Abstract
Mesenchymal stem cells (MSCs) are multipotential nonhematopoietic progenitors and are capable of differentiating into several tissues of mesenchymal origin. We have shown that bone marrow-derived MSCs from both SLE patients and lupus-prone MRL/lpr mice are defective structurally and functionally. Here we observe the long-term safety and efficacy of allogeneic MSC transplantation (MSCT) in treatment-resistant SLE patients. Eighty-seven patients with persistently active SLE who were refractory to standard treatment or had life-threatening visceral involvement were enrolled. Allogeneic bone marrow or umbilical cord-derived MSCs were harvested and infused intravenously (1 × 106 cells/kg of body weight). Primary outcomes were rates of survival, disease remission and relapse, as well as transplantation-related adverse events. Secondary outcomes included SLE disease activity index (SLEDAI) and serologic features. During the 4-year follow-up and with a mean follow-up period of 27 months, the overall rate of survival was 94% (82/87). Complete clinical remission rate was 28% at 1 year (23/83), 31% at 2 years (12/39), 42% at 3 years (5/12), and 50% at 4 years (3/6). Rates of relapse were 12% (10/83) at 1 year, 18% (7/39) at 2 years, 17% (2/12) at 3 years, and 17% (1/6) at 4 years. The overall rate of relapse was 23% (20/87). Disease activity declined as revealed by significant changes in the SLEDAI score, levels of serum autoantibodies, albumin, and complements. A total of five patients (6%) died after MSCT from non-treatment-related events in the 4-year follow-up, and no transplantation-related adverse event was observed. Allogeneic MSCT resulted in the induction of clinical remission and improvement in organ dysfunction in drug-resistant SLE patients.
Introduction
Systemic lupus erythematosus (SLE), a chronic autoimmune disease with significant morbidity and mortality, is characterized by highly diverse clinical manifestations and can affect any organs of the body (25). The conventio nal treatment of SLE mainly relies on high doses of corticosteroids, cyclophosphamide (CYC), and other immunosuppressive and biological agents. Although these drugs have led to markedly improved outcome in SLE (5,13), disease control remains unsatisfactory in a subset of patients (10,20). Moreover, the conventional therapeutic modalities may cause severe side effects such as infection, ovarian failure, and secondary malignancy (14,23,26), which remain as the important causes of mortality in SLE patients. Thus, more effective, but less toxic, treatments are needed.
Mesenchymal stem cells (MSCs), multipotential nonhematopoietic progenitors, can be isolated from various tissues and are capable of differentiating into several tissues of mesenchymal origin (4,15). These pluripotent cells can also differentiate into endoderm and neuroectoderm lineages, including neurons, hepatocytes, and cardiocytes (7,27,28). MSCs can modulate allogeneic immune cell responses including T-cells, B-cells, natural killer (NK) cells, and dendritic cells (12,22,30). In addition, inherently low immunogenicity due to lack of costimulatory molecule expression enables MSCs to escape alloantigen recognition. These properties make MSCs promising candidate cells for preventing rejection in organ transplantation and also as a promising potential treatment of autoimmune disease, including SLE.
In previous investigations, we showed that bone marrow-derived mesenchymal stem cells (BM-MSCs) from both SLE patients and lupus-prone Murphy Roths Large-lymphoproliferation spontaneous mutation (MRL/lpr) mice are defective structurally and functionally (32,34), which has been also confirmed by other studies (6,24). We therefore hypothesized that defects in lupus MSCs may contribute to disease pathology and consequently present a novel therapy target. If this hypothesis is true, then allogeneic, rather than autologous, MSC transplantation might prove to be an effective treatment for SLE. Consistent with our hypothesis, Carrion et al. showed that autologous bone marrow MSC treatment did not improve disease activity in two SLE patients (3). Consequently, after our preclinical assessments verified the potential therapeutic effects and possible mechanisms of human BM-MSC transplantation (BM-MSCT) on ameliorating autoimmune progression in MRL/lpr mice (32,35), we initiated the studies of allogeneic MSCT for lupus treatment, and the results of two short-term follow-up studies were encouraging (19,33), but the small number of patients and the short-term follow-up limit any conclusions regarding long-term safety and efficacy. We therefore carried out this 4-year follow-up study that included 87 severe and refractory SLE patients.
