Abstract
We previously reported the therapeutic potential of human peripheral blood (hPB) CD34+ cells for bone fracture healing via vasculogenesis/angiogenesis and osteogenesis. Transplantation of not only hPB CD34+ cells but also hPB total mononuclear cells (MNCs) has shown their therapeutic efficiency for enhancing ischemic neovascularization. Compared with transplantation of purified hPB CD34+ cells, transplantation of hPB MNCs is more attractive due to its simple method of cell isolation and inexpensive cost performance in the clinical setting. Thus, in this report, we attempted to test a hypothesis that granulocyte colony-stimulating factor-mobilized (GM) hPB MNC transplantation could also contribute to fracture healing via vasculogenesis/angiogenesis and osteogenesis. Nude rats with unhealing fractures received local administration of the following materials with atelocollagen: 1 × 107 GM hPB MNCs (Hi group), 1 × 106 GM hPB MNCs (Lo group), or PBS (PBS group). Immunohistochemistry and real-time reverse transcriptase-polymerase chain reaction (RT-PCR) demonstrated human cell-derived vasculogenesis and osteogenesis in the Hi and Lo groups, but not in the PBS group at week 1. Intrinsic angiogenesis and osteogenesis assessed by rat capillary, osteoblast density, and real-time RT-PCR analysis was significantly enhanced in the Hi group compared to the other groups. Blood flow assessment by laser doppler perfusion imaging showed a significantly higher blood flow ratio at week 1 in the Hi group compared with the other groups. Morphological fracture healing was radiographically and histologically confirmed in about 30% of animals in the Hi group at week 8, whereas all animals in the other groups resulted in nonunion. Local transplantation of GM hPB MNCs contributes to fracture healing via vasculogenesis/angiogenesis and osteogenesis.
Introduction
Recent progress in human embryonic and adult stem cell research has been reported in various fields, and bone formation and regeneration has received much attention as a target for regenerative medicine because of the capacity of stem cells to self-renew and differentiate into various types of adult cells or tissues (1,37,49).
Deficient bone formation after fracture is mainly caused by the impairment of neovascularization and results in delayed union or persistent nonunion, leading to morbidity and functional limitations for patients (8,13,30,31,38). Recently, advancements in microsurgical techniques led to the development of vascularized bone grafting, which is more successful in fracture healing than traditional autologous bone grafting. However, this procedure is technically challenging and invasive. Therefore, reduced invasion and more effective treatments for bone repair are still required.
We previously reported that transplantation of human peripheral blood (hPB) CD34+ cells, an endothelial/ hematopoietic progenitor cell-enriched cell population (4), contributes to fracture healing via vasculogenesis/ angiogenesis and osteogenesis in an immunodeficient rat model of an unhealing bone fracture (31,33,34). We demonstrated that hPB CD34+ cells, which contain osteo/endothelial progenitor cells and were recruited to the fracture site following systemic delivery, developed a favorable environment for fracture healing by enhancing vasculogenesis and osteogenesis, and finally led to functional repair from fracture (31). Moreover, local administration of granulocyte colony-stimulating factor-mobilized (GM) hPB CD34+ cells also demonstrated a contribution to fracture healing via vasculogenesis and osteogenesis in a dose-dependent manner (34).
In the field of revascularization, transplantation of not only hPB CD34+ cells but also hPB total mononuclear cells (MNCs) for hind limb ischemia or myocardial ischemia has shown their therapeutic efficiency in enhancing neovascularization at ischemic sites (22,43). In the clinical setting, the use of hPB MNCs, which can be easily collected in a short time and at low cost, is more attractive than hPB CD34+ cells. However, some reports have shown a higher therapeutic potency for ischemic neovascularization in CD34+ cells than total MNCs (24,40).
Based on this controversy, we first performed experiments to prove the hypothesis that hPB MNC transplantation may also contribute to fracture healing via vasculogenesis/angiogenesis and osteogenesis. To this end, by comparing the findings in the current study to our previous studies we can discuss which cells are more suitable for cell therapy in fracture healing.
Materials and Methods
Preparation of Human Cells
We purchased GM hPB MNCs of healthy men (21 years old, African-American; 23 years old, African-American; 27 years old, African-American) from Lonza (Valais, Switzerland) and used these cells at random for experiments.
Flow Cytometry Studies
We characterized GM hPB MNCs to assess the content of CD34+ cells by fluorescence-activated cell sorting (FACS) Calibur analyzer (BD Biosciences, San Diego) and CELLQuest software (Becton, Dickinson and Company, Mountain View, CA) as described previously (29).
Animal Model of Unhealing Bone Fracture
Female athymic nude rats (F344/N Jcl rnu/rnu, CLEA Japan, Inc. Tokyo, Japan) aged 8–12 weeks and weighing 150–170 g were used in this study. The rats were fed a standard maintenance diet and provided with water ad libitum.
All surgical procedures were performed under anesthesia and normal sterile conditions. As described previously, a reproducible model of an unhealing bone fracture was created in the femurs of nude rats by cauterizing the periosteum on each side of the fracture according to the methods of Einhorn (11) and Kokubu et al. (26), which led to the nonunion at 8 weeks postfracture.
