Abstract
A great deal of interest has arisen recently with respect to human mesenchymal stem cells (MSCs), due to their broad therapeutic potential. However, the safety and efficacy of MSCs expanded ex vivo for clinical applications remain a concern. In this article, we establish a standardized process for manufacture of human umbilical cord-derived MSCs (UC-MSCs), which encompasses donor screening and testing, recovery, two-stage expansion, and administration. The biological properties and safety of UC-MSCs were then characterized and tested. The safety data from use in human patients have also been reported. After clinical-scale expansion, a yield of 1.03–3.78 × 108 MSCs was achieved in 10 batch manufacturing runs. The biological properties, such as plastic adherence, morphology, specific surface antigen (CD105, CD73, CD90, positive ≥ 95%; CD45, CD34, CD31, CD11b, CD19, HLA-DR, negative ≤2%), and multipotent differentiation potential (osteogenesis and adipogenesis) were retained. Bacterial and mycoplasma tests were negative and endotoxin levels were lower than 2 EU/ml. No adverse events were noted in two patients treated with intravenously and/or intrathecally administered MSCs. The data obtained indicate that banking UC-MSCs for clinical use is feasible.
Introduction
Human multipotent mesenchymal stromal cells (MSCs), also known as mesenchymal stem cells, are of great therapeutic potential due to their capacity for self-renewal, multilineage differentiation, hypoimmunogenicity, immune modulation, stromal support, paracrine, migration, and genetic stability (2–4,6,7,16,19,25). At present, a variety of clinical trials with human MSCs (autologous or allogeneic) are ongoing for the treatment of various incurable diseases such as graft versus host disease (GVHD), multiple sclerosis, Crohn's Disease, liver failure/cirrhosis, myocardial infarction, myocardial ischemia, burn, osteogenesis imperfect, and leukemia (http://www.clinicaltrials.gov). Some of these studies are now showing promising results (8,10,11,15,18,20,27).
Bone marrow (BM) is currently the main source of MSCs for clinical investigation. Over the last few years, it has been reported that MSCs could be isolated from various tissues. Among those, umbilical cord (UC) and umbilical cord blood (UCB) may be ideal sources due to their accessibility, painless procedures to donors, promising sources for autologous cell therapy, and lower risk of viral contamination. However, MSCs are a rare population in umbilical cord blood mononuclear cells. In addition, the process of isolation of MSCs is at the expense of losing hematopoietic stem/progenitor cells in cord blood. Thus, umbilical cord should be focused on as an alternative source of MSCs.
Recently, we successfully isolated MSCs from human full-term umbilical cords and demonstrated the similarities of their biological properties to those of BM-MSCs (24), which is consistent with the observation of several other groups (1,13,14,28). Regardless of the success achieved in isolation and expansion of MSCs at laboratory scale, manufacturing a therapeutic cell-based product is and will continue to be challenging. For example, MSC manufacture is currently an open, time consuming, and labor intensive process. The extended period of expansion further introduces the risks of microbial contamination or changes in biologic properties. Here, we establish a standardized process for manufacture of the therapeutic UC-MSCs, which encompasses donor screening and testing, recovery, two-stage expansion and administration, and evaluate the safety and efficacy of those MSCs expanded ex vivo for clinical applications. Our preliminary results indicated that banking UC-MSCs for clinical use is feasible.
Materials and Methods
Collection and Transport of Umbilical Cord
Human umbilical cords were obtained from full-term cesarean section deliveries with informed consent of the mother. A screen of the mother's medical history was performed and a blood sample was tested for specific human pathogens, such as human immunodeficiency virus 1/2 (HIV1/2), Hepatitis B and C virus (HBV, HCV), and syphilis. Tissue collection was approved by the Institutional Medical Research Ethics Committee of the local maternity hospital. Cords (15 cm long) were immersed in D-MEM/F-12 (1:1) (Gibco) supplemented with gentamicin (100 μg/ml) and amphotericin-B (5 μg/ml) (Sigma) and immediately transferred to the laboratory. The shipment temperature was 4–10°C.
