Abstract
Parkinson's disease (PD) is a neurodegenerative disorder characterized by the degeneration of dopaminergic (DA) neurons in the midbrain. Induced pluripotent stem (iPS) cells have shown potential for differentiation and may become a resource of functional neurons for the treatment of PD. However, teratoma formation is a major concern for transplantation-based therapies. This study examined whether functional neurons could be efficiently generated from iPS cells using a five-step induction procedure combined with docosahexaenoic acid (DHA) treatment. We demonstrated that DHA, a ligand for the RXR/Nurr1 heterodimer, significantly activated expression of the Nurr1 gene and the Nurr1-related pathway in iPS cells. DHA treatment facilitated iPS differentiation into tyrosine hydroxylase (TH)-positive neurons in vitro and in vivo and functionally increased dopamine release in transplanted grafts in PD-like animals. Furthermore, DHA dramatically upregulated the endogenous expression levels of neuroprotective genes (Bcl-2, Bcl-xl, brain-derived neurotrophic factor, and glial cell-derived neurotrophic factor) and protected against 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP)-induced apoptosis in iPS-derived neuronal precursor cells. DHA-treated iPS cells significantly improved the behavior of 6-hydroxydopamine (6-OHDA)-treated PD-like rats compared to control or eicosapentaenoic acid-treated group. Importantly, the in vivo experiment suggests that DHA induces the differentiation of functional dopaminergic precursors and improves the abnormal behavior of 6-OHDA-treated PD-like rats by 4 months after transplantation. Furthermore, we found that DHA treatment in iPS cell-grafted rats significantly downregulated the mRNA expression of embryonic stem cell-specific genes (Oct-4 and c-Myc) in the graft and effectively blocked teratoma formation. Importantly, 3 Tesla-magnetic resonance imaging and ex vivo green fluorescence protein imaging revealed that no teratomas were present in transplanted grafts of DHA-treated iPS-derived DA neurons 4 months after implantation. Therefore, our data suggest that DHA plays a crucial role in iPS differentiation into functional DA neurons and that this approach could provide a novel therapeutic approach for PD treatment.
Keywords
Introduction
Parkinson's disease (PD) is characterized by the progressive loss of a specific population of dopaminergic (DA) neurons in the brain, which leads to debilitating impairments in the control of movement and eventual death (6,31,40). As PD results from the loss of DA neurons, the prospect of utilizing cell replacement therapies for this disease has attracted substantial interest. Mesencephalic DA neurons dissected from human embryos have been used to demonstrate that successful cell replacement therapy is clearly beneficial (25,34). Although embryonic pig neural cells were shown to survive in a PD patient, the number of surviving cells was very low (14,42). Stem cells of human origin are currently thought to be the most promising source of DA neurons for cell-based therapies for PD (1,2,26,44). Recent evidence has shown that DA neurons derived from embryonic stem (ES) cells and bone marrow-derived neural progenitors are functional when grafted into parkinsonian rats (18,38,53). However, teratomas were found to have developed in the rodents' brains following ES-derived DA neuron transplantation (4).
Recently, Dr. Yamanaka and colleagues demonstrated that induced pluripotent stem (iPS) cells could be generated from mouse embryonic fibroblasts and from adult human fibroblasts using retrovirus-mediated transfection of four transcription factors: octamer binding transcription factor 3/4 (Oct3/4), sex determining region Y box 2 (Sox2), c-Myc, and Kruppel-like factor 4 (Klf4) (46,47). These studies further suggested that iPS cells were indistinguishable from ES cells in morphology, proliferative abilities, surface antigens, gene expression, epigenetic status of pluripotent cell-specific genes, and telomerase activity. Wernig et al. demonstrated that neuronal and glial cell types could be derived from iPS cells in vitro and that transplantation of iPS-derived neuronal cells into the brain was able to improve behavior in a rat model of PD (50). Although the therapeutic potential of iPS cells has been demonstrated in animal models (50), the regulation of neurogenic differentiation is not well understood. Moreover, the ability to form teratomas in vivo has been a landmark and routine assay for evaluating the pluripotency of ES and iPS cells (47,52). Preventing teratoma formation has become an emergent issue in cell transplantation, especially in cases of ES or iPS cell transplantation (3,17,20,36,38).
Docosahexaenoic acid (DHA; C22:6) is the major ω-3 polyunsaturated fatty acid (PUFA) component in normal brain tissue (19) and has been used in neurological and psychiatric therapies (29). A recent study demonstrated that DHA protected against dendritic pathology and behavioral deficits and increased the antiapoptotic BCL2-associated agonist of cell death (BAD) phosphorylation associated with protection against Alzheimer's disease (5). Furthermore, DHA has been used to prevent decreases of DA and serotoninergic neurotransmitters in the frontal cortex of piglets fed linoleic acid-deficient diets (15). Notably, recent studies have demonstrated that DHA has the potential to promote neuronal differentiation of neural stem cells in vitro and to increase neurogenesis in aged rats (16,23). We have previously demonstrated that iPS cells are capable of multilineage differentiation (22) and exhibit the potential to specifically differentiate into astroglial-like and neuronal-like cells that display functional electrophysiological properties (9). Further study in vivo showed that the direct injection of iPS cells into damaged areas of rat cortex significantly decreased the infarct size and improved the motor function in rats with middle cerebral artery occlusion (9). In the present study, we examined whether functional neurons could be efficiently generated from iPS cells using a five-step induction procedure combined with DHA or another PUFA analogue, eicosapentaenoic acid (EPA) (5,29). We focused on the ability of DHA and EPA to increase DA differentiation and the potential for preventing teratoma formation during iPS treatments. Compared to EPA, our results suggest that DHA is superior for promoting the differentiation of iPS cells into DA neurons, and that DHA treatment results in functional improvements in the behavior of animal models of PD. Importantly, our data indicate that DHA is not only an effective agent for facilitating DA differentiation, but that it also plays a key role in preventing the formation of tumors after in vivo iPS cell transplantation.
Materials and Methods
iPS Cell Culture
Murine induced pluripotent stem cells were generated from mouse embryonic fibroblasts derived from 13.5-day-old embryos of C57/B6 mice. The iPS cells were reprogrammed by the transduction of retroviral vectors encoding four transcription factors, Oct-4, Sox2, c-Myc, and Klf4, as described previously (47). Briefly, undifferentiated iPS cells were routinely cultured and expanded on mitotically inactivated murine embryonic fibroblasts (MEFs; 50,000 cells/cm2) in six-well culture plates (BD Technology) in the presence of 0.3% leukemia inhibitory factor in an iPS medium consisting of Dulbecco's modified Eagle's medium (DMEM; Sigma) supplemented with 15% fetal bovine serum (FBS; Invitrogen, Carlsbad, CA, USA), 100 mM minimal essential medium (MEM), nonessential amino acids (Sigma), 0.55 mM 2-mercaptoethanol (Gibco), and antibiotics (Invitrogen). The mouse iPS cells were transfected with pCX-enhanced green fluorescence protein (GFP) genes to express green fluorescence constitutively and were maintained and differentiated in vitro as described previously (22). To evaluate the pluripotent capacity of iPS cells to differentiate into different cell types of all three germ layers, several differentiation protocols were applied as described previously (9). As for hepatocyte differentiation, the protocol using a two-step procedure was applied as described previously (45).
