Abstract
This study was designed to explore whether liver sinusoidal endothelial cells (SECs) play a pathological role in liver injury of heatstroke (HS) in rats. An HS rat model was prepared in a pre-warmed incubator. Rats were randomized into four groups: HS-sham group (SHAM group), the 39°C group, the 42°C group, and the HS group. The serum concentrations of SEC injury biomarkers including hyaluronic acid (HA), von Willebrand factor (vWF), thrombomodulin (TM), were measured. Plasma alanine aminotransferase (ALT) and aspartate aminotransferase (AST) activities and endothelium-derived vasoactive substances including endothelin-1 (ET-1) and nitric oxide (NO) were determined using a commercially available kit. Hepatic tissues were obtained for histopathological examination, electron microscopy examination, immunohistochemistry, and reverse transcription polymerase chain reaction (PCR) analysis. Our study team found increased levels of plasma ALT/AST during the course of HS. We were also able to detect microcirculation changes and inflammatory injury of the liver (especially in the sinusoidal areas). In addition, markers of SEC injury were significantly elevated. Thrombosis-related markers including vWF and TF expression levels were significantly upregulated and TM levels downregulated. Furthermore, imbalance between ET-1 and NO levels were detected. In conclusion, damage of SECs could result in microcirculation disturbances and pro-inflammatory injury in the liver during HS, which could prove to be a potential pathogenic mechanism of liver injury in HS.
Keywords
Introduction
Heatstroke (HS), according to its pathophysiology, can be defined as a form of hyperthermia associated with a systemic inflammatory response leading to a syndrome of multi-organ dysfunction (MODS) in which encephalopathy dominates. 1 Studies has shown that thrombosis and inflammation may be the major pathophysiological changes in HS. 2 Hyperthermic stress can directly induce endothelial cell death by apoptosis, widespread hemorrhage, thrombosis, and transmural migration of leukocytes in association with microvascular endothelial injury that are also the prominent features of HS. 2 Current knowledge on HS suggests that excessive activation/injury and apoptosis of the endothelial cells (ECs) might activate thrombotic and fibrinolytic responses to induce disseminated intravascular coagulation (DIC) or to develop pro-inflammatory phenotype leading to inflammatory injury of organs in HS.3–5 Therefore, studying endothelial cells may help in further investigations of the pathogenesis of HS.
Clinical and experimental studies have consistently demonstrated that liver injury is a common phenomenon in HS,6–9 but its mechanism remains elusive. Some studies show vascular congestion, thrombosis, increased inflammatory cells, and disruption of architecture in the liver, which are mainly displayed around the sinusoidal area.10,11
Liver sinusoidal endothelial cells (SECs), with special fenestrated structure, act as a filter between blood and hepatocytes and play important roles in liver physiology and disease. 12 SECs injury may activate microcirculatory dysfunction in the liver and subsequently disrupt the integrity of the endothelium, allowing for infiltration of immune cells from the blood into hepatic parenchyma, thereby leading to liver tissue injury.13–15 Emerging evidence suggests that SECs are a direct target in liver injury during clinically relevant stresses, such as ischemia/reperfusion (I/R) injury, 16 sepsis, 17 and hepatic toxicants. 18 In addition, SECs are sensitive to temperature changes and can be easily damaged during cold preservation for liver transplantation. 19 Studies have also shown that SECs can be damaged by bacterial lipopolysaccharides (LPS).17–20 Interestingly, evidence from HS patients and animal models demonstrate high LPS levels in the portal vein and systemic circulation.21–23 In summary, SECs are the key components of hepatocyte–blood barrier. Hence, we hypothesize that damage to these cells due to heat stress could be an important event in liver injury of HS. However, no study has yet focused on the effect of heat stress on SECs.
In our study, we focused on the changing characteristics of liver damage in rat HS models. We investigated whether heat stress could induce SEC injury. We also investigated the effects of activated SECs on the microenvironment and inflammatory injury in liver, during HS. These results will help to further understand the mechanisms of liver injury during HS and to design a potential therapy.
