Abstract
One endpoint of clinical islet cell transplantation for type 1 diabetic patients is the elimination or reduction of hypoglycemia. We previously developed a simple tool to evaluate islet graft function: the secretory unit of islet transplant objects (SUITO) index. The aim of this study is to clarify the association between the SUITO index and hypoglycemic episodes. Data from 310 clinical evaluations of 11 islet recipients were included in this study. Fasting plasma C-peptide and glucose levels were measured at every evaluation. The SUITO index was calculated according to the following formula: 1500 × C-peptide level (ng/ml)/[blood glucose level (mg/dl) – 63]. The number of hypoglycemic events (<3.8 mmol/L) and severe hypoglycemic events (<2.2 mmol/L or hypoglycemic unawareness) was assessed on the basis of interviews and self-monitoring of blood glucose (SMBG). Receiver operating characteristic (ROC) analysis was performed to determine the cut-off values of the SUITO index for hypoglycemic events. Based on the ROC study, follow-up data after transplantations were divided into the following three groups: low-SUITO (SUITO index <10, n = 91), middle-SUITO (10 ≤SUITO index <26, n = 83), high-SUITO (SUITO index ≤26, n = 125). The frequency of total hypoglycemia in the high-SUITO group was significantly decreased when compared to the other groups (value with Kruskal-Wallis test p < 0.001). The frequency of total severe hypoglycemia was significantly decreased in the low-SUITO group compared to pretransplant status and further decreased in the middle- and high-SUITO group. Spearman correlation coefficients were −0.663 (p < 0.001) between the number of total hypoglycemic events per one month and the SUITO index and −0.521 (p < 0.001) between that of severe events and the SUITO index. The SUITO index could predict the severity of hypoglycemic episodes in type 1 diabetic patients who received islet cell transplantations.
Introduction
According to the Diabetes Control and Complications Trial (DCCT), intensive insulin injection therapy serves to prevent diabetic microvascular complications for patients with type 1 diabetes, whereas severe hypoglycemic episodes increase approximately threefold compared to the conventional treatment group (31). Especially “brittle” type 1 diabetic patients with lack of endogenous insulin secretion have difficulty reaching favorable glycemic control and elimination of hypoglycemic episodes (2). Hypoglycemic episodes could not only limit one's social life, but also cause psychological fear, resulting in a significantly decreased quality of life (QoL) (3,6,9,26). Ideally, intensive insulin therapy for type 1 diabetes is supposed to achieve favorable glycemic control with higher QoL for a long term, while avoiding adverse events such as severe hypoglycemic events.
Islet cell transplantation (ICT) a promising treatment for unstable type 1 diabetic patients. ICT could restore glycemic control as well as prevent severe hypoglycemic episodes by replacement of β-cells with a minimally invasive procedure. However, the limited duration of insulin independence is an important problem (8,18). The University of Alberta group demonstrated that the number of hypoglycemic episodes was significantly improved up to 4 years after ICTs using the Edmonton protocol (24). The University of Miami group also showed ICT could prevent hypoglycemic episodes with approximately 4 years of follow-up (18). It was reported that excellent QoL scores were associated with an improved hypoglycemic scale in islet recipients (33).
A well-designed and easily accessible graft index is highly desirable to manage islet recipients clinically, because health care providers have to control immunosuppression. They must also consider additional ICTs when islet grafts encounter significant loss of function. Some indices that can be obtained from one-time blood sampling have been proposed for islet graft function: β score and C-peptide/glucose ratio (CP/G) (7,25). However, limited information is available on relationship between these clinical graft indices and the frequency or severity of hypoglycemic episodes. We developed the secretory unit of islet transplant objects (SUITO) index to assess transplanted islet function using fasting blood samples (19,21,23). It was previously reported that the SUITO index is associated with daily insulin dose, M-value, HbA1c, intravenous glucose tolerance test (IVGTT), and scores from QoL questionnaires (21,28–30). However, there is no evidence for a link between the SUITO index and hypoglycemic episodes. We hypothesized that the SUITO index should be related to the frequency and severity of hypoglycemic events. In this report, we demonstrated the association between the frequency and severity of hypoglycemic events and the SUITO index in type 1 diabetic patients with ICT.
