Abstract
Alzheimer's disease (AD) is characterized by degeneration and loss of neurons and synapses throughout the brain, causing the progressive decline in cognitive function leading to dementia. No effective treatment is currently available. Nerve growth factor (NGF) therapy has been proposed as a potential treatment of preventing degeneration of basal forebrain cholinergic neurons in AD. In a previous study, AD patient's own fibroblasts genetically modified to produce NGF were transplanted directly into the brain and protected cholinergic neurons from degeneration and improved cognitive function in AD patients. In the present study, human neural stem cells (NSCs) are used in place of fibroblasts to deliver NGF in ibotenic acid-induced learning-deficit rats. Intrahippocampal injection of ibotenic acid caused severe neuronal loss, resulting in learning and memory deficit. NGF protein released by F3.NGF human NSCs in culture medium is 10-fold over the control F3 naive NSCs at 1.2 μg/106 cells/day. Overexpression of NGF in F3.NGF cells induced improved survival of NSCs from cytotoxic agents H2O2, Aβ, or ibotenic acid in vitro. Intrahippocampal transplantation of F3.NGF cells was found to express NGF and fully improved the learning and memory function of ibotenic acid-challenged animals. Transplanted F3.NGF cells were found all over the brain and differentiated into neurons and astrocytes. The present study demonstrates that human NSCs overexpressing NGF improve cognitive function of learning-deficit model mice.
Keywords
Introduction
Alzheimer's disease (AD) is characterized by degeneration and loss of neurons and synapses throughout the brain, causing the progressive decline of cognitive function leading to dementia (42). In AD patients, dysfunction of cholinergic system is one of the primary causes of cognitive disorders (6,48), in which decreased activity of choline acetyltransferase (ChAT) responsible for acetyl-choline (ACh) synthesis is noted (43). To date, only five drugs have been approved for treatment of AD patients, and AD drug therapy is largely based on small molecule designed to increase ACh concentration by inhibiting acetyl cholinesterase (AChE), an ACh-degrading enzyme (31,43). Since drug therapy is only palliative, there is a substantial need for effective therapies for patients with AD, and stem cell-based therapeutic approach targeting AD should fulfill this requirement.
It is well established that nerve growth factor (NGF) enhances the function and survival of cholinergic neurons in basal forebrain in rats (7,10,22) and primates (20, 21,44–46). NGF has been delivered intraventricularly in these studies and resulted in adverse NGF effects including aberrant sympathetic and sensory neurite sprouting (33,50) and weight loss (49). Transplantation of cells genetically modified to produce and secrete NGF directly into the brain could achieve direct and well-targeted delivery of NGF to injured neurons, and previous studies in rodents have shown to protect cholinergic neurons from degeneration in chemical lesions (33,37) or aged brain (5,30). Similarly, cell-based NGF treatment using fibroblasts encoding NGF gene in the primate brain demonstrated to rescue degenerating basal forebrain cholinergic neurons (46). Based on these preclinical studies, a Phase I clinical trial in AD patients was undertaken to examine efficacy of ex vivo NGF gene therapy. Skin fibroblasts isolated from AD patients were transduced to produce NGF and stereotaxically transplanted into multiple locations within the forebrain of AD patients. Following a period of 22 months, the degree of cognitive decline was reduced, and no adverse effect of surgery was found (47). It should be noted that the successful application of in vivo gene transfer to the CNS will depend on the identification of suitable cells that can serve as carriers for therapeutic transgenes and provide platforms for efficient expression and secretion of transgene products. Immortalized CNS-derived NSCs have recently been introduced as potentially valuable candidates for this purpose.
NSCs have provided the basis for the development of potentially powerful new therapeutic strategies for a broad spectrum of human neurological disorders including AD, Parkinson's disease (PD), Huntington's disease (HD), amyotrophic lateral sclerosis (ALS), multiple sclerosis, stroke, spinal cord injury, and brain tumors (4,8,9,15,16,28). We have previously demonstrated that transplantation of human NSCs expressing diverse functional genes, especially encoding growth factors, preserves host cells and restored function in animal models of AD, PD, HD, ALS, stroke, and spinal cord injury (12,14,18,23–27,32,34,53).
Considering the evidence of improvement in cognition and rescue of neurons in lesioned basal forebrain cholinergic neurons after brain transplantation of fibroblasts expressing NGF, we wished to investigate in this study whether the human NSCs overexpressing NGF can lead to the increased neuronal survival and improvement in cognition in ibotenic acid-induced learning-deficit mouse model.