Materials and Methods
Study Design and Patient Enrollment
From March 2007 to April 2010, 87 SLE patients refractory to standard therapies were enrolled in an allogeneic MSCT trial at the Nanjing Drum Tower Hospital after signing an informed consent (Table 1). The study was approved by the Ethics Committee at The Drum Tower Hospital of Nanjing University Medical School. All enrolled patients had fulfilled at least 4 of 11 American College of Rheumatology criteria for the classification of SLE (29). The eligibility criteria included progressive and active disease with the modified Safety of Estrogens in Lupus Erythematosus National Assessment version of the Systemic Lupus Erythematosus Disease Activity Index (SELENA-SLEDAI) score of ≥8 (1). All the patients were unresponsive to conventional immunosuppressive drugs [CYC (Jiangsu Hengrui Medicine, Co. Ltd., Jiangsu, China) 500–1,000 mg/m2/month, mycophenolate mofetil 2 g/day (F. Hoffmann-La Roche Ltd., Shanghai, China), azathioprine 200 mg/day, leflunomide 20 mg/day (Soochow Changzheng-Cinkate Corporation Co. Ltd., Soochow, China), alone or in combination for more than 6 months] or had continuing requirement for daily dosage of ≥20 mg of prednisone (Tianjin Pharmaceutical Group Co., Ltd., Tianjin, China) or its equivalent. Patients were also included if they underwent severe and refractory cytopenia, although with a SELENA-SLEDAI score of <8. Patients were excluded from this study if they had uncontrolled infection, mean pulmonary artery pressure of ≥50 mmHg, failure of one or more organs, pregnant, or lactating.
MSC Transplantation
Bone marrow MSCs were isolated from bone marrow aspirates obtained from healthy donors (26 donors: 11 female and 15 female, average age 30.4±9.0 years; range 16–43 years) with informed consent (32). Briefly, 20 ml of bone marrow was aspirated on the posterior iliac crest. Bone marrow mononuclear cells were separated by Ficoll (Tianjin Haoyang Biotech, Inc., Tianjin, China) density centrifugation. Mononuclear cells were cultured in a 175-cm2 flask (Corning, Inc., Corning, NY, USA) with low-glucose Dulbecco's modified Eagle's medium (DMEM-LG; Gibco, Gaithersburg, MD, USA) containing 10% fetal bovine serum (FBS; Hyclone, Logan, UT, USA) and 1% penicillin–streptomycin (Gibco) in a humidified incubator at 37°C under 5% CO2. Nonadherent cells were removed when the medium was exchanged at the third day. When the primary MSCs had expanded to 80% confluence, they were harvested and expanded to reach treatment dose based on the body weight of the recipient. Umbilical cord (UC) MSCs (11 females, 5 males) were prepared by the Stem Cell Center of Jiangsu Province. Fresh UC was obtained from 16 informed healthy mothers (ages 20–30 years) in local maternity hospitals after normal deliveries (33). The cords were rinsed by phosphate-buffered saline (PBS; Wuhan Boster Biological Technology, Co. Ltd., Wuhan, China) in penicillin and streptomycin, the cord blood being removed during this process. The washed cords were cut into 1-mm2-sized pieces and floated in DMEM-LG containing 10% FBS. The pieces of cord were subsequently incubated at 37°C in humid air with 5% CO2. Nonadherent cells were removed by washing. The medium was replaced every 3 days after the initial plating. When well-developed colonies of fibroblast-like cells appeared after 10 days, the cultures were trypsinized (Gibco) and passaged into a new flask for further expansion. At about 80–85% confluency, the adherent cells were detached by treatment with 0.125% trypsin and 0.1% EDTA (Gibco). The cells were analyzed for the following surface markers by flow cytometry: cluster of differentiation 73 (CD73), CD105, CD90, CD29, human leukocyte antigen (HLA)-DR, CD45, CD34, CD14, and CD79 (all the antibodies were from eBioscience, Inc., San Diego, CA, USA).
To induce adipogenic differentiation, MSCs were cultured in culture medium supplemented with 1 × 106 M dexamethasone, 0.02 mg/ml indomethacin, and 10 μg/ml insulin (all Sigma-Aldrich). After 12 days of culture, cell differentiation into adipocytes was confirmed by Oil Red O staining (Sigma-Aldrich).