Cell Transplantation
Following the FACS analysis mentioned above, to adjust CD34+ cell number to the effective dose outlined in the previous study (34), two doses of MNC groups and the PBS group were determined. After the creation of the unhealing fracture, the rats received local administration of the following materials with atelocollagen gel (Koken, Tokyo, Japan), which was used as a bioabsorbable scaffold to retain the cells in the transplanted site (16,18): high-dose GM hPB MNCs (Hi group, 1 × 107) containing 1 × 105 CD34+ cells; low-dose GM hPB MNCs (Lo group, 1 × 106) containing 1 × 104 CD34+ cells suspended in 100 μl of phosphate-buffered saline (PBS); or the same volume of PBS alone (PBS group) (n = 27 in each).
Tissue Harvesting
Three rats in each group were randomly selected and were euthanatized at week 1, 2, and 4. The remaining rats receiving local transplantation were sacrificed at week 8. Bilateral femurs were harvested and quickly embedded as described previously (31,34).
Real-Time Reverse Transcriptase-Polymerase Chain Reaction (RT-PCR) Analysis of RNA Isolated From Tissue Samples at the Perifracture Site
Total RNA was obtained from the rat tissues at perifracture sites at week 1 or 2 using Trizol kit (Life Technologies, Gaithersburg, MD) according to the manufacturer's instructions. RNA isolated at week 1 was analyzed to detect human-specific cell markers, and RNA isolated at week 2 was analyzed to detect expressions of cytokines in recipients. After the first-strand cDNA was synthesized using a RNA LA PCR Kit Ver1.1 (TAKARA BIO Inc., Shiga, Japan), the converted cDNA samples (2 μl) were amplified in triplicate by real-time PCR (ABI PRISM 7700, Applied Biosystems, CA) in a final volume of 20 μl using SYBR Green Master Mix reagent (Applied Biosystems) with gene-specific primers listed in Table 1. Melting curve analysis was performed with Dissociation Curves software (Applied Biosystems). Results were obtained using sequence detection software (ABI PRISM 7700) and the mean cycle threshold (Ct) values were used to calculate gene expressions with normalization to human or rat glyceraldehyde-3-phosphate dehydrogenase (GAPDH).
Specific Primers for Real-Time RT-PCR Amplifications

rVEGF, rat vascular endothelial growth factor; rAng-1, rat angiopoietin-1; rBMP-2, rat bone morphogenetic protein-2; rBMP-4, rat bone morphogenetic protein-4; reNOs, rat endothelial nitric oxide synthase; rbFGF, rat fibroblast growth factor 2; rBMP-7, rat bone morphogenetic protein-7; rGAPDH, rat glyceraldehyde-3-phosphate dehydrogenase; hVE-cad, human vascular endothelial cadherin; hPECAM, human platelet endothelial cell adhesion molecule; hOC, human osteocalcin; hCol1A1, human collagen 1α 1; hGAPDH, human glyceraldehyde-3-phosphate dehydrogenase; bp, base pair.
Immunohistochemical Staining
To detect the transplanted human cells in the rat perifracture sites and to evaluate capillary density and osteoblast (OB) density, immunohistochemistry was performed with the following human- or rat-specific antibodies: biotinylated ulex europaeus agglutinin 1 (UEA-1) (Vector Laboratories, Burlingame, CA) for human endothelial cells (hECs), fluorescein-labeled isolectin B4 (Vector Laboratories) for rat ECs (rECs), human osteocalcin (hOC) (Biomedical Technologies, Inc., Stoughton, MA) for human OBs, and rat osteocalcin (v-19) (rOC) (Santa Cruz Biotechnology, Inc., Santa Cruz, CA) for rat OBs (rOB). The secondary antibodies for each immunostaining are as follows: Alexa Fluor 594-conjugated streptavidin (Jackson Immunoresearch Laboratories, West Grove, PA) for UEA-1 staining, Alexa Fluor 488-conjugated goat anti-rabbit IgG1 (Molecular Probes, Invitrogen Japan K.K., Tokyo, Japan) for hOC, and Alexa Fluor 594-conjugated rabbit anti-goat IgG1 (Molecular Probes) for rOC. 4′,6-Diamidino-2-phenylindole (DAPI) solution was applied for 5 min for nuclear staining.
Capillary or OB density was morphometrically evaluated as the average value in four randomly selected fields of soft tissue in the perifracture site. OBs were recognized as lining or floating cells positive for OC on the new bone surface. All morphometric studies were performed by two blinded examiners.
Physiological Assessment of Tissue Perfusion by Laser Doppler Perfusion Imaging (LDPI)
LDPI (Moor Instrument, Wilmington, DE) (29,46) was used to measure serial blood flow in both legs at 0, 1, 2, and 3 weeks postfracture. The ratio of fractured/intact (contralateral) blood flow was calculated to evaluate the serial blood flow recovery after fracture. The measurement was done under anesthesia with the animals supine and both limbs fully extended.
Radiographic Assessment of the Fracture Healing
Radiographs of the fractured hindlimbs were serially taken on weeks 0, 2, 4, and 8 following creation of the fracture under anesthesia with the animal supine and the limb fully extended. Fracture union was identified by the presence of a bridging callus on two cortices. Radiographic evaluations for fracture healing were examined by three blinded observers.