Materials and Methods
F3 Human Neural Stem Cell Line
The stable clonal human NSC line HB1.F3 (F3) was generated by retroviral transduction of primary fetal human NSCs (derived from fetal brain of 14 weeks gestation) with an avian v-myc cell cycle regulatory gene as previously reported (17,23). F3 human NSCs expressed phenotypes specific for NSCs including ABCG2, Musashi1, and nestin. F3 NSCs were grown in a serum-free medium (DM4) consisting of Dulbecco's modified Eagle medium with high glucose (DMEM) containing 10 μg/ml insulin, 10 μg/ml transferrin, 30 nM sodium selenate, 50 nM hydrocortisone, 0.3 nM triiodothyronine, and 20 μg/ml gentamicin (19). Recombinant human FGF-2 (10 ng/ml; PeproTech, Rocky Hill, NJ) was supplemented to the DM4 during the routine feeding. All chemicals except FGF-2 were obtained from Sigma (St. Louis, MO).
Transfection of Human NGF Gene Into F3 NSCs
PG13 mouse packaging cell line (Clontech, Mountain View, CA) was transfected with pLPCX.NGF vector (Clontech) (Fig. 1A) using LipofectAMINE (Invitrogen, Carlsbad, CA), and stable PG13 cells were selected using 5 μg/ml puromycin for 3 days. Replication-incompetent retroviral vector collected from PG13.NGF cells were collected and used for transfection of F3 NSCs. Puromycinresistant clones were isolated, screened, expanded, and used for the transplantation. Expression of NGF in the F3.NGF cell line was analyzed by RT-PCR, ELISA, and immmunofluorescence microscopy.
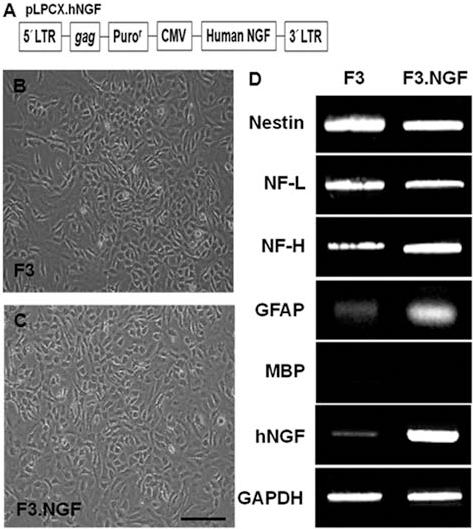
Characterization of human neural stem cell (NSC) lines. (A) The retroviral vector encoding NGF (pLPCX.NGF) used in this study for the generation of F3.NGF human NSC line. (B, C) Phase contrast microscopy of F3 and F3.NGF human NSCs. (D) Gene expression of cell type-specific markers as studied by RT-PCR in F3 and F3.NGF NSCs. Both of F3 and F3.NGF cells express cell type-specific markers nestin (for neural stem cells), NF-L and NF-H (for neurons), GFAP (for astrocytes), MBP (for oligodendrocytes), and human NGF. Scale bar: 50 μm.
RT-PCR
Total RNA was extracted from F3 and F3.NGF NSC cultures using TRIzol (BRL, Gaithersburg, MD). Complimentary DNA templates from each sample were prepared from 1 mg of total RNA primed with oligo dT primers using 400 U of MMLV (Moloney murine leukemia virus) reverse transcriptase (Promega, Madison, WI) followed by 30 PCR cycles, and RT-PCR products were separated electrophoretically on 1.2% agarose gel containing ethidium bromide and visualized under UV light. The primers used for the RT-PCR of NGF, nestin, neurofilament low-molecular-weight protein (NF-L), neurofilament high-molecular-weight protein (NF-H), glial fibrillary acidic protein (GFAP), human myelin basic protein (MBP), and glyceraldehyde 3-phosphate dehydrogenase are listed in Table 1.