Materials and methods
Animals
Because estrogen may play a protective role during HS and organ injury, 24 only pathogen-free adult male Wistar rats, weighing 250–320 g, were used in this study (Experimental Animal Center of Guangzhou General Hospital of Guangzhou Military Command our hospital, animal quality certification number: SCXK2006-0015, Guangzhou, China). The institutional Animal Care and Use Committee approved all procedures involving experimental animals.
HS model
The study rats were anesthetized by intraperitoneal injection with sodium pentobarbital (50 mg/kg body weight). Anesthetized rats were placed inside a temperature- and humidity-controlled chamber (pre-set at 40°C ± 0.5°C and a relative humidity of 60% ± 5%) and a trocar (24G) was cannulated into the right femoral artery of the rats to monitor mean arterial pressure (MAP). Rectal temperature (Tc) was continuously monitored at 10-min intervals using a Multi-parameter Physiological Monitor (Infinity Delta XL: Dräger, Germany) and the moment at which MAP dropped from the peak to a value of 25 mmHg and their core temperature was more than 42°C and was considered as the onset of HS. 25 Rats were randomly assigned into four groups (n = 12 per group): the SHAM group (subjects were sham-heated in the same chamber preset at 25°C and humidity of 35% ± 5% throughout the experiment); a core temperature 39°C group (once the Tc reached 39°C, rats were taken out for blood and tissue sampling collection); the 42°C group (once the Tc reached 42°C, rats were taken out for blood and tissue sampling collection); and the HS group. Our pilot study showed that the time of HS onset in rats was found to be about (76.88 ± 1.46 min); 26 therefore, HS group rats were exposed to heat stress for exactly 77 min in this experiment.
Blood and tissue analysis
After induction of HS, the liver was exposed by midline laparotomy; the blood samples (6 mL) were acquired immediately from the inferior vena cava using vacuum blood collection tube (von Willebrand factor (vWF), thrombomodulin (TM)) and heparinized tubes (nitric oxide (NO), endothelin (ET)-1, hyaluronic acid (HA)). Blood samples were centrifuged at 1500g for 15 min to acquire plasma; serum was centrifuged at 1000g for 20 min. Aliquots of the serum and plasma were stored at −80°C until assayed. Liver tissues were obtained at autopsy for pathology and immunohistochemical (IHC) examination. The liver tissue homogenate was used for reverse transcription polymerase chain reaction (PCR) analysis.
Aspartate aminotransferase and alanine aminotransferase analysis
Plasma alanine aminotransferase (ALT) and aspartate aminotransferase (AST) activities were determined using a commercially available kit (Nanjing Jiancheng Bioengineering Institute, Nanjing, China) following the manufacturer’s instructions.
Measurement of serum levels of TM, vWF, NO, ET-1, and HA in plasma
The concentration of TM, NO, ET-1, and HA in the serum and vWF in plasma were measured with commercially available enzyme-linked immunosorbent assay (ELISA) kits (Cusabio Biotech, Wuhan, China) following the manufacturer’s instructions. Samples with values below the detection limit were excluded.
Histological examination of liver sections
For histological analysis, liver tissues were fixed in buffered formalin solution overnight with paraffin embedded. Sections were stained with hematoxylin and eosin (H&E) using standard protocol and images were obtained under a light microscope (OLYMPUS BX51, Japan).
Electron microscopy of liver sections
The 1–2 mm3-sized tissues for transmission electron microscopy (TEM) were prefixed in glutaraldehyde and post-fixed in osmium tetroxide. After gradient dehydration by ethoxide and acetone, the tissues were embedded by epoxy resin Epon812 and examined under a transmission electron microscope (Hitachi Ltd, Tokyo, Japan).