Generation of iPS-Derived Functional Neurons and DHA Treatment
Every 3–4 days, iPS colonies were detached with 0.2% collagenase IV (Invitrogen), dissociated into single cells with 0.025% trypsin (Sigma-Aldrich) and 0.1% chicken serum (Invitrogen) in phosphate buffered saline (PBS), and plated again onto MEFs (stage 1). For embryoid body (EB) formation (stage 2), iPS cells were dissociated into a single cell suspension by 0.25% trypsin-EDTA and plated onto nonadherent culture dishes in DMEM with 15% FBS, 100 mM MEM nonessential amino acids, 0.55 mM 2-mercaptoethanol, and antibiotics at a density of 2 × 106 cells/100-mm plate. After 4 days as a floating culture, EBs were transferred onto gelatin-coated plates and maintained in the same medium for 24 h. The selection of nestin-positive cells (stage 3) was initiated by replacing the medium with DMEM/F12 (Gibco) supplemented with insulin-transferrin-selenium (ITS) (Gibco), 5 μg/ml fibronectin (Sigma), and antibiotics. After 6–10 days of selection, cells were trypsinized to remove remnant EBs and plated onto glass coverslips or culture dishes precoated with polyornithine (15 μg/ml; Sigma) and fibronectin (1 μg/ml; Sigma or Gibco) at a concentration of 2 × 105 cells/cm2. Neural precursor cells were expanded in N2 medium 3 containing 20 ng/ml basic fibroblast growth factor (bFGF), a murine N-terminal fragment of sonic hedgehog (SHH-N), FGF8-b (all from R&D Systems), and 200 μM ascorbic acid (AA; Sigma) for 4 days (stage 4). Terminal differentiation (stage 5) was induced by withdrawing bFGF, SHH-N, and FGF8-b. Cells were kept in differentiation conditions for 10–15 days. To investigate the role of DHA in neuronal differentiation, DHA was added at stage 3, 4, or 5 to evaluate its effects. To determine the optimal dosages and durations of DHA treatment for all experiments, differentiated iPS cells were treated with different dosages (20–60 μM) of DHA for different time (12–48 h).
Cell Viability Assay
iPS cells were seeded on 24-well plates at a density of 2 × 104 cells/well in medium and submitted to a methyl thiazol tetrazolium assay (MTT assay; Sigma-Aldrich). iPS cells were incubated with 0.25 mg/ml MTT for 4 h at 37°C, and the reaction was terminated by the addition of 100% isopropanol (14). The amount of MTT formazon product was determined by using a microplate reader, and absorbance was measured at 560 nm (SpectraMax 250, Molecular Devices, Sunnyvale, CA, USA).
Immunofluorescence Staining
An avidin–biotin complex-based method was used for immunohistochemical staining of differentiated iPS cells. Following washes with 3% hydrogen peroxide, sodium azide and antigenicities were retrieved using a microwave. Each slide was then treated with antibodies for Bcl-2, Tuj1 (Upstate Biotechnology, Waltham, MA, USA), nuclear receptor related 1 protein (Nurr1), microtubule-associated protein 2 (MAP2), and tyrosine hydroxylase (TH) (Chemicon, Temecula, CA, USA). Immunoreactive signals were detected with a mixture of biotinylated rabbit anti-mouse IgG and Fluoresave (Calbiochem, La Jolla, CA, USA) and a confocal microscope (Olympus, FV300).
Microarray Analysis and Bioinformatics
Total RNA was extracted from cells using Trizol reagent (Life Technologies, Bethesda, MD, USA) and the Qiagen RNAeasy (Qiagen, Valencia, CA, USA) column for purification. Total RNA was reverse-transcribed with Superscript II RNase H-reverse transcriptase (Gibco BRL) to generate Cy3- and Cy5-labeled (Amersham Biosciences Co., Piscataway, NJ, USA) cDNA probes for the control and treated samples, respectively. The labeled probes were hybridized to a cDNA microarray containing 10,000 gene clone immobilized cDNA fragments. Fluorescence intensities of Cy3 and Cy5 targets were measured and scanned separately using a GenePix 4000B Array Scanner (Axon Instruments, Burlingame, CA, USA). Data analysis was performed using GenePix Pro 3.0.5.56 (Axon Instruments, USA) and GeneSpring GX 7.3.1 software (Agilent, Palo Alto, CA). The average linkage distance was used to assess the similarity between two groups of gene expression profiles as described below. The difference in distance between two groups of sample expression profiles to a third was assessed by comparing the corresponding average linkage distances [the mean of all pair-wise distances (linkages) between members of the two groups concerned]. The error of such a comparison was estimated by combining the standard errors (the standard deviation of pair-wise linkages divided by the square root of the number of linkages) of the average linkage distances involved. Classical multidimensional scaling (MDS) was performed using the standard function of the R program to provide a visual impression of how the various sample groups are related.
Real-Time Reverse Transcription-Polymerase Chain Reaction (RT-PCR)
For real-time RT-PCR, the total RNA was extracted using the RNAeasy kit (Qiagen, Valencia, CA, USA) as previously described (14,26). Briefly, total RNA (1 μg) of each sample was reversely transcribed in 20 μl using 0.5 μg of oligo(dT) and 200 U Superscript II RT (Invitrogen). The amplification was carried out in a total volume of 20 μl containing 0.5 μM of each primer, 4 mM MgCl2, 2 μl LightCycler™-FastStart DNA Master SYBR green I (Roche Molecular Systems, Alameda, CA, USA), and 2 μl of 1:10 diluted cDNA. The quantification in the unknown samples was performed by the LightCycler Relative Quantification Software version 3.3 (Roche Molecular Systems). In each experiment, the glyceraldehyde 3-phosphate dehydrogenase (GAPDH) housekeeping gene was amplified as a reference standard. The primers sequences of target genes were showed in Table 1. Reactions were prepared in duplicate and heated to 95°C for 10 min followed by 40 cycles of denaturation at 95°C for 10 s, annealing at 55°C for 5 s, and extension at 72°C for 20 s. All PCR reactions were performed in duplicate. Standard curves (cycle threshold values versus template concentration) were prepared for each target gene and for the endogenous reference (GAPDH) in each sample. To confirm the specificity of the PCR reaction, PCR products were electrophoresed on a 1.2% agrose gel.
The Sequences for the Primers of Quantitative Reverse Transcription-Polymerase Chain Reaction
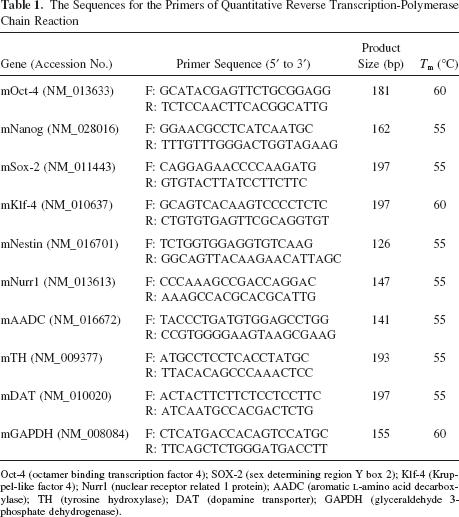
Oct-4 (octamer binding transcription factor 4); SOX-2 (sex determining region Y box 2); Klf-4 (Kruppel-like factor 4); Nurr1 (nuclear receptor related 1 protein); AADC (aromatic L-amino acid decarboxylase); TH (tyrosine hydroxylase); DAT (dopamine transporter); GAPDH (glyceraldehyde 3-phosphate dehydrogenase).