Isolation and Primary Expansion
MSCs were isolated from umbilical cord based on methods previously described by our group, with slight modifications (24). Briefly, after rinsing with phosphate buffered saline (PBS) twice and then disinfecting in 75% ethanol for 30 s, umbilical cord was minced and digested. Cells were plated at a density of 10,000 to 16,000 cells/cm2 in a T75 cell culture flask (Corning) in D-MEM/F-12 culture medium supplemented with 10% fetal bovine serum (FBS, Hyclone) and 10 ng/ml epidermal growth factor (Sigma). Once confluence had been reached, adherent cells (passage 0) were detached with 0.125% trypsin and passaged at a 1:3 split ratio in the T75 flask. Cells from passage 2 or 3 were harvested during the first expansion period.
Cryopreservation
Prior to freezing, cells from the same donor were combined and an aliquot was removed and tested (Table 1). The remaining cells were resuspended in a cryoprotectant solution composed of 90% FBS and 10% dimethyl sulfoxide (DMSO) (Edwards Lifesciences) and stored at −80°C overnight, then transferred to the vapor phase of a liquid nitrogen tank. Cryopreservation was maintained for 1 year.
In-Process Test and Cryopreservation Criteria

Thawing and Clinical-Scale Expansion
Frozen cells were thawed for clinical-scale expansion using the same protocol as described for primary expansion. Cells from passage 5 to 7 were harvested during the second expansion period. After rinsing twice with Plasmalyte-A (Baxter), cell pellets were resuspended in Plasmalyte-A containing 1% or 5% human serum albumin (Hualan Biological Engineering Inc.). Cell aliquots from each donor batch were removed and tested (Table 2). The majority of remaining cells were packaged in 25-ml disposable plastic blood bags (10 million cells in 10 ml volume) (Shandong Weigao Group Medical Polymer Company Limited) and then transported to the clinical site for infusion within 12 h or for research, while a fraction of the cells were seeded for further expansion until passage 15 (approximately 35 doublings) and used for research.
Final Product Release Criteria

In-Process and Release Testing
Cell Counts and Viability
Cell numbers were determined using a hemocytometer and viability was assessed by trypan blue exclusion.
Sterility and Mycoplasma Assay
A 14-day sterility culture and 28-day mycoplasma culture were performed in accordance with methods set forth in the Chinese Pharmacopoeia. To test for sterility, an additional Gram stain was performed to detect any gross contamination before release of the final product.
Endotoxin Assay
Endotoxin levels were determined by the gel clot limulus amebocyte lysate test method, in compliance with the Chinese Pharmacopoeia.
Phenotype and Purity
Cells were stained with phycoerythrin (PE)-conjugated antibodies against CD11b, CD45, CD73, CD90, CD105, and human leukocyte antigen (HLA)-DR, or fluorescein isothiocyanate (FITC)-conjugated antibodies against CD19 and CD34. Mouse isotypic antibodies served as the control. Antibodies used were purchased from Becton Dickinson, except for CD105 (Serotech). Cells were stained with a single label and then analyzed by flow cytometry with a FACScan (Becton Dickinson).
In Vitro Differentiation
In vitro osteogenic and adipogenic differentiation were examined using a MSC Osteogenesis Kit (CHEMICON) and a MSC Adipogenesis Kit (CHEMICON) following the manufacturer's protocol for each kit. Osteogenic differentiation was assessed by staining cells with von Kossa. Adipogenic differentiation was assessed by staining cells with Oil Red O Solution.
Administration
Patient 1
A 55-year-old female patient was diagnosed with primary progressive multiple sclerosis in December 2007. The patient showed little response to either high-dose methylprednisolone (1000 mg/day for 3 days) or prednisone (40 mg/day for more than 2 months) and remained quadriplegic 8 months after disease onset. After informed consent and approval by the Ethics Committee at the local hospital, she received a UC-MSCs therapy comprised of intrathecal injection of 1 times; 107 cells and intravenous injection of 2 times; 107 cells, which followed preconditioning with cyclophosphamide (600 mg daily for 3 days) (22).
