Abstract
Alzheimer disease (AD) is a progressive neurodegenerative disease, which is characterized by loss of memory and cognitive function. In AD patients dysfunction of the cholinergic system is the main cause of cognitive disorders, and decreased activity of choline acetyltransferase (ChAT), an enzyme responsible for acetylcholine (ACh) synthesis, is observed. In the present study we investigated if brain transplantation of human neural stem cells (NSCs) genetically modified to encode ChAT gene improves cognitive function of kainic acid (KA)-induced learning deficit rats. Intrahippocampal injection of KA to hippocampal CA3 region caused severe neuronal loss, resulting in profound learning and memory deficit. F3.ChAT human NSCs transplanted intracerebroventricularly improved fully the learning and memory function of KA-induced learning deficit animals, in parallel with the elevation of ACh levels in cerebrospinal fluid. F3.ChAT human NSCs migrated to the KA-induced injury site (CA3) and differentiated into neurons and astrocytes. The present study demonstrates that human NSCs expressing ChAT have lesion-tropic property and improve cognitive function of learning deficit model rats with hippocampal injury by increasing ACh level.
Keywords
Introduction
Alzheimer disease (AD) is a progressive neurodegenerative disease, which is characterized by degeneration and loss of neurons and synapses throughout the brain, particularly in the basal forebrain, amygdala, hippocampus, and cortical area. Memory and cognitive function of patients progressively decline, and patients become demented and die prematurely (24). In AD patients, dysfunction of the presynaptic cholinergic system is one of the causes of cognitive disorders (4,27), where decreased activity of choline acetyltransferase (ChAT), which is responsible for acetylcholine (ACh) synthesis, is observed (10,23). To date, AD therapy has largely been based on small molecules designed to increase ACh concentration by inhibiting acetylcholinesterase (AChE) (17,23). Since therapies with these drugs is only palliative without potential protection against progressive tissue destruction, there is a need for effective therapies for patients with AD, and stem cell-based therapeutic approaches targeting AD should fulfill this requirement. We investigated if brain transplantation of NSCs genetically modified to encode ChAT gene improves cognitive function of kainic acid (KA)-induced learning deficit rats. Intrahippocampal injection of KA to hippocampal CA3 region causes severe neuronal loss, resulting in profound learning and memory deficit (21).
NSC-based cell therapy has recently been noted as novel strategies to treat neurological disorders such as Parkinson's disease (PD), Huntington's disease, amyotrophic lateral sclerosis (ALS), stroke, and spinal cord injury (5,12,14). In contrast to a transient improvement of body function by pharmaceuticals, stem cells may prevent or delay host cell death and restore injured tissue (3,5,12,14). We previously demonstrated that transplantation of human NSCs expressing diverse functional genes preserve host cells and recover body function in animal models of stroke, PD, ALS, and spinal cord injury (9,13,22,31). Immortalized human NSCs have emerged as a highly effective source of cells for genetic manipulation and gene transfer into the central nervous system ex vivo, where genetically modified NSCs survive, integrate into host tissues, and differentiate into neurons and glial cells after transplantation (12).
Materials and Methods
Human ChAT Cloning and cDNA Constructs
Full-length human ChAT cDNA was obtained by polymerase chain reaction (PCR) from human small intestine Marathon-Ready cDNA (Clontech, Mountain View, CA) following the procedures previously described for rat peripheral type ChAT (15). Sequencing of plasmid DNA was determined with ABI 3100 DNA sequencer (Applied Biosystems, Foster City, CA).
Human NSC Lines
An immortalized NSC line, HB1.F3 (F3), was established from primary cultures of a 15-week gestational human fetal brain by infecting it with a retroviral vector encoding v-myc oncogene (11–13). The F3 NSC line was infected with a retrovirus encoding human ChAT gene. Plasmid pLPCX.ChAT containing the full-length human ChAT cDNA was used. PG13 mouse packaging cell line was transfected with pLPCX.ChAT vector using SuperFect reagent and stable PG13 cell line were selected using 5 μg/ml puromycin for 7 days (31). Replication-incompetent retroviral vector collected from PG13.ChAT cells was used for infection of F3 human NSCs and puromycin-resistant F3.ChAT clones were isolated, and the clone F3.ChAT.D2 was expanded and used for the transplantation.