PCR Primer Sequences for Cell Type-Specific Markers (All Human)

Immunocytochemistry
Immunocytochemical demonstration of NGF protein in F3.NGF human NSCs was performed. F3.NGF cells plated on poly-l-lysine-coated Aclar plastic coverslips (9 mm in diameter) were grown in DM4 serum-free medium supplemented with FGF-2 for 3–5 days, rinsed in PBS, and fixed in cold acid alcohol (5% glacial acetic acid in 95% ethanol) for 10 min at −20°C. Fixed cultures were incubated in blocking solution that consisted of PBS containing 10% normal goat serum and 3% Triton-X at room temperature (RT) for 20 min, incubated in primary antibody specific for human NGF (1:200, mouse monoclonal, Chemicon, Temecula, CA) at RT for 1 h, followed by Alexa Fluor 594 anti-mouse, rabbit IgG (Molecular Probes, Eugene, OR) for 1 h at RT, rinsed in PBS, and mounted onto slides with Gelvatol.
Protection Against Cytotoxic Agents Provided by NSCs
F3 and F3.NGF cells were plated 1 × 104 cells per well in 96-well plates (Falcon, Becton Dickinson, Franklin Lakes, NJ) with DMEM containing 5% FBS and incubated in medium containing varying concentrations of H2O2 (0–500 μM, Sigma-Aldrich, St. Louis, MO), amyloid β1–42 (Aβ1–42, 0–50 μM, BioSource, Camarillo, CA), and ibotenic acid (0–100 μg/ml, Sigma-Aldrich) for 12 h. Untreated controls contained normal media alone. Cells were then processed for viability assay and Western blot analysis. Cell viability was assessed with CCK-8 kit (Dojindo Laboratories, Kumamoto, Japan) following instructions given by the manufacturer. The absorbance was measured at 450 nm using a microplate reader.
F3 and F3.NGF cells were lysed in RIPA buffer (150 mM NaCl, 1% NP-40, 0.5% sodium deoxycholate, 0.1% SDS, 50 mM Tris, pH 8.0) and centrifuged at 14,000 rpm for 5 min. The supernatant was harvested, and 20–30 μg proteins were subject to SDS-polyacrylamide gel electrophoresis. The proteins on the gel were transferred electrophoretically onto Immobilon-P membranes, and Western analysis was performed with primary antibodies against caspase 3 (1:500, Chemicon, Temecula, CA), phospho-Akt (1:500, Cell Signaling Technology, Danvers, MA), and Akt1 (1:500, Chemicon). Secondary antibodies (Amersham, Chicago, IL) and a chemiluminescence kit (Amersham) were used for immunodetection.
Mouse Cognitive Dysfunction Model
All experimental procedures were approved and carried out in accordance with the Institutional Animal Care and Use Committee of the Laboratory Animal Research Center at Korea University. Mouse cognitive dysfunction model (CDM) was induced by stereotaxic, intrastriatal administration of ibotenic acid by previously described methods (41). After an intraperitoneal injection of 1% ketamine (30 mg/kg) and xylazine hydrochloride (4 mg/kg), ICR mice (male, 25–30 g; n = 32) were placed in a stereotaxic frame (Kopf Instruments, Tujunga, CA). A burr hole was made, and a 30-gauge needle was inserted through the burr hole into the freshly prepared ibotenic acid solution (1 μl saline containing 0.5 μg, Sigma) was infused into the left and right CA3 hippocampi (stereotaxic coordinates from bregma: posterior 3.7 mm, lateral ±4.1 mm, and ventral 4.5 mm) over a period of 5 min. The needle was removed after remaining in place for another 5 min.
Brain Transplantation
F3 and F3.NGF NSCs grown in culture flasks were dissociated into single cells by a brief trypsin treatment and suspended in PBS at 4 × 107 cells/0.1 ml and kept on ice until transplanted. One week after CDM surgery, randomly selected CDM mice received 2 μl of PBS containing 2 × 105 cell suspension of F3 (n = 9) or F3.NGF cells (n = 9). In control sham group, 2 μl of PBS without NSCs was injected into the CDM mice (n = 7). PBS or PBS-containing NSCs were injected slowly for 5 min into overlying cortex of the hippocampal lesion (3.7 mm posterior, ±4.1 mm ventral, and 2.0 mm lateral to the bregma). The needle was removed after remaining in place for another 5 min. Immunosuppressant was not used in any of the animals.