Histological Assessment of the Fracture Healing
Histological evaluation of the process of endochondral ossification was performed by toluidine blue staining at weeks 2, 4, and 8. The degree of fracture healing was evaluated using a 5-point scale proposed by Allen et al. (3).
DiI Labeling of MNCs and Histochemical Staining in Internal Organ
In order to histologically trace the transplanted cells, we labeled the GM hPB MNCs with fluorescent 1,1′-dioctadecyl-3,3,3′,3′-tetramethylindocarbocyanine iodide (DiI; Molecular Probes) dye. Briefly, GM hPB MNCs were incubated in DiI solution diluted in Hanks' balanced salt solution (HBSS) (Invitrogen) for 10 min at 37°C. The DiI-labeled 1 × 107 MNCs were transplanted with atelocollagen into unhealing fracture sites within the femur of nude rats (n = 3). The rats were euthanatized at week 1 postsurgery, and fractured femurs and internal organs (lung, liver, and spleen) were harvested and quickly embedded in OCT compound to generate fresh frozen samples. The samples were stained by DAPI and DiI-positive cells detected by microscopy.
Biomechanical Analysis of Fracture Healing
Biomechanical evaluation was performed with nude rats of the Hi, Lo, and PBS groups at week 8 postfracture in Kureha Special Laboratory (KSL, Tokyo, Japan). Moreover, we formed another group of animals with transplantation of 1 × 105 CD34+ cells (CD34+ group) (n = 4). We purchased GM hPB CD34+ cells obtained from healthy men (20 years old, Caucasian; 24 years old, Caucasian; 22 years old, African-American) from Lonza (Valais, Switzerland) and used them randomly for the experiment. Briefly, fractured femurs and the contralateral nonfractured femurs were prepared and intramedullary fixation pins were removed prior to the bending test. The standardized 3-point bending test was performed using load torsion and bending tester “MZ-500S” (Maruto Instrument Co., Ltd., Tokyo, Japan). The bending force was applied with cross-head at a speed of 2 mm/min until rupture occurred. The ultimate stress (N), the extrinsic stiffness (N/mm), and the failure energy (N x mm) were interpreted and calculated from the load deflection curve. The relative ratio of the fractured (right) femur to nonfractured (left) femur was calculated in each group and averaged.
Statistical Analysis
The results were statistically analyzed using a software package (Statview 5.0, Abacus Concepts Inc, Berkeley, CA). All values were expressed as mean ± SE. The multiple comparisons among groups were made using a one-way analysis of variance (ANOVA). Post hoc analysis was performed by Fisher's Protected Least Significant Difference test. The comparison of radiological results was performed with a chi-square test. A probability value of p < 0.05 was considered to denote statistical significance.
All experimental procedures were conducted in accordance with the Japanese Physiological Society Guidelines for the care and Use of Laboratory Animals and the study protocol was approved by the Ethics Committee in RIKEN Center for Developmental Biology.
Results
Characterization of GM hPB MNCs
GM hPB MNCs from healthy men used in this study were analyzed by flow cytometry. About 1% of GM hPB MNC fraction was positive for CD34 (Fig. 1). On the basis of this data, we decided that the number of MNCs of the Hi group was 1 × 107 containing 1 × 105 CD34+ cells, which was shown to be an effective dose of CD34+ cells in the previous study (34).

Characterization of granulocyte colony-stimulating factor-mobilized human peripheral blood mononuclear cells (GM hPB MNCs). GM hPB MNCs from the healthy males used in this study were analyzed by flow cytometry. About 1% of the GM hPB MNC fraction was positive for CD34.
Human Cell-Derived Vasculogenesis and Osteogenesis
We performed experiments to characterize the transplanted GM hPB MNCs incorporated into the fracture sites. To histologically validate the phenomenon of human cell-derived vasculogenesis, histochemical staining for UEA-1, a human-specific EC marker, was performed using the tissue samples obtained 1 week after local cell transplantation. Differentiated human ECs in the vasculature of the perifracture area were detected as UEA-1-positive cells in the Hi group but not in the Lo and PBS groups (Fig. 2A–C). These findings suggest that GM hPB MNCs have potential to differentiate into ECs. We performed similar staining using samples obtained 2 weeks after surgery, but could not detect UEA-1-positive cells in all groups. To further verify this phenomenon, real-time RT-PCR analysis of tissue RNA isolated from the perifracture site for human-specific EC markers [human vascular endothelial-cadherin (hVE-cad) and human platelet endothelial cell adhesion molecule (hPECAM)] was performed. The expression level of hVE-cad significantly augmented the Hi and Lo groups compared to the PBS group (Hi, 4746.7 ± 687.6; Lo, 4259.3 ± 637.6; PBS, 0.0 ± 0.00; p < 0.01 for Hi or Lo vs. PBS group) (Fig. 2D). The expression level of hPECAM also significantly augmented the Hi and Lo groups compared to the PBS group (Hi, 4448.1 ± 1121.9; Lo, 4222.8 ± 1550.4; PBS, 0.0 ± 0.00; p < 0.01 for Hi or Lo vs. PBS group) (Fig. 2E).

