments, thus affecting acid–base balance.
Before oxygenation, pH in the control medium was 7.44 and pO2 was 25.6 kPa; in PFOB emulsion-supplemented medium pH was slightly lower at 7.33 and the pO2 was comparable with the control medium at 25.9 kPa.
Following oxygenation, pH in the control medium rose to 8.31 and the pO2 increased to 114 kPa. In the medium supplemented with PFOB emulsion pH also increased following oxygenation to reach 8.07 and pO2 rose to 112 kPa.
The acid–base balance of culture media returned to normal values within 10 min of placing the test tubes in the cell culture incubator (21% O2, 5% CO2, 37°C).
Partial pressure of oxygen remained high in both hyperoxygenated control medium and PFOB emulsion-supplemented medium for about 15 min and then dropped to 50% of its initial value within 60 min and returned to preoxygenation values within 120 min.
Quantification of β-Cell Number in the Presence of PFOB Emulsion in Culture Medium
The number of cells observed in PFOB emulsion supplemented medium was similar to the number in standard condition. Indeed, the ratio was very close to 100% either at day 1 or day 3 (Fig. 1).

Influence of perfluorooctyl bromide (PFOB) emulsion on the β-cells number. Quantification of RINm5f β-cells number in the presence of PFOB emulsion compared to standard condition using Cell Titer®. Results were expressed as the ratio between the OD in PFOB emulsion supplemented medium and the OD in standard condition (n = 6).
Influence of the PFOB Emulsion on β-Cell Functionality
At day 1 (Fig. 2), pseudoislets cultured in the PFOB emulsion-supplemented medium secreted more insulin than the control islets to stimulation by 3.3 mmol/L glucose (13.4 ± 1.9 vs. 7.1 ± 1.7 μg insulin/mg total protein, respectively, n = 6, p < 0.05). In response to stimulation by 27.5 mmol/L glucose, no difference between the two culture conditions was observed (14.4 = 6.1 vs. 8.8 = 1.8 μg insulin/mg total protein, respectively, n = 6). At this step of the study, the stimulation index was very low (around 1.1). At day 3, no significant difference of insulin secretion was observed between controls and pseudoislets in the presence of PFOB emulsion. Indeed, at 27.5 mmol/L glucose, the latter secreted 32.5 = 7.7 μg insulin/mg total proteins compared to the former 23.3 = 10.3 μg of insulin/mg total protein for controls. There was a significant increase of insulin secretion in all conditions (e.g., in control 3.3 mmol/L glucose, 15.1 = 2.7 μg of insulin/mg total protein at day 3 vs. 7.1 = 1.7 μg of insulin/mg total protein at day 1, n = 6, p < 0.05) except for 3.3 mmol/L glucose (10.9 = 5.5 μg of insulin/mg total protein vs. 13.4 = 1.9 μg of insulin/mg total protein, n = 6) compared to day 1. There was a significant difference in insulin secretion between 3.3 and 27.5 mmol/L glucose by pseudoislets in the presence of the PFOB emulsions (10.9 = 5.5 vs. 32.5 = 7.6 μg insulin/mg total protein, n = 6, p < 0.05). At the opposite no significant difference was observed between the insulin secretion in the presence of low- and high-glucose concentration in control pseudoislets (15.1 = 2.7 vs. 23.3 = 10.3 g insulin/mg total protein, n = 6).

Influence of PFOB emulsions on β-cell functionality. Effect of culture on glucose-stimulated insulin release of pseudoislets in the presence or not of PFOB emulsion at 1 and 3 days of culture. Results are expressed as means of insulin secretion toward total protein quantity (SD of six independent experiments). $$p < 0.05, 27.5 mmol/L glucose versus 3.3 mmol/L glucose; *,**p < 0.05, 3.3 mmol/L glucose versus control 3.3 mmol/L glucose at day 1; #,###p < 0.05, 27.5 mmol/L glucose versus control day 1 at 27.5 mmol/L glucose.
