Abstract
Cell-based therapies as an alternative to liver transplantation have been anticipated for the treatment of potentially fatal liver diseases. Not only mature hepatocytes (MHs) but also hepatic stem/progenitor cells are considered as candidate cell sources. However, whether the stem/progenitor cells have an advantage to engraft and repopulate the recipient liver compared with MHs has not been comprehensively assessed. Therefore, we used Thy1+ (oval) and CD44+ (small hepatocytes) cells isolated from GalN-treated rat livers as hepatic stem and progenitor cells, respectively. Cells from dipeptidylpeptidase IV (DPPIV)+ rat livers were transplanted into DPPIV” livers treated with retrorsine following partial hepatectomy. Both stem and progenitor cells could differentiate into hepatocytes in host livers. In addition, the growth of the progenitor cells was faster than that of MHs until days 14. However, their repopulation efficiency in the long term was very low, since the survival period of the progenitor cells was much shorter than that of MHs. Most foci derived from Thy1+ cells disappeared within 2 months. Many cells expressed senescence-associated β-galactosidase in 33% of CD44-derived foci at day 60, whereas the expression was observed in 13% of MH-derived ones. The short life of the cells may be due to their cellular senescence. On the other hand, the incorporation of sinusoidal endothelial cells into foci and sinusoid formation, which might be correlated to hepatic maturation, was completed faster in MH-derived foci than in CD44-derived ones. The survival of donor cells may have a close relation to not only early integration into hepatic plates but also the differentiated state of the cells at the time of transplantation.
Introduction
As an alternative to organ transplantation, much attention has been paid to the potential applicability of the transplantation of mature hepatocytes (MHs) for the treatment of liver diseases (10,47). Transplanted MHs engraft in the recipient liver and morphologically as well as functionally connect with the surrounding cells (16,48). In animal models with liver injury and/or selective growth advantage, it is known that engrafted cells respond to growth stimuli and repopulate recipient livers (12,15,49). Furthermore, elucidation of the molecular pathways of liver regeneration and extensive pre-clinical cell transplantation experiments in animals have led to the application of hepatocyte transplantation in a limited number of patients with hereditary metabolic liver disease and acute liver failure (10,12,39). However, the shortage of cell sources and the difficulties of cryopreservation and long-term culture of MHs have limited the clinical application of cell-based therapies.
Stem or progenitor cells have been considered as candidate cell sources, because they can expand in vitro and be cryopreserved for a long term (4,32). The cells derived from adult and fetal livers and other organs have been shown to differentiate into cells with hepatic characteristics in vitro (31). In addition, most of the cells could differentiate into hepatocytes in the recipient livers of genetically altered mice (26,34,36,48) and in toxic injury models (21,25). On the other hand, it is also known that hepatic stem/progenitor cells in adult livers are activated when the proliferation of MHs is inhibited by hepatotoxins (9,11,40). Among them, oval cells and small hepatocytes (SHs) are well recognized as stem and progenitor cells, respectively. Oval cells, named for their possession of ovoid nuclei, are known to express markers for cell membrane proteins such as CD34, c-kit, and Thy-1, shared hematopoietic stem cell markers (35). SHs are a subpopulation of hepatocytes (28). These cells can clonally proliferate (27) and mature by interacting with hepatic nonparenchymal cells (NPCs) (30) or as a result of treatment with Matrigel (44).
The CD44 gene encodes for a family of alternative spliced, multifunctional molecules, and CD44 plays a role in adhesion of cells to extracellular matrices such as hyaluronic acid, collagen, and fibronectin (13). CD44 standard form is composed of a short cytoplasmic tail, a transmembrane region, and two extracellular domains, and 10 variant forms (v1–v10) exist (7). In normal livers, MHs do not express CD44, whereas some hepatic NPCs, including hepatic stem cells, liver sinusoidal endothelial cells, Kupffer cells, biliary epithelial cells (BECs), and fibroblasts in Glisson's sheath express CD44 standard form and no CD44v6+ cells exist (18,38). Recently, we reported that CD44 standard form and v6 were specifically expressed in cultured SHs isolated from an adult rat (22) and human liver (37). In addition, in injured livers, hepatocytes expressing CD44 transiently appeared around the periportal area (22). Furthermore, Thy1+ cells isolated from the liver injured by D-galactosamine (GalN) could differentiate into hepatocytes through CD44+ SHs (21). Both transplanted Thy1+ and CD44+ cells sorted from GalN-treated rat livers could survive, proliferate, and differentiate into hepatocytes in retrorsine/partial hepatectomy (Ret/PH)-treated livers. Therefore, when the stem/progenitor cells are transplanted into injured livers, rapid growth and expanded repopulation of donor cells will be expected. However, Haridass et al. (17) recently reported that MHs could generate liver tissue more efficiently than cells derived from fetal tissue or embryonic stem cells in an albumin-urokinase plasminogen activator (Alb-uPA) transgenic mouse model. Therefore, the issue of whether stem/progenitor cells have an advantage over MHs as cell sources for cell-based therapies should be resolved.