Fracture site vasculogenesis and osteogenesis derived from GM hPB MNCs. (A–C) Representative double immunostaining for ulex europaeus agglutinin 1 (UEA-1; red) and isolectin B4 (green) using tissue samples of the fracture site at week 1 (original magnification 200x). Differentiated human endothelial cells (ECs) in the vasculature of the perifracture area were detected as UEA-1-positive cells in the Hi (1 × 107 cells) group (A) but not in the Lo [1 × 106 cells) (B) and PBS (C) groups. (D, E) Realtime RT-PCR analysis of tissue RNA isolated from the perifracture site for human-specific EC markers [human vascular endothelial-cadherin (hVE-cad) and human platelet endothelial cell adhesion molecule (hPECAM)]. The expression level of hVE-cad significantly augmented the the Hi and Lo groups compared to the PBS group. There were no significant differences between the Hi and Lo groups (p < 0.01 for Hi or Lo vs. PBS group) (D). The expression level hPECAM also significantly augmented the Hi and Lo groups compared to the PBS group. There were no significant differences between the Hi and Lo groups (p < 0.01 for Hi or Lo vs. PBS group) (E). (F–H) Representative immunostaining for human osteocalcin (hOC; green) using tissue samples of the fracture site at week 1 (original magnification 100x). Differentiated human osteoblasts (OBs) derived from high (F) and low (G) dose of GM hPB MNCs were detected as hOC-positive cells (arrows) in the perifracture area, while hOC-positive cells were not identified in the PBS group (H). (I, J) Real-time RT-PCR analysis of tissue RNA isolated from the perifracture site for human-specific bone related markers [hOC and human collagen, type 1α (hCol1A1)]. The expression level of hOC significantly augmented the Hi and Lo groups compared to the PBS group (p < 0.01 for Hi or Lo vs. PBS group) (I). The expression level of hCol1A1 was also significantly augmented in the Hi and Lo groups compared to the PBS group (p < 0.01 for Hi or Lo vs. PBS group) (J).
To identify osteogenic differentiation of GM hPB MNCs, immunohistochemical staining for human-specific OC, an OB marker, was performed using tissue samples obtained 1 week after cell transplantation. Differentiated human OBs derived from high and low doses of GM hPB MNCs were detected as hOC-positive cells in the perifracture area, while hOC-positive cells were not identified in the PBS group (Fig. 2F–H). We could not detect hOC-positive cells in samples obtained 2 weeks after surgery in all groups. Real-time RT-PCR analysis also demonstrated expression of human-specific bone-related markers [hOC and human collagen type Iα 1 (hCol1A1)] following GM hPB MNC transplantation. The expression level of hOC significantly augmented the Hi and Lo groups compared to the PBS group (Hi, 4452.0 ± 856.3; Lo, 3623.8 ± 805.1; PBS, 0.0 ± 0.00; p < 0.01 for Hi or Lo vs. PBS group) (Fig. 2I). The expression level of hCol1A1 also significantly augmented the Hi and Lo groups compared to the PBS group (Hi, 361.0 ± 129.8; Lo, 307.5 ± 102.2; PBS, 0.0 ± 0.00; p < 0.01 for Hi or Lo vs. PBS group) (Fig. 2J).
These results indicate GM hPB MNCs differentiate into both EC and OB lineages in the fracture-induced environment.
Enhancement of Intrinsic Angiogenesis and Osteogenesis
Enhanced angiogenesis and osteogenesis by the paracrine effect of the transplanted cells on recipients' cells were confirmed by immunostaining for rat-specific markers. Vascular staining with isolectin B4, a rat-specific marker for EC using tissue samples collected 2 weeks postfracture, demonstrated enhancement of intrinsic neovascularization around the endochondral ossification area in the animals treated with high doses of GM hPB MNCs (Fig. 3A). Capillary density was significantly greater in the Hi group compared to the other groups (Hi, 895.8 ± 15.4; Lo, 708.2 ± 30.8; PBS, 634.7 ± 10.0/ mm2; p < 0.05 for Hi vs. Lo or PBS group) (Fig. 3B).

Enhancement of angiogenesis and osteogenesis by recipient cells following GM hPB MNC transplantation. (A) Representative vascular staining with isolectin B4 using tissue samples of perifracture sites collected at week 2 in Hi, Lo, and PBS groups (original magnification 100x). (B) Angiogenesis assessed by capillary density at week 2 was significantly greater in the Hi group compared to the other groups (p < 0.05 for Hi vs. Lo or PBS group) (n = 4 in each group). (C) Representative osteoblast staining with anti-rat osteocalcin using tissue samples of perifracture sites collected at week 2 in Hi, Lo, and PBS groups (original magnification 200x). (D) OB density was significantly greater in the Hi group than in the other groups (p < 0.05 for Hi vs. Lo or PBS group) (n = 4 in each group). (E–H) Gene expression of intrinsic cytokines for angiogenesis and osteogenesis at week 2 (n = 4 in each group). The expression level of rat vascular endothelial growth factor (rVEGF) and rat angiopoietin 1 (rAng1) at week 2 was significantly greater in the Hi group compared with the PBS group, but there was no significant difference between the Lo and PBS groups (p < 0.01 for Hi vs. PBS group) (E, F). The expression level of rat bone morphogenetic protein 2 (rBMP-2) at week 2 was also significantly greater in the Hi group compared with the PBS group, and the Lo group was significantly higher than the PBS group (p < 0.01 for Hi vs. Lo or PBS group, p < 0.05 for Lo vs. PBS group) (G). The expression level of rBMP-4 at week 2 was greater in the Hi group compared with the other groups, but there were no significant differences among all groups (p = ns) (H).