Patient 2
A 35-year-old male patient was diagnosed with chronic myeloid leukemia-chronic phase in June 2005. After treatment via conditioning regimes including antithymocyte globulin described by Lu et al. (23), this patient received a haplo-peripheral blood hematopoietic stem cell transplantation donated by his sister. Two months after transplantation, the patient had developed grade III acute graft versus host disease (GVHD), including diarrhea, an increased level of liver enzymes, and a continuing pancytopenia with lung and left knee joint infections. Besides being treated for repeated bacterial, viral, and fungal infections, he showed a poor response to either steroids or other immunosuppressors. After the informed consent by the patient and approval by the Ethics Committee of the local hospital, the patient received 4 times; 107 UC-MSCs intravenously on September 18, 2006 and October 25, 2006.
For intravenous injection, UC-MSCs resuspended in Plasmalyte-A containing 5% human serum albumin were infused for 10 min per 1 times; 107 cells. For intrathecal injection, 1 times; 107 cells resuspended in Plasmalyte-A containing 1% human serum albumin were drawn into a 20-ml syringe. After the lumbar puncture, about 10 ml of cerebrospinal fluid (CSF) was withdrawn and immediately the syringe was attached to the lumbar puncture needle and an identical volume of UC-MSCs was slowly injected. The patient was monitored for 30 min after the administration to watch for possible allergic reactions.
Statistical Analysis
Data are presented as mean ± SD. Comparisons of continuous variables between more than two groups were performed by a one-way ANOVA. If the F distribution was significant, Bonferroni or Games-Howell was used to specify differences between groups. A value of p < 0.05 was considered statistically significant. The SPSS software package was used for the statistical tests.
Results
Two-Stage MSC Expansion
Approximately 1.87–3.10 × 106 nucleated cells were obtained from umbilical cords (n = 10, range 19–36 cm) after enzyme digestion. More than 1 × 106 of total nucleated cells from each donor were used for cell surface phenotype analysis. The remaining cells were seeded into 1–2 T75 flasks. Adherent cells with fibroblastic morphology could be observed as early as 24 h after seeding. The primary cultures reached confluence after 7–12 days, when 3.74–7.38 × 106 adherent cells were harvested. After successive passages with a 1:3 split ratio, 3.3 × 107–1.2 × 108 P2 or P3 MSCs were harvested and cryopreserved at the end of first expansion period. An aliquot of cryopreserved cells was used for each donor batch manufacturing run. Duration of culture ranged from 7 to 10 days in ten clinical-scale MSCs expansions. A yield of 1.03–3.78 × 108 MSCs (P5 to P7) was harvested and approximately 1010 P7 cells could be harvested from one umbilical cord using our methods. No evidence was provided that the number of harvested cells per cm2 was changed after clinical-scale expansion (p < 0.05). Viability of the freshly harvested cells was greater than 90% in all cases. Basic data for the two-stage MSC expansions are shown in Table 3.
Basic Data of Two-Stage MSC Expansion

More than 1 × 106 of total nucleated cells used for cell surface phenotype analysis. The remaining cells seeded in 1–2 T75 flasks.
No evidence was provided that harvested cell density (between P0, P2/P3, and P5/P7) was changed after clinical-scale expansion (p > 0.05). The p-value was analyzed by one-way ANOVA.