To induce osteogenic differentiation, adherent cells were grown at 3 × 104 cells/cm2 in culture medium with 10−7 M dexamethasone, 50 μg/ml ascorbic acid, and 10 mM β-glycerophosphate (all Sigma-Aldrich). After 21 days of culture, calcium deposits were detected by Alizarin Red staining (Sigma-Aldrich).
Criteria for release of MSCs for clinical use included absence of visible clumps, spindle-shaped morphology, and absence of contamination by pathogens (as documented by aerobic and anaerobic cultures) as well as by virus for hepatitis B surface antigen, hepatitis B core antibody, hepatitis C virus antibody, human immunodeficiency virus antibodies I and II, cytomegalovirus IgM, and syphilis antibody [as determined by enzyme-linked immunosorbent assays (ELISA); Dakewe Biotech Inc., Co. Ltd., Shenzhen, China], cell viability greater than 92% [as determined by trypan blue (Sigma-Aldrich, St. Louis, MO, USA) testing]. We used good manufacturing practice (GMP) conditions and clinical-grade reagents for preparation of the cells. The trial was conducted in compliance with current good clinical practice standards and in accordance with the principles set forth under the Declaration of Helsinki (1989).
Before allogeneic MSC treatment, patients were administered with CYC (10 mg/kg/day; Jiangsu Hengrui Medicine, Co. Ltd.) intravenously on days −4, −3, and −2 to inhibit active lymphocytes responses. Serum levels of albumin and creatinine were detected by automatic biochemical analyzer (Beckman Coulter, Inc., Brea, CA, USA), with normal ranges from 3.5 to 5.5 g/dl for albumin and from 0.5 to 1.2 mg/dl for creatinine, respectively. White blood cell number was detected by automatic cell analyzer (Hanfang Medical Instrument, Co. Ltd., Wuhan, China) and with 4,000–10,000/μl as a normal range. If the patient underwent severe baseline disease condition such as low serum albumin (<2.5 g/dl), or high serum creatinine (>3.4 mg/dl), or severe leucopenia (white blood cell count < 2,000/μl), the dose of CYC would be discontinued. All the patients received a single intravenous infusion of BM- or UC-derived MSCs (1 × 106 cells/kg of body weight per infusion). All the infused MSCs were derived from passage 2 to passage 5. Multiple infusions of MSCs were permitted if disease activity was not controlled satisfactorily or with lupus relapse, employing the same dose (but not necessarily the same source) of allogeneic MSCs each infusion.
Assessment
After MSCT, all patients returned for scheduled follow-up at 1, 3, 6, and 12 months and then yearly thereafter. Physical examinations, including but not limited to temperature, blood pressures, pulse, swelling and pain of joints, myodynamia, and erythema, were performed. Serologic tests, including serum levels of creatinine, albumin, urea nitrogen, protein [all by automatic biochemical analyzer (Beckman Coulter)], anti- double-stranded DNA (dsDNA), and antinuclear antibody (ANA) (both by ELISA, Euroimmun Medical Laboratory Diagnostics Stock Company, Lubeck, Germany) were determined at each visit time. In addition, regular blood and urine tests were also assessed by automatic analyzer (Hanfang Medical Instrument, Co. Ltd.). Technetium-99m-diethylenetriaminepentacetic acid (99mTc-DTPA; 6–7 mIU; Nanjing Shengke Biotech. Co. Ltd., Nanjing, China) was infused intravenously into the patients and then the speed and quantity of its clearance were measured and collected dynamically. Glomerular filtration rate (GFR) was calculated by analyzing the collected images and data. Primary outcomes were overall survival and disease remission. Complete clinical remission was defined as SELENA-SLEDAI score of <3 and steroid requirement of ≤10 mg/day of prednisone or its equivalent, combined with British Isles Lupus Assessment Group (BILAG) D scores or better in all organs but not hematological system (11,21). Complete remission for hematological system was defined as white blood cell count > 4,000/μl, hemoglobin > 11 g/dl, platelet count > 100,000/μl, and steroid maintenance ≤10 mg/day of prednisone or its equivalent. Disease relapse was defined as an increase in SELENA-SLEDAI score of ≥3 from the previous visit or experience one new domain with a BILAG A score or two new domains with a BILAG B score after a previous response (11,21). Secondary characteristics included SELENA-SLEDAI score, lupus serologic changes, systemic evaluations such as renal functional indexes, and hematological involvements. Transplantation-related mortality included all deaths associated with MSC transplantation, except those related to recurrence of underlying disease. The investigators assessed and recorded adverse events and their severity throughout the study.
