Abstract
Objectives: Exposure of spleen tissues to ionizing radiation during radiotherapy can induce cellular stress and immune-dysfunction leading to cellular senescence. Introduction: The process of a cancerous development is facilitated by the accumulation of senescent cells. This justifies the incorporation of anti-senescent medications during splenic irradiation (SI). Methods: In this study senescence was induced in the spleen of male albino rats by radiation exposure (5Gy-single whole body gamma-irradiation) then after 2 weeks, oral astaxanthin regimen was started once daily in a dose of 25 mg/kg for 7 consecutive days. Concurrent control groups were carried out. Results: the present data reflected that irradiation provoked an increase in the oxidative stress biomarkers (nitric oxide, lipid peroxidation and total reactive oxygen species levels)and the inflammatory biomarkers (Myeloperoxidase and interleukin-6). In addition irradiation led to the over expression of stimulator of interferon genes (cGAS-STING), mammalian target of rapamycin (mTOR) and Toll-like receptor 4 (TLR4) along with the lactate dehydrogenase (LDH), cyclin-dependent kinase inhibitor 1 (p21) cyclin-dependent kinase inhibitor 2A (p16) increment with elevation of tumor suppressor protein (p53) level. However, reduced glutathione contents and catalase activity were reduced post irradiation in spleen tissues, all these changes reflecting induction of cellular senescence. Astaxanthin treatment showed an improvement in the antioxidant/oxidative stress balance, inflammatory biomarkers, histopathological examination and immunohistochemical expressions of the tested proteins in the irradiated rats. Conclusion: the current findings offer a new insight into the senomorphic effect of astaxanthin following radiation-induced spleen senescence via STING, mTOR, and TLR4 signalling pathways.
Keywords
Introduction
Cell cycle senescence is defined as a stable and permanent state of cell cycle blockage whereby cells lack the ability to divide, regardless of ideally suited conditions for proliferation and impulses for mitosis. It occurs in most tissues and organisms. Cellular senescence emerges as an outcome of several intrinsic or extrinsic stressors and triggers. For example, DNA damage accumulation, telomere attrition, oncogene stimulation, organelle stress, irradiation and chemotherapy. Furthermore, it has been connected to a number of processes, including the suppression of tumours, healing of tissues, embryogenesis, and ageing of organisms.1–3
Radiotherapy (RT) is considered as a primary option for many types of cancer, frequently used alone or in conjunction with surgery and chemotherapy. But its effectiveness is restricted as it may cause serious side effects.4,5 RT causes fibrosis and inflammation along with apoptosis and senescence in many cases usually through DNA damage as a first mechanism to cell cycle arrest.6,7 The oxidative stress and inflammation are interrelated; as well they lead to the initial events of senescence. Senescent cell build-up produces a microenvironment that allows for the growth of tumours. 8
The spleen is a lymphoid organ that hosts a wide range of immunological roles along with its functions in haematopoiesis and red blood cell clearance. 9 Splenic irradiation (SI) is used as a restorative treatment strategy for splenomegaly associated symptoms, or secondary to hematologic and abdominal malignancies and disorders, or as an alternative to splenectomy. 10 As well, SI induces apoptosis which increased the level of senescence-associated β-galactosidase, p16, and p19 besides the reduction of phagocytosis activity and over expression of senescence-associated secretory phenotype (SASP) in the spleen. 11
Nevertheless, senescent cells can be identified by DNA damage, lake of proliferation and expression of oncogenes. None of these traits is completely comprehensive for senescence, and it is a must to examine several biomarkers concurrently to rationale this state. Since DNA damage is the main driver of senescent stress, it triggers the DNA damage response (DDR) and the definitive p53-p21 pathway. While p21 blocks the cyclin–cyclin-dependent kinase that control cell cycle progression. Evidences suggest thatp16 maintains the cellular senescence; p21 is particularly triggered in the initial stages of senescence.1,12 Involving mammalian target of rapamycin (mTOR) pathway in studying aging and senescence has been discussed extensively, while the mechanisms remain indefinable. mTOR is a protein kinase, serves as a feed sensor that influences cellular processes related to survival, growth, and proliferation.13,14 cGAS-STING (cyclic GMP-AMP synthase linked to a stimulator of interferon genes), activates the transcriptional factors included in the secretion of pro-inflammatory mediators. 15 It is considered as a connecting point between DNA damage and immune response. 16 Moreover, it is involved in the mechanistic role of protection against stimulators like irradiation. 17 Damaged DNA in the cytoplasm induced cGAS-STING/TLR4 pathway, which has a role in activation of the SASP. Extrinsic inducers of innate immune responses, like irradiation, also impact SASP induction leading to tissue inflammaging and senescence.8,18
The molecular name for Astaxanthin (ASX), a xanthophyll oxygenated carotenoid, is 3,3′-dihydroxy-β,β-carotene-4,4′-dione. 19 It is primarily biosynthesized by various algae, bacteria and fungi and consumed by marine animals. 20 Though structurally comparable to β-carotene, ASX exhibits antioxidant action that is 40 times greater, and over 500 times more effective than that of α-tocopherol. 21 ASX has an anticancer activity and antioxidant properties, 22 it can stimulate the immune system and is used to treat diabetes, neurological diseases, and cardiovascular conditions.23–26 In addition, ASX has an anti-aging role attributed to its antioxidant and anti-inflammatory properties.27,28 The current study was designed to evaluate the significance of employing astaxanthin as spleen senomorphic drug in radiation-induced senescence model. This aim was achieved by assessment of cGAS-STING, mTOR and TLR4 signalling pathway while also paying attention to its connection to oxidative/ antioxidant balance, inflammation, histopathology and cell integrity.