Characterization and Test
Of the primary nucleated cells initially isolated from umbilical cords, 24.57 ± 9.51% and 12.52 ± 3.34% cells expressed CD73 and CD105, respectively, whereas 10.94 ± 2.97%, 6.86 ± 3.46%, and 5.11 ± 2.11% cells expressed CD34, CD45, and HLA-DR, respectively. Based on plastic adherence, however, MSCs were enriched and purified during cell culture and passage as at the end of primary culture, the cells expressing CD73 and CD105 were significantly increased (90.74 ± 10.58% and 68.94 ± 22.02%, respectively, p < 0.05), while the cells positive for CD34, CD45, and HLA-DR were significantly decreased (1.42 ± 2.05%, 1.15 ± 1.16%, and 0.92 ± 0.50%, respectively, p < 0.05). From passage 1 onwards, flow cytometry showed homogeneous reactivity of consistently more than 95% of cells with antibodies against CD73, CD90, CD105, and less than 1% reactivity with CD45, CD34, CD11b, CD19, and HLA-DR (Fig. 1A). This level of purity was not changed even at the end of passage 15 (Fig. 1B).
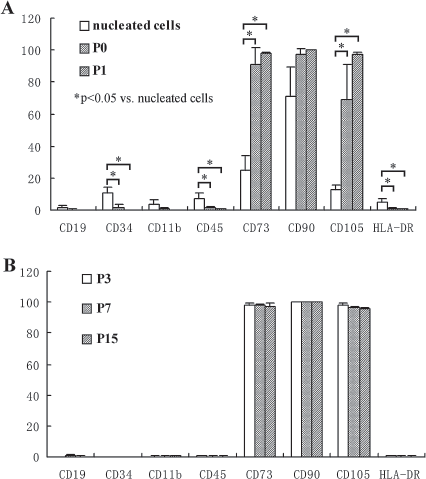
A set of typical surface phenotypes that define human mesenchymal stem cells (MSCs) (5) and demonstrate the level of MSC purity was detected by flow cytometric analysis. (A) Of the primary nucleated cells initially isolated from umbilical cords, 24.57 ± 9.51% and 12.52 ± 3.34% cells expressed cluster of differentiation 73 (CD73) and CD105, respectively, whereas 10.94 ± 2.97%, 6.86 ± 3.46%, and 5.11 ± 2.11% cells expressed CD34, CD45, and human leukocyte antigen (HLA)-DR, respectively. At the end of primary culture, the cells expressing CD73 and CD105 were significantly increased (90.74 ± 10.58% and 68.94 ± 22.02%, respectively, p < 0.05), while the cells positive for CD34, CD45, and HLA-DR were significantly decreased (1.42 ± 2.05%, 1.15 ± 1.16%, and 0.92 ± 0.50%, respectively, p < 0.05). From passage 1 onwards, flow cytometry showed homogeneous reactivity of consistently more than 95% of cells with antibodies against CD73, CD90, CD105, and less than 1% reactivity with CD45, CD34, CD11b, CD19, and HLA-DR. This demonstrated that MSCs were purified and that the contaminating endothelial cells (CD34+) and cord blood cells (CD45+) rapidly disappeared during cell culture and passage. (B) The level of MSC purity was fairly stable between P3, P7, and P15 cell population. The p value was analyzed by one-way ANOVA.
The in vitro differentiation capacity of MSCs was retained after clinical-scale expansion. Osteogenic and adipogenic induction could be confirmed by staining cells with von Kossa and Oil Red O Solution, respectively (Fig. 2).

Adipogenic and osteogenic differentiation of P3, P7, and P15 umbilical cord (UC)-MSCs demonstrated by staining with Oil Red O Solution, von Kossa respectively (original magnification as indicated by scale bars).
Sterility and mycoplasma assays were performed before cryopreservation and release, as well as by random testing of culture supernatants in the course of the expansion, to exclude microbiological contamination. An additional Gram stain would be performed to detect any gross contamination before release of final product. Endotoxin levels of final products were lower than 2 EU/ml.