We previously used a GalN-injured liver model to show the sequential differentiation of hepatic stem/progenitor cells and demonstrated that some of the Thy1+ cells that appeared at day 3 after GalN injection were bipotential hepatic stem cells and could differentiate into hepatocytes through CD44+ SHs (21). In addition, isolated Thy1+ and CD44+ cells could generate foci in recipient livers after transplantation. In the present study we examined whether transplanted hepatic stem and progenitor cells could repopulate in host livers faster than MHs. For donor cells, we used Thy1+ (oval) and CD44+ (SH) cells isolated from GalN-treated rat livers as hepatic stem and progenitor cells, respectively. Large foci derived from MHs and CD44+ cells were observed even 1 year after transplantation, whereas most foci derived from Thy1+ cells disappeared within 2 months. CD44-derived cells could proliferate faster and form larger foci than MH-derived ones until day 14 after transplantation. However, MH-derived cells gradually repopulated the recipient livers until 1 year, whereas CD44-derived ones decreased with time. Low expression of CCAAT/enhancer binding protein (C/EBP)-α was observed in the cells having foci without sinusoidal endothelial cells (SECs). On the other hand, many cells expressed senescence-associated β-galactosidase (SA-β-Gal) in more than 30% of CD44-derived foci at day 60, whereas the expression was observed in about 10% of MH-derived foci. CD44-derived cells may show cellular senescence much earlier than MH-derived ones in host livers. These results indicate that MHs may be superior to stem/progenitor cells to repopulate the injured liver in the long run when they are directly transplanted.
Materials and Methods
Animals and Liver Injury Model
Male F344 rats [dipeptidylpeptidase IV (DPPIV)+ strain; Sankyo Lab Service Corporation, Inc., Tokyo, Japan] weighing 150–200 g were used. All animals received humane care and the experimental protocol was approved by the Committee of Laboratory Animals according to Sapporo Medical University guidelines. For GalN-injured livers, GalN (Acros, Geel, Belgium; 75 mg/100 g body weight dissolved in PBS) was intraperitoneally administered (22). For the transplantation experiment, female F344 rats (DPPIV- strain; Charles River Japan, Yokohama, Japan) were intraperitoneally given two injections of retrorsine (30 mg/kg body weight; Sigma Chemical Co., St. Louis, MO), 2 weeks apart (24). Four weeks after the second injection, two thirds partial hepatectomy was performed. Sorted Thy1+ and CD44+ cells and normal MHs (5 times; 105 cells) were transplanted into Ret/PH livers (DPPIV-) via the spleen (three to five rats per group).
Isolation of Cells From Liver
Rats were used to isolate hepatic cells by the collagenase perfusion method as previously described (6). After the perfusion, the cell suspension was centrifuged at 50 times; g for 1 min. The supernatant and the precipitate were used for sorting Thy1+ (GalND2, 2 days after GalN treatment) and CD44+ (GalND4) cells and preparing MHs (normal adult, 10–12 weeks old), respectively (21).
Cell Sorting
The supernatant was centrifuged at 50 times; g for 1 min again. After this procedure was repeated, the supernatant was centrifuged at 50 times; g for 5 min. The precipitate was suspended in PBS containing 2 mM EDTA and 0.5% BSA. Antibodies used for cell sorting are listed in Table 1. The details were previously described (21).