Materials and methods
Chemicals and kits
The Sigma-Aldrich chemical company (Saint Louis, Missouri, USA) provided the astaxanthin, chemicals, and reagents used in this investigation. We obtained the Enzyme Linked Immunosorbent Assay (ELISA) kits from MyBioSource San Diego, CA, USA, for the estimation of p21, p16, p53 and IL-6. The source of the monoclonal antibodies against mTOR and TLR4 was Dako Corp, Carpentaria, CA, USA. Measurements of serum lactate dehydrogenase and spleen catalase were conducted in compliance with the manufacturer’s kit instructions (Biodiagnostic Co., Dokki, Giza, Egypt).
Animal procedures and gamma irradiation
The National Research Centre in Giza, Egypt’s animal breeding unit provided the male albino rats, which ranged in weight from 150 to 180 g. Before being employed, rats were housed for a week in the animal facility of the National Centre for Radiation Research and Technology (NCRRT)-Atomic Energy Authority, Cairo, Egypt. They were housed in a plastic cage with a 12-h light-dark cycle, room temperature (25°C ± 2°C), and humidity (60% ± 10%). They were given a regular pellet feed and unrestricted access to water. All of the rats were cared for by competent staff members, and every attempt was made to minimize the quantity of animals used as well as their suffering. Following the published criteria of the National Research Council (US) Institute for Laboratory Animal Research (2011), the parameters of health and well-being as well as markers of pain and distress were monitored over the experiment time. The study was conducted in agreement with the U.K. Animals (Scientific Procedures) Act, 1986 and associated guidelines; EU Directive 2010/63/EU for animal experiments, Moreover, the Research Ethics Committee of the NCRRT-EAEA has accepted the experimental procedures under Permit No: 11 A/23. This study’s reporting complies with ARRIVE 2.0 requirements. 29
Rats were subjected to whole body irradiation at the NCRRT utilising the Gamma Cell-40 biological irradiator (Atomic Energy of Canada Limited; Sheridan Science and Technology Park, Mississauga, Ontario, Canada) with a 137 Caesium source. The irradiation chamber measured 40 cm in diameter and 10 cm in height, large enough to hold a whole animal group at once. The dosage rate was 0.47 Gy/min and the radiation dose level was 5 Gy.
Experimental design
Four groups of rats were used (6 rats per each) and allocated as follow: Group 1: control → normal rats received corn oil. Group 2: ASX→ rats were received a daily oral dose of ASX (25 mg/kg) according to Alam, Hossain 30 dissolved in corn oil for 7 days. Group 3: senescence (Sc) group → rats were exposed to acute whole body gamma radiation in a dose of 5 Gy for induction of senescence at the 1stday from the beginning of the experiment. 31 Group 4: Sc + ASX group →rats were exposed to 5 Gy as in group 3, then after 2 weeks rats were treated with ASX (25 mg/kg/day, orally) for 7 consecutive days. All rats were weighed and sacrificed after 3 weeks from irradiation. Blood samples were collected by decapitation under deep urethane anaesthesia prior to scarification, and spleen tissue samples were separated and prepared for further biochemical analysis as well as histopathological and immunohistochemical examinations.
Blood and tissue sampling
Blood samples were collected, left for 1 h at room temperature and then centrifuged at 3000 rpm for 15 min to separate sera, which were kept at-20°C till use for LDH determination. On the other hand, each rat’s spleen was promptly removed, cleaned with regular saline, and blotted with filter paper. The spleen tissue was divided into two portions; the first one was weighted and certain volume of saline was added to get 20% homogenate then homogenized. The homogenate was placed in a cooling centrifuge and processed at 10,000 rpm for 10 min at 4°C. The supernatant was then prepared and separated for biochemical analysis. The second part of spleen was immersed in 10% formalin for histopathological and immunohistochemical examinations.
Biochemical assays
Analysis of oxidative stress markers
Lipid peroxidation in spleen tissue was assessed by determining the concentration of thiobarbituric acid reactive substances (TBARS), which have been identified as malondialdehyde (MDA) according to Mihara and Uchiyama. 32 Reduced glutathione (GSH) and Total nitrate/nitrite (NOx) were determined in spleen tissue homogenates in normal saline following Beutler, Duron33,34 assay methods, respectively. A ready-to-use kit was employed to measure the catalase activity (CAT) with regard to the manufacturer’s instructions. The absorbance of pervious mentioned parameters were measured using UV/V spectrophotometer (Unicam 8625, Cambridge, UK).