OB staining with anti-rat OC using tissue samples collected 2 weeks postfracture revealed augmentation of intrinsic osteogenesis in the area of new bone formation in the animals treated with the high dose of GM hPB MNCs (Fig. 3C). OB density was significantly greater in the Hi group than in the other groups (Hi, 378.1 ± 36.8; Lo, 228.5 ± 6.7; PBS, 193.2 ± 14.0/mm2; p < 0.05 for Hi vs. Lo or PBS group) (Fig. 3D).
A possible explanation for the enhancement of intrinsic angiogenesis and osteogenesis following MNC therapy is the upregulation of angiogenesis and osteogenesis-related cytokines at the perifracture site. Accordingly, we performed real-time RT-PCR to quantify the expression of rat vascular endothelial growth factor (rVEGF), rat angiopoietin 1 (rAng1), rat bone morphogenic protein 2 (rBMP-2), and rBMP-4 around the fracture sites. The expression level of rVEGF at week 2 was significantly greater in the animals of the Hi group compared with the PBS group, but there was no significant difference between the Lo and PBS groups (Hi, 6541.0 ± 467.5; Lo, 1903.8 ± 187.7; PBS, 1472.1 ± 138.5; p < 0.01 for Hi vs. PBS group) (Fig. 3E). The expression level of rAng1 at week 2 was significantly greater in animals of the Hi group compared with the PBS group, but there was no significant difference between the Lo and PBS groups (Hi, 5781.7 ± 195.4; Lo, 1064.4 ± 227.1; PBS, 865.1 ± 142.1; p < 0.01 for Hi vs. PBS group) (Fig. 3F). The expression level of rBMP-2 at week 2 was also significantly greater in the animals of the Hi group compared with the PBS group, and the Lo group was significantly higher than the PBS group (Hi, 563.8 ± 41.1; Lo, 291.0 ± 9.58; PBS, 205.3 ± 25.9; p < 0.01 for Hi vs. PBS group, p < 0.05 for Lo vs. PBS group) (Fig. 3G). The expression level of rBMP-4 at week 2 was greater in the animals of the Hi group compared with the other groups, but there were no significant differences among the three groups (Hi, 146.8 ± 18.5; Lo, 126.8 ± 15.4; PBS, 118.1 ± 26.5; p = ns) (Fig. 3H).
These results indicate a high dose of GM hPB MNCs enhances both intrinsic angiogenesis and osteogenesis, at least in part by upregulating rVEGF, rAng1, and rBMP-2 at the fracture sites.
Serial Improvement of Blood Flow at Fracture Site
To evaluate blood flow recovery at the fracture sites, LDPI was serially examined after fracture. LDPI analysis demonstrated severely low blood flow at the fracture site 1 h after fracture creation (week 0) and subsequent recovery at week 1, 2, and 3 in all groups (Fig. 4A). In all groups, severe reduction of the blood flow was observed 1 h after fracture. There was no significant difference in the blood flow ratio of fractured to intact (contralateral) limbs 1 h after fracture creation among any group, while the ratio at week 1 was significantly higher in the animals of the Hi group compared to the other groups (Hi, 1.714 ± 0.141; Lo, 1.239 ± 0.060; PBS, 1.170 ± 0.041; p < 0.01 for Hi vs. Lo or PBS group). After week 2 the flow ratio was similar in each group (Fig. 4B).
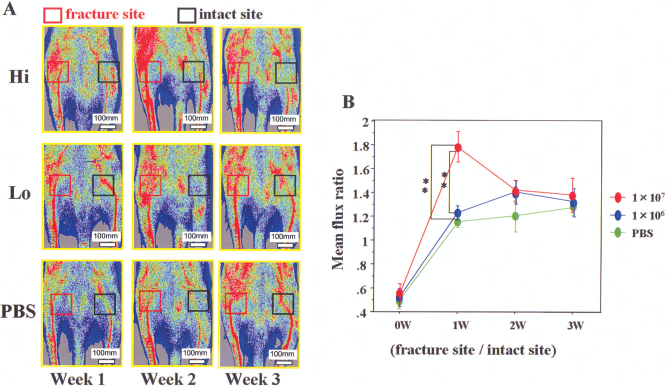
Blood flow improvement at fracture site. (A) Representative laser Doppler perfusion imaging at week 0 (1 h after fracture), 1, 2, and 3 is shown. In these digital color-coded images, maximum perfusion values are indicated in white, medium values in green to yellow, and lowest values in dark blue. The skin blood flow within fracture site (red square) and intact contralateral site (black square) were evaluated as mean flux, and the ratio of the mean flux in the fractured site to that in the contralateral site (mean flux ratio) was calculated. (B) In all groups, severe reduction of the blood flow was observed 1 h after fracture. There was no significant difference in the blood flow ratio of fractured to intact (contralateral) limbs 1 h after fracture creation among any group, while the ratio at week 1 was significantly higher in the Hi group compared to the other groups (p < 0.01 for Hi vs. Lo or PBS groups). After week 2, the flow ratio was similar in all groups.
These results indicate that local transplantation of Hi dose GM hPB MNCs contributes to the rapid improvement of tissue perfusion at the fracture site.