Antibodies Used in the Present Experiments

Histochemistry
After cell transplantation, recipient rats were sacrificed at days 1, 7, 10, 14, 21, 30, 60, 90, and 365. Removed livers were immediately sliced 5 mm thick. Some slices were frozen and the others were fixed in 10% paraformaldehyde solution. Frozen sections were used for enzyme histochemisty and for immunohistochemistry. Livers embedded in Tissue-Tek® (Sakura Finetechnical Co, Tokyo, Japan) were frozen using isopentane/liquid nitrogen and the samples were kept at −80°C until use. For identifying donor cells, enzyme histochemistry for DPPIV was carried out. DPPIV enzyme activity was detected as previously described (42). DPPIV+ foci were photographed using a microscope equipped with a CCD camera and the area of each focus was measured using Image J (http://rsb.info.nih.gov/ij/index.html).
Immunohistochemistry
Antibodies used are listed in Table 1. For immunohistochemistry, the liver sections were fixed in absolute cold ethanol after washing with PBS. The details have been previously described (21). Triple staining was carried out for enzyme histochemistry of DPPIV and immunohistochemistry for Ki67 and p16. Triple staining for enzyme histochemistry of DPPIV and immunohistochemistry for both SE-1 (a marker of SECs) and C/EBPα (a marker of highly differentiated hepatocytes) was also performed. Finally, samples were embedded with 90% glycerol including 0.01% p-phenylenediamine and 4,6-diamidino-2-phenylindole (DAPI).
Senescence-Associated β-Galactosidase Staining
SA-β-Gal staining was performed using the method reported by Dimri et al. (8). When more than 10% of cells in the DPPIV+ focus were intensively stained with SA-β-Gal, the focus was regarded as a positive one. The ratio of the positive foci per DPPIV+ ones was calculated.
Statistical Analysis
All data were analyzed by ANOVA followed by the Turkey-Kramer multiple comparison test. Statistical significance was accepted atp < 0.05. Experimental results were expressed as the average ± SD.
Results
Transplantation of Sorted Cells
The details about the characterization of the Thy1+ and CD44+ cells were previously described (21). The viability of both Thy1+ and CD44+ cells was over 75%, and that of MHs was over 95%. The ratios of enzymatically DPPIV+ cells in Thy1+ and CD44+ cell fractions and MHs were 62.0 ± 14.1%, 54.0 ± 4.3%, and 100%, respectively. We reported that Thy1+ cell fractions included polygonal (epithelial-like)- and spindle (fibroblast-like)-shaped cells (21). Most polygonal-shaped cells showed DPPIV positivity and some of them could form colonies. However, spindle-shaped cells did not express DPPIV positivity and their shape could not change to that of epithelial-like cells. Many CD44+ cells were morphologically polygonal and looked like epithelial cells. Although we conducted detailed examination, only about 55% of the cells were DPPIV+. It may be enzyme cytochemically difficult to find a single cell that faintly expresses DPPIV. However, we have never observed that cells without features of epithelial-like cells can form a colony.
Thy1+ cells from GalND2, CD44+ cells from GalND4, and MHs from a normal rat were transplanted into Ret/PH rat livers through the spleen. Some donor cells attached in lobules of recipient livers and then proliferated to form foci, which could be distinguished from resident hepatocytes by the expression of DPPIV. The typical DPPIV+ foci formed by the transplanted Thy1+ cells, CD44+ cells, and MHs are shown in Figure 1. As previously reported (21), not only MHs but also both Thy1+ and CD44+ cells could attach and differentiate into hepatocytes. Since the number of Thy1–derived foci was not large enough to measure their growth ability, the growth ability of transplanted cells was limitedly examined in CD44- and MH-derived ones.

Transplantation of sorted cells from D-galactosamine (GalN)-treated rat livers and of normal mature hepatocytes (MHs) into retrorsine/partial hepatectomy (Ret/PH)-treated livers. (A) At day 30 after 5 × 105 sorted Thy1+ cells (GalN-D2) were transplanted. (B) At day 60 after 5 × 105 sorted CD44+ cells (GalN-D4) were transplanted. (C) At day 90 after 5 × 105 normal MHs were transplanted. Scale bars: 200 μm (A, B), 1.0 mm (C).
As shown in Figure 2A, CD44-derived foci expanded with time after transplantation and their size was significantly larger than that of MH-derived ones until day 14. At day 14 the numbers of cells per foci were 31.1 and 16.3, respectively. However, at day 21, no difference was observed in the average size of MH- and CD44-derived foci (Fig. 2B). After day 60, the size of MH-derived foci was clearly bigger than that of CD44-derived ones. As shown in Table 2, after day 30, the number and the size of foci per liver fluctuated from rat to rat and from lobe to lobe. However, the differences among the three types of donor cells became clear with time after transplantation. Only a few foci were observed in the livers with Thy1+ cell transplantation at days 30 and 60, and thereafter no focus was found. The number and the repopulated area of CD44-derived foci obviously decreased after day 90. On the other hand, the number of MH-derived foci was unchanged and the repopulated area enlarged until day 90. Thereafter, the repopulated area did not change until day 365.