Reverse Transcriptase (RT)-PCR Analysis
Total RNA was extracted from NSC cultures using TRIzol (BRL, Gaithersburg, MD). Complimentary DNA templates were prepared from 1 μg of total RNA primed with oligo dT primers using 400 U of Moloney Murine Leukemia Virus reverse transcriptase (Promega, Madison, WI) for 1 h at 42°C, inactivated for 10 min at 95°C, and cooled to 4°C. The cDNA was amplified using 25 PCR cycles and RT-PCR products were separated electrophoretically on 1.2% agarose gel containing ethidium bromide and visualized under UV light. The primers used for the ChAT RT-PCR are following: Sense: 5′-CTGTGCCCCCTTCTAGAGC-3′; Antisense: 5′-CAAGGTTGGTGTCCCTGG-3′.
Immunocytochemistry of NSCs
F3.ChAT cells were plated on poly-L-lysine-coated Aclar plastic cover slips and fixed in 4% paraformaldehyde in 0.1 M phosphate buffer for 5 min at room temperature. Fixed cultures were incubated in primary antibody specific for human ChAT (1:100, rabbit polyclonal, AB143, Chemicon, Temecula, CA) for 24 h at 4°C, followed by Alexa Fluor 488-conjugated anti-rabbit IgG (1:1,000, Molecular Probes, Eugene, OR) for 1 h at room temperature. Cells were counterstained with 4′,6-diamino-2-phenylindole (Sigma, St. Louis, MO) to identify cellular nuclei. Cells were mounted on glass slides using antifade mounting solution and viewed under an Olympus laser-scanning confocal microscope.
AD Model and NSCs Transplantation
Male Sprague-Dawley rats (Orient-Bio, Seongnam, Korea) weighing 220–230 g were anesthetized with enflurane and positioned in a stereotaxic frame. After incision of the skin and drilling a hole, freshly prepared KA (Sigma) solution (0.4 μg/2 μl/rat) was infused into the left CA3 hippocampus at the following stereotaxic coordinates from bregma: posterior 3.7 mm, lateral 4.1 mm, and ventral 4.5 mm, at a flow rate of 0.2 μl/min (21). Four weeks later, F3 or F3.ChAT cells (1 × 106 cells/rat) were transplanted in rats showing loss of learning and memory functions (n = 14/group) via left lateral intracerebroventricular (ICV) injection at the following coordinates: posterior 0.9 mm, lateral 1.4 mm, and ventral 3.9 mm. All experimental procedures were approved by the Animal Care Committee of the Chungbuk National University.
Learning/Memory Testing
The rats were subjected to learning and memory function tests immediately before (4 weeks after KA injection) and 2, 4, and 6 weeks after transplantation of the cells (n = 8/group). The passive avoidance trials were performed once a day for 4 consecutive days. The latency time of stay in the light room from light-on was recorded following experience of electric shock (1 mA for 2 s) in the dark compartment. The end point was set at 300 s, denoting full acquisition of memory. Water-maze trials were performed in a circular water bath filled with water maintained at 22 ± 2°C. The bath was divided into four quadrants and a hidden escape platform (10 cm in diameter) was submerged in the center of one quadrant. The rats were trained to learn to find the hidden platform, based on several cues external to the maze. Three trials were conducted on each day with 5-min intervals for the 4 consecutive days. The mean time spent to escape onto the platform was recorded.
ACh Analysis in Cerebrospinal Fluid (CSF)
The rats were sacrificed at the end of learning/memory testing, and CSF was collected to analyze ACh contents (n = 6/group). ACh concentration in CSF was measured with the Amplex Red acetylcholine/acetylcholinesterase assay kit (Molecular Probes) according to the manufacturer's instructions. In this assay, ACh is hydrolyzed by AChE to release choline, which is then oxidized by choline oxidase to betaine and H2O2. H2O2 interacts with Amplex Red (7-dihydroxyphenoxazine) in the presence of horseradish peroxidase to generate the highly fluorescent resorufin. The resulting fluorescence was measured in a fluorescence microplate reader using excitation in the range of 530–560 nm and emission at ~590 nm.