Analysis of inflammatory markers
MPO activity was estimated in spleen homogenates according to the method of Bradley, Christensen. 35 On the other hand, the IL-6 was measured by ELISA technique using a commercially available kit according to the manufacturer’s guidelines.
Evaluation of cellular integrity and senescence markers
Using a kinetic ultraviolet (UV) kit, serum LDH was measured as an indicator of cellular integrity using the Gay, McComb 36 approach. The procedure was followed in compliance with the kit manufacturer’s directions. With a commercially available kit, the p21, p16, and p53 levels in the spleen tissue homogenate were assessed using the ELISA technique in accordance with the manufacturer’s procedures.
Estimation of STING gene expression using real-time quantitative PCR (RT–qPCR)
RNeasy Purification Reagent (Qiagen, Valencia, CA, USA) was used to isolate total RNA from spleen tissue homogenate in compliance with the directions provided by the manufacturer. Using spectrophotometry, the extracted RNA was estimated at 260 nm. The amplification of STING-specific fragments was carried out by PCR utilising the reverse transcription mixtures and the primer pairs F: 5′GGACAGTCGCTGACCGAGGTT3′ and R: 5′GCTTGTTGGGCCAGTCCTGATG3′ (gene bank accession number 025142159.1). The QuantiTectSYBRgreen PCR Kit (Qiagen, Germany) was used to conduct the real-time PCR in compliance with the manufacturer’s instructions, which were provided by Applied Biosystems 7500 Instrument, USA. As an invariant endogenous control, all values were normalised to the β-actin genes (F: 5′AGGCATCCTCACCCTGAAGTA3′ R: 5′CACACG-CAGCTCATTGTAGA3′) (gene bank accession number NM 001009945.1). The fold changes were then used to compute the relative quantification using the following equation: ∆CT = ∆CTsamples − ∆CTcontrol.
Histopathological examinations
Spleen samples of all groups underwent regular processing to create paraffin blocks from formalin-fixed specimens, Following this, 4-5 μm serial slices were made and stained with hematoxylin and eosin (H&E) according to Bancroft, Stevens. 37 Rats that had exposed to radiation as well as those that had received treatment had their spleens subjected to a qualitative analysis to determine the severity of the lesions. Severity scores ranging from 0 to 4 were utilized to measure specific observable criteria. 38
Immunohistochemical studies
Immunohistochemical detection of TLR4, IL-6, and mTOR expression in spleen of all groups was carried out utilizing Sigma Chemical Co.'s avidin-biotin peroxidase method. Paraffin slices from every group’s spleen were exposed to monoclonal antibodies against TLR4, IL-6, and mTOR (Abcam, ab22048, ab271042, and ab32028 respectively), Cambridge, MA, USA) at 1:200, 1:400, and 1:200 dilutions, respectively. Following this, sections were exposed to reagents for the avidin-biotin-peroxidase method. (Vactastain ABC peroxidase kit, Vector Laboratories). DAB (Chromagen 3,3 – diamino benzidine tetrahydrochloride, Sigma Chemical Co.'s) was utilized for visualization of both markers expression. 39 Quantitative image analysis of the optical density of the positive brown hue in five microscopic fields was conducted using Image J, 1.46a, NIH, USA
Statistical analysis