Immunohistochemistry
For immunohistochemistry, serial sections were deparaffinized, rehydrated, and incubated with different antibodies at 4°C overnight according to the manufacturer’s directions. Thrombomodulin (TM) Antibody (H-300; sc-9162; Santa Cruz Biotechnology, Santa Cruz, CA, USA), tissue factor (TF) Antibody (FL-294; sc-30201; Santa Cruz Biotechnology), and anti-von Willebrand Factor (AB7356, Millipore Germany) were used, and images were obtained under a light microscope. The optical densities (ODs) of the positive areas were randomly counted in six fields using Image-Pro Plus ver. 6.3 software (Media Cybernetics, Silver Spring, MD, USA), under a light microscope. At the same time, the OD value of tissue on the same slice was measured as background. By subtracting the OD of the background on the same tissue, the calibrated optical densities (COD) was obtained, which was then used for one-way analysis of variance (ANOVA).
Reverse transcription PCR
Intercellular adhesion molecule-1 (ICAM-1), TF, and IL-6 expression levels in the liver tissue homogenate were measured by reverse transcription PCR using a model Chromo 4 reverse transcription PCR detection system (Bio-Rad, Hercules, CA, USA). Primers used were as follows: 5′-GCAGGTGAACTGCTCTTCCT-3′ and 5′-GTCTTCCCCAATGTCGCTCA-3′ for ICAM-1; 5′-AATGTGGAGAGCACCGGTTC-3′ and 5′-CGTTCATCTTCTACGGTCACATTC-3′ for TF; and 5′-GACTGATGTTGTTGACAGCCACTGC-3′ and 5′-TAGCCACTCCTTCTGTGACTCTAACT-3′ for IL-6. Glyceraldehydes-3-phosphate dehydrogenase (GAPDH) was used as the endogenous reference gene to normalize the data.
Statistical analysis
All data were analyzed using the SPSS 17.0 software (IBM, USA). Data are presented as means ± SEM. Comparison between each group was performed using single-factor ANOVA (one-way ANOVA). P < 0.05 is considered statistically significant.
Results
Effects of heat stress on liver function
We examined the levels of hepatic enzymogram at the onset of HS. Higher temperature led to the upregulation of plasma ALT and AST levels. Compared with the Sham-heated group, both the 42°C group and the HS group showed significant elevation in AST and ALT levels (P < 0.05, Figure 1).

Effects of heat stress on liver function. Plasma concentration levels of AST (black bars) and ALT (gray bars) are shown for the four treatment groups. * and # indicate significant difference (P < 0.01) in comparison to Sham-heated groups (baseline) for AST and ALT values, respectively.
Heat stress–induced histopathologic changes in the liver
To investigate whether heat stress initiates histopathological damage in the liver, we examined the liver section by microscopy at the onset of HS. As expected, no significant morbid changes were observed in both SHAM and 39°C groups, while mild thrombosis was seen in the 42°C group. Following the onset of HS, widespread microthrombus formation was seen in the liver, especially around the sinusoidal area (Figure 2).

Comparison of heatstroke-induced histopathological changes in the liver. All liver sections were stained with hematoxylin and eosin. (a) Sham-heated rats were used as control; (b) no significant morbid changes were observed in both a and b groups in 39°C group; (c) mild sinusoidal congestion was observed in 42°C group; and d) after HS was onset, severe sinusoidal congestion, inflammatory cell infiltration, and hepatocellular (hepatocyte) necrosis were observed in HS group. Neutrophils accumulated in the sinusoids and the venules and extravasated into the parenchymal tissue (magnification 400×).
Heat stress–induced expression of inflammatory injury and thrombosis-related markers
Rats were exposed to designated temperatures and expression of messenger RNA (mRNA) with increasing temperature was examined by transcription PCR. Values were normalized to sham group (set to 1). Heat exposure resulted in a progressive increase in mRNA expression of TF, IL-6, and ICAM-1 (P < 0.05, Figure 3).