Materials and Methods
Patients
Eleven patients who received a total of 20 ICTs at our hospitals (Baylor University Medical Center at Dallas and Baylor All Saints Medical Center at Fort Worth, TX) from March 2005 to February 2010 were included in this study. Islet preparations were performed as previously described (16,20,22) except for the first seven transplantations in which islets were isolated at a remote center (15). The protocol and consent forms were approved by the institutional review board of Baylor Research Institute (Dallas, TX). Written consent forms were obtained from all participants.
Clinical Follow-up
The islet recipients attended an outpatient clinic every month, and blood samples were taken to measure fasting C-peptide and glucose levels. Patients also reported the frequency of hypoglycemic episodes including hypoglycemic unawareness at every outpatient visit. The records of 310 clinical evaluations from pre-and post-ICTs were analyzed. The median follow-up period was 20.9 months (interquartile range: 20.1–29.2).
Calculation of Clinical Graft Indices for Islet Function
Plasma fasting C-peptide and glucose levels were used to calculate the SUITO index according to the following formula: 1500 × C-peptide level (ng/ml)/[blood glucose level (mg/dl) – 63] (19,21,23). Also, C-peptide/glucose ratio [C-peptide (ng/ml) × 100/glucose (mg/dl)] and β score were calculated at the same time points of SUITO index evaluations (7,25).
Monitoring of Blood Glucose and the Count of Hypoglycemic Events
Self-monitoring of blood glucose (SMBG) was performed with an OneTouch Ultra meter (LifeScan, Milpitas, CA). The GlucoMON-ADMS system (Diabetech, Dallas, TX) transferred SMBG data to investigators automatically. The frequencies of hypoglycemic events per month were counted based on patient reports and SMBG levels below 3.8 mmol/L. Hypoglycemic events were classified as severe if either hypoglycemic unawareness occurred or SMBG levels fell below 2.2 mmol/L. The low blood glucose index (LBGI), which could predict the occurrence of severe hypoglycemia, was calculated using SMBG data for 3 days immediately before every outpatient visit (17).
Receiver-Operating Characteristic Analysis
Receiver-operating characteristic (ROC) analysis is a useful tool to assess biomarker accuracy and to chose the cut-off point (11,27). Because the data were collected in a longitudinal format with measurements made on the same subject over time, a generalized linear mixed model (GLMM) was applied to adjust for the correlation of repeated observations (1,4). A random intercept was included in the GLMM to account for both within- and between-subject variations. Then, the logistic regression model was employed to produce ROC curves based on the GLMM results. The area under the curve (AUC) of ROC analysis with the 95% confidence interval (CI) and the p-value under a null hypothesis of AUC = 0.5 were calculated. The criterion of AUC was considered as follows: acceptable discrimination (0.7 ≤ AUC < 0.8), excellent discrimination (0.8 ≤ AUC < 0.9), and outstanding discrimination (0.9 ≤ AUC) (14). The sensitivity and specificity of the SUITO index's ability to predict the occurrence of total and severe hypoglycemic events were determined by using an ROC plot. Positive and negative predictive values were also calculated.
Statistics
The number of hypoglycemic events per month was compared among multiple groups using the Kruskal-Wallis test followed by the Scheffé post hoc test as a nonparametric technique. Spearman coefficient was used to investigate the correlation between the frequency of hypoglycemic events, SUITO index, and insulin dose per body weight. The comparisons for multiple time points of the SUITO index and number of hypoglycemic events were performed with repeated measures analysis of variance (ANOVA) with Bonferroni correction. Box plots demonstrate the median values with 25% and 75% interquartiles. The whiskers extend from the minimum to the maximum values after excluding the outliers. Statistical significance was considered when two-sided p < 0.05 in all statistical tests. Statistical analysis was performed using PASW statistics 18.0.2 (SPSS Inc., Chicago, IL) and SAS 9.22 (SAS Institute, Cary, NC) for multiple comparisons of ROC analysis (10).
Results
Patient Characteristics and Clinical Course
Basic characteristics of patients with type 1 diabetes before first ICT were as follows [median (interquartile range)]: age (year) —46 (35–53), body weight (kg)—64.3 (54.9–79.3), body mass index (kg/m2)—23.6 (21.2–27.9), disease duration (year)—33 (23–42), daily insulin requirements per body weight (unit/kg)—0.47 (0.43–0.58), fasting plasma C-peptide (ng/ml)—0.0 (0.0–0.0), HbA1C (%)—8.3 (7.1–8.6), and SUITO index—0.0 (0.0–0.0).