Influence of the PFOB Emulsion on β-Cell Hypoxia
Hypoxia markers were analyzed in order to assess the impact of PFOB emulsions as oxygen carriers (Fig. 3A). In pseudoislets incubated in the PFOB emulsion-supplemented medium, HIF-1α mRNA expression remained constant during the experimentation (0.49 ± 0.09 at day 1 and 0.64 ± 0.19 at day 3, n = 6). At the opposite, HIF-1α protein content significantly increased in control pseudoislets between day 1 and day 3 (0.52 ± 0.09 vs. 1.03 ± 0.07, respectively, n = 6, p < 0.001). HIF-1α mRNA expression is significantly reduced in pseudoislets cultured in PFOB emulsion-supplemented medium compared to controls (0.64 ± 0.19 vs. 1.03 ± 0.07, n = 6, p < 0.05).

Influence of PFOB emulsion on β-cell hypoxia. (A) Study of hypoxia-inducible factor (HIF)-1α protein expression by Western blot during 1 and 3 days after the beginning of experiment. (B) Study of vascular endothelial growth factor (VEGF) mRNA expression by pseudoislets in the presence or absence of PFOB emulsions during the experiment was performed by semiquantitative RT-PCR. Bands were quantified and results expressed toward β-actin expression and reported in the histogram. Data are expressed as means ± SD of six independent experiments. ***p < 0.05, control day 3 versus control day 1; ##p < 0.05, PFOB emulsion versus control day 3.
VEGF mRNA levels, used as hypoxia marker as well, were similar in control pseudoislets and in those cultured in PFOB emulsion-supplemented medium at day 1 (0.68 ± 0.09 vs. 0.75 ± 0.07, n = 3) (Fig. 3B). At day 3, a significant decrease of VEGF mRNA was observed in pseudoislets cultured in PFOB emulsion-supplemented medium compared to controls (1.15 ± 0.30 vs. 0.66 ± 0.17, n = 5, p < 0.01).
Influence of PFOB Emulsion and Oxygen on Pancreatic Islet Viability
We performed FDA/PI tests on rat islets cultured for 1 and 3 days in four different conditions (control, PFOB emulsion, oxygenated control, oxygenated PFOB emulsion) (Fig. 4A). There was no difference between the various culture conditions after 1 day of culture. The viability was maintained at 80%. The same observation was made after 3 days of culture. The number of islets remained constant throughout the experimentation (data not shown). The study of pro/antiapoptotic markers was then studied (Fig. 4B). The charts represent the ratio between Bcl2 mRNA expression and Bax mRNA expression. Consequently a ratio above 1 means that the survival pathways are more likely activated in these cells. At day 1 (Fig. 4B), in medium without overoxygenation the ratio was under 1 (control: 0.75 ± 0.20, PFOB emulsion: 0.71 ± 0.17). The addition of oxygen to the medium changes the trend. Islets cultured in PFOB emulsion-supplemented medium exhibited a ratio of 1.03 ± 0.28, slightly above controls, even though the pH was above 8 (0.75 ± 0.20). At the opposite the overoxygenation of medium alone slightly decreased the ratio compared to control (0.51 ± 0.10 vs. 0.75 ± 0.20, n = 5). After 3 days of culture (Fig. 4C), control islets and islets cultured in oxygenated PFOB emulsion-supplemented medium showed a ratio around 2 (controls: 2.19 ± 0.71 and oxygenated PFOB emulsion: 1.72 ± 0.9, n = 3). A slightly decrease of the ratio was observed in islets cultured in PFOB emulsion-supplemented medium (1.05 ± 0.40, n = 3). Oxygenated control exhibited a reduction trend of the ratio (0.41 ± 0.14, n = 3).

Influence of PFOB emulsion and oxygen on islet cells viability. (A) Viability of islets after 1 and 3 days of culture incubated in control conditions or with PFOB emulsion, oxygenated or not. Islets stained with fluorescein diacetate/propidium iodide (FDA/PI). Scale bars: 50 μm. Percentage of viability of islets was represented in function of time and culture medium. The graph represents mean from six independent isolations and experiments.(B) B cell lymphoma-2 (Bcl-2) and Bcl-2-asociated X protein (Bax) mRNA expression using semiquantitative RT-PCR in islets after 1 and 3 days of culture in control conditions or with PFOB emulsion, oxygenated or not. β-Actin was used as the housekeeping gene.