Immunohistochemistry in Brain Sections
To pursue the migration of NSCs from the transplanted site to KA-lesioned site, the rats were sacrificed 2 days and 1, 2, 3, 4, and 6 weeks after transplantation (n = 6/group). The rat brains were perfusion fixed with 4% paraformaldehyde solution and postfixed for 48 h, followed by cryoprotection in 30% sucrose for 72 h. Coronal cryosections in 30-μm thickness were prepared and processed for double immunostaining of human mitochondria (hmito) and ChAT, neurofilament heavy chain (NF-H, for neurons) or glial fibrillary acidic protein (GFAP, for astrocytes) using antibodies specific for hmito (1:100, MAB1273, mouse monoclonal, Chemicon), ChAT (1:100, AB143, rabbit polyclonal, Chemicon), NF-H (1:100, AB1989, rabbit polyclonal, Chemicon), or GFAP (1:100, AB1540, rabbit polyclonal, Chemicon). Brain sections were incubated with primary antibodies overnight at 40°C and with secondary antibodies conjugated with Alexa Fluor 488 or 594 (1:1,000, Molecular Probes) for 1 h at room temperature.
Statistical Analysis
Data are presented as mean ± SEM. The statistical significance between group comparisons for behavioral data was determined by one-way analysis of variance (ANOVA), followed by post hoc Tukey's multiple comparison test. Values of p < 0.05 were considered to be statistically significant.
Results
We recently established an immortalized human NSC line HB1.F3 that overexpresses the human ChAT gene (F3.ChAT). Transfection of F3 human NSCs with the ChAT gene (Fig. 1A) was confirmed by RT-PCR analysis (Fig. 1B). The F3.ChAT cells showed similar morphology to their parental F3 cells (Fig. 1C) and were highly immunoreactions positive for ChAT antibody (Fig. 1D).

Establishment and ChAT expression of F3.ChAT human neural stem cell line. (A) F3.ChAT was constructed by infecting F3 human neural stem cells with a retroviral vector encoding the human choline acetyltransferase (ChAT) gene. (B) RT-PCR analysis of ChAT mRNA expression. (C) Morphology of F3 and F3.ChAT cells. (D) Immunocytochemical identification of ChAT protein (green) in F3 and F3.ChAT cells. Scale bar: 50 μm.
At 1, 4, and 6 weeks after ICV transplantation of F3.ChAT cells (1 × 106 cells/rat) in rats pretreated with KA (0.4 μg/rat) into the hippocampal CA3 region, ChAT immnuoreactivity was detected in the ventricles and hippocampus (Fig. 2). In order to confirm that the transplanted F3.ChAT cells express ChAT in vivo, a double immunostaining specific for hmito and ChAT was performed. The results of this experiment indicate that most of the hmito-positive F3.ChAT cells strongly express ChAT up to 6 weeks posttransplantation (Fig. 2, upper panels at each time point). F3.ChAT cells double positive for hmito and GFAP as well as double positive for hmito and NF-H were observed, indicating that F3.ChAT cells differentiated into astrocytes (Fig. 2, middle panels) and neurons (Fig. 2, lower panels) as early as 1 week posttransplantation.
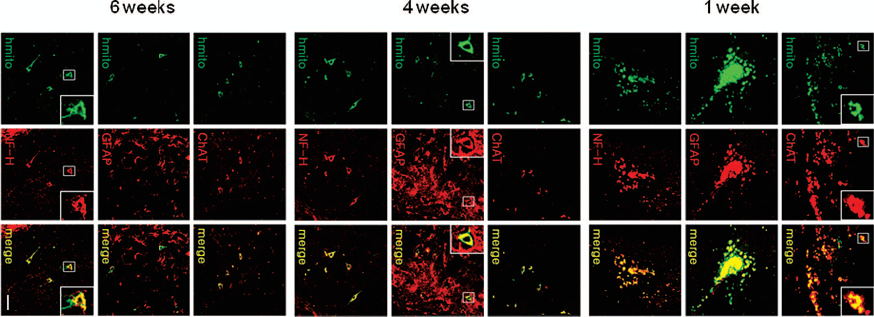
Immunohistochemical identification of ChAT expression and differentiation of F3.ChAT cells (human mitochondria [hmito]-stained) into astrocytes (glial fibrillary acid protein [GFAP]-stained) and neurons (neurofilament [NF]-H-stained) 1, 4, and 6 weeks after intracerebroventricular (ICV) transplantation. Insets are magnifications of the boxes in each photograph. Scale bar: 50 μm.