Numbers and Areas of Foci in Ret/PH Rat Livers After Cell Transplantation

The numbers in parentheses are the numbers of rats. Three samples were analyzed per rat. The results are shown as the average ± SD. Ret/PH, retrorsine and partial hepatectomy; Tx, transplant; MH, mature hepatocytes.
p < 0.05, significant difference from the CD44+ cells group.
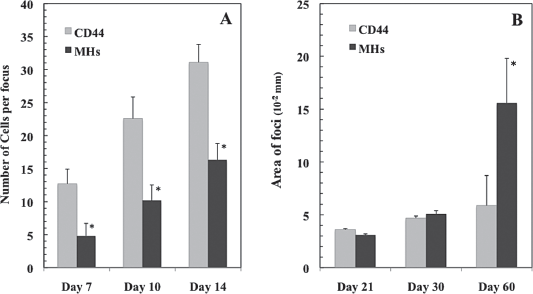
The growth of transplanted CD44- and MH-derived cells in host rat livers treated with retrorsine following partial hepatectomy (PH). (A) The numbers of cells in foci were counted at days 7, 10, and 14 after transplantation. More than three rats per group and two slices per liver were examined. The columns and bars show the average and SD, respectively. *Significantly different from CD44, p < 0.05. (B) The dipeptidylpeptidase IV (DPPIV+) area of each focus was measured. More than 50 foci per group were examined. The columns and bars show the average and SD, respectively. *Significantly different from CD44, p < 0.05.
The results suggested that the growth of the engrafted cells was divided into two phases, before and after day 21. Before day 21, the growth of CD44-derived cells was apparently faster than that of MH-derived ones. However, after day 21, their growth rate was markedly decreased. In contrast to CD44-derived cells, MH-derived cells constantly proliferated and the average size of the foci reached that of CD44-derived foci at day 21. The size of MH-derived foci exceeded that of CD44-derived ones after day 60. To clarify why the development of foci was different between the donor cells, the characteristics of the foci were examined. Five factors were analyzed: 1) growth activity, 2) integration into hepatic plates, 3) differentiation status, 4) disappearance of donor cells, and 5) cellular senescence. First, to examine the proliferative activity of the engrafted cells, immunohistochemistry for Ki67 and p16 was performed in DPPIV-stained samples (Fig. 3). At days 14, 30, and 60, many Ki67+ nuclei in the livers were observed within the DPPIV+ foci, which were derived from both CD44 and MHs. As shown in Figure 3I, the ratios of Ki67+ cells in foci derived from CD44+ cells at days 14, 30, and 60 were 17.7 ± 2.0%, 5.7 ± 1.7%, and 4.1 ± 4.0%, respectively, whereas those of Ki67+ cells from MHs were 9.6 ± 2.0%, 1.7 ± 2.2%, and 4.4 ± 2.1%, respectively. The ratio of Ki67+ cells was significantly larger in CD44-derived foci than in MH-derived ones at days 14 and 30. On the other hand, as shown in Figure 3J, the ratios of p16+ cells in foci derived from CD44+ cells at days 14, 30, and 60 were 15.8 ± 2.0%, 33.8 ± 1.7%, and 27.8 ± 2.2%, respectively, whereas those of p16+ cells from MHs were 18.4 ± 2.1%, 22.0 ± 1.7%, and 23.2 ± 2.0%, respectively. The number of p16+ cells was increased significantly more in CD44-derived foci than in MH-derived ones at days 30 and 60.

Expression of Ki67 and p16 in DPPIV+ cells at days 14, 30, and 60 after transplantation. CD44+ cells (A–D) and MHs (E–H) were transplanted into rat livers treated with retrorsine following partial hepatectomy (PH). Enzyme histochemistry for DPPIV (A, E), immunohistochemistry for Ki67 (B, F), p16 (C, G), and merge images (D, H) at day 30. Nuclei were stained with DAPI. All photos are the same magnification. Scale bars: 100 μm. Graph (I) shows the ratios of Ki67 in DPPIV+ cells at days 14, 30, and 60. Graph (J) shows the ratios of p16 in DPPIV+ cells at days 14, 30, and 60.