Influence of PFOB Emulsions and Oxygen on Islet Functionality
Figure 5 shows the results obtained on islet functionality. In basal conditions, insulin secretion was comparable in controls (0.18 ± 0.04 μg insulin/L), in the presence of PFOB emulsions (0.31 ± 0.09 μg insulin/L), in oxygenated medium (0.23 ± 0.08 μg insulin/L), and in oxygenated PFOB emulsion-supplemented medium (0.21 ± 0.03 μg insulin/L, n = 5).

Influence of PFOB emulsions and oxygen on islet function. Glucose stimulation test was performed on islets after 1 (A) and 3 days (B) of culture after incubation in control or PFOB emulsion with or without preoxygenation. Results were expressed as mean ± SD of insulin secreted by five islets from three independent experiments. *p < 0.05, 27.5 mmol/L glucose versus 3.3 mmol/L glucose, n = 5. (C) Stimulation index of islets after 1 and 3 days of culture in the four different media. ##p < 0.05 culture condition versus control day 1; *p < 0.05 culture conditions versus control day 3, n = 6.
In the stimulated condition (27.5 mmol/L glucose), insulin secretion was comparable in all the conditions tested (control: 0.51 ± 0.18 μg insulin/L; PFOB emulsion: 0.53 ± 0.14 μg insulin/L; control O2: 0.56 ± 0.17 μg insulin/L; PFOB emulsion + O2, 0.81 ± 0.16 μg insulin/L, n = 5, ns) and significantly higher than the basal condition (3.3 mmol/L glucose, n = 5, p < 0.05) (Fig. 5A).
At day 3 (Fig. 5B), no significant difference between the four conditions was measured on insulin secretion in basal condition (3.3 mmol/L glucose): around 0.2 μg insulin/L was measured. At 27.5 mmol/L of glucose, a loss of ability to respond to a high-glucose concentration by insulin secretion was observed in control conditions (control: 0.21 ± 0.13 μg insulin/L; control + O2: 0.18 ± 0.04 μg insulin/L). The ability to respond to a high-glucose concentration was maintained after 3 days of culture in islets cultured in the presence of PFOB emulsion independently of the oxygenation (PFOB emulsion: 0.41 ± 0.16 μg insulin/L, PFOB emulsion + O2: 0.75 ± 0.24 μg insulin/L). Insulin secretion was maintained compared to day 1 in the PFOB emulsion-supplemented medium.
A decrease in secretion was observed in islets cultured in M199 control, independently of oxygenation compared to 1 day (control: 0.21 ± 0.13 μg insulin/L at day 3 vs. 0.51 ± 0.18 μg insulin/L at day 1; oxygenated control: 0.18 ± 0.04 μg insulin/L at day 3 vs. 0.56 ± 0.17 μg insulin/L at day 1). In the presence of PFOB emulsion, insulin secretion is maintained throughout the experiment (PFOB emulsion at day 3: 0.41 ± 0.16 μg insulin/L vs. day 1: 0.53 ± 0.14 μg insulin/L; PFOB emulsion + O2: 0.75 ± 0.24 μg insulin/L vs. day 1 0.81 ± 0.16 μg insulin/L).
In accordance with these results, the analysis of stimulation index (Fig. 5C) showed that pancreatic islets were functional after 1 day of culture in controls, in the presence of PFOB emulsions, and in the presence of oxygenated medium with a stimulation index of 1.94 ± 0.4, 2.00 ± 0.3, and 1.79 ± 0.3, respectively. Islets incubated in the presence of oxygenated PFOB emulsions exhibited a significant increase of the stimulation index at day 1 (4.3 ± 1.2, n = 6, p < 0.01) compared to control conditions. The stimulation index was maintained after 3 days of culture at the level of the one observed at day 1 in islets cultured in oxygenated PFOB emulsion-supplemented medium (3.6 ± 0.7). The culture of islets with oxygenated control seems to be deleterious for islet functionality because the stimulation index was lower after 3 days of culture (1.7 ± 0.3 and 1.0 ± 0.1, respectively).