To examine the migration of transplanted NSCs, serial brain sections were prepared and hmito-positive cells were examined 2 days and 1, 2, 3, 4, and 6 weeks after transplantation. F3.ChAT cells moved from the transplanted site (left ventricle) to the KA-lesioned site (left hippocampus) (Fig. 3). Interestingly, a remarkable migration of the NSCs to severely damaged hippocampal CA3 region was observed at 1 week, and the cells were detected only at CA1 and CA3 lesion sites (alveus, oriens, and pyramidal cell layers) 4 weeks posttransplantation.

Time course of F3.ChAT cell migration. Note movement of F3.ChAT cells from ICV transplantation site (Cells) to lesion sites induced by intrahippocampal injection of kainic acid (KA). Scale bar: 5 mm.
Rats challenged with KA displayed severe impairment of learning and memory functions as measured by passive avoidance performances 4 (prior to cell transplantation, Fig. 4A), 6 (Fig. 4B), 8 (Fig. 4C), and 10 (Fig. 4D) weeks after KA injection. Notably, KA rats that received F3.ChAT cells fully recovered learning and memory functions as early as 2 weeks posttransplantation and lasted to 6 weeks, whereas control F3 cells were ineffective. Eight and 10 weeks after intrahippocampal injection of KA, the ACh concentration in CSF significantly decreased compared with saline-administered control rats (Figs. 4E, F). Such decreases in ACh levels were fully restored in brains receiving F3.ChAT cell transplantation 4 weeks after KA challenge, while the ACh level remained low in KA-challenged animals receiving control F3 cells.
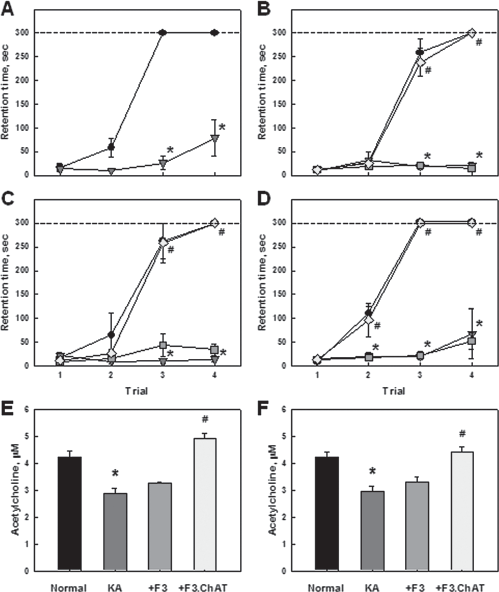
Recovery by F3.ChAT of cognitive function and cerebrospinal ACh levels in KA-challenged animals. (A-D) Passive avoidance performances of rats immediately before (A, 4 weeks after KA injection) and 2 (B), 4 (C), and 6 (D) weeks after transplantation of F3 or F3.ChAT (1 × 106 cells/rat) following 4-week pretreatment with KA (0.4 μg/rat). Circle: normal control; triangle: KA alone; square: KA + F3; diamond: KA + F3.ChAT. (E, F) Acetylcholine (Ach) concentration in rat CSF 4 (E) and 6 (F) weeks posttransplantation. Data are presented as mean ± SEM. *Significantly different from control, p < 0.05. #Significantly different from KA alone, p < 0.05.