Statistical Analysis
Rates of overall survival, complete remission, and disease relapse at different visit times were analyzed by a Kaplan–Meier survival curve. Significance of influential factors for survival was assessed by Cox regression. Pairwise comparisons of pre- and post-MSCT variables were analyzed by paired t test analysis. Serial data were compared within and between groups by repeated measures one-way analysis of variance (ANOVA, with or without CYC pretreatment, BM- or UC-derived MSC infusion), all using statistical software (SPSS 13.0; IBM, Armonk, NY, USA). Statistical significance was set at p < 0.05 and was adjusted by the Bonferroni method to allow for multiple comparisons.
Results
Cell Characteristics
Immunophenotype analysis indicated that the cultured MSCs had high expression of CD73, CD105, CD90, and CD29 (>90%) and low expression of CD45, CD34, CD14, CD79, and HLA-DR (<2%). In vitro osteogenic and adipogenic induction of MSCs was also confirmed.
Participant Characteristics
Eighty-seven patients were enrolled in this trial, and all patients underwent MSCT. The mean follow-up period was 27 months (range 12–48 months). The patient demographics and medication history are shown in Table 1. Fifty-one patients (59%) were given intravenous CYC for pretreatment before MSC infusion, and 36 (41%) patients received no CYC. At the time of data collection, 69 patients had received a single MSCT (23 from BM and 46 from UC), and the other 18 had received multiple MSCT (initially 3 from BM and 15 from UC).
Patient Demographics and Pretransplantation History

GFR, glomerular filtration rate.
Clinical Outcomes
With a mean follow-up period of 27 months, the overall survival rate for all patients was 94% (82/87). Survival rate was not correlated with disease duration (p = 0.60, 95% confidence interval = 0.99–1.01), initial SELENA-SLEDAI score (p = 0.24, 95% confidence interval = 0.96– 1.20), BM- or UC-derived MSC infusions (p = 0.93, 95% confidence interval = 0.11–11.34), or CYC conditioning regimen (p = 0.22, 95% confidence interval = 0.04–2.08) by Cox regression. The probability of complete clinical remission was 28% at 1 year (23/83), 31% at 2 years (12/39), 42% at 3 years (5/12), and 50% at 4 years (3/6), respectively (Fig. 1). The longest continuous duration of remission was 42 months. Time to remission varied from 3 to 24 months (mean 9.4 months).
Rate of relapse was 12% (10/83) at 1 year, 18% (7/39) at 2 years, 17% (2/12) at 3 years, and 17% (1/6) at 4 years. The overall rate of relapse was 23% (20/87), and the time to relapse differed from 3 to 42 months (mean 16 months). Disease relapse was not correlated with duration (p = 0.54, 95% confidence interval = 0.98–1.01), baseline SELENA-SLEDAI score (p = 0.17, 95% confidence interval = 0.97–1.16), or CYC conditioning regimen (p = 0.14, 95% confidence interval = 0.13–1.31) by Cox regression analysis. For all 20 relapse events, 15 had suspected underlying causes (infection in nine patients, drug withdrawal in five patients, and bone fracture in one patient). Only a minority of patients (7/87, 8%) had no clinical response to allogeneic MSCT during 1–4 years of follow-up.
Eighteen of the patients who did not have sustained responses or had relapse after the first MSC infusion were treated with subsequent infusions of the same dose, but not necessarily the same type of infusion, each time, without CYC for pretreatment (13 had previously been CYC pretreated). The average time of the second MSCT is 10.0±6.3 months (3–28 months). Sixteen patients received two transplants (4 received BM followed by UC, 1 received UC followed by BM, and 11 received two UC transplants), one patient received three transplants (all UC), and one patient received four transplants (two BM followed by two UC) from different donors. Two patients died; two other patients achieved complete clinical remission 3 and 6 months after the second MSCT; seven patients showed response but did not reach the criteria of complete clinical remission, while an additional seven patients were refractory to multiple doses or underwent lupus relapses.

Cumulative survival and remission after MSCT. Probability of survival (A) and clinical remission (B) in lupus patients undergoing mesenchymal stem cell transplantation (MSCT) by Kaplan–Meier survival analysis. “No. at Risk” refers to number of patients at that time point.