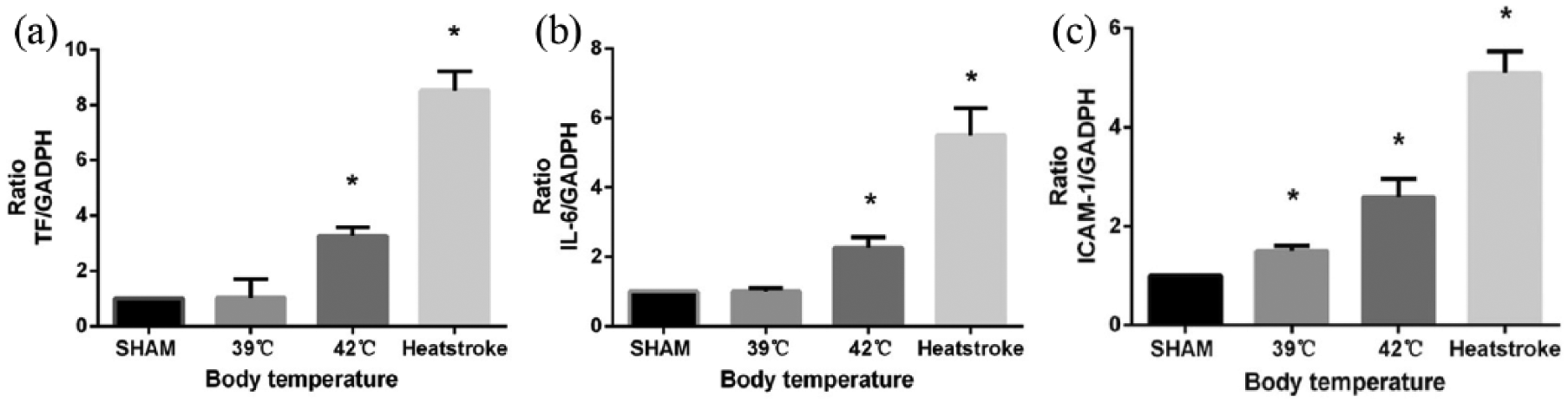
Effect of temperature on TF, IL-6, and ICAM-1 gene expression. Liver tissue homogenate were collected and subjected to real-time PCR analysis for the indicated genes reverse transcription PCR. (a) Heat exposure caused a significant increase in expression of TF, especially in HS group; (b) level of ICAM-1 expression was not affected in SHAM group and 39°C group, but increase was observed when exposed to 42°C group, especially in HS group; (c) the exposure to heat produced a progressive increase in IL-6 expression; Values were normalized to sham group (set to 1). Results are reported as means ± SD, n = 3 per group. *P < 0.05 compared to sham group. Glyceraldehydes-3-phosphate dehydrogenase (GAPDH) was used as internal control.
Heat stress–induced marked SECs injury
We measured the plasma markers of SECs to assess the injury in each group exposed to different temperatures (Figure 4). In this model, elevated plasma concentration of HA was observed early in heat-treated rats. Furthermore, the levels of HA and vWF were much higher in the HS group than the other groups. The heat exposure also resulted in a significant decrease in sTM level (P < 0.05), suggesting heat stress can induce severe SEC injury in HS. In addition, ultrastructural findings of liver by transmission electron microscopy further confirmed SEC injury following heat stress. Sinusoidal narrowing, severe sinusoidal congestion, and hepatocyte necrosis were observed along with endothelial gap formation and structural disorder in the HS group. Conversely, no significant structural changes were seen in both the SHAM and the 39°C groups (Figure 5). These results indicate that the heat stress could directly damage SECs, which may play an important role in the microthrombus formation during HS.

Effect of temperature on the expression markers of SEC injury. Compared to the sham group, gently elevated serum TM was displayed in the 39°C group, but subsequently decreased levels were observed in the 42°C and HS groups (a). Heat exposure caused a significant increase in the expressions of both HA and vWF (b and c). (*P < 0.05), n = 12 per group.