Case Report
Patient 1
There was no immediate toxicity in response to UC-MSC therapy. After UC-MSC transplantation, the patient successively showed alleviation of sensory impairment, her muscle strength increased, and objective improvement was noted in neurological signs. During a 2-month follow-up, the patient showed that her Expanded Disability Status Scale (EDSS) score decreased by at least 2 points from the baseline of 8.5 and a significant reduction in the T2 lesion load was confirmed by magnetic resonance imaging. The patient is still in our follow up and remains in stable condition.
Patient 2
The patient showed an unexpected response to this therapy (Fig. 3). His liver enzymes dropped sharply and pancytopenia was markedly improved. The physical exam emphasized that both his lung and left knee joint infections were cured. On November 17, 2006, the patient was released and we lost follow-up.

Effects of two UC-MSC transfusion in a human patient. The alanine transaminase (ALT) level decreased from more than 200 IU/L to 78.03 IU/L (reference value 10~40 IU/L) and the gamma glutamyl transpeptidase (GGT) from 1594.6 IU/L to 671.72 IU/L (reference value 0~51 IU/L) after two transfusions of UC-MSCs. Either hemoglobulin or white blood cells and platelet counts rose and were close to normal levels. The first transfusion of UC-MSCs corresponds to day 1. *Indicates the day the patient received UC-MSCs transfusion.
Discussion
Research into MSCs has risen dramatically over the last 2 decades. The most attractive aspect of MSCs is that allogeneic MSCs can be transplanted without the need for a major histocompatibility complex (MHC) match. MSCs from one single donor can be used for multiple patients. The concept of “off-the-shelf” stem cell therapies has become reality. However, MSC manufacture currently is an open, time-consuming and labor-intensive process. The extended period of expansion introduces the risk of microbial contamination and changes in biologic properties. Establishing a cell bank operating in accordance with Good Manufacturing Practice standards is an advisable choice (34). A standardized manufacturing process and extensive testing can ensure the safety, identity, purity, and potency of the final product.
Our previous study indicates that UC represents a relatively large reservoir of MSCs (24). In this study, we establish a standardized manufacturing process that encompasses donor eligibility, collection of UC and isolation, expansion, cryopreservation, thawing, characterization, release, and administration of the UC-MSCs. A two-step donor screening procedure was developed. Using a standard health questionnaire the detailed personal and family history of genetic disease, malignancy, and communicable diseases is taken to assess whether the mother or newborn is eligible for UC donation. It also allows the opportunity for donor self-exclusion. A sample of maternal blood was then taken for further screening of infectious diseases. We only select UCs obtained from cesarean section because more than a 50% bacterial contamination rate from vaginal delivery was observed, while the rate from cesarean section was lower than 1%. Mechanical chopping of the cord was performed before enzymatic digestion to improve the efficiency of digestion and shorten the time required for isolation process. MSCs were enriched and purified by virtue of plastic adherence in our optimum culture conditions. After two-stage expansion, a yield of 1.03–3.78 × 108 MSCs was harvested. This quantity represented one to six putative application doses for a 60-kg patient, according to a transplantation of 1 × 106 cells per kg body weight.
The biologic properties of UC-MSCs, such as plastic adherence, morphology, specific surface antigens, and multipotent differentiation potential, were retained after clinical-scale expansion. The cells formed a monolayer of homogenous bipolar spindle-like cells with a whirlpool-like array. Cell size underwent no obvious change, as evidenced by the consistent harvested cell density after clinical-scale expansion. A set of typical surface phenotype analysis by flow cytometry confirmed that more than 95% of the cells reacted with antibodies against CD73, CD90, and CD105 and less than 1% reacted with CD45, CD34, CD11b, CD19, and HLA-DR. These cells could be induced to differentiate along both osteogenic and adipogenic lineages. These results are in accordance with the criteria proposed by the International Society for Cellular Therapy (ISCT) to define human MSCs (5).