Serum albumin and complement 3 levels improved after MSCT. Levels of serum albumin normalized at 3 months posttransplantation for most patients and remained stable thereafter [mean (SD): 2.56 (0.47) g/dl before MSCT, 2.92 (0.48) g/dl at 3 months, 3.14 (0.42) g/dl at 6 months, 3.38 (0.50) g/dl at 1 year, 3.59 (0.56) g/dl at 2 years, 3.50 (0.57) g/dl at 3 years, 3.83 (0.66) g/dl at 4 years, p < 0.001 at 3 months, 6 months,1 year, and 2 years, p = 0.010 at 3 years] (Fig. 2A). Complement 3 increased at 3 months post-MSCT (p = 0.001) and achieved nearly normal levels at 6 months visit [mean (SD): 49.05 (16.50) mg/dl before MSCT, 62.13 (21.61) mg/dl at 3 months, 70.67 (21.35) mg/dl at 6 months, 76.06 (21.75) mg/dl at 1 year, 80.13 (17.06) mg/dl at 2 years, 87.71 (17.90) mg/dl at 3 years, 90.00 (20.83) mg/dl at 4 months, p < 0.001 at 6 months, 1 year, and 2 years, p = 0.002 at 3 years] (Fig. 2B). Serum anti-dsDNA antibody decreased significantly at 3 and 6 months of follow-up [mean (SEM): 543.4 (110.8) U/ml before MSCT, 398.2 (98.9) U/ml at 3 months, 480.1 (125.6) U/ml at 6 months, 475.5 (163.6) U/ml at 1 year, 394.0 (110.9) U/ml at 2 years, p = 0.021 at 3 months, p = 0.019 at 6 months] (Fig. 2C), along with decreased antinuclear antibody (ANA) at 6 months [p = 0.045, mean (SEM): 5.81 (0.44) before MSCT, 4.99 (0.46) at 3 months, 4.86 (0.59) at 6 months, 4.37 (0.64) at 1 year, 3.95 (0.68) at 2 years] (Fig. 2D).

Serology and complement levels before and after (MSCT). Serum albumin (A) and complement C3 (B) increased after MSCT. Serum anti-double-strand DNA (dsDNA) antibody and antinuclear antibody (ANA) declined after MSCT (C and D). The black bars represent mean value. *p < 0.05, **p < 0.01.
SELENA-SLEDAI scores significantly improved for up to 4 years after MSCT (Fig. 3A). Renal involvement assessed by 24-h proteinuria improved 6 months, 1 year, 2 years, 3 years, and 4 years after MSCT [mean (SD): 2.71 (1.19) g before MSCT, 2.02 (1.10) g at 1 month, 1.79 (1.29) g at 3 months, 1.52 (0.960) g at 6 months, 1.52 (1.01) g at 1 year, 1.53(1.02) at 2 years, 1.12 (1.02) g at 3 years, 1.32 (1.52) g at 4 years] (Fig. 3B). For those with abnormal levels of serum creatinine and urea nitrogen at baseline (n = 31), these index improved significantly 1 and 2 years after MSCT [mean (SEM), for serum creatinine: 1.98 (0.26) mg/dl versus 2.28 (0.19) mg/dl at 1 year, n = 31, p = 0.029; 2.02 (0.25) mg/dl versus 2.37 (0.26) mg/dl at 2 years, n = 22, p = 0.026; for serum urea nitrogen: 11.73 (0.91) mol/l versus 18.27 (1.70) mol/l at 1 year, n = 31, p < 0.001; 11.07 (1.01) mol/l versus 17.35 (2.11) mol/l at 2 years, n = 21, p < 0.001] (Fig. 3C, D). Glomerular filtration rate (GFR) was calculated in 63 patients before MSCT, in which 10 patients had normal levels at baseline and did not have additional assessment after MSCT. GFR for the other 53 patients improved significantly 1 year after MSCT [mean (SD), 51.5 (21.5) ml/min before MSCT, 63.4 (26.2) ml/min at 1 year, p < 0.01]. For 25 patients who conducted GFR at 2 years of follow-up, we also found a statistical improvement [mean (SD), 56.1 (20.6) ml/min before MSCT, 67.8 (28.6) ml/min at 2 years, p = 0.03].