Western Blot Assay
At the end of cell culture, proteins were extracted from the cells, followed by immunoblotting analysis. In brief, 15 μl of protein samples were separated on a 10% SDS-polyacrylamide gel electrophoresis. The proteins were then transferred to Hybond-ECL nitrocellulose paper or polyvinylidene fluoride membranes (Amersham, Arlington Heights, IL) by a wet-transfer system. Primary and secondary antibodies were added as indicated. The reactive protein bands were detected by the ECL detection system (Amersham).
Dopamine Determination by High-Performance Liquid Chromatography and Electrochemical Detection (HPLC-ED)
Dopamine levels in cell lysates were determined in cells at day 14 of the fifth stage. An HPLC-ED system (HTEC-500; Eicom, Kyoto, Japan) consisting of a three-electrode cell with an Ag/AgCl electrode as the reference electrode, a counter electrode, and a graphite electrode as the working electrode was used to measure levels of serotonin (5-HT). The graphite working electrode had a 25-μm gasket, and the applied potential was set at +450 mV versus Ag/AgCl. The column used for separation at room temperature (25°C) was a PP-ODS (4.6 × 30 mm; Eicom, Kyoto, Japan). The mobile phase, containing 0.1 M sodium phosphate buffer (0.1 M NaH2PO4; 0.1 M Na2HPO4 = 1000:160, v/v), 1% methanol, 500 mg/L sodium decanesulfonate, and 50 mg/L EDTA, was adjusted to pH 6.0 with 5 M NaOH at a flow rate of 0.5 ml/min. A volume of 10 μll of each lysate was injected manually into the chromatographic system and assayed on the same sampling day (35).
Animal Surgery and Transplantation
All surgical procedures were performed according to the rules set by the Ethical Committee for Use of Laboratory Animals at Taipei Veterans General Hospital. Adult Sprague-Dawley rats (male, 8 weeks old, approximately 250 g) were used in this study. Under chloride hydrate anesthesia (400 mg/kg, IP), the rats were placed in a stereotaxic frame. The DA-innervated striatum was unilaterally lesioned by administering injections of 6-hydroxydopamine (6-OHDA) into the median forebrain bundle (AP, −4.3 mm; R/L, +1.6 mm; H, −8.2 mm; and AP, −4.0 mm; R/L, +1.8 mm; H, −8.0 mm) (8). Each rat received 30 μg of 6–OHDA dissolved in 5 μl of physiological saline containing 0.02% ascorbic acid. Amphetamine-induced rotational behavior was assessed at 4, 8, 12, 16, and 20 weeks after 6-OHDA injection. For that assessment, the rats were placed in individual plastic hemispherical bowls and allowed to habituate for 10 min before being injected with a subcutaneous dose of amphetamine (5 mg/kg). Left and right full-body turns were counted. Amphetamine-induced net rotation over a period of 60 min, starting 30 min after injection, was quantified. Animals showing >360 turns per hour ipsilaterally toward the lesioned side after a single dose of amphetamine were considered successful parkinsonian models and were selected for grafting. All behavioral tests were performed in a closed room to avoid environmental disturbance and were assessed by an independent observer blind to the treatments. The day before grafting, animals were immunosuppressed (15 mg/kg daily, cyclosporine, Novartis, Switzerland) and then received cyclosporine daily (10 mg/kg) for the duration of the study. The Sprague-Dawley rats were divided into three groups of 12 animals each. One month after 6-OHDA injection, rats in the control group received an injection of PBS into their dopamine-denervated striatum. Rats in group 2 received a suspension of 105 grafted iPS cells that had been cultured using the five-step induction procedure only (stage 5 cells), whereas rats in group 3 received a suspension of 105 stage 5 grafted iPS cells that had also been treated with DHA. Rats were imaged during the 4-week period of stem cell growth using a 3 Tesla-magnetic resonance imaging (3T-MRI) Biospect system (Bruker, Germany) combined with a mini-quadrature coil (12-cm inner diameter) for radiofrequency transmission and reception of MRI signals (24).
In experiments on teratoma formation ability of iPS cells to determine the pluripotency, a total of 1,000,000 undifferentiated iPS cells suspended in 100 μl PBS were subcutaneously injected into the back skin of nude mice. Six weeks after cell transplantation, grafts were surgically dissected from the mice, fixed, and sectioned, followed by hematoxylin and eosin (H&E) staining of the sections.
In Vivo Analysis of Tumor Growth
All procedures involving animals were in accordance with the institutional animal welfare guidelines of Taipei Veterans General Hospital. In vivo GFP imaging was performed using an illuminating device [LT-9500 Illumatool/TLS equipped with excitation illuminating source (470 nm) and filter plate (515 nm)]. Tumor size was measured with calipers and the tumor volume was calculated using the formula (length × width2)/2. The integrated optical density of green fluorescence intensity was captured and analyzed using Image Pro-plus software.
Enzyme-Linked Immunosorbent Assay (ELISA), Terminal dUTP Nick-End Labeling (TUNEL) Assay, and Immunohistochemistry
The activities of caspase-3 were determined using an ELISA kit (R&D Systems) and quantified at 490 nm (MRX; Dynatech Laboratories). Each individual sample was analyzed in triplicate. Furthermore, apoptotic cells were identified by the terminal dUTP nick-end labeling (TUNEL) method, following the directions provided by the manufacturer (In situ Cell Death Detection Kit, POD, Roche Boehringer Mannheim Corp., IN). TH immunohistochemistry was employed for the evaluation of dopaminergic neuronal degeneration. Anti-TH antibody (Chemicon, CA, USA; 1:500 dilution) was used for the experiments. Loss of neurons in the substantia nigra of treated mice was determined by serial section analysis of the total number of TH-positive neurons as described previously (3,4). For detection of doublecortin (Dcx) by immunohistochemistry, the sections were first reacted with primary antibodies against Dcx (1:1000; Abcam, Cambridge, UK) followed by incubation with biotinylated secondary antibodies. Detection was accomplished using streptavidin-peroxidase conjugate and diaminobenzidine as a substrate (LAB Vision, Fremont, CA). Counterstaining was carried out with hematoxylin. Finally, the slides were mounted and analyzed using an optical microscope.
In Vitro and In Vivo 1-Methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP)-Induced Neuropathy
To examine the cytoprotective roles of DHA and EPA in MPTP-induced apoptosis, 500 μM MPTP (Sigma-Aldrich) was added to the cell culture 40 h after pretreatment with 20 μM DHA or 40 μM EPA, followed by measurement of cell viability as well as apoptotic activity 24 h later. As regards in vivo experiments, the male severe combined immunodeficiency (SCID) mice (8 weeks old; around 25 g) were kept at room temperature under a 12-h light/dark cycle and had free access to food and water. Four days prior to transplantation, animals from each group received one intraperitoneal injection of MPTP-HCl (15 mg/kg free base suspended in saline) or saline every 2 h for a total of four doses over an 8-h period in 1 day. And then mice were randomly divided into four treatment groups: PBS, iPS-5th, iPS-5th-EPA and iPS-5th-DHA group. On the day of transplantation, cells were harvested and resuspended in PBS at a concentration of 5 times; 105/μl. Each group was unilaterally injected with 5 times; 106 cells into the striatum using the following coordinates in relation to bregma: 0.1 mm posterior, 2.4 mm mediolateral, and 3.6 mm dorsal. The number of animals studied with striatal transplantation was: PBS (n = 8), iPS-5th (n = 8), iPS-5th-EPA (n = 8), and iPS-5th-DHA (n = 8).