Discussion
Transplantation of F3.ChAT human NSCs fully restored the ACh level in the brain (Fig. 4E, F) and improved the learning and memory function of KA-induced learning-deficit rats up to the levels comparable to normal animals (Fig. 3B–D). Previous studies have demonstrated that NSC transplantation improves cognitive function of AD model animals by enhancing synaptic density in hippocampus which is mediated by brain-derived neurotrophic factor (BDNF) (3,29). Similarly, NSCs expressing nerve growth factor (NGF) restored learning and memory function in AD model animals (21,28). Previously, a phase 1 clinical trial of ex vivo NGF gene delivery was performed in eight mild AD patients, implanting autologous fibroblasts genetically modified to express human NGF into the forebrain. After a mean follow-up of 22 months in six subjects, long-term adverse effects were not found and improvement in the rate of cognitive decline was achieved. Serial PET scans showed significant increases in cortical fluorodeoxyglucose after treatment (26).
In our previous studies, F3 human NSCs expressing neurotrophic factors including NGF, BDNF, neurotrophin-3 (NT-3), glial cell line-derived growth factor (GDNF), and vascular endothelial cell growth factor (VEGF) protected host neurons in animal models of hemorrhagic stroke (13), PD (31), and ALS (9). Therefore, neurotrophic factors may play roles in stem cell proliferation, differentiation, and neuroprotection (2,8,19). In the present study, parental F3 NSCs did not improve cognitive function in the KA-treated rats, while F3 cells encoding ChAT fully restored the learning and memory function in the animals, suggestive of a key role of ChAT.
Septal cholinergic neurons project into the hippocampus where they release ACh (18). Intrahippocampal administration of KA causes excitotoxic degeneration of hippocampal pyramidal neurons, mainly the cholinergic subtype (6), which may be mediated by calpain activation and tau phosphorylation (7). KA administration reduced ChAT immunoreactivity and cholinergic receptor binding, which induced cognitive impairments including memory and learning deficits (21). Thus, we adopted the KA-injected rats as an animal model of memory and learning deficits for NSC-based gene therapy in the present study. Previous studies have shown that a decrease of 25–30% in ACh levels leads to severe memory loss in β-amyloid transgenic (Tg2576) or cholinotoxin (AF64A)-challenged mice (1,25). In the present study, animals injected with KA demonstrated a similar level of ACh in the brain (Fig. 4E, F).
Choline uptake enhancers are also effective in improving memory function (23), since choline is taken up by cholinergic neurons and used for the synthesis of ACh mediated by ChAT (18). F3.ChAT cells demonstrate much higher mRNA expression and protein synthesis of ChAT than parental F3 cells (Fig. 1B, D). ChAT protein is detected in transplanted F3.ChAT cells distributed from the hippocampal lesion sites 6 weeks posttransplantation (Fig. 2). Interestingly, migration of the transplanted cells started within 1 week and fully moved to the lesion sites within 4 weeks (Fig. 3). The pathology tropism of F3 NSCs might be triggered by chemoattractants such as hepatocyte growth factor (HGF), stromal cell-derived factor-1 (SDF-1), VEGF, and stem cell factor (12,20,22), or by inflammatory mediators such as tumor necrosis factor-α (TNF-α) (16). Notably, intrahippocampally injected mouse NSCs also exhibit lesion-tropic property and long-term (4.5 months) survival in mice with diphtheria toxin-induced neuronal loss in hippocampus and cortex (30), which is in good agreement with our observations in rats ICV transplanted with F3.ChAT human NSCs following KA-induced hippocampal injury.
F3 and F3.ChAT cells migrated to lesion sites, differentiated into neurons and astrocytes, and survived up to 6 weeks posttransplantation (Fig. 2). It was confirmed that most of the hmito-positive F3.ChAT cells expressed ChAT (Fig. 2), indicating that the transplanted F3.ChAT cells functioned in the host brain tissue, which was further supported by the increased ACh levels in CSF (Fig. 4E, F). In conclusion, the present study demonstrates that the human NSCs expressing ChAT ameliorate the cognitive deficiencies associated with KA-induced hippocampal CA3 lesions via increased ACh production. These results suggest that human NSCs genetically engineered to encode ChAT gene are one of the promising candidates for the treatment of human AD patients.
Footnotes
Acknowledgments
This work was supported by Priority Research Centers Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology (2009-0094035). The authors declare no conflicts of interest.