Learning/Memory Testing
To evaluate learning and memory functions, the mice were subjected to Morris water maze test. Water maze trials were performed in a circular water bath (180 cm in diameter) filled with water (27 cm in depth) maintained at 22 ± 2°C. The bath was divided into four quadrants, and a hidden escape platform (10 cm in diameter, 25 cm in height) was submerged in the center of one quadrant, 2 cm below the surface of the water. The mice were trained to learn to find the hidden platform based on several cues external to the maze. The position of the cues was unchanged throughout the experiments. Mice were subjected to three training trials of 5 min each per day for four consecutive days followed by fifth performance 1 week later. One week later, mice were processed for cognitive dysfunction model (CDM) by stereotaxic, intrastriatal administration of ibotenic acid. Each mouse was given a maximum of 90 s to find the hidden platform and then allowed to remain on it for another 30 s. The mean time spent to escape onto the platform during training trials, mean escape latency, was calculated and compared. If a mouse failed to escape from the water within 90 s, it was guided to the platform and kept to stay there for another 30 s.
Immunohistochemistry in Brain Sections
At the end of behavioral testing, each animal was anesthetized and perfused through the heart with 4% paraformaldehyde in 0.1 M phosphate buffer, the brains were postfixed in the same fixative for 24 h, followed with cryoprotection in 30% sucrose for 24 h, and then coronal sections were prepared on a cryostat. Serial brain sections 30-μm thick through the needle entry site, which was identifiable on the brain surface, and sites 1.0 mm anterior and 1.0 mm posterior to plane were prepared. This generally yielded six or seven sections in a series. Brain sections were processed for double immunostaining of human NGF and cell type-specific markers using antibodies specific for human NGF (1:200, mouse monoclonal, Chemicon, Temecula, CA), ChAT (1:200, rabbit monoclonal, Chemicon), GFAP (1:1,000, rabbit monoclonal, Chemicon), or NF (1:200, rabbit monoclonal, Chemicon). To determine whether grafted F3 and F3.NGF cells continue to proliferate in vivo, brain sections were processed for double immunofluorescence staining of human nuclear matrix antigen (hNuMA, 1:200, mouse monoclonal, Abcam, Cambridge, MA). Brain sections were incubated in primary antibodies overnight at 4°C as free floating sections, followed by Alexa Fluor 594 anti-mouse, rabbit IgG (Molecular Probes, Eugene, OR) for 1 h at RT. Negative control sections were prepared in an identical manner, except that the primary antibodies were omitted. Stained sections were then examined under an Olympus laser confocal fluorescence microscope.
Statistical Analysis
Two-way ANOVA and the post hoc Tukey test were used for analysis of stem cell transplantation. Data are presented as means ± SE.
Results
Stable Human Neural Stem Cell Line Expressing Human NGF
F3 human NSCs were infected with a retroviral vector-encoding human NGF gene (Fig. 1A), and clones resistant to puromycin were selected and expanded. One of the clones was chosen and used in the present study. The morphology of the selected F3.NGF cells does not differ from the parental F3 NSCs with bipolar or multipolar morphology (Fig. 1B, C). Results of RT-PCR analysis of mRNAs isolated from F3 and F3.NGF cells are shown in Figure 1D. Transcripts for nestin (an NSC specific marker), neurofilament triplet proteins (NF-L and NF-H, cell type-specific markers for neurons), glial fibrillary acidic protein (GFAP, a specific marker for astrocytes), and NGF are all expressed by both F3 and F3.NGF cells. However, transcript for MBP, structural protein, and a specific cell type-specific marker for oligodendrocytes was not demonstrated. In addition, transcriptional level of human NGF gene is much higher in F3.NGF cells as compared to parental F3 cells.
Levels of NGF in the spent media of cultured F3 and F3.NGF cell lines are shown in Figure 2A. ELISA analyses indicated that human NGF released by F3.NGF NSCs in culture media is 10-fold over the control F3 parental NSCs at 1203.0 ± 107.3 ng/106 cells/day (mean ± SEM) versus 120.9 ± 36.3 ng/106 cells/day (p < 0.05). All F3.NGF cells expressed NGF-positive immunoreactions, while only a small number of parental F3 cells express it (Fig. 2B, C).