Locomotor Behavior Evaluation
Apomorphine-induced rotations were analyzed 2 days before and 1, 4, 8, and 12 weeks after transplantation, to evaluate the effects of transplantation on symmetry of motor function (11,13). Mice were tested for rotation in response to an intraperitoneal injection of 1 mg/kg apomorphine in PBS. Rotation was measured in an open field box for 30 min at 50 lux. For monitoring the locomotor behavior, EthoVision video tracking system (Noldus Information Technology) was used for recording and processing the data of each group mice. The relative meander was calculated based on the net ipsilateral turning angle divided by distance.
Striatal Dopamine Levels
A commercial kit [Alpco, Dopamine (Research) ELISA] was used to measure the dopamine levels in the striatum ipsilateral and contralateral to the grafted side. One, 2, and 4 months after transplantation, mice were sacrificed and samples were dissected and homogenized. Protein concentrations were measured using BCA Protein Assay Kit (Pierce). Samples were processed according to the manufacturer's instructions.
Statistical Analysis
The data are expressed as means ± SD. Statistical analysis was performed using the one-way or two-way ANOVA test followed by Tukey's test. Values of p < 0.05 were considered statistically significant.
Results
Characterization of MEF-Derived iPS Cells
We introduced retroviruses containing mouse Oct3/4, Sox2, Klf4, and c-Myc into MEFs. Seven days after infection, cells were harvested and plated onto mitomycin C-treated MEF feeder cells (22). Ten days later, some flat and round colonies were observed (Fig. 1A). Undifferentiated murine iPS cells were cultured on inactivated mouse embryonic fibroblasts (Fig. 1A) and formed colonies very similar to murine ES cells (22). These iPS clones were positive for alkaline phosphatase (red color) and stage-specific embryonic antigen-1 (SSEA-1) (red fluorescence; Fig. 1A). We performed RT-PCR to examine whether ES marker genes were expressed in iPS cells. We used primers that only amplify endogenous genes (Oct-4, Sox2, c-Myc, and Klf4) and not transgenes from the retroviruses (Fig. 1B). The RT-PCR results showed that the iPS cells expressed a similar gene signature comprising ESC marker genes, including Oct-4, Nanog, Sox2, Klf-4, F box protein 15 (Fbx15), ES-Ras (ERas), and embryonal stem cell-specific gene 1 (Esg1) (Fig. 1B). The expression levels of these ES-related genes were significantly higher in iPS cells than in MEFs (data not shown). We investigated the pluripotency of the iPS cells reprogrammed from MEFs using EB formation and various differentiation protocols (14). The iPS cells could be differentiated into cartilaginous, adipocyte-like (mesodermal), and hepatocyte-like (endodermal) cells, as confirmed by positive Safranin-O, Oil Red, and albumin staining, respectively (Fig. 1C). After in vitro differentiation, immunostaining detected cells positive for α-smooth muscle actin and cardiac troponin-T (mesoderm markers), α-fetal protein (endoderm marker), and β-tubulin (ectoderm marker; data not shown). The iPS cells could be differentiated into neuronal-like cells (Fig. 1C). Furthermore, under cardiac inductive conditions (14), cells that had migrated from attached EBs began to exhibit spontaneous beating (Fig. 1C). Our data demonstrate that, like ES cells, our MEF-derived iPS cells expressed stem cell marker genes and possessed a pluripotent capacity to differentiate into different cell types of all three germ layers (Fig. 1C). We further demonstrated that signals from GFP retrovirally transduced into iPS cells was consistently detected in EBs and differentiated cells (Fig. 1D). We further investigated the in vivo pluripotency of these GFP-positive iPS cells (Fig. 1E). Xenotransplantion analysis was used to detect the three germ layer-like structure of teratoma formation in iPS-transplanted nude mice (Figs. 1E, F).

Derivation of induced pluripotent stem (iPS) cells and analysis of their pluripotency in vitro and in vivo. (A) Phase contrast microscopic images of iPS cells. Positive alkaline phosphate stain and stage-specific embryonic antigen-1 (SSEA1) of iPS cells. (B) Reverse transcription-polymerase chain reaction (RT-PCR) of stem cell marker gene expression in iPS cells. Note that the expression of stem cell genes [Oct-4 (octamer binding transcription factor 4), Nanog, Sox-2 (sex determining region Y box 2), Klf-4 (Kruppel-like factor 4), Fbx15 (F box protein 15), ERas (ES-Ras), and Esg1 (embryonal stem cell-specific gene 1)] was similar for embryonic stem (ES) and iPS cells. –: RNA-free; +: RNA-positive. (C) iPS cells were differentiated into tissues of all three germ layers, including cartilage, adipocytes, cardiomyocytes (mesodermal), hepatocyte-like (endodermal) cells, and neuronal cells (ectodermal), as confirmed by positive staining with Safranin-O, Oil Red, albumin, and neurofilament, respectively. (D) iPS cells with constitutive expression of green fluorescence protein (GFP) were cultured on a feeder layer of undifferentiated murine iPS cells and undifferentiated colonies on mitotically inactivated murine embryonic fibroblasts (MEFs) (left panel). Embryoid bodies with GFP expression (right panel). (E) iPS cells (106) were implanted into the subcutaneous of nude mice. Teratoma formation after 4 weeks was indicated by arrows. (F) After 4 weeks, ex vivo biopsies and histological analysis revealed teratoma formation in the subcutaneous grafts of iPS cells. Scale bar: 100 μm.
DHA Promotes Neurodifferentiation and Facilitates iPS Differentiation Into DA Neurons
To determine the appropriate induction system for our study, we tested the differentiation method for DA neuron differentiation using a five-stage induction protocol (Fig. 2A). Undifferentiated murine iPS cells constitutively expressing GFP were cultured on inactivated mouse embryonic fibroblasts (Fig. 2A, stage 1). First, DHA was added at stage 3, 4, or 5 to evaluate its effects (Fig. 2). To determine the optimal dosages and durations of DHA treatment for all experiments, differentiated iPS cells were treated with different dosages (20–60 μM) of DHA for different time (12–48 h). Relative to untreated cells, the viability of the differentiated iPS cells treated with 20 μM DHA at 48 h was higher than for those treated with other dosages and treatment durations (Fig. 2E). As 20 μM of DHA was the most effective dose to increase cell viability (Fig. 2E), this concentration was used for all further experiments. In order to confirm the specificity of DHA's impact, an ω-3 PUFA highly similar in structure to DHA, EPA, was used as a control compound (5,29). The result showed that DHA significantly improves cell viability compared with EPA (Fig. 2E). The data also support the hypothesis that DHA treatment improves the cell viability of DA-differentiated iPS cells during stage 5 induction compared to those treated with EPA (Fig. 2E). Recent reports have suggested that Nurr1 plays an important role in the development of mesencephalic DA progenitor cells (41,54). The results of immunofluorescence study found that DHA or EPA treatment increased the percentage of Nurr1-positive iPS cells at stage 4 and 5 relative to control cells (both p < 0.001) (Fig. 2D).