The data provided was presented as mean ± standard error of the mean (S.E), and the Tukey-Kramer multiple comparison tests were conducted after one way analysis of variance (ANOVA). These statistical tests were performed using Graph Pad Software Instant (version 8). At p < .05, the statistical significance level was established. The sample size of animals was calculated according to Charan and Kantharia , Arifin and Zahiruddin.40,41
Results
ASX protects against oxidative stress induced by irradiation in spleen
Excessive reactive oxygen and nitrogen species (RONS) production are checkpoints for cellular damage. Irradiation of rats led to a marked decrease in reduced GSH and catalase activity (markers of anti-oxidant capacity) as compared to control group, GSH and catalase level were decreased to 32% , 92% respectively (Figure1(A) and (B)). Treatment of ASX protected against these changes. Alternatively, acute irradiation led to an increase in the splenic content of TBARS from 10.6 ± 0.7643 nmol/g. tissue to19.81 ± 0.47 nmol/g.tissue. On the other hand, NOx contents showed significant increase from 21.35 ± 2.588 µmol/g. tissue to 74.88 ± 6.73 µmol/g.tissue in irradiated group as compared to control group. The ASX treatment post irradiation guarded against the elevation in NOx and TBARS. The group received ASX alone showed normal levels of oxidative stress parameters. (Figure 1(C) and (D)). Astaxanthin ameliorates oxidative stress in spleen tissues of irradiated rats in senescence-induced model. A: GSH, B: Catalase, C: TBARS, D: NOx. Values are given as mean ± S.E, n = 6 statistically differences were assessed by one –way ANOVA followed by Tukey-Kramer multiple comparison test.*: significant change from control at p < .05, #: significant change from Sc group at p < .05.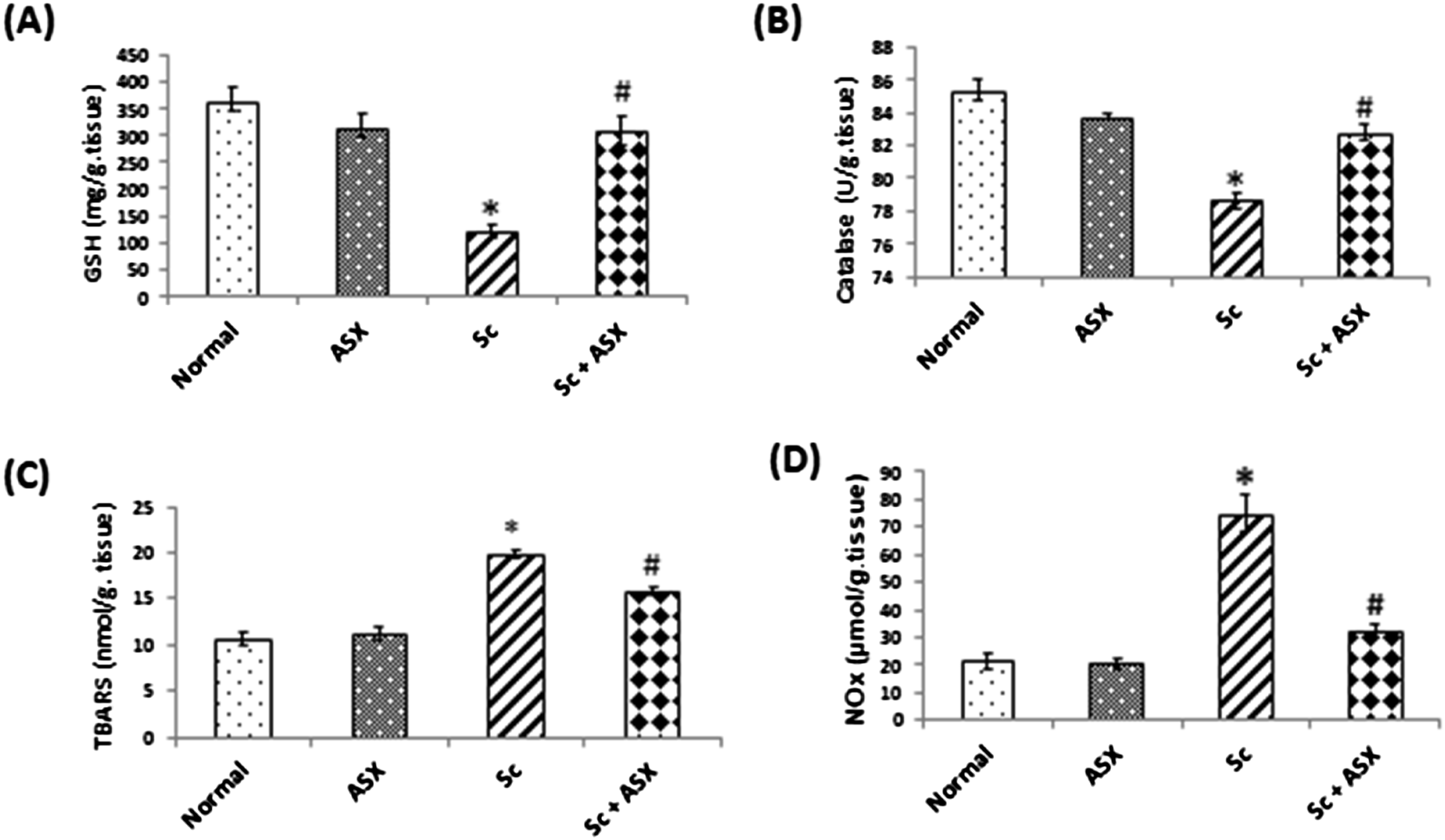
ASX ameliorates inflammation induced by irradiation in spleen
Irradiation of animals led to increment of MPO as an inflammatory marker, MPO was significantly increased to 348.6 % in senescence- induced rats (Figure 2(A)). As well as, irradiation of animal caused a significant elevation in IL-6 to 185.8% (Figure 2(B)). While the treated group with ASX showed marked decrease in elevated MPO and IL-6. The administration of ASX alone didn’t show any significant change in the measured inflammatory markers when compared to control. The increment of IL-6 was confirmed using immunohistochemical study, the immune expression of IL-6 showed intense diffuse immune-expression in Sc group compared to control group while ASX treatment could significantly diminish the expression of IL-6 (Figure 2(C)). Astaxanthin ameliorates inflammation induced by irradiation in spleen in senescence-induced model. A: MPO, B: IL6, C: IL6 expression, D: photomicrographs of immune-stained spleen sections IL-6; showing negative expression of IL-6 , in control and ASX group (a&b), diffuse intense expression in irradiated group (c), and significant decreased expression of IL-6 in ASX treated group (d). Image analysis of the optical density of the positive brown color. Values are given as mean ± S.E, statistically differences were assessed by one –way ANOVA followed by Tukey-Kramer multiple comparison test.*: significant change from control at p < .05, #: significant change from Sc at p < .05.