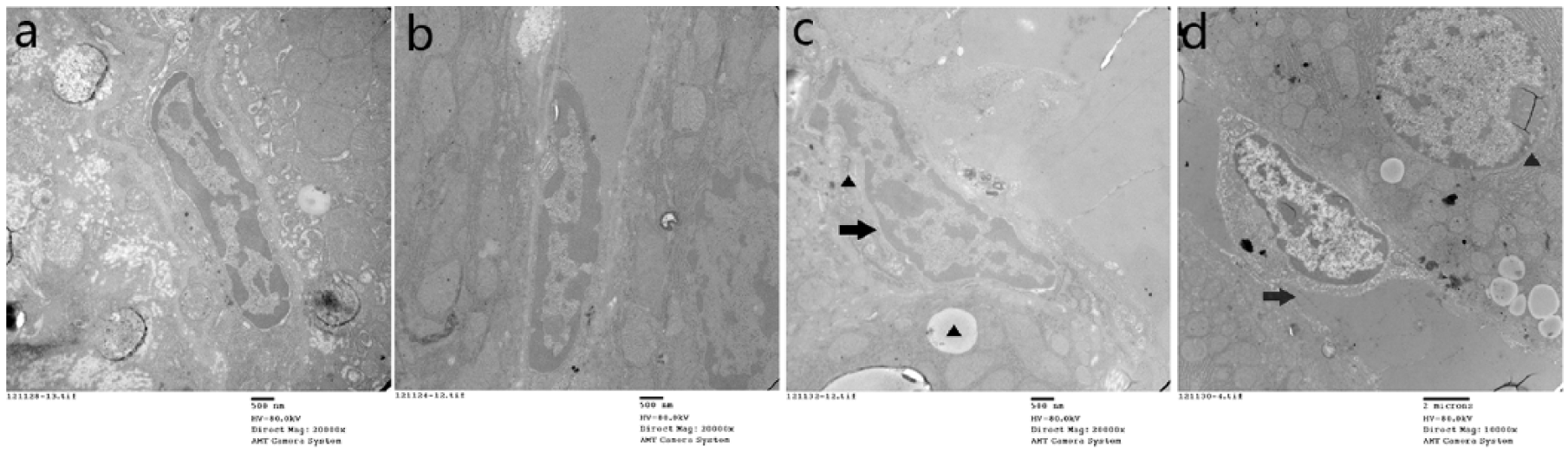
Electron microscopic comparison of heatstroke-induced SEC changes in rats. (a) Sham-heated; (b) in 39°C group, no significant morbid changes were observed in both group a and b, maintained integrity of the sinusoidal lining and endothelium; (c) in 42°C group, RBCs and platelets accumulated into the sinusoidal lumen, narrowing of lumen and SEC swelling were found, microvilli and cytoplasm ballooning were observed in SEC; (d) in HS group, sinusoidal narrowing, severe sinusoidal congestion, hepatocyte necrosis, and endothelial gap formation with RBCs accumulation in the extra sinusoidal space were also observed. Original magnifications: 20,000× (a, b, and c) and 10,000× (d).
Heat stress–induced endothelium-derived vasoactive substances imbalance in intrahepatic microcirculation
The levels of ET-1 were very low in the sham-heated and 39°C groups, whereas significant increase was seen in the 42°C and the HS groups. However, concentration of the NO showed no significant difference in the sham group and the 39°C group. Interestingly, a distinct decrease in NO levels was observed in the 42°C and HS groups (Figure 6).

Effects of heat stress on vasoactive substances expression levels in the intrahepatic microcirculation of rats. Heat stress induces imbalance between ET-1 and NO levels in the liver. Serum levels of ET-1 were significantly higher in the heatstroke group than in the sham-heated group (*P < 0.05). NO levels showed a decrease compared to the sham-heated animals (*P < 0.05). The difference between the groups is significant (*P < 0.05), n = 12 per group.
Heat stress-induced thrombosis-related marker expression in SECs
During the temperature course of HS, VWF and TF expression levels were significantly upregulated, while TM levels were downregulated (P < 0.05). (Figure 6)
Discussion
HS has high mortality and morbidity with no specific targeted treatment and unclear pathophysiology. An earlier study shows that microvascular injury, thrombosis, and inflammation may be the major pathophysiological changes in HS. 2 Liver injury is a common phenomenon in HS, but its mechanism is not fully understood. Several mechanisms can contribute to hepatic injury, including inflammation and microcirculatory dysfunction. We observed functional and morphological changes of the liver during heat stress or severe HS. Similar to the major pathophysiological changes in HS, our results showed elevated levels of plasma AST/ALT in the HS group (P < 0.05) indicating heat stress could directly promote severe liver injury during the onset of heat shock (Figure 1). By measuring the morphological changes in the liver during HS, severe microcirculation disturbances and inflammatory injury of the liver were observed in our study, which displayed as severe hepatic sinusoid congestion and hemorrhage, as well as thrombic and inflammatory cell infiltration in the HS group (Figures 2 and 5).