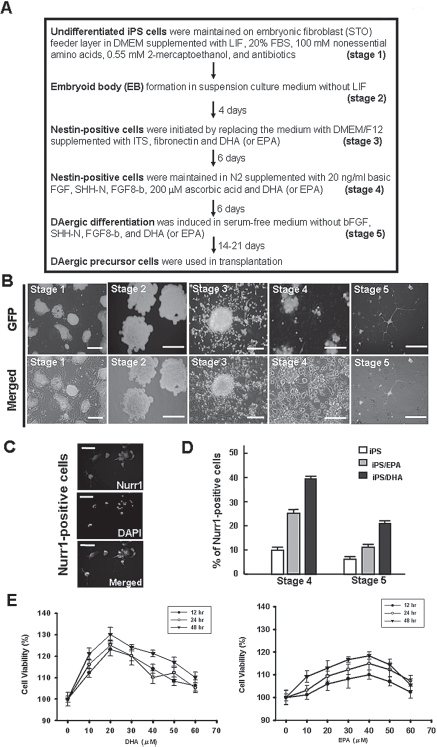
In vitro dopaminergic neuron differentiation of iPS cells and the effect of docosahexaenoic acid (DHA). (A) Five-step induction protocol for producing dopaminergic neuron from iPS cells. DHA was added in steps 3 to 5. (B) The undifferentiated iPS cells constitutively expressed GFP under a nanog-specific promoter in stage 1. Embryoid bodies formed in stage 2. Fibroblast growth factor (FGF)2-responsive neural precursor cells in stage 3. Differentiated neural morphologies were noted 7 days after growth factor withdrawal. After adding sonic hedgehog (SHH) and FGF8 in stage 4, tyrosine hydroxylase (TH)-positive dopaminergic (DA) neurons were observed in stage 5. Scale bar: 50 μm. (C) Immunohistochemistry of DHA-treated iPS-derived cells stained for Nurr1 (nuclear receptor related 1 protein) demonstrate that DHA increased the expression of Nurr1. (D) Expression of Nurr1 in DHA-treated iPS-derived cells was significantly increased compared with controls and stage 4 and 5 eicosapentaenoic acid (EPA)-treated iPS cells during differentiation. (E) Cell viability of iPS cells in response to DHA or EPA treatment.
We next examined the genomic traits associated with DHA (20 μM)- or EPA (50 μM)-induced neurodifferentiation of iPS cells using gene expression microarray analysis. The gene expression profiles and the Gene Ontology (GO) database of neural progenitor cells (NPCs) or ES cells were compared to those of iPS or ES cells to determine the relationship between undifferentiated iPS/ES cells, NPCs, and DA neurons (Fig. 3A). Microarray analysis showed that the expression of 1039 probe sets were significantly altered in DHA- or EPA-treated iPS cells compared with undifferentiated iPS/ES cells when examined by the hierarchical clustering method (Fig. 3A). The predominant upregulated processes in undifferentiated iPS/ES cells included those pertaining to nervous system development cadherin, neurogenesis, and others (Fig. 3A). In contrast, downregulated genes included those related to hexose metabolic process, RNA processing, and ribonucleoprotein complex, (Fig. 3A). Microarray data indicated that DHA and EPA have the potential to promote ES/iPS cell gene patterns to shift to resemble neural progenitors as well as DA lineage neurons (Fig. 3B). Moreover, multidimensional scaling analysis (MDS) further showed that DHA-induced neurodifferentiation in iPS or ES cells was more similar to DA neurons and NPCs than parental and neural induction iPS cells (Fig. 3C). In addition, we used a literature-based network analysis of all MEDLINE records (title and abstract) and Cytoscape software to group the target linkage genes from our microarray data using a Natural Language Processing (NLP) regimen for gene and protein names. We identified the network genes that were involved in Nestin, Tuj1, orthodenticle homeobox 1 (OTX1), OTX2, LIM homeobox transcription factor 1-beta (LMX1B), retinoid X receptor, alpha (RXRA), Nurr1, aromatic L-amino acid decarboxylase (AADC), TH, and dopamine transporter (DAT)-related pathways in DHA- or EPA-treated iPS/ES cells compared to undifferentiated iPS/ES cells (Fig. 3D). Consistent with these findings, the results of quantitative RT-PCR further showed that the expression levels of Nestin, Nurr1, and TH were significantly increased in DHA-treated iPS cells at stage 5 compared to untreated control cells (p < 0.001) (Fig. 4A and data not shown). In contrast, compared to other groups, the ESC-related stemness genes-Oct4 and Nanog were dramatically suppressed in DHA-treated iPS cells at stage 5 (p < 0.001) (Fig. 4A).
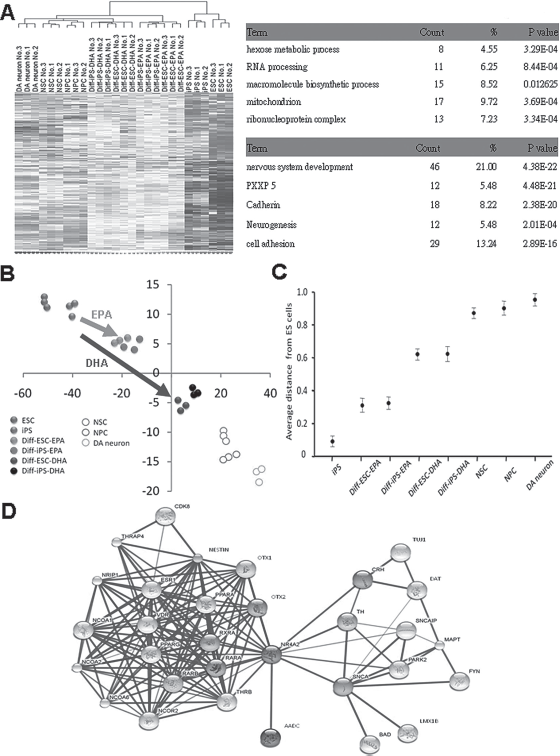
Genomic traits associated with DHA- or EPA-induced neurodifferentiation of iPS cells by gene expression microarray analysis. (A) Microarray data (left; the hierarchical clustering heat map was identified by GenSpring GX Gene Tree Clustering). (B) Principle component analysis (PCA) (right; PCA; PC2: vertical axis; PC1: horizontal axis; Unit: D-chip unit) was used to measure the expression profiling between iPS, ES, differentiated ES and iPS, neural progenitor cells (NPC), and dopamine neurons using bioinformatics method. DHA- or EPA-induced neurodifferentiation of iPS cells was more similar to dopaminergic neurons and NPCs than parental and neural-induced iPS cells. (C) Average linkage distance to DA neuron (multidimensional scaling, MDS); Diff-iPS, differentiated iPS cells; DA, dopamine; NPC, neural progenitor cell. (D) Literature-based network for dopamineric-related genes in DHA-related gene profiling by Cytoscape software. Data shown here are the mean ± SD of three independent experiments.