As shown in Figure 4, Thy1-derived foci consisted of irregularly shaped and sized cells, and the DPPIV+ cells were randomly distributed even at day 60 (Fig. 4A), whereas, in MH-derived foci, DPPIV+ cells were linearly arranged and sinusoids had already been formed along the hepatic plates at day 10 (Fig. 4C). In small CD44-derived foci, many cells were irregularly shaped and the sinusoids were not easily found (Fig. 4B), whereas in the large ones, most cells were similar in shape and the formation of sinusoids was easily recognized. These results suggested that hepatocytes in large foci were arranged to form plates and that SECs might integrate into the foci. Therefore, to investigate whether the development of sinusoids was related to the size of foci and the maturation of hepatocytes, double immunohistochemistry for SE-1 and C/EBPα was carried out in slices stained for DPPIV enzyme histochemistry. The DPPIV+ foci were divided into two groups by the number of cells in the foci. The results are summarized in Figure 4L and M. In small foci consisting of less than 20 cells, a few or no SE-1+ cells were distributed inside CD44-derived foci (Fig. 4E), whereas SE-1+ cells forming networks of sinusoids were observed in about 77% of MH-derived foci. In foci consisting of more than 20 cells, linear arrangement of SE-1+ cells was observed in more than 80% of CD44-derived foci, whereas all MH-derived foci integrated SE-1+ cells (Fig. 4I). On the other hand, in spite of the incorporation of SE-1+ cells into CD44-derived foci, less than 30% of the cells possessed C/EBPα+ nuclei (Fig. 4M). In MH-derived foci with well-developed sinusoids, more than 70% of cells expressed C/EBPα in their nuclei (Fig. 4J), whereas in those with poorly developed sinusoids the positivity decreased to about 40%. At day 30, all foci integrated SE-1+ cells regardless of the cellular origin, and about 72% and 87% of the cells were C/EBPα+ in CD44-derived and MH-derived foci, respectively. Since the ratio was the same as in the surrounding hepatocytes, all the integrated cells derived from SH as well as MHs differentiated to MHs by day 30.

Characteristics of DPPIV+ foci formed by transplanted cells. Thy1+ cells (A), CD44+ cells (B, D–G), and MHs (C, H–K) were transplanted into Ret/PH-treated rat livers. Enzyme histochemistry for DPPIV (A–D, H), and immunohistochemistry for SE-1 (sinusoidal endothelial cells; E, I) and C/EBPα (CCAAT/enhancer binding protein; F, J). Nuclei are stained with DAPI. The photos (G, K) are merge images of DPPIV, SE-1, C/EBPα, and DAPI. The graph (L) shows the ratio of the foci integrated with SE-1+ SECs at day 14. The graph (M) shows that the ratio of C/EBPα+ hepatocytes per focus at day 14 is compared between SE-1+ and SE-1- foci. The columns and bars show the average and SD, respectively. *Significant difference between CD44 and MH, p < 0.05. The areas surrounded by a line (D-K) are the corresponding foci. At days 60 (A), 14 (B, D–K), and 10 (C) after transplantation. Scale bars: 100 μm.
Finally, the cause of the cell loss in the foci was analyzed. We first examined whether an increase of apoptotic cells was observed in CD44- and MH-derived foci. Although terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) staining and immunohistochemistry for cleaved caspase 3 and Annexin V were carried out in the foci at day 14, no increase of the positive cells was found in either CD44- or MH-derived foci compared with surrounding hepatocytes (data not shown). However, as the number of CD44-derived foci obviously decreased after day 60, SA-β-Gal staining was carried out to examine whether CD44-derived cells might undergo cellular senescence. The expression of SA-β-Gal was not as evident in CD44-derived cells as in MH-derived ones at day 14 (data not shown). At day 60, in more than a few CD44-derived foci, cells intensively expressing SA-β-Gal occupied most parts of the foci, whereas only a small number of cells were stained in many MH-derived ones (Fig. 5). The ratio of the foci with SA-β-Gal+ cells was significantly larger in CD44-derived foci (33.0 ± 9.4%) than in MH-derived foci (13.3 ± 6.7%).