Disease activity score and serum measurements following MSCT. Systemic lupus erythematosus (SLE) Disease Activity Score (Safety of Estrogens in Lupus Erythematosus National Assessment version of the Systemic Lupus Erythematosus Disease Activity Index; SELENA-SLEDAI) (A) and proteinuria (B) improved before and after MSCT. Serum urea nitrogen (C) and creatinine (D) decreased significantly 1 and 2 years after MSCT. The black bars represent mean value. *p < 0.05, **p < 0.01.
Thirty-six patients had refractory hypohemoglobinemia, in which 29 combined with severe thrombocytopenia. Hemoglobin and platelet counts were found increased since 1 month after MSCT and remained stable for more than 2 years (Fig. 4). Seizure did not recur in three patients following MSCT, in parallel with amelioration of lupus nephritis and satisfactory control of hypertension. Aminotransferase levels remained normal in three patients with autoimmune hepatitis after MSC infusion including no recurrence of upper gastrointestinal hemorrhage in one patient who had liver cirrhosis and experienced two episodes of severe hemorrhage 1 month before MSCT. Diffuse alveolar hemorrhage (DAH) associated with active lupus in three patients was satisfactorily controlled by MSC transplantation. An additional two patients received MSCT for interstitial pneumonia: one had improvement in clinical symptoms, and the other required a second MSCT for unsatisfactory control of the disease. No differences were found in disease remission, SELENA-SLEDAI score, proteinuria, and serum albumin between those with or without CYC pretreatment, those infused with bone marrow- or umbilical cord-derived MSCs (Figs. 5 and 6).

Hemoglobin and platelet counts following MSCT. Hemoglobin level (A) and platelet (PLT) counts (B) increased significantly after MSCT. The black bars represent mean value. *p < 0.05, **p < 0.01.

Effect of cyclophosphamide (CYC) on disease and serum parameters. No differences in disease remission (A), SELENA-SLEDAI score (B), proteinuria (C), and serum albumin (D) were found between those with or without CYC pretreatment.
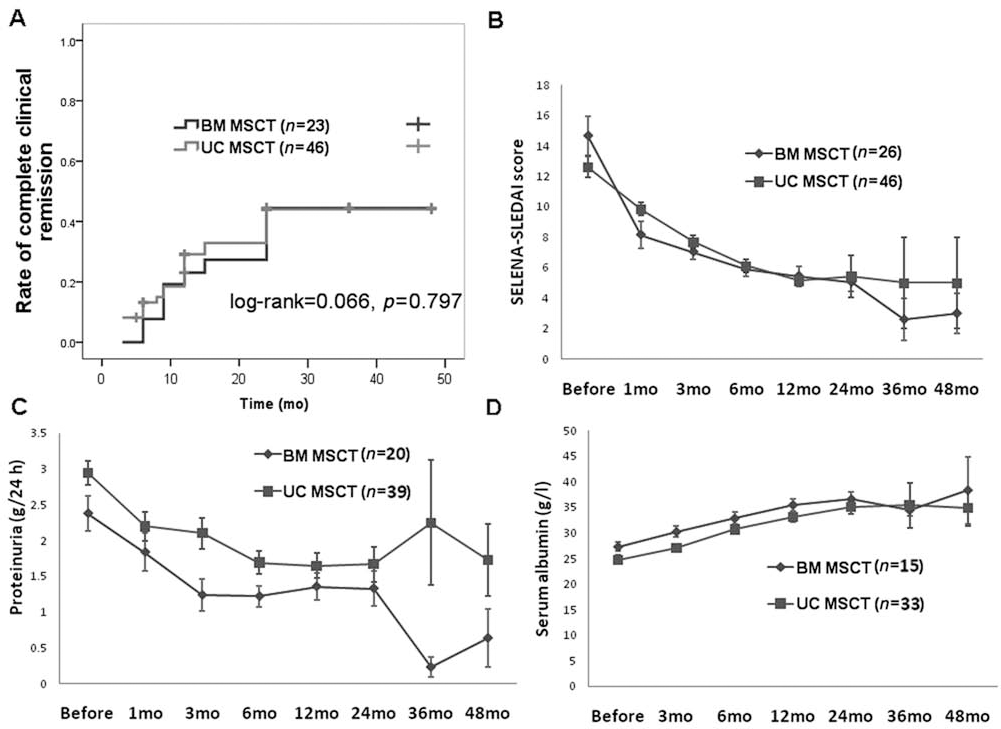
Effect of source of MSCs on disease and serum parameters. Comparisons of disease remission (A), SELENA-SLEDAI score (B), proteinuria (C), and serum albumin (D) between patients transplanted with bone marrow (BM)- or umbilical cord (UC)-derived MSCs.