Four, five, and two patients received one, two, and three islet infusions, respectively. The average islet yield per patient's body weight (IE/kg) was 12,096 (11,535–16,726). Eight patients achieved insulin independence for more than 2 weeks after one ICT in three patients and two ICTs in five patients. Four patients kept insulin independence at the time of analysis, while three patients could not achieve insulin independence for the entire observation periods.
Longitudinal Course of the SUITO Index and Number of Hypoglycemic Events
Longitudinal course of the SUITO index as well as number of hypoglycemic events after the last ICT was shown in Figure 1. Eight patients were followed up over 14 months after the last ICT. Among those patients, significant increase of SUITO index and significant decrease of the numbers of both total and severe hypoglycemic events were observed within 14 months when compared to pretransplant data using repeated-measure ANOVA (p < 0.05).

Longitudinal course of the SUITO index and number of hypoglycemic events in ICT. The changes of averages and the SE for the SUITO index, number of total hypoglycemic events, and that of severe events are shown.
ROC Analysis for Hypoglycemic Events
ROC curves determined the clinical indices' ability to predict the occurrence of hypoglycemic events. AUCs of ROC curves to determine the occurrence of total and severe hypoglycemic events are shown in Table 1. All clinical indices had significant discrimination with outstanding accuracy for both total and severe hypoglycemic events (p < 0.001). These ROC curves are plotted in Figure 2. There were no significant differences among AUCs for SUITO index, CP/G, β score, and LBGI regarding the discrimination of both total and severe hypoglycemic events.

ROC analysis for prediction of hypoglycemic events. ROC curves of clinical indices to predict the occurrence of total hypoglycemic events (A) and severe hypoglycemic events (B) are shown. Dots in (A) and (B) represent points of 26 and 10 on the SUITO index, respectively.
AUC of ROC Curve for the Prediction of Hypoglycemic Events

Null hypothesis; the true area of AUC = 0.5.
The SUITO index had a sensitivity of 88.3%, specificity of 93.2%, positive predictive value of 95.1%, and negative predictive value of 81.0% for the occurrence of total hypoglycemic events when the cut-off value was 26 (Fig. 2A). The cut-off point of SUITO index value = 10 for severe hypoglycemic events had 90.0%, 82.0%, 55.6%, and 97.0% for sensitivity, specificity, positive prediction value, and negative prediction value, respectively (Fig. 2B).
Relationship Between Hypoglycemic Events, Insulin Dose, and SUITO Index
Follow-up data after ICTs were divided on the basis of ROC analysis described above into the following three groups: low-SUITO group (SUITO index < 10, n = 91), middle-SUITO group (10 ≤ SUITO index < 26, n = 83), and high-SUITO group (26 ≤ SUITO index, n = 125). Basic statistics on the number of total hypoglycemic events per month [median (interquartile range)] are given as follows: 11 (6–20) in the low-SUITO group, 8 (3–14) in the middle-SUITO group, and 0 (0–3) in the high-SUITO group (Fig. 3A). The statistics for severe hypoglycemic events were: 0 (0–1) in the low-SUITO group, 0 (0–0) in the middle-SUITO and the high-SUITO group (Fig. 3B). Also, those for daily insulin requirements per body weight (unit/day/kg) were: 0.36 (0.29–0.49) in the low-SUITO group, 0.19 (0.07–0.30) in the middle-SUITO group, and 0 (0.00–0.00) in the high-SUITO group (Fig. 3C). Importantly there were no severe hypoglycemic events in middle and high SUITO groups. The frequency of both total and severe hypoglycemic events had significant differences among the four groups, including the pretransplant group (value with Kruskal-Wallis test p < 0.001) (Fig. 3A, B). In post hoc analysis, there were significant differences for total hypoglycemic events between the middle versus high-SUITO group (p < 0.001), the low- versus high-SUITO group (p < 0.001), and the pretransplant versus high-SUITO group (p < 0.001), although no significant difference was observed between the low- versus middle-SUITO group (p = 0.22) and the pretransplant versus low-SUITO group (p = 0.41). The significant differences were also observed for severe hypoglycemic events between the low- versus middle-SUITO group (p < 0.001) and the low- versus high-SUITO group (p < 0.001), although no significant difference was observed between the middle-and high-SUITO groups (p = 0.41). The frequencies of both total and severe hypoglycemic events were inversely correlated with the SUITO index (Spearman r = −0.663, p < 0.001, and r = −0.521, p < 0.001).