Hepatic microcirculation consists of special capillary system called sinusoids with a fenestrated endothelium that allows contact between hepatocytes and the blood. 27 During the early phase of inflammation, hepatic sinusoidal endothelium participates in host defense mechanisms and blood flow regulation by forming a functional barrier to separate the hepatic parenchyma from blood constituents passing the liver. Furthermore, SEC is a major target for injury and has the capacity to produce immunoregulatory and proinflammatory cytokines, such as IL-1 and IL-6.28–30 Moreover, SECs produce important regulators of vascular tone, including NO and ET. 30 SECs are the barrier between hepatocytes and blood. Therefore, it is extremely noteworthy to study the exact role of SECs in liver injury of HS.
In this study, markers of SECs injury were measured at different temperature conditions. Histological analysis showed that the extent of the damage of the SECs was associated with the degree of heat stress. ELISA measurements revealed that levels of both plasma HA and serum vWF were significantly elevated following heat stress, while the concentration of TM showed significant decrease in the 42°C group and HS group (Figure 4). HA is a marker of hepatic SEC function. More severe injury to SEC corresponds to the higher concentration of HA in the plasma. 31 Serum vWF is also an indicator of SEC injury, as the levels are elevated with exposure of the rats to increased temperature as shown in our study. The current view suggests that TM levels are significantly decreased in SECs isolated from endotoxin-treated rats. After treatment with endotoxin and tumor necrosis factor (TNF)-α in vitro, TM expression in cultured SECs was also reduced. Accordingly, recombinant TM is capable of attenuating increased levels of serum fibrin degradation products, fibrin deposition within liver sinusoids, injury to SECs, and liver dysfunction after endotoxin exposure. 32 Similar to endotoxin, our results showed that TM increased mildly at 39°C but decreased significantly in the 42°C group and HS group (Figure 4). Furthermore, IHC staining with TM and vWF confirmed the same elevated expression markers of SEC injury (Figure 7). Ultrastructural findings of SEC by TEM further confirmed the extent of SEC injury and that it was aggravated during the induction of HS, with its chromatin staining deeply and nuclear chromatin condensation, narrowing and SEC swelling, and microvilli and cytoplasm ballooning (Figure 5). These results indicate that the SEC is an initial target of injury induced by heat stress, which may play a crucial role in liver injury during HS.
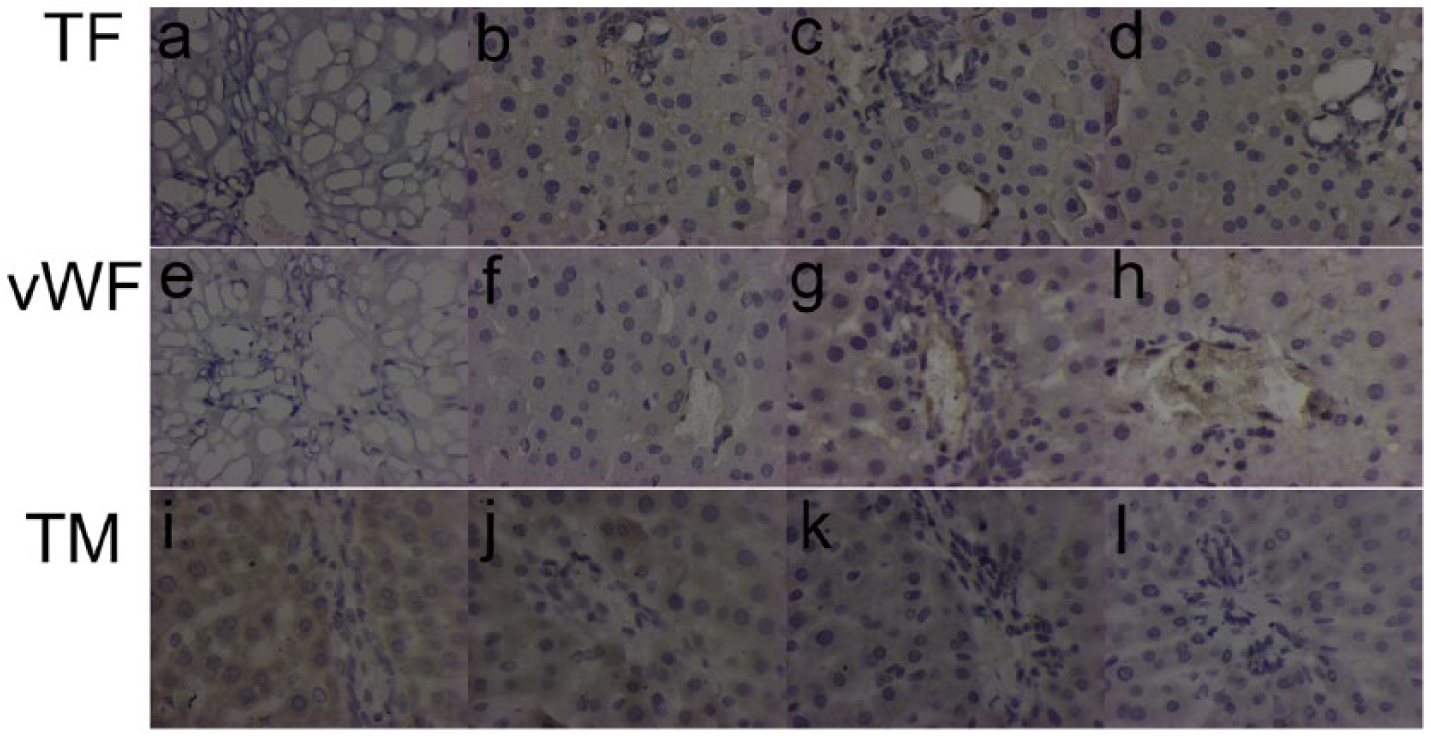
Immunohistochemical localization and expression. Immunohistochemical staining with an anti-tissue factor (TF) antibody. (a) and (b) No TF expression was detected in the sinusoids of the control group; (c) TF expression was detected in the sinusoidal endothelial cells; (d) TF expression was higher in the sinusoids of the HS group; (e) and (f) immunostaining for vWF antigen. No expression of vWF was found; (g) and (h) vWF expression in the microvasculature barrier and around the sinusoids; (i) and (j) immunostaining for TM. TM expression was detected in both sham-heated group and the 39°C, group; (k) and (l) TM was downregulated in the 42°C group and the heatstroke group compared to controls. Positive staining appears as brown color. COD was measured and compared with Sham group (*P < 0.05).