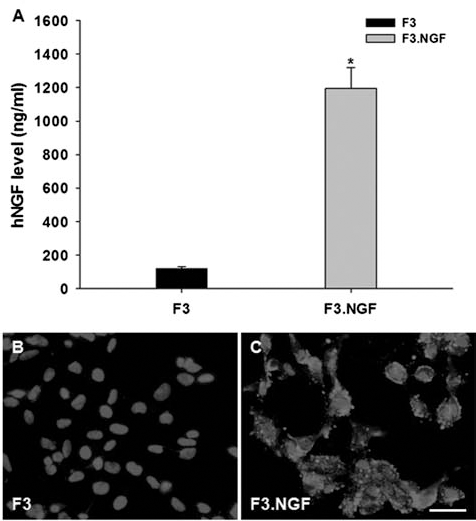
Detection of immunoreactive human NGF and quantitative analysis of NGF levels in vitro. (A) ELISA assay for human NGF in spent media of F3 and F3.NGF human NSCs. F3.NGF cells secrete 10 times NGF amount over parental F3 cells (∗p < 0.05). (B, C) Immunocytochemical demonstration of NGF immnureactivity in F3 and F3.NGF cells. All F3.NGF cells express NGF-positive reaction, while only a small number of F3 cells express it. Scale bar: 50 μm.
Protection Against Cytotoxic Agents Provided by NSCs Expressing NGF
F3 and F3.NGF NSCs in culture were exposed to varying concentration of H2O2, Aβ1–42, or ibotenic acid to determine the protective effects of NGF on cell death induced by these cytotoxic agents. F3.NGF cells showed a higher survival rate to H2O2-, Ab1–42-, or ibotenic acid-induced cell death as compared to the F3 controls (Figs. 3 and 4). Survival rate of F3 parental cells exposed to 100 μM H2O2 for 12 h was 15%, while in F3.NGF cells, it was 85%. Similarly, in F3 cells treated with 5 μM Aβ1–42, survival was 40%, while in F3.NGF cells, it was 85%. In F3 cells exposed to 25 μg/ml ibotenic acid for 12 h, the survival rate was 47%, but in F3.NGF cells, the survival rate increased to 70% (Fig. 3).

Cell viability of F3 and F3.NGF human NSCs following treatment with H2O2, Ab, or ibotenic acid. The rate of cell survival in F3.NGF cells was much higher as compared to parental F3 cells following treatment with cytotoxic agents H2O2, Ab, or ibotenic acid for 12–24 h.
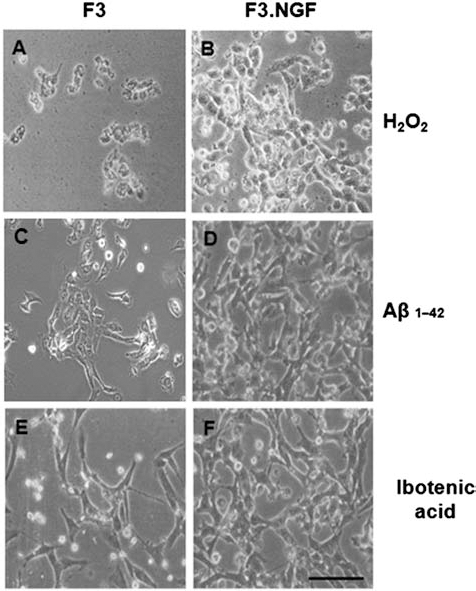
Phase contrast microscopy of F3 and F3.NGF human NSCs following exposure to 100 μM H2O2 (A, B), 5 μM Ab (C, D), or 25 μg/ml ibotenic acid (E, F) for 12 h. F3.NGF cells survived well as compared to the parental F3 cells. Scale bar: 50 μm.
Whether cytotoxic agents H2O2, Aβ1–42, or ibotenic acid induces changes in caspase 3 cleavage and phosphorylation status of Akt1 in F3 and F3.NGF cells was investigated. When F3 and F3.NGF cells were exposed to H2O2 at 100 μM, Aβ1–42 at 5 μM, and ibotenic acid at 25 μg/ml for 12 h, an increase in active fragment of caspase 3 (20 kDa) was found in F3 cells, while in F3.NGF cells, the increase was considerably attenuated (Fig. 5). Following treatment with these cytotoxic agents, phosphorylated form of Akt1 was found to increase in F3.NGF cells, while F3 cells showed the opposite expression pattern (Fig. 5). Since Akt1 is a critical mediator of growth factor-induced neural survival, an increase in phosphorylated Akt1 is the source of higher survival rate in F3.NGF cells following exposure to cytotoxic agents. These results indicate that the increase in Akt1 phosphorylated form and better cell survival are due mainly from production and secretion of NGF and its phosphorylation in F3.NGF cells.