ASX protects against cellular integrity damage and senescence markers in spleen of irradiated rats
Ionizing radiation in senescence group led to loss of cellular integrity in spleen tissues, signified by an increase in serum LDH activity to approximately 4 folds increase from control group (Figure 3(A)). Moreover, the induction of senescence by irradiation, as evidenced by a marked elevation in the spleen content of p21, p53 and p16 reaching 2 folds, 5 folds and 3 folds increase respectively. Moreover, irradiation of animal with the mentioned regimen led to over expression of STING to approximately7 folds increase. Elevated p21, p53, p16 and activated STING were down regulated in the irradiated rats via ASX administration post irradiation. Treatment with ASX alone had hardly any effect on the former mentioned parameters (Figure 3(B) and (C)). Astaxanthin protects against cellular integrity damage and senescence markers in spleen of irradiated rats. A: LDH, B: P21, C: STING relative gene expression, D: p16, E:P53. Values are given as mean ± S.E, n = 6 statistically differences were assessed by one –way ANOVA followed by Tukey-Kramer multiple comparison test.*: significant change from control at p < .05, #: significant change from Sc at p < .05.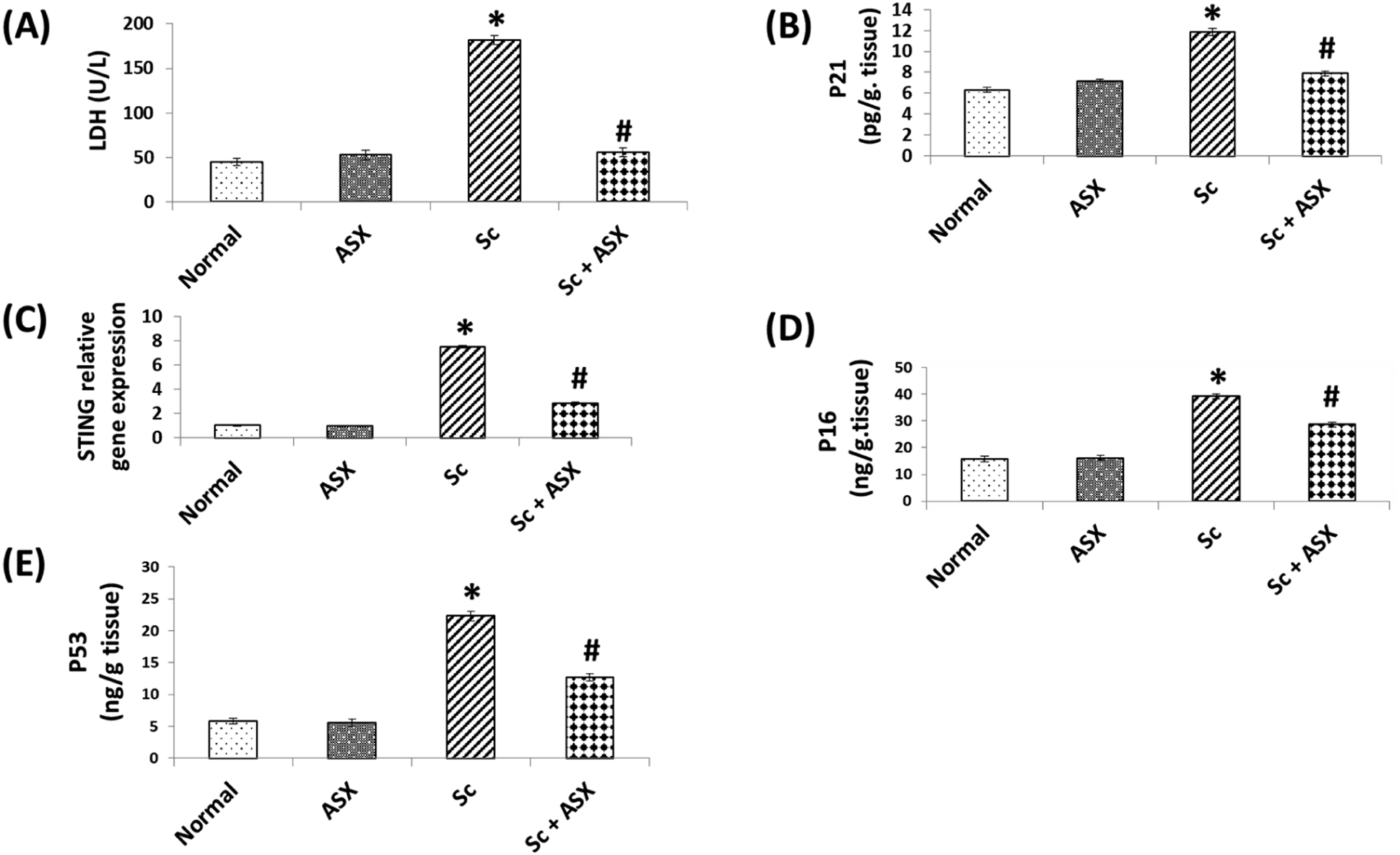
ASX ameliorates histopathological changes of splenic tissue in radiation induced senescence model
Splenic tissue sections of control (Figure 4(a)) and ASX administrated rats (Figure 4(b)) revealed normal capsule and trabeculae, as well as typical histological architecture of red pulp and white pulp follicles, with lighter marginal zones encircling the germinal centers. On the other hand, the rats in the group that were exposed to radiation showed congested