Senescence-associated (SA)-β-galactosidase (Gal)-positive cells in recipient livers at day 60 after transplantation. CD44+ cells (A–C) and MHs (D–F) were transplanted into Ret/PH-treated rat livers. Enzyme histochemistry for DPPIV (A, D) and SA-β-Gal (B, E), and merge images (C, F). Nuclei were stained with DAPI. All photos are the same magnification. Bars: 100 μm.
Discussion
In the present experiment, to examine whether hepatic stem/progenitor cells could be used as a source for cell-based therapies, Thy1+ and CD44+ cells isolated from GalN-treated rat livers were transplanted into livers of Ret/PH model rats. The repopulation efficiency and the survival of the cells were compared to those of MHs. Both transplanted Thy1+ and CD44+ cells could form hepatic foci in the recipient livers, but the frequency of CD44-derived foci was much higher than that of Thy1-derived ones. The results were consistent with our previous results obtained from cells that were examined at day 30 after transplantation (21). In this study we found that Thy1-derived hepatocytes could not survive for a long time; almost all the cells disappeared within 2 months. In addition, although the survival and maintenance of CD44-derived hepatocytes were much better than those of Thy1-derived ones, many CD44-derived foci could not survive for a long time. One year later, the number and area of the CD44-derived foci were remarkably decreased to 1/30 and 1/20 of those at day 60, respectively. In contrast, MH-derived hepatocytes gradually repopulated the recipient livers and, even when a year passed, the number and area were maintained.
The capabilities for hepatic differentiation and repopulation of other stem/progenitor cells derived from a different tissue have been reported. When enriched Thy1+ bone marrow (BM) cells were transplanted directly into rat livers treated with monocrotalin and 2-acetylaminofluorene following two thirds PH, hepatocytic differentiation of the cells was observed at 60 days after transplantation (2). Furthermore, β2m-/Thy1+ BM cells integrated into the host parenchyma and repopulated the recipient livers when they were transplanted into rat livers under strong selection pressure and continuous host liver injury (1). However, those experiments revealed that only a few Thy1+ BM cells could differentiate into hepatocyte-like cells and repopulate the liver. The issues of the low repopulation efficiency and the short longevity in recipient livers have also been raised in the transplantation of other hepatic stem cells. Murine and human ES cell-derived hepatocytes, which express typical marker proteins of hepatocytes, were transplanted into livers of immune-deficient mice and/or of rodents with severe liver injuries (3,17,19,46). The efficiency of hepatic repopulation was at most 2%. Furthermore, in an experiment using bipotential mouse embryonic liver stem cell lines, although the cells could differentiate into hepatocytes and BECs in Alb-uPA/SCID mice (severe combined immunodeficient mice with murine urokinase genes under control of an albumin promoter) after transplantation, they could not repopulate a large area of the recipient liver (43). To our knowledge, most studies have been focused on proving that candidates for hepatic stem cells possess bipotential to differentiate into hepatocytes and/or BECs. Shafritz et al. (41) summarized previous transplantation experiments using hepatic stem/progenitor cells, and found that the cells, except ED14 fetal hepatocytes, showed very low efficiency of engraftment and repopulation regardless of the condition of the recipient liver. These results suggest that induced hepatocytes from ES cells and other stem/progenitor cells have not yet matured to the stage at which they can efficiently repopulate the liver of an adult.
The fact that hepatocytes are not a uniform population has been reported. SHs, which possess high growth activity, were first found in the culture of primary hepatocytes (28) and then their clonal expansion was shown (27,45). Since the sizes of MHs and SHs are markedly different, the growth activity and repopulation capability have been investigated between subpopulations of hepatocytes divided by size. Mouse hepatocytes were separated by cell density, and then three distinct populations that differed in cell size, small (16 μm), medium (21 μm), and large (27 μm) cells, were transplanted into livers of fumarylacetoacetate hydrolase (FAH)-deficient mice and the ability to repopulate was compared at 8 weeks after transplantation (33). Unexpectedly, the results demonstrated that small, mononucleate hepatocytes repopulated significantly less well than larger hepatocytes. In addition, the most abundant size fraction (21 μm), constituting approximately two thirds of all cells isolated by collagenase perfusion, had the best repopulation properties. They also showed that serial transplantation neither enhanced nor diminished the repopulation capacity of the cells to any significant degree. On the other hand, Katayama et al. (20) found the opposite. Subfractionated SHs, SH-R2s (22.5 μm) and SH-R3s (17 μm), were transplanted into livers of Ret/PH model rats and 21 days after transplantation the numbers of cells comprising repopulated foci were compared. The results showed that the growth potential of SH-R3s was about three times higher than that of SH-R2s. Although these two reports seem to be inconsistent concerning the repopulation capacity of small-sized hepatocytes, this discrepancy may be explained by our present results; SHs (CD44+ cells) showed high growth activity until about 2 weeks after their transplantation, and thereafter their growth activity clearly decreased with time. Until day 14, the number of cells in CD44-derived foci was much higher than that in MH-derived ones, whereas at day 60 the size of MH-derived foci became obviously larger than that of CD44-derived ones. In addition, the area repopulated by MH-derived cells constantly increased with time after transplantation. Tetraploid and octaploid hepatocytes were reported to form proliferating foci as large as diploid ones in uPA-transgenic mouse livers (48). Taking these findings together, it is feasible to think that the nature of small-sized hepatocytes is different from that of large hepatocytes (typical MHs). However, it may be difficult to find any significant difference of the growth ability among large hepatocytes (33).