Western blot analyses of protein levels of caspase 3 and phospho-Akt1 enzymes in F3 and F3.NGF human NSCs following treatment with H2O2, Ab, or ibotenic acid. Active fragment of caspase 3 was increased in F3 cells, while in F3.NGF cells, the increase was considerably attenuated and the levels of activated Akt1 phosphorylation in F3.NGF cells increased following treatment with H2O2, Ab, or ibotenic acid.
Learning/Memory Performance in CDM Mice Following F3 NSC Transplantation
Learning/memory performance of CDM mice receiving PBS, F3, or F3.NGF NSCs was determined by the Morris water maze (Fig. 6). The CDM mice receiving F3.NGF cells showed improved learning/memory performance on the Morris water maze as compared with the animals injected with PBS or F3 cells, and the effect persisted for at least up to 4 weeks posttransplantation (the point at which animals were sacrificed) (Fig. 6). Significant difference in Morris water maze test performance in F3.NGF versus F3 groups was evident during the period of 1–4 weeks posttransplantation (p < 0.05).
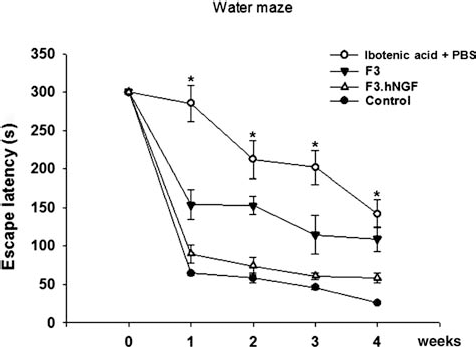
Learning/memory improvement demonstrated in learning-deficit mice transplanted with F3 or F3.NGF human NSCs. Morris water maze test. F3.NGF-transplanted group showed considerable improvement in water maze test close to normal controls. Improved performance continued up to 4 weeks posttransplantation (∗p < 0.05). ●, Control; ○, Ibotenic acid + PBS; ▼, Ibotenic acid + F3; Δ, Ibotenic acid + F3.NGF.
Transplanted F3.NGF NSCs Differentiate Into Neurons and Astrocytes
Following transplantation into lesioned mouse cortex overlying hippocampal lesion site, hNuMA+ human NSCs migrated selectively to the hippocampus and also located on the border of the lesion and further away from the injection sites (Fig. 7A–C). A large number of transplanted hNuMA+ F3.NGF cells differentiated into neurofilament+ (Fig. 7G–I) (35–45%) or ChAT+ (Fig. 7M–O) (1–3%) neurons in the CA1, CA3 hippocampal regions. Only a small number of transplanted hNuMA+ F3.NGF cells were GFAP+ astrocytes (Fig. 7J–L) (3–5%), and the hNuMA+/GFAP+ double-positive cells were found along the border of CA3. These results indicate that a majority of grafted F3.NGF NSCs differentiate into either neurons or astrocytes in response to signals from the local microenvironment provided by the host hippocampal lesion site.

Distribution of human nuclear matrix antigen (hNuMA) immunostaining-positive F3.NGF NSCs in ibotenic acid-injected mouse brain. Four weeks posttransplantation. (A) Schematic drawing indicating the localization of hNuMA-positive F3.NGF cells. (B, C) hNuMA-positive F3.NFG cells in ibotenic acid-injected mouse brain shown in (A). (D–F) F3.NGF cells producing NGF protein in the mouse brain. (G–I) F3.NGF cells differentiate into neurons in the mouse brain as shown by NF reaction. (J–L) F3.NGF cells differentiate into astrocytes in the mouse brain as shown by GFAP reaction. Scale bar: 50 μm.
Discussion
Gene therapy became a potential way of achieving direct intracerebral delivery of neurotrophic factors such as NGF in the injured or diseased brain (16,28). In previous studies, fibroblasts genetically modified to encode NGF gene were transplanted in the brain with cholinergic lesions or aged brain and resulted in protecting cholinergic neurons from degeneration (33,37). Recent studies have also utilized a replication-defective adeno-associated virus type 2 (AAV2) vector containing NGF cDNA to deliver NGF into lesioned basal forebrain and prevented degeneration of cholinergic neurons (29,51). The FDA has approved a Phase I clinical trial based on these AAV2-based in vivo animal studies to evaluate the safety and efficacy of AAV2 vector encoding NGF in Alzheimer's disease (AD) patients (1,3). Effectiveness of this viral injection approach is limited by the viral dosage that can be delivered and the slow spread of the virus through the brain. In contrast to these shortcomings of viral vectors, the genetically modified NSCs encoding NGF gene migrate selectively and widely to pathology site and provide protection of cholinergic neurons by producing NGF continuously.