In addition, we observed the pro-coagulant function in the microenvironment of activated SECs. The microcirculation of the liver is affected by several factors such as microvascular nutrition, neutrophil accumulation in the sinusoids, and alteration of the sinusoidal lumen. First, we focused on whether sinusoidal vasomotor dysfunction occurred in HS. Endothelium-derived vasoactive substances play an important role in the regulation of intrahepatic microcirculation. Current knowledge suggests that the most two potent vasoactive substances in the liver are ET-1 that primarily induces vasoconstriction and NO, which causes vasodilatation in the hepatic sinusoids. 33 The balance of the vasoactive effects between ET-1 and NO is crucial to maintain normal hepatic microcirculation. The imbalance between ET and NO leads to active vasoconstriction, narrowing of sinusoidal lumen, and subsequent microcirculatory failure. 34 In this study, severe unbalance of vasoactive substances was seen and the concentration of ET-1 was elevated in the 42°C and HS groups. On the contrary, the NO level showed a decreased association with the degree of heat stress (Figure 6). These results indicate that the imbalance between ET-1 and NO levels associated with the injury of SECs is an important factor, which contributes to the microcirculatory derangement during HS. Meanwhile, the expression of TF and TM further explained the pro-coagulant function of SECs. Studies have demonstrated the correlation between SECs and the incidence of sinusoidal thrombosis within the septic liver tissue. 35 Our study demonstrated that SEC injury could cause elevated expression of TF (Figures 3 and 7) and vWF (Figures 4 and 7) and decreased expression of sTM (Figures 4 and 7) implicating that these markers have a close relationship with microcirculation changes. TF is a well-established trigger of the coagulation cascade in many pathological conditions36,37 and TF expression demonstrates severe SEC damage. 35 Recent studies revealed that TF mediates coagulation activation in a baboon HS model, 38 but there are no studies on TF expression in rat model of HS. In this study, using IHC analysis, high level of TF expression was detected in the sinusoidal area (Figure 7) and significantly elevated TF mRNA level was observed by reverse transcription PCR (Figure 3). These results suggest that TF pathway could be responsible for the microcirculation changes in HS. Due to the limitations in this study, further exploration using pharmacological inhibition or TF blocking antibodies are needed to confirm these findings. On the other hand, TM is a receptor for thrombin, which is mainly located on the surface of endothelial cells and plays an important role as an anticoagulant protein. In sepsis, endothelial cell expression of TM is strongly downregulated. Such downregulation causes an impaired activation of protein C that is central in the modulation of coagulation activation and inflammatory processes. 