splenic blood vessels, and uneven thickness of the splenic capsule. The white pulp follicles displayed marked atrophy with severe lymphocytic depletion (Figure 4(c)) especially in the peri-arteriolar sheath, follicle, and the marginal zone where the reticular mash’s underpinning is visible with an increase in tingible-body macrophages (Figure 4(d)). The central arteriole showed thickened arteriolar wall with multifocal hyalinization with marked periarteriolar lymphocytic pyknosis, necrosis and debris (Figure 4(e)). Many megakaryocytes were observed in the vicinity of the narcotic debris with appearance of the underlying reticular cells and fibers (Figure 4(f)). The blood sinusoids in the red pulp were congested. Astaxanthin improves the histological alterations in the splenic tissue in a rat-model of radiation-induced senescence . Photomicrographs of spleen sections stained with H&E.(a and b) Control and ASX-administrated groups; showing normal histologic structure, normal white pulp follicles (Fo), periarteriolsheeth (PA), mantel zone (MZ), and red pulp (RP). (c-f) SC group showing (c) marked white pulp follicles atrophy and lymphocytic depletion (square), (d) many tangible body macrophages (arrow and insert) and lymphocytolysis in the follicles and in (e) the periarteriolar area (PA), with the reticular mash underneath appearance (insert). The ASX treated group exhibited a significant recovery in the size and intensity of the mantel zone (MZ) of white pulp follicles, along with a modest to moderate degree of depletion of follicular (F) lymphocytes and appearance of a few tingible body macrophages (arrow).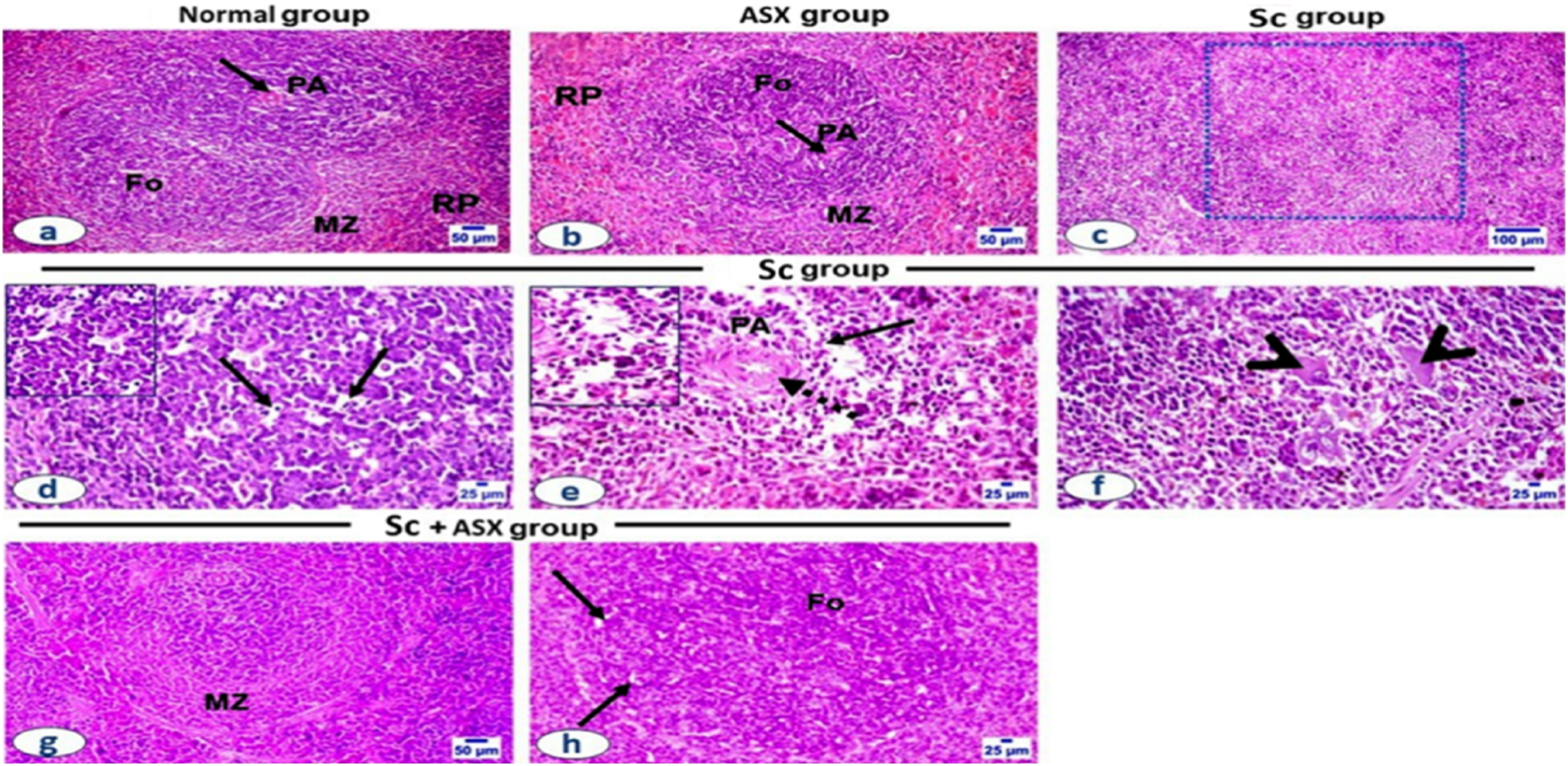
A summary of the histopathology results across different groups, along with its scoring.