The dose of prednisone was tapered at 5–10 mg every 2 weeks in the first month following transplantation according to clinical status and laboratory indicators of disease amelioration. During 4 years of visits, all the patients underwent steroid and immunosuppressive drugs tapering after MSC transplantation. Six of 82 (7%) patients had discontinued immunosuppressants and only took prednisone at 5–10 mg/day for maintenance therapy. Dose of immunosuppressive drugs were tapered to maintenance level (CYC 0.6–0.8 g/2–3months, or mycophenolate mofetil 0.5 g/day or leflunomide 10 mg/day) for 53/82 (65%) patients at the last follow-up (Table 2).
Treatments Used Pre- and Postallogeneic MSCT

MSCT, mesenchymal stem cell transplantation.
Two patients combined with CYC 0.6 g/month.
Two patients combined with CYC, 0.6 g/month and 0.8 g/month, respectively.
Safety
Five of 87 patients died after MSCT, all judged to be from non-treatment-related events. One patient died 3 months after the second transplantation, associated with acute gastroenteritis and heart failure. A second patient succumbed to a disseminated pulmonary infection and uncontrolled lupus nephritis 6 months after MSCT. One patient had a lupus relapse 8 months after MSC infusion with pulmonary hypertension and died from right-sided heart failure. A fourth patient had had a remission since 3 months of follow-up with a significant decrease in proteinuria and normalized serum albumin, but her disease relapsed 8 months later; she underwent a second MSCT and also achieved remission, but she died from pulmonary embolism 9 months after the second MSCT without evidence of active SLE. The fifth patient, who had active lupus with malar rash, arthralgia, uncontrolled hypertension, rapid deterioration of renal function, hypoproteinemia, and severe proteinuria, died 1 week post-MSCT from uncontrolled progressive disease and acute heart failure.
Two patients experienced diarrhea 3 and 4 months after MSCT, respectively. Two additional patients had moderate herpesvirus infection at 1 and 6 months after MSCT, respectively. One patient experienced agranulocytosis and oral fungus infection 7 days after CYC pretreatment, and her condition improved after conventional therapy. Tuberculosis infection in the lung was found in one patient 9 months after MSCT, and she was given antituberculosis drugs for 9 months. One patient had bone fracture on L3 in an accident 2 months after her third MSCT. One patient underwent myocardial infarction 14 months after MSCT and responded to treatment by percutaneous coronary intervention (PCI), with no lupus relapse. All the adverse events were not considered to be related to the MSC infusions.
Discussion
Systemic delivery of autologous or allogeneic MSCs has been evaluated in patients with graft versus host disease (GVHD), acute myocardial infarction, ischemic stroke, etc., based on their properties of immunomodulation and tissue repair (9,16,17). Our preliminary small-scale and short-term data suggested that allogeneic bone marrow- or umbilical cord-derived MSC transplantation may be a feasible and safe therapy for refractory SLE patients (19,33). However, a larger-scale and longer term study is required to assess safety and efficacy.
Among the 87 SLE patients enrolled, 73 (84%) had nephritis, and a small portion had other systemic involvements. The overall survival rate was 94% (82/87), with a mean follow-up duration of 27 months. Five patients died of active lupus that was refractory to conventional therapies as well as MSCT. No major toxicity related to MSCT was observed, although some adverse events may have occurred, associated with the CYC conditioning regimen. The study met its primary objective demonstrating the safety of allogeneic MSCT during the infusion and long-term follow-up. In addition, the results of this study provided evidence that allogeneic MSCT demonstrated clinical efficacy in SLE patient refractory to conventional therapies by the decline of disease activity score, improvement of renal function, and systemic involvement.
This study is the first to report long-term follow-up data for allogeneic MSCT in refractory SLE patients. With a similar mean follow-up time (27 months) compared to two studies of autologous stem cell transplantation (ASCT) for SLE (29 months and 26 months, respectively) (2,11), the rate of complete clinical remission was similar [50% at 4 years in allogeneic MSCT; 50% at 5 years in ASCT of Burt et al. study (2); 66% at 6 months in ASCT of Jayne et al. study (11)]. Importantly, the overall survival rate was higher in the current study (94% compared to 84% and 62% in ASCT, respectively), and there was no transplantation-related mortality (0 in MSCT compared to 4% and 12% in ASCT, respectively). Additionally, the overall rate of relapse was lower (23% in MSCT compared to 28% and 32% in ASCT, respectively); all indicated that allogeneic MSCT possesses therapeutic potential to induce refractory lupus remission and higher safety. However, all the three studies were nonrandomized, and it was difficult to compare exactly due to the heterogeneous disease manifestation at baseline.