Association between number of hypoglycemic events, daily insulin requirements and SUITO index. Box plots of the number of total hypoglycemic events (A), severe hypoglycemic events (B), and daily insulin requirements (C) are shown where pretransplant group, low, middle, and high SUITO group were classified. *p < 0.05, **p < 0.001 with the Scheffé post hoc test.
Similarly, daily insulin requirements per body weight were significantly different among the four groups (value with Kruskal-Walllis test p < 0.0001) (Fig. 3C). Significant differences were observed in all pairs except for the comparison between pretransplant and low-SUITO group (p = 0.001 except forp = 1.0 between pretransplant versus low-SUITO group). The daily insulin amounts per body weight were inversely correlated with the SUITO index (Spearman r = −0.776, p < 0.001).
The daily insulin requirements per body weight were positively correlated with the frequency of both total and severe hypoglycemic events (Spearman r = 0.649 and 0.392, value for both correlationsp < 0.001) (Fig. 4). No severe hypoglycemic events were observed in insulin independent recipients (Fig. 4B), although moderate hypoglycemic events with 3.7 mmol/L (3.5–3.8) were found in five patients with insulin independence after ICTs (y axis of Fig. 4A).
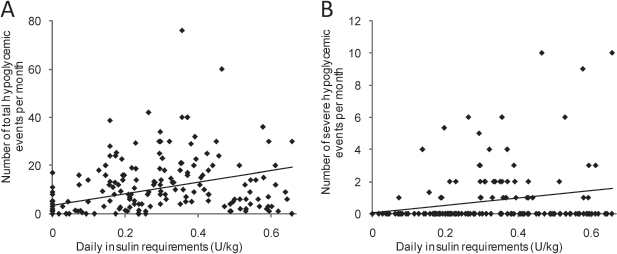
Association between hypoglycemic events and insulin requirements. Dot plots of the frequency per month of total hypoglycemic events (A) and severe events (B) versus daily insulin requirements (unit/day/kg) are shown. The line shows linear regression.
Discussion
This study is the first report to show the association between the SUITO index and hypoglycemic events after ICTs for type 1 diabetic patients. One of the main purposes of ICT is to eliminate hypoglycemic episodes, which have a negative impact on QoL (6,9). Since the SUITO index can predict engrafted islet function as previously reported (19,21,23), findings in this report also support the association between graft function and hypoglycemic episodes. Similar to previous reports from other ICT centers (12,18), our patients significantly reduced hypoglycemic events after ICTs. Significant increase of the SUITO index was also observed after ICTs. The significance was confirmed up to 14 months and the SUITO index was slightly reduced 8 months after the last ICT. Longer improvement of SUITO index would be expected if engrafted islet function can be maintained for a longer term. These results demonstrate that ICTs could lead to reduced risk of hypoglycemic events and enhance the role of ICT in the treatment of patients with unstable type 1 diabetes.
ROC analysis showed the accuracy of the clinical graft indices in predicting hypoglycemic episodes. AUCs of ROC curves of the SUITO index for the occurrence of total and severe hypoglycemic events indicated outstanding accuracy (AUC > 0.9) as well as those of CP/G, β score, and LBGI. The discriminating accuracy of the SUITO index for hypoglycemic events were almost the same as CP/G, which would be explained by the use of the same components and the similar formula for their calculations: fasting serum C-peptide and glucose level in the numerator and denominator, respectively. The β score also showed outstanding accuracies for the occurrence of hypoglycemic events, but the score requires additional data on HbA1c and daily insulin amounts compared to SUITO index and CP/G. The similar AUCs among the SUITO index, CP/G, β score, and LBGI suggest that the islet graft indices would be useful as a predictor for hypoglycemia because LBGI had been demonstrated as a clinical landmark for hypoglycemic profiles (17).