Activation of the DA pathway and functional DA release from iPS-derived DA differentiated neurons. (A) Real-time quantitative RT-PCR of Oct-4, Nanog, Nestin, TH, aromatic L-amino acid decarboxylase (AADC), and dopamine transporter (DAT) expression in DHA-treated, EPA-treated, and untreated iPS cells during the differentiation stage 5. (B) A Western blot of iPS-derived neural cells demonstrates that protein levels of TH, DAT, and ADCC (DA production-related enzymes) were detected in iPS cells, stage 5 iPS (iPS-5th) cells, EPA-treated iPS-5th cells, and DHA-treated iPS-5th cells. (C) Immunofluorescence of iPS-derived neurons stained for Tuj-1 and TH demonstrate that DHA increased the induction of neuronal expression of Tuj-1 and TH. The right panel shows a quantification plot of the left panel. Scale bar: 30 μm; *p < 0.001. (D) High-performance liquid chromatography and electrochemical detection (HPLC-ED) was used to detect DA levels in stage 5 iPS-derived neurons. (E) A quantification plot demonstrates that DHA upregulated DA neuron differentiation by 3.6-fold relative to untreated stage 4 controls. DHA and EPA upregulated the amount of dopamine released in the culture medium of iPS-5th cells more than that of untreated control iPS cells. Scale bar: 100 μm; *p < 0.001. Data shown here are the mean ± SD of three independent experiments.
DHA Upregulated the DA Pathway and DA Release From Differentiated iPS-Derived Neurons
To further investigate the role of DHA in the DA pathway of treated iPS cells, the RNA and protein levels of TH, DAT, and AADC were determined in stage 5 iPS (iPS-5th) cells treated with DHA (Fig. 4A, B). As shown in Figure 4B, Western blotting quantification results confirmed the significantly increased expression of TH, DAT, and AADC in DHA-treated iPS-5th compared to control iPS cells, iPS-5th cells, and EPA-treated iPS-5th cells (p < 0.001) (Fig. 4B). Next, we examined whether the catecholaminergic neuron-specific markers Tuj-1 and TH were expressed in those cells. Using fluorescence microscopy, we found high positive signals for both Tuj-1 and TH in DHA-treated iPS-5th cells (Fig. 4C). To further confirm that iPS cells were differentiated into functional DA neurons, we examined their ability to release dopamine using a real-time, dynamic liquid chromatography system coupled with electrochemical detection (HPLC-ED) and a microdialysis device (10). Figure 4D shows the quantification of DA expression. The results showed that treatment with DHA effectively upregulated the amount of dopamine in the culture medium of iPS-4th cells (stage 4) 6 days after induction. It is important to note that DHA upregulated the amount (40 × 10–7 ng/ml) of dopamine released in the culture medium of iPS-5th cells 14 days after induction to 3.6-fold more than that of untreated control iPS cells (Fig. 4E). These results suggest that DHA can activate the DA pathway in differentiated iPS cells and further promote DA production and secretion. EPA also can promote DA production, but the EPA-induced dopamine level is less than the DHA-induced level (Fig. 4E).
DHA Facilitated the Expression of Cytoprotective Genes and Was Protective Against MPTP-Induced Apoptosis in Differentiated iPS-Derived Neurons
The chemical MPTP has been suggested to be a parkinsonian agent in humans and was demonstrated to exert similar effects in a number of other primates and rodents. We used MPTP to mimic PD in murine cells in vitro. To explore the cytoprotective role of DHA in iPS-5th cells, we examined the expression of neuroprotective genes such as Bcl-2, Bcl-xL, brain-derived neurotrophic factor (BDNF), glial cell-derived neurotrophic factor (GDNF), and MAP2 in DHA-treated iPS-5th cells. Using quantitative real-time RT-PCR, we found that the mRNA levels of these genes were significantly increased relative to untreated iPS and EPA-treated iPS cells (p < 0.001) (Fig. 5A). We further investigated the role of DHA in preventing MPTP-induced cell toxicity, and our results showed that the mortality of MPTP-treated precursor cells was reduced with DHA treatment compared to EPA treated iPS-5th cells and untreated iPS-5th (control) cells (Fig. 5B). These results indicated that DHA is capable of protecting iPS cells and differentiated neural precursors from MPTP-induced cell death. Moreover, the results of immunofluorescence studies clearly indicated that DHA could significantly increase the expression level of Bcl-2 in the cytoplasm of MPTP- and DHA-treated iPS cells relative to MPTP-treated iPS cells without DHA treatment (p < 0.001) (Fig. 5C). We then used MPTP to induce apoptosis in precursor cells. Using an ELISA assay, we found that the activity of caspase-3 was significantly increased in MPTP-treated iPS cells compared with untreated iPS cells (p < 0.001) (Fig. 5D). Similar results were obtained by TUNEL assay (Fig. 5E). DHA and EPA can decrease MPTP-induced apoptosis, but DHA is more effective than EPA. Therefore, these results suggest that DHA stimulated endogenous levels of cytoprotective genes and thus protected against MPTP-induced apoptosis in iPS-derived neuronal precursor cells.

Detection of Bcl-2 expression and antiapoptotic activity in DHA-treated iPS cells by real-time RT-PCR and immunofluorescence. (A) Real time RT-PCR results demonstrate that the mRNA expression of Bcl-xL, Bcl-2, brain-derived neurotrophic factor (BDNF), glial cell-derived neurotrophic factor (GDNF), and microtubule-associated protein 2 (MAP2) at DHA-treated iPS-5th cells were significantly increased compared to untreated iPS cells; *p < 0.001. (B) Plotting the survival of 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP)-treated cells at iPS-5th cells relative to control cells. The mortality of MPTP-treated precursor cells was reduced with DHA and EPA treatment compared to control cells; *p < 0.001 versus control. (C) Immunofluorescence of iPS-derived DA neurons showed Bcl-2 expression. Scale bar: 40 μm. The right panel shows a quantification plot of the left panel. An apoptosis study in different groups using (D) caspase-3, in which the ratio of non-MPTP-treated iPS was defined as 0.1 and the relative activities of caspase 3 in the different treatment groups were compared to non-MPTP-treated iPS cells, and (E) Terminal dUTP nick-end labeling (TUNEL) assays are demonstrated. Data shown here are the mean ± SD of three independent experiments. *p < 0.001 versus iPS group.