In the present experiment we used Ret/PH model rats as recipients. In these model rats it is well known that small hepatocyte-like progenitor cells (SHPCs) appear in the liver (14). As the morphology of SHPCs is quite similar to that of the SHs that we use, it is important to clarify whether SHPCs are the same as SHs. We use CD44 expression as a marker of SHs. In the early period (1–2 weeks) after Ret/PH treatment, some clusters consisting of SHPCs were immunohistochemically positive for CD44, but not all SHPCs were stained (data not shown). In addition, although CD44+ hepatocytes appeared in the liver treated with GalN, the expression disappeared soon. Moreover, the expression of CD44 was suppressed in DPPIV+ foci derived from CD44+ cells. On the other hand, when the maturation of cultured SHs occurs via the interaction of NPCs or Matrigel treatment, CD44 expression of SHs rapidly disappears (22). Taken together, these findings suggest that, although CD44 is a marker for SHs, the expression may be repressed due to the interaction with neighboring MHs. Therefore, it is plausible to think that the intrinsic SHs proliferate to form the cluster of SHPCs under specific conditions in which the proliferation of MHs is strongly inhibited.
The present results raise the question of why it is difficult for hepatic stem/progenitor cells to survive for a long time and to differentiate into fully matured hepatocytes. When the histology of DPPIV+ foci was examined in detail, we realized that the irregularity of the size and shape of cells was more evident in Thy1- and CD44-derived than in MH-derived ones, especially in the livers within 2 weeks after transplantation. Furthermore, in some Thy1- and CD44-derived foci, the distribution of the DPPIV+ membrane was irregular, which means that bile canaliculi (BCs) were not connected among cells, and sinusoids were indistinct. In fact, in such irregular foci, especially in smaller ones, invasion of SECs (SE-1+ cells) was not obvious and sinusoids were not reconstructed. However, even when the deficiency of SECs was prominent in Thy1- and CD44-derived foci, many MH-derived foci had already incorporated SECs and reformed sinusoids. In addition, at that time more than 70% of hepatocytes in the foci expressed C/EBPα in their nuclei, a percentage similar to that in the surrounding host liver. Although no relationship between hepatic maturation and sinusoid formation in the donor-derived foci has been reported, a delay of maturation was shown in the urokinase-type plasminogen activator transgene (uPA)/recombination activation gene 2 knockout (uPA/RAG-2) mouse liver into which fetal hepatic cells were transplanted (5). In spite of the fact that transplanted MHs could form gap junctions and BCs within 1 week, it took more than a month for the transplanted fetal cells to express albumin and α-fetoprotein (AFP) mRNAs and DPPIV protein (16). Furthermore, in all CD44-derived foci that survived for a month or more, SECs were integrated into the foci and many of the cells expressed C/EBPα protein. The ratio of the positive cells in CD44-derived foci was as large as that of the positive cells in both MH-derived ones and their surroundings (70–90% at day 30). Thus, it may take time for progenitor cells to differentiate into MHs, and their survival and maturation may have a close relationship with sinusoid formation. In other words, the lack of integration of donor cells into resident hepatic plates may result in the early loss of the foci. Considering the finding that many transplanted hepatic stem/progenitor cells could not survive in the recipient livers for a long time and that engrafted MHs continued to proliferate with a minimum loss of the cells, both the integration into hepatic plates and the nature of the cells themselves may be very important to survive and expand.