Values are given as mean ± S.E, n = 5. *: significant change from control #: significant change from Sc.
ASX administration down regulates the splenic mTOR and TLR4 expression in irradiated rats (immunohistochemical study)
Figure 5 displays the immune expression of TLR4 and mTOR in the splenic tissue of different groups. Here, the irradiation group’s splenic tissue exhibited strong diffuse immune-expression of these markers in comparison to the control and other treated groups. ASX treatment could significantly diminish the expression of the TLR4 and mTOR in the spleens of treated rats as estimated by the image analysis software. Astaxanthin administration down regulates the splenic mTOR (A) and TLR4 (B) expression splenic tissue in radiation induced senescence model (immunohistochemical study). (C) Photomicrographs of immune-stained spleen sections for TLR4 and mTOR; showing negative expression of TLR4 and mild expression of mTOR in control and ASX groups, diffuse intense expression in irradiated group, and significant decreased expression of the two markers proteins in ASX treated groups, Image analysis of the optical density of the positive brown color. Values are expressed as mean ± SE. Data were analyzed by using one –way ANOVA followed by Tukey-Kramer multiple comparison test.*: significant change from control at p < .05, #: significant change from Sc at p < .05.
Discussion
Splenic irradiation (SI) has been used in various treatment strategies in symptomatic splenomegaly, secondary to hematologic and abdominal malignancies, or as an alternative therapy to splenectomy. Ionizing radiation directly affects DNA structure inducing a series of cellular DNA damage. A persistent DNA damage signal is essential for triggering cellular senescence; which compelled our interest to study the potential effect of ASX as a senomorphic supplement in spleen of irradiated rats.
Spleen senescence was associated with different mechanisms like oxidative stress and inflammation. Our study showed that gamma irradiation led to an increase in the oxidative stress biomarkers (TBARS, NOx), and a decrease in the antioxidant biomarkers (catalase activity and the reduced glutathione contents) in spleen homogenates. Altogether, there was an induction of splenic stress, as shown in similar studies of Wei, Wang, 42 Mohye El-Din, Abdelrazzak 43 and Singh, Yashavarddhan. 44 This increase in oxidative stress was attributed to the excessive production of hydrogen peroxide radical post irradiation, leading to a chain reaction of reactive oxygen and nitrogen species, contributing to disruption of intercellular organelles and breaking of proteins.45,46 The reduction of catalase activity and GSH content in irradiated rats could be the result of their overuse to combat the production of ROS.7,47 ASX administration to the senescence-induced rats caused an improvement in the antioxidant parameters accompanied with inhibition of the oxidative stress biomarkers. This was explained by the ROS scavenging activity of ASX and its ability to activate antioxidant enzymes. 48
Moreover, the effect of irradiation on spleen is related to several modifications to the immunomodulatory effects, such as the activation of chemokines, cytokines, and mediators of inflammation. In addition to that, it induces apoptosis during the inflammatory process.46,49–51 The MPO activity and IL-6 level was increased after irradiation in this study; they play a role in regulating inflammatory and immune responses. Radiation triggered the oxidative damage and neutrophil accumulation that were related to a rise in MPO activity. 52 MPO is an intrinsic enzyme that catalyses the synthesis of hypochlorous acid/hypochlorite together with other oxidising radicals. It is released when neutrophils are activated. 53 Our results showed a great impact of ASX on restoring the MPO activity and IL-6 levels to the normal ranges. Accordingly, ASX was reported to reduce inflammation through decreasing the levels of MPO in many organs rather than spleen.54–56
In such context, the histopathological results supported the biochemical data of oxidative stress and inflammation parameters contributed to the initial events of senescence. It had manifested tissue impairment and reflected a severe mutilation in the splenic compartments with haemorrhage and appearance of many megakaryocytes in the vicinity of the necrotic debris, as well as, the presence of reticular cells and fibres. However, ASX administration to the senescence-induced rats restored the normal configuration of spleen tissues. The splenic follicle’s structure and size also restored, reflecting a great improvement upon ASX treatment. In conjunction, we found that LDH was increased in the irradiated spleen tissues. The serum LDH activity increased as a result of impairment of cellular integrity due to exposure to radiation which increases the release of LDH enzyme. 57 In agreement with Kim, Lee 58 ASX treatment could antagonise such cellular insult.