Behavioral Recovery by Transplantation of iPS-Derived DA Precursor Cells in 6-OHDA-Treated Rats with Parkinson-Like Pathology
Selective neurotoxic disruption of dopaminergic pathways can be reproduced by injection of 6-OHDA or MPTP whereas depleting drugs and oxidative-damaging chemicals may also reproduce specific features of PD in rodents (12,51). Unlike MPTP, 6-OHDA lesions cause massive irreversible neuronal loss, and can be uni- or bilateral. Thus, 6-OHDA lesion model was utilized to evaluate DHA effect on iPS cells. We demonstrated that GFP was retrovirally transduced into iPS cells and evaluated DHA-induced DA differentiation. Transplanted 6-OHDA-treated rats were spontaneously delivered and analyzed in their first, second, and fourth months after surgery by 3T-magnetic resonance imaging (3T-MRI) (Fig. 6A, left panel). The results of ex vivo GFP imaging demonstrated that strong, positive GFP signals were detected in transplanted DHA-treated differentiated iPS-5th cells (Fig. 6A, middle panel). We next examined the behavior of control rats and rats grafted with DA neurons derived from iPS-5th cells. Amphetamine stimulation of animals lesioned unilaterally with 6-OHDA induced a movement bias ipsilateral to the injection site (n = 12 each group). The transplanted animals in the iPS-5th only, the iPS-5th with EPA, and the iPS-5th with DHA groups showed a marked recovery of the rotation behavior 4 months after transplantation (Fig. 6B). At the site of the transplanted graft, histological and immunohistochemical studies showed that DHA-treated iPS-5th cells differentiated into neuronal-like cells and presented a high percentage of TH-positive signals (Fig. 6C). Importantly, the results of double-staining immunofluorescences suggested that DHA-treated iPS-5th cells were further differentiated in vivo into TH-positive cells displaying GFP and improved the behavior of 6-OHDA-treated rats with PD after 4 months (Fig. 6C). Furthermore, relative to the control group (PBS injection) and iPS-5th cells only, the percentage of both TH- and GFP-positive cells in the EPA-treated and DHA-treated iPS-5th cells in the transplanted graft were significantly higher 4 months after transplantation (p < 0.05) (Fig. 6D). Furthermore, the concentration of dopamine released in the transplanted grafts of DHA-treated iPS-5th cells was significantly increased compared with the iPS only, iPS-5th, and EPA-treated iPS-5th groups (p < 0.05) (Fig. 6E).

Behavioral recovery by transplantation of iPS-derived DA precursor cells in Parkinson's disease (PD)-like mice. (A) The left panel demonstrates a 3 Tesla-magnetic resonance imaging (3T-MRI) image of a transplanted brain to evaluate iPS growth. GFP was retrovirally transduced into iPS cells, and DHA-induced DA differentiation was evaluated. The right panel demonstrates ex vivo GFP imaging (scale bar: 2 mm) showing the iPS-GFP cells proliferation at the injection site. (B) Behavioral recovery was evaluated by amphetamine-induced net rotation in control, EPA, and DHA treatment groups. The behavioral test was performed in the PBS, iPS-5th, DHA-treated iPS-5th, and EPA-treated iPS-5th cells groups after 1, 2, and 4 months in a 6-hydroxydopamine (6-OHDA)-lesioned animal model (n = 12 for each group). Transplantation of DHA-treated iPS-5th cells into 6-OHDA-treated rats improved behavior compared to other groups. (C) The results of hematoxylin and eosin (H&E) staining (left upper) and immunohistochemistry (IHC) for staining TH (left lower) showed that DHA-treated iPS cells differentiated into neuronal-like cells and presented a high percentage of TH-positive signals (scale bar: 100 μm). The immunofluorescent study (middle and right) showed that GFP-positive and TH-positive signals were co-localized in iPS-derived DA neurons of transplanted grafts. (D) Ratios of cell survival TH/GFP-positive cells 1, 2, and 4 months after transplantation demonstrate that the cell survival ratio of DHA-treated iPS-5th cells was higher than the untreated control or iPS-5th group. Scale bar: 15 μm. (E) The in vivo DA concentrations of transplanted grafts were measured in every group. (F) The results of the behavioral test with different treatment groups after 1, 2, and 4 months in MPTP-treated PD-like severe combined immunodeficiency mice is presented. Data shown here are the mean ± SD of three independent experiments. *p < 0.05, iPS-5th induction versus undifferentiated/untreated (control) iPS cells.
As interactions between grafted cells and host can now be studied more thoroughly in mice rather than in rats (12,51), so we used MPTP-lesioned mice to test the effect on locomotor behavior. We analyzed apomorphine-induced rotation in MPTP-lesioned mice with unilateral grafts of iPS cells, iPS-5th cells, iPS-5th cells with EPA, iPS-5th cells with DHA, or mice sham-injected with PBS. As expected, the animals showed no rotational bias after lesioning and 1 day before transplantation, since systemic MPTP application induces symmetric cell death of the DA neurons in the substantia nigra. Transplantation of iPS cells, iPS-5th cells, iPS-5th cells with EPA, and iPS-5th cells with DHA led to a marked recovery of apomorphine-induced rotation behavior on a timescale of months after transplantation compared with control animals. The transplanted animals in both the iPS-5th only, the iPS-5th with DHA, and the iPS-5th with DHA groups showed marked recovery of the rotation behavior 1 month after transplantation (Fig. 6F). Our results suggest that DHA induced the differentiation of functional DA precursors and improved the abnormal behavior in PD-like animal models after transplantation.
Inhibition of Teratoma-Like or Tumor-Like Formation by Treatment with DHA
Previous studies have shown that transplanted iPS cells are likely to form teratomas in vivo, a feature also found in ES cells (47). We used MRI and ex vivo GFP imaging techniques to evaluate iPS cell growth and teratoma formation without 5-step induction or DHA treatment in transplanted 6-OHDA-treated rats (Fig. 7A). Furthermore, we found that, compared with stage 3 or stage 4 iPS cells, transplantation of well-differentiating stage 5 cells into 6-OHDA-treated rats decreased the incidence of teratoma-like (tridermal layer structure) or tumor-like formation (Fig. 7B). Interestingly, the frequency of tumor-like formation (percent of animals showing tumors in the engrafted cells.) was lower in 6-OHDA-treated rats with DHA-treated iPS cells compared with rats treated with untreated cells or EPA-treated iPS cells (Fig. 7B). Importantly, the results of quantitative RT-PCR further confirmed that the expression of Oct-4 (Fig. 7C) and c-Myc (Fig. 7D) was significantly suppressed in grafts of 6-OHDA-treated rats transplanted with DHA-induced iPS stage 5 cells. To further confirm the undifferentiated tumor-like (neuro-epithelial rosette-like) formation in the transplanted grafts, the positive signals of Dcx (an early neuroblast marker) in the different stages and different induction groups were detected by immunhistochemistry (Fig. 7E). Importantly, no teratoma-like or tumor-like (undifferentiated rosette-like) formations were noted in the grafts of rats transplanted with DHA-induced differentiated stage 5 iPS cells after 4 months of transplantation (Fig. 7B, E). Taken together, these results demonstrate that DHA effectively promoted neurogenic differentiation and significantly inhibited tumorigenicity in vivo. Even though EPA is similar in effect to DHA, the efficacy of EPA is less than that of DHA.

Inhibition of teratoma formation by DHA treatment. (A) Histological evaluation of iPS cell growth in transplanted mice demonstrated teratoma formation (black arrows). Note that iPS cells without DHA or EPA treatment had increased teratoma formation. Scale bar: 150 μm. (B) The incidences of teratoma formation in cells transplanted at different differentiation stages is quantified (%: the incidence of teratoma-formation rats in total rats; each group n = 12). Note that more differentiated cells, such as DHA-treated iPS-derived neural cells in stage 5, showed the lowest rate of teratoma formation. (C, D) The RT-PCR results of the expression of (C) Oct-4 and (D) c-Myc are quantified (each group n = 12). (E) The undifferentiated tumor-like (rosette-like; arrows) formations in iPS cells at different differentiation stages with or without EPA and DHA treatment were confirmed by the immunohistochemical detection of doublecortin (Dcx, early neuroblasts). Scale bar: 50 μm (each group n = 12). Data shown here are the mean± SD of three independent experiments. *p < 0.05 versus iPS group; *p < 0.05, iPS stage 5 cells versus undifferentiated/untreated (control) iPS.