In the present study, F3 and F3.NGF NSCs in culture were exposed to varying concentration of H2O2, Ab1–42, or ibotenic acid to determine the protective effects NGF on cell death induced by these cytotoxic agents. F3.NGF cells showed a considerably higher survival rate to H2O2-, Aβ1–42-, or ibotenic acid-induced cell death as compared to the F3 controls. When F3 and F3.NGF cells were exposed to H2O2, Ab1–42, or ibotenic acid, an increase in caspase 3 was found in F3 cells, while in F3.NGF cells, the increase was considerably attenuated. In contrast, phosphorylated form of Akt1 was found to increase in F3.NGF cells, while in F3 cells, it was decreased. Since Akt1 is a critical mediator of growth factor-induced neural survival, an increase in phosphorylated Akt1 is mediated by the production and secretion of NGF and its phosphorylation in F3.NGF cells, and this increase in Akt1 further resulted in higher survival of F3.NGF cells following exposure to cytotoxic agents.
NGF secreted by F3.NGF human NSCs protected hippocampal cholinergic neurons from degeneration in the brain and improved the learning and memory function of learning-deficit mice induced by injection of ibotenic acid. Previous studies have demonstrated that neural progenitor cells and fibroblasts expressing NGF restored learning and memory function in AD model animals (10,20,22,39,44). Neurotrophic factors that include NGF and BDNF play important roles in stem cell proliferation, differentiation, and neuroprotection (2,11,52). We have reported previously that F3 human NSCs overexpressing neurotrophic factors protected host neurons in animal models of neurological diseases (16). These include F3 NSCs expressing BDNF, glial cell line-derived growth factor (GDNF) and VEGF in hemorrhagic stroke animal models (23–26), F3.VEBF NSCs in ALS model (13), and F3.NT3 NSCs in spinal cord injury (12). In the present study, naïve parental F3 NSCs did not exhibit notable effect on the cognitive function of the cognitive impaired rats with severe memory impairment induced by ibotenic acid excitotoxin injection 4 weeks earlier. However, transplantation of F3.NGF cells fully restored the learning and memory function in the animals.
Homing of F3 NSCs to tumor or injury site is triggered by chemoattractants such as vascular endothelial cell growth factor (VEGF), hepatocyte growth factor (HGF), stromal cell-derived factor-1 (SDF-1), and stem cell factor (SCF) (14,36,38,40,41). In addition, tumor tropism (36,38) and injury site tropism (14) may be related to inflammatory mediators such as tumor necrosis factor-α (TNF-α) that also influences differentiation and survival of transplanted stem cells (16). Notably, intrahippocampally injected human NSCs overexpressing choline acetyltransferase (ChAT) exhibited injury site tropic property and long-term survival in rats with kainic acid-induced neuronal loss in hippocampus (32), which is very much similar to our observations in mice transplanted with F3.NGF human NSCs following ibotenic acid-induced hippocampal injury.
F3 and F3.NGF cells migrated to lesion sites, differentiated into neurons and astrocytes, and survived up to 4 weeks after transplantation. It was confirmed that F3.NGF human NSCs are double-positive for human-specific nuclear antigen and NGF, indicating that the transplanted F3.NGF cells survived and released NGF in the host brain tissue. In addition, F3.NGF cells were found to differentiate into NF-positive neurons or GFAP-positive astrocytes following transplantation into the brain. The present study demonstrated that human NSCs expressing NGF ameliorate the cognitive deficiencies associated with ibotenic acid-induced lesions in AD model mice via increased NGF production. These results indicate that human NSCs expressing NGF are promising candidates for the cell-based gene therapy for AD patients.
Footnotes
Acknowledgments
This research was supported by Research Program through the National Research Foundation (NRF) of Korea/ Ministry of Education, Science and Technology (20100023426, H.J.L.), Korea University Education and Research Foundation (K0830921, Y.K.), and Canadian Myelin Research Initiative (S.U.K.). Conceived and designed the experiments: H.J.L., Y.K., S.U.K. Performed the experiments: H.J.L., I.J.L. Analyzed the data: H.J.L., I.J.L., S.W.P., Y.B.K. Wrote the paper: H.J.L., S.U.K. The authors declare no conflicts of interest.