Radiographic and Histological Evidence of Fracture Healing
Morphological fracture healing was evaluated by radiographic and histological examinations. Animals receiving high dose of GM hPB MNCs demonstrated fracture healing with bridging callus formation radiographically in 19% at week 4 and 33% at week 8 and the others in the Hi group did not show fracture healing, although some showed callus formation. Fracture sites in all animals receiving the low dose of GM hPB MNCs or PBS showed no bridging callus formation and subsequently displayed nonunions after 8 weeks, which is consistent with previous reports of the natural course in this animal model (31,34) (Fig. 5A, B).
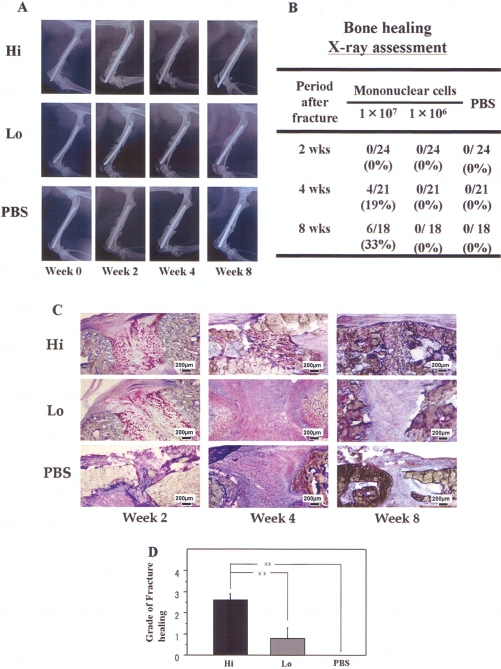
Radiographical and histological evidence of fracture healing. (A) Representative radiographs of fractured sites at weeks 0, 2, 4, and 8 in each group. (B) The fracture healing ratio in all groups. Nineteen percent of animals at week 4 and 33% of animals at week 8 that received a high dose of GM hPB MNCs demonstrated fractures that radiographically appeared healed with bridging callus formation. Fracture sites in all animals receiving a low dose of GM hPB MNCs or PBS showed no bridging callus formation and subsequently displayed nonunions after 8 weeks. (C) Histological evaluation with toluidine blue (original magnification 40x). The evaluation demonstrated enhanced enchondral ossification consisting of numerous chondrocytes and newly formed trabecular bone at week 2, bridging callus formation at week 4, and complete union at week 8 in the Hi group. In contrast, although thick callus formation was observed at week 2, the healing process stopped by week 4, and finally the callus was absorbed at week 8 in the Lo and PBS groups. (D) Allen and colleagues' (3) score of fracture healing at week 8. The degree of fracture healing at week 8 was assessed by Allen and colleagues' classification and was significantly higher in the Hi group compared to the other groups (p < 0.01 for Hi vs. Lo or PBS group).
Fracture healing was also histologically evaluated with toluidine blue staining. The evaluation demonstrated enhanced enchondral ossification consisting of numerous chondrocytes and newly formed trabecular bone at week 2, bridging callus formation at week 4, and complete union at week 8 in the Hi group. In contrast, although thick callus formation was observed at week 2, the healing process stopped by week 4, and finally the callus was absorbed at week 8 in the Lo and PBS groups (Fig. 5C). The degree of fracture healing assessed by Allen and colleagues' classification (3) at week 8 was significantly higher in the Hi group compared to the other groups (Hi, 2.8 ± 0.83; Lo, 0.6 ± 0.89; PBS, 0.0 ± 0.00; p < 0.01 for Hi vs. Lo or PBS group) (Fig. 5D). These results indicate that a nonhealing femoral fracture in a rat created by periosteal cauterization may heal by local administration of Hi dose GM hPB MNCs.
Fate and Kinetics of the Transplanted MNCs
One week after the transplantation, the DiI-labeled MNCs were identified in the intramuscular area, granulation area, and intramedullary area at the fracture site, but not in the remote organs such as lung, liver, and spleen (Fig. 6).

Fate and kinetics of the transplanted MNCs. One week after the transplantation, the 1,1′-dioctadecyl-3,3,3′,3′- tetramethylindocarbocyanine iodide (DiI)-labeled MNCs were identified in the intramuscular area, granulation area, and intramedullary area at the fracture site, but not in the remote organs such as lung, liver, and spleen.
These results suggest that the locally transplanted GM hPB MNCs mainly remained at the fracture sites without traveling to the remote organs.
Effectiveness of MNCs Compared to CD34+ Cell Transplantation: Functional Assessment for the Fracture Healing
Biomechanical examinations by 3-point bending test in CD34+ group (Fig. 7) showed a significantly increased ultimate stress ratio (CD34+, 68.53 ± 5.56; Hi, 20.08 ± 11.28; Lo, 9.05 ± 2.37; PBS, 5.93 ± 1.81; p < 0.01 for CD34+ vs. Hi, Lo, or PBS group, n = 4) and extrinsic stiffness ratio, compared with the other groups (CD34+, 45.03 ± 16.71; Hi, 3.98 ± 1.75; Lo, 3.10 ± 1.57; PBS, 2.70 ± 1.11; p < 0.05 for CD34+ vs. Hi, Lo, or PBS group, n = 4) (Fig. 7A, C). For the failure energy ratio, the CD34+ group demonstrated a significantly higher level compared with Lo and PBS groups but there was no significant difference between CD34+ and Hi groups (CD34+, 208.1 ± 49.8; Hi, 96.3 ± 47.9; Lo, 28.9 ± 8.8; PBS, 27.60 ± 11.1; p < 0.05 for CD34+ vs. Lo or PBS group, n = 4) (Fig. 7B).