The SUITO index also had excellent discriminations for insulin independence and impaired glucose tolerance (more than 10 mmol/l at 30 min in IGVTT) in previous studies: AUC = 0.907 and 0.839, respectively (29,30). While biopsy of engrafted islets in the liver might provide direct graft assessment (32), a simple indicator for graft function like SUITO index has practical value in clinical settings which require repeatable measurements, minimum burden to patients, and high reliability.
Values of 26 and 10 on the SUITO index provided reasonable cut-off points for the occurrence of total and severe hypoglycemic episodes, respectively, and it was consistent with previous results; SUITO index > 26 predicted insulin independence after ICTs (21) and SUITO index < 10 was associated with poor QoL (28). Similar findings were also observed in this study; significantly reduction of insulin amounts was observed in the high-SUITO group (SUITO ≥ 26) compared to the middle group and in the middle-SUITO group compared to the low group (SUITO < 10). Since some islet recipients still need to use the exogenous insulin injection to prevent hyperglycemic episodes, iatrogenic hypoglycemia due to excessive insulin might occur. A SUITO index value >26 indicates insulin independence; therefore, iatrogenic hypoglycemia should be discontinued. In addition, islet recipients with a SUITO index ≥ 26 had excellent glycemic profiles at IVGTT, indicating that such patients could maintain excellent glycemic control, thus avoiding hypoglycemic events (30). In contrast, the frequency of severe hypoglycemic events was significantly higher in the low-SUITO group (SUITO index ≤ 10) when compared to the other two groups where the SUITO was ≥ 10. The results of the previous IVGTT study provided similar findings; the low-SUITO group had prolonged hyperglycemia and larger AUC (30). Islet recipients with a SUITO index <10 have limited benefit, including poor metabolic control and QoL. Therefore, additional islet infusions and/or modification of immunosuppression should be considered for such patients. The low positive predictive value from a SUITO index of 10 for the cut-off value of severe hypoglycemic events suggests a low rate of severe hypoglycemic events in our patients, since predictive values reflect the prevalence as well as the accuracy of the test (13).
Excessive insulin administration is considered for one of the reasons of hypoglycemic events in patients with type 1 diabetes (5). Similar findings were found in this study; daily insulin amounts per body weight were positively correlated with the frequency of both total and severe hypoglycemic events. However, in the middle-SUITO group, there were no severe hypoglycemic episodes even when such patients used insulin injection (average daily dose approximately 0.2 U/kg). On the contrary, patients who used less than 0.2 U/kg insulin injections still experienced severe hypoglycemic episodes in the low-SUITO group. The explanation for this should be that transplanted islets in the middle-SUITO group could secret enough glucagon to avoid severe hypoglycemia when exogenous insulin dose was excessive and islet grafts in the low-SUITO group would have less function to restore severe hypoglycemia. Because islet grafts have a function to correct severe hypoglycemia, the SUITO index would be better a predictor for severe hypoglycemic episodes compared to daily insulin requirements.
Moderate hypoglycemic episodes were observed even when the patients achieved insulin independence, although no severe hypoglycemic episodes were found in such recipients. Most of the moderate hypoglycemic events were accompanied with gastrointestinal adverse events such as nausea and vomiting (data not shown) so that anorexia or decreased glucose intake might be associated with the hypoglycemic episodes. The immunosuppression protocol with less gastrointestinal adverse events would also contribute to reduce the risk of hypoglycemic episodes in islet recipients.
The study presented here demonstrated that the frequency and severity of hypoglycemic events were reduced after ICT for type 1 diabetic patients, and SUITO index could predict hypoglycemic episodes, including severe events, with excellent accuracy. Limitations of this study include the small number of subjects and retrospective design. Further prospective investigations with a larger group size will provide definitive conclusions.
Footnotes
Acknowledgments
This study was partially supported by All Saints Health Foundation. Dr Chujo has been supported by the proceedings of the W. W. Caruth Jr. Chair for Organ Transplantation Immunology to J. Banchereau, Ph.D. The authors thank Ms. Yoshiko Tamura and Mr. Greg S. Olsen for their technical support and Mr. Jeff SoRelle for English grammar. The authors declare no conflicts of interest.