Of 87 patients infused with a single allogeneic MSCT, only 8% of patients had no clinical response, suggesting that the current infused cell dose (106/kg of body weight) is sufficient to facilitate a clinical effect. We chose a low dose of CYC for precondition regimen to inhibit lymphocyte activation in these patients. The dose of CYC was much lower in MSCT (30 mg/kg) than in ASCT (200 mg/ kg), and white blood counts were not less than 1,000/μl after CYC treatment. Furthermore, a part of the population was not given CYC pre-MSCT for the severe disease conditions at baseline. They also responded well to MSC infusions, and no difference in disease remission was found between those with or without CYC pretreatment after 4 years of visits. Moreover, our previous study showed that the addition of CYC could not enhance therapeutic effect and immunomodulation in lupus mice (35), all suggesting that the clinical effect was not related to CYC.
While the present results provide reassurance regarding the safety and clinical efficacy of allogeneic MSCT for refractory SLE, especially for lupus nephritis, more studies are required to understand the mechanism of action of this approach. Our previous lupus animal study also demonstrated that carboxyfluorescein diacetate succinimidyl ester (CFSE)-labeled allogeneic MSCs could home to kidney, lung, liver, and spleen tissues of lupus-prone mice (8). Meanwhile, MSCs are reported to differentiate into functional renal tubular epithelial cells and hepatocytes in vitro and in vivo and contribute to tissue repair (18,31). Additionally, MSCs exerted complex paracrine and endocrine actions including the secretion of growth factors and cytokines, modulation of immune response, and antiapoptotic and anti-inflammatory effects. Our previous clinical studies had shown that allogeneic MSC transplantation could upregulate peripheral cluster of differentiation 4-positive forkhead box P3-positive (CD4+Foxp3+) regulatory T-cells in SLE patients (19,33), all of which may contribute to organ repair and immune regulation (22).
As this study was a clinical trial, we were unable to label MSCs, and thus, whether a positive response to MSCT is associated with specific tissue localization and healing is still unclear. Our previous work has shown that inhibition of Th2-mediated humoral immunity may also play a role in the effectiveness of MSCT (33), and we hypothesize that MSCs may play a role individually or synergistically in SLE patients. Certainly, further experiment is required to investigate the mechanistic issues surrounding the use of MSCT.
This study focused on the safety and efficacy of allogeneic MSCT for severe and drug refractory SLE patients but was not a randomized controlled trial. We could not compare quantitatively the effect of MSCT to the conventional treatments with corticosteroids and other immunosuppressive drugs. In our opinion, it is difficult to design an RCT study in refractory SLE patients. Next, we will consider to perform a randomized, double-blind, and multicenter-controlled study in China to assess the safety and efficacy of MSCT in newly diagnosed SLE patients or those without previous formal treatments to further examine whether the adding of MSCT is prior to conventional therapies only. In addition, this study involved only a single center with patients all being Chinese in ethnicity, and thus the results should not be extrapolated to SLE patients of other ethnic origins. However, based on our long-term follow-up and demonstrated safety profile of this cell-based therapy, more clinical trials in multiple centers are needed to determine the efficacy of MSCT for larger groups of SLE patients.
This trial provides evidence that allogeneic MSCT may be performed safely in treating refractory and active SLE patients, resulting in disease clinical remission and improvement of organ dysfunction. MSCT as a new cell therapy could be introduced for treatment of refractory and severe SLE patients.
Footnotes
Acknowledgment
Dr. Sun's work was supported by the Major International (Regional) Joint Research Project (No. 81120108021), National Natural Science Foundation of China (No. 30972736, No. 81273304), Jiangsu Province Natural Science Foundation (BK2009034), and Jiangsu Province Kejiao Xingwei Program. This work was supported in part by the Intramural Research Program of the National Institute of Dental and Craniofacial Research, National Institutes of Health (Dr. Chen). The authors declare no conflict of interest.