Discussion
The ultimate goal of cell therapy is to transplant functional cell types into patients with a specific disease, such as PD, diabetes, or cardiac infarction. A recent study from Redmond et al. showed the clinical benefits derived from transplantation of human neural stem cells into MPTP-treated nonhuman primates (37). However, avoiding immunorejection is an important concern in current transplantation medicine. One breakthrough study demonstrated that ectopic expression of four genes is sufficient to reprogram murine and human fibroblasts into inducible pluripotent stem cells, which can eliminate the immune rejection of the host (46,47). A recent study demonstrated that patient-specific iPS cells from dermal fibroblasts of patients with long QT syndrome can differentiate into functional cardiac myocytes, but still recapitulated the electrophysiological features of the disorder (32). Therefore, the major advantage of iPS cells over ES cells is that iPS cells can be derived from a patient's own somatic cells, thereby avoiding immune rejection after transplantation and the ethical concerns raised by using ES cells. However, iPS cells have been shown to form teratomas after transplantation. Eliminating this side effect is a critical issue for the field of iPS cell transplantation (30,47,52). Therefore, measures to overcome the tumorigenicity of iPS cells are crucial for the successful treatment of patients. Werning et al. demonstrated that transplanted grafts derived from SSEA-1-depleted mouse iPS cells were consistently smaller and did not develop teratomas up to 8 weeks after transplantation into 6-OHDA-lesioned rats (50). Importantly, Tsuji et al. have provided evidence that selected “safe” iPS clone-derived neural stem cells can produce functional neurons and promote locomotor function recovery without forming teratomas in a mouse spinal cord injury (SCI) model (48). In this study, we have demonstrated, for the first time, that a five-step induction procedure combined with DHA effectively promoted iPS differentiation into functional DA neurons (Figs. 2, 3, 4, and 6). Our in vivo transplantation study confirmed that DHA treatment promoted the development of TH-positive cells from the iPS cells, increased survival of iPS-derived DA neurons in 6-OHDA-lesioned rats (Fig. 6), and also decreased the rate of teratoma formation in the rat brain 4 months after transplantation (Fig. 7). Importantly, we found that DHA treatment in iPS cell-bearing rats significantly downregulated the mRNA expression of ES-specific genes (Oct-4 and c-Myc) in the graft and effectively blocked teratoma formation (Fig. 7). Since safety issues from this therapeutic approach may arise in the future, further studies are required to examine the role of DHA in the inhibition of tumorigenicity in various models of transplantation therapy using iPS cells. To our knowledge, this is the first report demonstrating the role of DHA not only in promoting iPS differentiation into DA neurons but also in inhibiting expression of ES-specific genes as well as teratoma formation in iPS-transplanted grafts.
Nurr1 (NR4A2), an orphan nuclear receptor, is expressed at high levels in the ventral mesencephalon and is involved in the maintenance of midbrain dopamine (7,41,49,54). Nurr1 activates transcription by interacting with the 9-cis-retinoic acid receptor (RXR) and forms heterodimers permissive to retinoids on multimerized AGGTCA response elements. Overexpression of Nurr1 in neural progenitors promotes the induction of DA-specific neurons that demonstrate in vitro presynaptic DA neuronal functionality, releasing DA neurotransmitter in response to specific DA reuptake and further reversing motor dysfunction in a rodent model for PD (43). A recent study showed that overexpression of Nurr1 in mouse ES cells alone promotes the development of TH-positive neurons by up to 50% (53). DHA is the ligand for the RXR/Nurr1 heterodimer, which plays a critical role in the development of the central nervous system. In this study, the transcriptome profiles and molecular signatures analyzed by microarray suggested that DHA drives iPS differentiation into DA progenitors (Fig. 3). Our results showed that DHA treatment dramatically upregulated the endogenous mRNA and protein levels of Nurr1, activated the DA pathway (Figs. 2 and 3), and further enhanced the production of dopamine in differentiated iPS cells (Fig. 4). Taken together, these results support our findings that DHA facilitates iPS differentiation into functional DA neurons, partly through the activation of Nurr1 and the DA pathway, and further promotes DA secretion in vitro as well as in vivo.
DHA and its analogue have been shown to upregulate Bcl-2 and Bcl-xL and decrease the expression of proapoptotic Bax and Bad to further protect neural stem cells from apoptosis (27). Neuroprotectin D1, a docosahexaenoic acid-derived docosatriene, was shown to promote brain survival via the induction of antiapoptotic and neuroprotective gene expression in models of Alzheimer's disease and retinal degeneration (27,33). In accordance with these findings, our results showed that DHA could further upregulate the mRNA and protein expression of Bcl-2 and Bcl-xL in treated iPS cells. DHA has been suggested to have the potential to reduce L-Dopa-induced dyskinesia in MPTP-treated monkeys (39). Mahmoudi et al. have demonstrated that treatment with L-Dopa and DHA can increase the mRNA levels of Nur77, a member of the Nur family, which also includes Nurr1 and Nor-1, in the anterior caudate putamen of MPTP-treated monkeys (28). Importantly, one recent study showed that sustained Nurr1 expression endowed the Nurr1/Akt-induced DA neurons with resistance to toxic stimuli, enhanced survival, and sustained DA phenotypes after transplantation (21). Moreover, the gain or loss of levels and activities of neurotrophic factors such as BDNF has been suggested to be involved in a number of neurodegenerative diseases, including PD (55). We demonstrated here that the expression levels of Bcl-2, Bcl-xL, BDNF, and GDNF were highly upregulated in iPS cells treated with DHA and MPTP (Fig. 5). Our data further showed that additional DHA effectively protected against MPTP-induced apoptosis in iPS cells (Fig. 5). Taken together, these observations indicate that DHA plays a key role in neuronal development and certain forms of neural plasticity and prevents neurotoxicity in the neural differentiation process of stem cells. Whether the upregulation of Bcl-2, BDNF, GDNF, and other genes are involved in the DHA- or Nurr1-related pathways and play a role in enhancing neuroprotection and neuroplasticity should be further investigated.
In conclusion, we demonstrate that DHA greatly enhanced the neurogenic and dopaminergic differentiation of iPS cells, effectively protected iPS cell-derived dopaminergic neurons, and prolonged graft survival in 6-OHDA-treated rats with Parkinson-like pathology. Furthermore, DHA treatment resulted in significantly increased development of iPS-derived DA neurons while preventing teratoma formation. In the future, DHA combined with cell therapy using nonviral vector or DNA-free iPS cells may improve the prognosis and provide an alternative treatment for PD.
Footnotes
Acknowledgments
This study was assisted in part by the Division of Experimental Surgery of the Department of Surgery, Taipei Veterans General Hospital. This study was supported by research grants from National Science Council (NSC-98-3111-B-010-005, NSC-98-2314-B-075-008-MY3, and NSC-98-2314-B-075-032-MY3), Taipei Veterans General Hospital (V97B1-006, E1-008, ER2-018, ER3-005, F-001, V99C1-145, and V99E1-009), the Joint Projects of UTVGH (VGHUST-98-G6-6), Yen-Tjing-Ling Medical Foundation, National Yang-Ming University (Ministry of Education, Aim for the Top University Plan), and Center of Excellence for Cancer Research at Taipei Veterans General Hospital (DOH99-TD-C-111-007), Taiwan. The authors declare no conflicts of interest.