Biomechanical assessment for fracture healing. (A) %Ultimate stress. CD34+ group demonstrated significantly higher values than other groups (p < 0.01 for CD34+ vs. Hi, Lo or PBS group). (B) %Fracture energy. CD34+ group demonstrated significantly higher values than Lo and PBS groups, but there was no significant difference between CD34+ and Hi groups (p < 0.05 for CD34+ vs. Lo or PBS group). (C) %Extrinsic stiffness. CD34+ group demonstrated significantly higher values than other groups (p < 0.05 for CD34+ vs. Hi, Lo, or PBS group).
These results suggest that total MNCs have an inferior effect on fracture healing compared with purified CD34+ cells.
Real-Time RT-PCR Analysis with Additional Growth Factor Primers
To investigate the mechanism of the inferior effect of fracture healing on total MNC transplantation, real-time RT-PCR analyses were performed with additional growth factors at the fracture sites after transplantation of GM hPB total MNC and GM hPB CD34+ cells. For this, we formed another group, CD34+ group, a group of animals receiving transplantation of 1 × 105 GM hPB CD34+ cells. RNA was isolated from tissue samples from animals of the Hi, Lo, PBS, and CD34+ groups and we performed real-time RT-PCR with those RNA to quantify the expression of rat endothelial nitric oxide synthase (reNOs), rat fibroblast growth factor 2 (rFGF2), rat bone morphogenic protein 7 (rBMP-7), and rat osteopontin (rOsteopontin) around the fracture sites.
The expression level of reNOs at week 2 was significantly greater in animals in the CD34+ group compared with the other groups, but there was no significant difference between the Hi, Lo, and PBS groups (CD34+, 593.5 ± 58.95; Hi, 398.0 ± 46.86; Lo, 339.3 ± 44.12; PBS, 345.1 ± 41.34; p < 0.05 for CD34+ vs. Hi and PBS group, p < 0.01 for CD34+ vs. Lo group) (Fig. 8). The expression level of rbFGF at week 2 showed no significant differences among the four groups (CD34+, 51.91 ± 9.41; Hi, 54.98 ± 20.05; Lo, 33.35 ± 4.54; PBS, 50.58 ± 5.04; p = ns) (Fig. 8B). The expression level of rBMP-7 at week 2 was also significantly greater in animals of the CD34+ and Hi groups compared with the Lo and PBS groups, but there were no significant differences between the CD34+ and Hi groups and between the Lo and PBS groups (CD34+, 83.28 ± 22.93; Hi, 76.33 ± 10.1; Lo, 30.81 ± 6.3; PBS, 31.24 ± 4.2; p < 0.05 for CD34+ vs. Lo or PBS group, p < 0.05 for Hi vs. Lo or PBS group) (Fig. 8C). The expression level of rOsteopontin at week 2 was greater in animals of the CD34+ and Hi groups compared with the other groups, but there were no significant differences between the three groups (CD34+, 5605.6 ± 913.7; Hi, 5201.4 ± 832.7; Lo, 2872.9 ± 678.0; PBS, 1674.0 ± 1149.0; p = ns) (Fig. 8D).

Gene expression of intrinsic cytokines for angiogenesis and osteogenesis at week 2 (n = 4 in each group). The expression level of rat endothelial nitric oxide synthase (reNOs) at week 2 was significantly greater in the CD34+ group compared with Hi, Lo, and PBS groups (p < 0.05 for CD34+ vs. Hi or PBS group, p < 0.01 for CD34+ vs. Lo group) (A). The expression level of rat basic fibroblast growth factor (rbFGF) at week 2 showed no significant differences among the four groups (CD34+, 51.91 ± 9.41; Hi, 54.98 ± 20.05; Lo, 33.35 ± 4.54; PBS, 50.58 ± 5.04; p = ns) (B). The expression level of rBMP-7 at week 2 was also significantly greater in the CD34+ and Hi groups compared with Lo and PBS groups, but there was no significant difference between CD34+ and Hi groups (p < 0.05 for CD34+ or Hi vs. Lo or PBS group) (C). The expression level of rOsteopontin at week 2 was greater in the CD34+ and Hi groups compared with the Lo and PBS groups, but there were no significant differences among all groups (p = ns) (D).
These results indicate that GM hPB CD34+ cells enhance both intrinsic angiogenesis and osteogenesis more than Hi doses of GM hPB total MNC, at least in part by upregulating reNOs and rBMP-7 at the fracture sites.
Discussion
Whereas most fractures can be healed with bridging callus by conservative or surgical treatments, 5–10% of fractures do not achieve union and culminate in delayed union or persistent nonunion (8,13,30,38). Four elements are advocated to be integral to fracture healing: an osteoconductive matrix, osteoinductive signals, osteogenic cells, and a sufficient blood supply, and deficient fracture healing is mainly caused by insufficient blood supply (6,8,31,34,45).