It is well known that SA-β-Gal is expressed in cells with cellular senescence and that the expression is histochemically detected at pH 6 (8). In the present study, although neither apoptotic nor SA-β-Gal+ cells in foci derived from any cells were evident at day 14, many SA-β-Gal+ cells appeared in more than 30% of CD44-derived foci at day 60. The frequency of the appearance of SA-β-Gal+ cells was much lower in MH-derived foci than in CD44-derived ones. One year after transplantation, the number of CD44-derived foci was remarkably decreased, whereas most MH-derived foci were maintained. Considering that CD44-derived hepatocytes proliferated more rapidly than MH-derived ones in the early period after transplantation, the engrafted CD44-derived cells may have reached the state of cellular senescence much earlier than MH-derived ones. Unfortunately, we could not clarify why Thy1+ and CD44+ cells are more prone to cellular senescence than MHs. However, we can hypothesize based on our results. As we reported in a previous paper (21), we think that most CD44+ cells are derived from MHs. In the adult rat liver less than 2% of MHs have the potential to become SHs (29). After acute hepatic injury by GalN, those cells may have already divided once or twice to become SHs. Furthermore, those cells may have experienced many more divisions than other MHs in the development. Therefore, the transplanted CD44+ cells must rapidly proliferate. In fact, in the foci within 2 weeks after transplantation, CD44-derived foci are clearly larger and cells in the foci show higher positivity of Ki67 than MH-derived ones. On the other hand, transplanted MHs slowly proliferate compared to CD44+ cells. At 1–2 months after transplantation, the accumulation of the number of divisions in CD44-derived cells may be much larger than that in MH-derived ones. In addition, the rapid proliferation of the cells may cause cellular senescence. On the other hand, at day 30 after transplantation, we certified that most foci derived from CD44+ cells were integrated into hepatic plates and that E-cadherin and connexin 32 (Cx32) were well expressed between DPPIV+ cells and resident hepatocytes (data not shown). The ratio of C/EBPα-positive cells in both CD44- and MH-derived foci was not different from those in the surroundings. Therefore, we do not think that “anoikis” occurs in the CD44-derived foci, which can survive until 30 days after transplantation.
We examined the expression of p16 in DPPIV+ foci as another marker of cellular senescence (23). At day 14, the ratio of Ki67 in the foci derived from CD44+ cells was higher than that in the foci derived from MHs, whereas there was no difference in the ratio of p16+ cells between the foci. However, at days 30 and 60, the ratio of p16+ cells in CD44-derived foci was higher than that in MH-derived ones. These results suggest that in CD44-derived foci the cells rapidly proliferate within 2–3 weeks after transplantation and that many of the cells enter growth arrest and finally die. On the other hand, in MH-derived foci the cells slowly proliferate and the growth speed may be constant. Although some cells fall into cellular senescence, the ratio may be constant and the number may be less than that of growing cells. Therefore, MH-derived foci may continue to proliferate and survive even one year after transplantation. Although the details were not well clarified in the present experiment, the stem/progenitor cells may die earlier than MHs and be immediately excluded from the host liver. Further experiments are required to clarify the mechanism by which this might occur.
Footnotes
Acknowledgments
We thank Dr. Tomoko Sonoda (Department of Public Health, Sapporo Medical University School of Medicine, Sapporo, Japan) for help with the statistical analysis. We also thank Ms. Minako Kuwano and Ms. Yumiko Tsukamoto for technical assistance and Mr. Kim Barrymore for help with the manuscript. This study was supported by a grant from the Ministry of Education, Culture, Sports, Science and Technology, Japan, Grants-in-Aid for Young Scientists (B) for J. Kon (19790294) and N. Ichinohe (22790385), and Grant-in-Aid for Scientific Research (B) for T. Mitaka (17390353/21390365); the Japan Science and Technology Agency, Japan, the Science and Technology Incubation Program in Advanced Regions for T. Mitaka; Program for developing the supporting system for upgrading the education and research for T. Mitaka; Ministry of Health, Labour and Welfare, Japan, Health and Labour Sciences Research Grants, Research on Advanced Medical Technology for T. Mitaka; Program for developing the supporting system for upgrading the education and research for T. Mitaka; the Suhara Memorial Foundation for T. Mitaka. The authors declare not conflicts of interest.