Influence of PFOB Emulsion and Oxygen on VEGF Expression in Islets
The measure of secretion of VEGF (Fig. 6) showed a comparable expression between islets cultured in the control medium (0.09 ± 0.04 μg VEGF/mg total protein) and islets in the oxygenated control medium (0.10 ± 0.0.06 μg VEGF/mg total protein). In contrast, a significant decrease in VEGF secretion was observed in the presence of PFOB emulsion (0.02 ± 0.02 μg VEGF/mg total protein, n = 4, p < 0.05). While VEGF secretion was increased in all the conditions tested at day 3 (n = 4, p < 0.05), this increase was significantly reduced in islets cultured in the PFOB emulsion-supplemented media with or without oxygen (0.10 ± 0.08 μg vs. 0.09 ± 0.03 μg VEGF/mg total protein, respectively) compared to controls (0.23 ± 0.04 μg VEGF/mg total protein, n = 4, p < 0.05). Finally, the oxygenation of M199 significantly increased the VEGF secretion of the pancreatic islets compared to controls (0.9 ± 0.1 μg VEGF/mg total protein vs. 0.33 ± 0.16 μg VEGF/mg total protein, n = 6, p < 0.001).
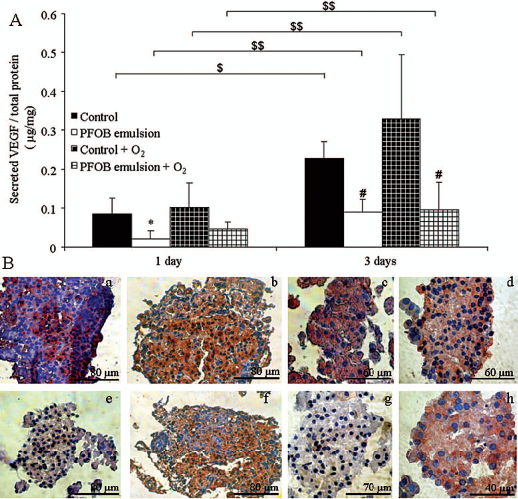
Influence of PFOB emulsion and oxygen on VEGF expression in islets. (A) VEGF secretion of rat pancreatic islets incubated in control conditions or with PFOB emulsion, oxygenated or not, after 1 and 3 days of culture. Results are expressed as mean of the ratio between secreted VEGF toward total protein content from four independent experiments. *p < 0.05, culture conditions versus control day 1; # p < 0.05, culture conditions versus control day 3; $,$$p < 0.05, day 3 versus day 1. (B) Staining of VEGF (red) inside islets incubated in control conditions (a, c, e, g) or with PFOB emulsion (b, d, f, h), oxygenated (c, d, g, h) or not (a, b, e, f), after 1 (a, b, c, d) and 3 days (e, f, g, h) of culture (n = 6).
Immunohistochemistry analysis of VEGF expression (Fig. 6B) showed a strong red staining of pancreatic islets at day 1 of culture in all the conditions tested with a comparable dyeing. After 3 days of culture, no VEGF staining was observed in control or oxygenated control islets. However, in the presence of PFOB emulsion independently of oxygenation, the staining seemed to be comparable to that seen on day 1.
Discussion
With the recent advances in islet isolation techniques and the introduction of more potent and less diabetogeneic immunosuppressive therapies, islet transplantation has progressed from research to clinical reality (38). Many transplant protocols incorporate a period of short-term (24–72 h) islet culture before transplantation to the recipient in order to facilitate recipient preparation and provide time for assessment of islet quality. Short-term islet culture indeed has some benefits, such a purification of islet preparation and immunomodulation (18), and hence may improve allograft survival. However, cultured islets are known to degrade rapidly (8,19) and lose viability and functional responsiveness to glucose stimulation with the extension of culturing time (10,37).
We studied effects of supplementation of culture media with PFOB emulsion on viability, apoptosis, stimulated insulin secretion, and expression of hypoxia markers in pseudoislets and isolated rat islets. Pseudoislets are three-dimentional clusters of β-cells secreting insulin in response to glucose stimulation in a similar manner as islets (14,24). Once they are fully developed and functional, pseudoislets might be prone to hypoxia during culture to a similar extent as isolated pancreatic islets. We used pseudoislets to simplify studied system by eliminating interaction of various cell types in culture to study response of only a β-cell fraction of the islets to different conditions.