39 Similarly, a decreased TM expression in SECs may result in intrasinusoidal microthrombus formation during sepsis. 39 Our study also demonstrated that the levels of TM were significantly decreased (Figures 4 and 7) when exposed to heat stress, which may result in intrasinusoidal microthrombus formation (Figures 2 and 5). These results suggest that decreased TM levels may be responsible for microcirculatory changes in HS. Moreover, a marked increase in staining of SECs with vWF-Ag and platelet accumulation was found in the liver during HS (Figure 7). It is known that vWF-Ag can facilitate the adherence of platelets to the subendothelium of injured ECs. 40 Thus, our results suggest a possible role of vWF in platelet-thrombus formation during HS.
To observe the pro-inflammatory effect of SECs under heat stress, we analyzed the impact of heat stress on enhancing the production of IL-6 by SECs. The IL-6 can directly increase endothelial permeability and play an essential role in organ polymorphonuclear leukocytes infiltration.41,42 Furthermore, activated SECs exert a pro-adhesive function by upregulating ICAM-1 expression, which further primes the sinusoids as a site for inflammatory leukocyte adhesion and transmigration. 43 After exposure to heat stress, we found that gene expressions of ICAM-1 and IL-6 increased significantly (Figure 3) along with the extent of SEC injury indicating that heat stress can induce the pro-inflammatory function by activating SECs. ICAM-1 is an endothelial- and leukocyte-associated transmembrane protein. In extensive liver injury models, elevated ICAM-1 levels can predict the microcirculatory disturbances of liver injury.44,45 Our study shows accumulation and adhesion of neutrophils (Figure 2) and elevated levels of ICAM-1 (Figure 3). These findings indicate that elevated levels associated with damage to SECs occurred before the accumulation and adhesion of neutrophils during the progression of HS.
In conclusion, injured SECs exert pro-inflammatory, pro-adhesive, and pro-coagulant properties, which could further aggravate liver injury. Our data suggest that the microcirculatory injury of liver is an early event in HS. Also, morphological and functional changes in SECs were observed at the onset of HS, which probably lead to a disturbed microcirculation and inflammatory injury of the liver.
There are some limitations to this study. The HS animal model does not ideally simulate the clinical course of HS. Direct experimental evidence of SEC damage in heat stress is not available in our study. We have demonstrated that heat stress can induce marked SECs injury and severe microcirculation disturbances and inflammatory injury in the liver as well as the expression of thrombosis-related markers on the SECs. However, the exact correlation between the SECs injury/activation and liver injury during HS needs to be further elucidated.
Footnotes
Acknowledgements
X.Z. and Y.C. contributed equally to this study and share first authorship.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the National Natural Science Foundation of China (Grant Nos 81503460, 81671896, and 81471839) and Science and Technology developing Project of Dongguan City (No. 2014108101012).