As previously reported, cellular senescence is triggered by accumulation of excessive DNA breaks and the manifestation of cell cycle arrest. 59 Exposure to gamma radiation during SI induces DNA damage, inflammatory response and fibrous tissue formation hence it leads to senescence in many pathological stages. 6 Upon sensing DNA damage and among the cell cycle checkpoints (G1/S, G2/M and S-phase),p53 is effectively stimulated which in turn increases the expression of p21. That has the ability to block any cyclin/CDK complexes leading to cell arrest; the key point for propagation of cellular senescence.60,61 Likewise, Di Micco and his colleagues revealed that the irradiation potential effect in induction of prolonged DDR conjugated with senescence-incurred by upregulation of p21 and p16 cell cycle regulators. 1 ASX could be considered as a senomorphic remedy regarding its inhibition of cell cycle arrest through decrease the expression of p21. ASX was found to inhibit the upregulation of H2O2-induced p21 expression, yet it prevents cell cycle arrest. 62
The present data showed that there was a significant expression of the STING, TLR4 and mTOR in the model of irradiated spleen. These findings are in line with many previous studies.17,63–65 Senescent cells have increased levels of damaged DNA in the cell’s cytoplasm triggering inflammaging and production of pro-inflammatory cytokines. 66 DNA-sensing molecules such as cGAS-STING, which promote transcriptional factors involved in the production of pro-inflammatory mediators, are capable of recognising free cytoplasmic DNA. The stimulation of innate immunity by such these cytoplasimic DNAs can lead to overexpression of factors like STING and TLR4; the key determinats of pro-inflammatory mediators activation like IL-6 and MPO.63,67 Actually, senescent cells exhibit abnormal activation of cGAS-STING, which is essential for the induction of senescence. 68
mTOR is a member of PI3K family which responsible for cell cycle arrest and DNA damage repair that associated with aging.13,69,70 Zhang, Guo 71 found that there was a crosstalk between TLR4 and PI3K management of the pro-inflammatory signalling cascades. Irradiation causes activation of mTOR signalling pathway by increasing the intracellular ROS accumulation which is mediated through the inhibition of mitochondrial antioxidant enzymes.64,72 Also, activation of mTORis associated with the over expression of TLR4 leads to stem cell exhaustion, apoptosis and shortened lifespans in different experimental models.73–75 Simultaneously, the activation of STING led to mTOR expression and caused aging of immune responses (immunosenescence). 76 Thus, there was a crosstalk between STING, TLR4 and mTOR signalling.18,77
The deleterious effects displayed in senescence strongly related to the senescence-associated secretory phenotype (SASP). The SASP conjunctly related to induction of many cytokines and proteases which in turn promotes the signalling of inflammation, angiogenesis, tissue degeneration, tumorigenesis and senescence. 13 In course of irradiation, activation of STING/mTOR pathway could induce SASP activation.18,77 Another activator of SASP is radiation-induced NF-κB which increases the senescence marker proteins, such as CDKs and IL-6. 78 Here, we postulate that radiation-induced damage through STING/mTOR/TLR4 pathway could trigger the pathological changes of immunosenescence and inflammaging.
In the current study, ASX showed a significant amelioration in spleen senescence through regulation of STING, TLR4 and mTOR pathway. ASX was able to enhance the innate immunity may be through stimulation of protective mechanisms that could be achieved either by reducing direct and indirect DNA damage or by enhancing antioxidants and DNA double-strand break repair. Therefore, it can protect DNA from radiation-induced damage which eventually supresses the overexpression of STING. The findings of previous studies have demonstrated that TLR4 plays a pivotal role in mediating anti-inflammatory and the neuroprotective effect of ASX primarily depends on suppressing the TLR4 signalling pathway, which was parallel to the current study. 79 Furthermore, ASX significantly down regulates mTOR expression which plays an important role in promoting SASP of senescent cells; and its inhibition was shown to prevent stem cell senescence.80,81 The inhibition of mTOR could suppress the IL-1 receptor-dependent SASP by preventing the translation of IL-1α mRNA, which reduces translocation of NF-κB to the nucleus. 82 Under stressful conditions, it’s possible that mTOR promotes the rapid rise in ROS and mitochondrial DNA, which is linked to stress-induced senescence. 83 Notably, it is important to take in consideration some limitations of our work. The main limitation of this study is that further research on molecular mechanisms involving comprehensive in-depth analysis of a wide range of indicators regarding senescence markers like SASP, SA-βgal, p19 and autophagy markers are recommended in the future work.
Conclusion
Finally, our findings imply that spleen senescence is a multifaceted process and that the administration of ASX can be used as senomorphic supplement against radiation which exacerbates the senescence insults. ASX efficiently could protect against oxidative stress and inflammation. In addition, it positively regulates cell cycle arrest as well as cellular integrity. Here, we proposed that potential modification in expressions of proteins like STING, TLR4 and mTOR by ASX may explain the underlined cellular pathways. In general, the preclinical studies on radiation-induced senescence using senomorphics provide a promising therapeutic strategy in clinical settings, aiming to improve the longevity of cancer patients during radiotherapy regimen.
Footnotes
Acknowledgments
The authors express their gratitude to the NCRRT’s gamma irradiation unit team members for their assistance with the irradiation process.
Author contributions
Similar contributions have been made by each author in the areas of conception, data curation, formal analysis, research, methodology, software, and original draft writing and editing. Each author has reviewed the published version of the manuscript and given their approval.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