We demonstrated that presence of PFOB emulsion in the medium improves function of pseudoislets cultured for 3 days compared with pseudoislets cultured in standard conditions. We also found that expression of HIF-1α and VEGF increased significantly in pseudoislets during this period of culture in standard conditions, whereas it remained stable in pseudoislets cultured in PFOB supplemented medium.
Culture of isolated rat islets in standard conditions for 72 h resulted in decreased response to glucose stimulation and reduced viability. This potentially reflects relative hypoxia, when oxygen demand exceeds speed of its diffusion resulting in decreased islet viability (9,12) and function (11,23,42). Supplementation of the culture medium with PFOB preserved secretory function of cultured islets. Although we have not provided direct evidence of better diffusion of oxygen to islets' core, a lack of increase in VEGF expression in islets cultured in nonhyperoxygenated PFOB emulsion with concomitant preservation of their viability and secretory function may suggest that perfluorocarbon emulsion facilitates diffusion of oxygen to the islets even at normal oxygen partial pressures, as demonstrated previously (17). However, we cannot exclude involvement of a completely different mechanism, which is not related to oxygen delivery.
Surprisingly, preoxygenation of the culture medium had a detrimental effect on islets. This manifested itself as significantly decreased stimulation index, with exceptionally abnormal insulin secretion profile, and increased expression of proapoptotic factors after 3-day culture. Although oxygen partial pressure in the culture medium was elevated for only about 60–120 min, as estimated in our initial experiments without the cells, it is likely that preoxygenation resulted in oxygen toxicity and release of free radicals that islets are not well equipped to deal with (28). Interestingly, expression of VEGF in these islets was also elevated. This has to be interpreted with caution as we measured levels of VEGF protein rather than mRNA levels (39). VEGF is a potent stimulant of angiogenesis and its expression is regulated at different levels in the context of hypoxia rather than hyperoxia. It is stored in extracellular matrix bound to glycosaminoglycan (GAG)-heparan sulphate. It is possible that free radicals formed during brief exposure to oxygen in high concentrations could disrupt GAGs and release VEGF to the culture medium (27,39,43). There is evidence showing that fluctuation in pO2 from hyperoxia to hypoxia triggers disproportionately higher increase in VEGF expression than change from normoxia to hypoxia (27,43). This could explain why in our experiments we observed much higher levels of VEGF in a group with preoxygenated medium compared with control.
As discussed above, observed effects of preoxygenation were most likely related to oxygen toxicity. They were markedly prevented by addition of PFOB emulsion. We observed a significant improvement in secretory function in this group, which was associated with decrease in VEGF expression, compared with preoxygenated medium without PFOB. It was suggested previously that perfluorocarbons might act as a sump for toxic metabolites or indeed free radical scavengers (3). Nevertheless, this mechanism cannot on its own fully explain why the presence of PFOB in the hyperoxic environment improved islet secretory function. It is therefore possible that compartmentalization of oxygen in the PFOB emulsion could prevent exposure of islets to excessive oxygen concentrations and thus reduce formation of free radicals and deliver the benefit of improved oxygenation during recovery period following islet isolation.
In conclusion, our results showed that PFC emulsions can reduce hypoxia, cell adhesion, and apoptosis. They did not need to be oxygenated in order to decrease hypoxia in pancreatic islets and diffusion of atmospheric oxygen was sufficient. Improved secretory function and decreased apoptosis were observed in the presence of oxygenated PFOB emulsion, implying that PFOB exerts its protective effect potentially as free radicals scavenger while delivering sufficient oxygenation (3). The quality of isolated islets is of utmost importance for successful clinical transplantation and therefore preservation of their viability and function during pretransplant culture should be maintained. Further studies simulating clinical scenarios, including high cell densities and high metabolic demand of isolated human islets, are required to prove usefulness of perfluorocarbons in clinical islet transplantation.
Footnotes
Acknowledgments
We thank the European Commission for financial support (Grant BARP+: STREP#NMP3-CT-2003-505614). The authors gratefully acknowledge A. Belcourt for his involvement in this work during the coordination of the European project. The authors declare no conflicts of interest.