No antibiotics were added in any isolation or expansion medium, except for the transfer medium. Sterility and mycoplasma assays were routinely performed and microbiological contamination was consistently excluded. There were no bacterial, fungal, or mycoplasma contaminations observed in any of our 10 cultures. This may largely be attributed to the fact that we employed a rigorous donor screening procedure, umbilical cords that were obtained from cesarean section deliveries only, and a simplistic isolation method, all of which minimized the risk of microbial contamination. Endotoxin levels of the 10 final products were lower than 2 EU/ml. The low endotoxin content and an additional Gram stain might help to improve safety of the final product that may be released before obtaining final bacterial culture result. Fetal bovine serum sourced from New Zealand was still used for therapeutic-scale expansion of MSCs. It remains to be determined whether FBS can be replaced by serum-free growth media.
Another safety concern relates to malignant transformation of ex vivo cultured MSCs. Human adipose tissue-derived MSCs can undergo spontaneous transformation after long-term in vitro culture (30). In contrast, there are conflicting reports regarding human BM-derived MSCs (2,29). In addition, genomic instability has not yet been reported in other human tissue-derived MSCs. Recently, a set of safety evaluation assays for our ex vivo cultured UC-MSCs was performed by a qualified independent laboratory. The results showed that MSCs from different donors at different passages (P3, P7, and P15) did not grow in soft agar and form tumors in immune-deficient mice. Furthermore, they were able to inhibit the growth of tumors in tumor-bearing nude mice, established by subcutaneously injecting K562 leukemia cell lines. In our previous study, the genetic characterization of MSCs, including karyotype, telomerase activity, and the expression of human telomerase reverse transcriptase (hTERT), v-Ha-ras Harvey rat sarcoma viral oncogene homolog (H-ras), myelocytomatosis viral oncogene homolog (c-myc), BMI1 polycomb ring finger oncogene (Bmi-1), cyclin-dependent kinase inhibitor 2A (p16), and tumor protein p53 (p53) was investigated. No abnormal signs were observed in any sample (unpublished data).
In comparison with MSCs derived from BM, banking of UC-MSCs has several advantages, such as an abundant tissue source, fewer ethical issues, painless procedures, and lower risk of viral contamination. Our previous study indicates that the colony-forming unit fibroblast (CFU-F) frequency is significantly higher in freshly isolated nucleated cells of umbilical cord than it is in bone marrow mononuclear cells (24). The high frequency of MSCs and shorter doubling time minimize the duration of purity and expansion to yield a reasonable number of potent cells. Furthermore, several experiments have also indicated that MSCs derived from different tissue sources possess different potencies (21,31). Banking of MSCs from different sources for research or future therapeutic use is important. In addition, umbilical cord is a rich source of stem/progenitor cells (9). In addition to MSCs, endothelial colony forming progenitor cells have also been isolated from umbilical cords (26), and these also show promising therapeutic potential in regenerative medicine (12).
With a small number of cells and a limited number of injections, the administration of UC-MSCs was demonstrated intravenously and intrathecally without any immediate adverse events. Given the safety of UC-MSCs for clinical applications, it is necessary to do a longer follow up. The patient with multiple sclerosis reported in the study and some expanded samples in a clinical trial for multiple sclerosis (ClinicalTrials.gov Identifier: NCT00962923) are all under our tracking so far. Similar to previous reports (10,17,32,33), there was no signs of immunologic response and no evidences in ectopic tissue formation with the dosage escalation and frequences of UC-MSCs used in these patients.
In summary, umbilical cords represent an appealing source of MSCs for cell therapy. We established a standardized process for banking UC-MSCs for clinical use. The data obtained indicate that the banked UC-MSCs are homologous, genetically stable, safe, and biologically active.
Footnotes
Acknowledgments
This study was supported by 863 projects from Ministry Science & Technology of China (2006AA02A110), National Natural Science Foundation of China (30570357, 30700285, and 30600238), Tianjin University of Commerce introduced talent funding (100101Q), and Tianjin Municipal Science and Technology Commission (06YFSZSF01300, 07JCYBJC11200, and 08ZCKFGX09400). The authors declare no conflicts of interest.