Since Asahara et al. (4) reported that circulating CD34+ cells isolated from the adult human peripheral blood contain endothelial progenitor cells (EPCs) and contribute to vasculogenesis, the transplantation of EPCs was performed in preclinical and clinical studies to develop a therapy for ischemic diseases (17,19,20,23, 25,35,36,39,42,47). Meanwhile, in the field of skeletal biology, several reports suggest that the EPC-rich cell populations are committed to osteogenic differentiation (5,7,9,10,28,44). Based on these evidences, we previously investigated and reported the therapeutic potency of intravenous and local administration of CD34+ cells for bone fracture via vasculogenesis/angiogenesis and osteogenesis (31,34). Moreover, we reported the pivotal role of EPCs on their mobilization and incorporation for fracture healing (32).
In the field of revascularization, MNC transplantation has also shown its therapeutic efficiency in enhancing neovascularization at ischemic sites in preclinical and clinical studies (12,14,21,27,41,43). Compared with the isolation methods of purified CD34+ cells, hPB MNCs do not require magnetic cell sorting and therefore can be performed in a shorter time and at a lower cost. We performed the current study to investigate the efficacy of hPB MNC local transplantation for bone fracture.
In the present study, we first measured the ratio of CD34+ cells in GM hPB MNCs by FACS analysis, which demonstrated that GM hPB MNCs contained about 1% of CD34+ cells. On the basis of the data, we calculated that out of the 1 × 107 MNCs in the Hi group, 1 × 105 were CD34+ cells, which was the effective dose of CD34+ cells in the previous study (34).
Differentiation of GM hPB MNCs into ECs and OBs was identified by immunohistochemical staining and real-time RT-PCR with human-specific markers in the Hi and Lo groups. Additionally, immunofluorescent staining and real-time RT-PCR with rat-specific ECs and OBs markers revealed significant enhancement of intrinsic angiogenesis and osteogenesis in the Hi and Lo groups, which indicated the paracrine effect of the transplanted GM hPB MNCs. We actually performed experiments at each time point of 1, 2, and 3 weeks after transplantation, but the time points (human cell-derived vasculogenesis and osteogenesis 1 week; intrinsic angiogenesis and osteogenesis, 2 weeks) represented in this article showed significant differences among the groups, indicating the contribution of paracrine, angiogenic, and osteogenic effects following early direct differentiation to osteoblast and endothelial cells. In addition to these mechanisms of MNC transplantation indicating direct differentiation and intrinsic contributions for bone fracture healing, physiological assessment by LDPI showed significant recovery of blood flow at the fracture sites in the Hi group, but not in the other groups, and morphological assessment of fracture healing showed significantly better repair in the Hi group compared with the other groups.
The current study revealed the potency of transplanted GM hPB MNC for repairing bone fracture by enhancing vasculogenesis/angiogenesis and osteogensis in an autocrine/paracrine manner, as well as in the previous studies using CD34+ cells (31,34). However, there was a discrepancy between CD34+ cells and GM hPB MNCs in the radiological/histological findings after transplantation. In 33% of animals in the Hi group, fractures radiographically healed with bridging callus formation, while the fracture site of all animals in the other groups showed no bridging callus formation and all fell into nonunion at week 8. These results were consistent with the histological evaluation, which showed the degree of fracture healing assessed by the classification of Allen et al. (3). It is notable that the healing ratio in the Hi group in the present study is much lower than that in the previous study (34), in which all animals in the group receiving local transplantation of 1 × 105 CD34+ cells achieved fracture union, although all animals in both groups were effectively transplanted with the same number (1 × 105) of CD34+ cells (1 × 107 MNCs vs. 1 × 105 CD34+ cells). To compare total MNCs with CD34+ cells in their efficacy for fracture healing, biomechanical analysis, which was not carried out in our previous study of the local transplantation of 1 × 105 GM hPB CD34+ cells (34), was performed in four groups (the Hi, Lo, PBS, and CD34+ groups), and the highest value for all parameters was observed in the CD34+ group. These results indicated the reduced ability of total MNCs to achieve mechanical fracture healing compared to CD34+ cells. The mechanism that causes MNC transplantation to be inferior to CD34+ cells in their therapeutic effect for fracture healing is still being investigated. Total MNCs are mixed cell populations, and lymphocytes and monocytes/macrophages are the most prevalent mononuclear cells (40). These inflammatory cells could have a negative influence on osteogenesis (2,15,48). The recent report of our group about intramyocardial transplantation of GM hPB total MNCs showed the possible risk of severe hemorrhagic myocardial infarction in nude rats through the excessive inflammation induced by transplanted cells (24). We speculate that inflammation after transplantation of total MNCs could interfere with the long-term survival and differentiation of transplanted cells.
Conclusion
Local transplantation of GM hPB MNCs contributes to fracture healing via vasculogenesis/angiogenesis and osteogenesis. However, when compared with purified CD34+ cells, the therapeutic potential of GM hPB MNCs for fracture healing could be inferior to that of purified CD34+ cells even if GM hPB MNCs contained the same number of CD34+ cells.
Footnotes
Acknowledgments
We would like to thank Ms. Janina Tubby for her editing assistance in preparing this manuscript. The authors declare no conflicts of interest.
