Abstract
Duchenne muscular dystrophy is a recessive X-linked genetic disease caused by dystrophin gene mutations. Cell therapy can be a potential approach aiming to introduce a functional dystrophin in the dystrophic patient myofibers. However, this strategy produced so far limited results. Transforming growth factor-β (TGF-β) is a negative regulator of skeletal muscle development and is responsible for limiting myogenic regeneration. The combination of TGF-β signaling inhibition with myoblast transplantation can be an effective therapeutic approach in dystrophin-deficient patients. Our aim was to verify whether the success of human myoblast transplantation in immunodeficient dystrophic mice is enhanced with losartan, a molecule that downregulates TGF-β expression. In vitro, blocking TGF-β activity with losartan increased proliferation and fusion and decreased apoptosis in human myoblasts. In vivo, human myoblasts were transplanted in mice treated with oral losartan. Immunodetection of human dystrophin in tibialis anterior cross sections 1 month posttransplantation revealed more human dystrophin-positive myofibers in these mice than in nontreated dystrophic mice. Thus, blocking the TGF-β signal with losartan treatment improved the success of myoblast transplantation probably by increasing myoblast proliferation and fusion, decreasing macrophage activation, and changing the expression of myogenic regulator factors.
Keywords
Introduction
The super family of transforming growth factor-β (TGF-β) cytokines exerts a potent level of control over muscle gene expression. In 1981, Roberts and colleagues (27) reported that TGF-β induced the proliferation of rat kidney fibroblasts. Since then, TGF-β has been found to have a plethora of effects, such as regulation of cell growth, proliferation, differentiation, adhesion, migration, and apoptosis (21). Its aberrant expression has been implicated in fibrotic and inflammatory conditions in kidney, liver, and lung (6,16,32). Recently, an in vivo role for TGF-β1 in skeletal muscle regeneration was confirmed (9). Mice deficient for the extracellular matrix protein fibrillin-1 have increased TGF-β1 signaling activity that causes failure of skeletal muscle regeneration (9). These mice are used as a model for the human Marfan syndrome, which is caused by a mutation of the fibrillin-1 gene (11).
There is recent evidence that elevated TGF-β1 signaling also limits regeneration in muscular dystrophies. Cohn et al. (9) investigated this possibility in mdx mice, a model for Duchenne muscular dystrophy (DMD). DMD is caused by mutations in the dystrophin gene, which leads to a severe deficiency of the dystrophin protein in the muscle. Mutations within the dystrophin gene can either be inherited or occur spontaneously during germ line transmission, affecting 1 in 3,500 male births. Dystrophin, the dystrophin-associated glycoprotein complex, and laminin α-2 form a link between the intracellular cytoskeleton and the extracellular matrix. This link is crucial to maintain the structural integrity of myofibers (5,25). The main consequence of dystrophin absence in skeletal muscle is sarcolemmal instability leading to increased myofiber vulnerability to mechanical stress, resulting in myofiber necrosis, followed by regeneration as long as the regenerative capacity is not annihilated. The frequent cycles of myofiber necrosis and regeneration lead to fibrosis and fat infiltration, which progressively replace the muscle tissue.
Cohn et al. (9) found that muscles from mdx mice expressed elevated levels of proteins induced by TGF-β1 signaling, and also elevated levels of thrombospondin-1 (TSP-1), which activates latent TGF-β1 in response to signaling through the angiotensin II type 1 receptor (AT1). Losartan, an FDA-approved drug, is a potent inhibitor of AT1 activation, thus blocking TGF-β1 activation by inhibiting TSP-1 production. This blockade restored the regenerative capacity and improved muscle function in mouse models of Marfan syndrome and DMD (9).
Many older DMD patients are currently being treated with losartan for the presumptive cardiac benefits and fibrosis reduction induced by angiotensin II-type 1 receptor blockers. A clinical trial of losartan in children with DMD to evaluate its effects on skeletal muscle strength and function was anticipated to begin enrollment in 2010.
DMD treatment may eventually require a combination of several therapeutic approaches. Our research group has recently demonstrated that dystrophin expression following the transplantation of normal myoblasts protects mdx muscle from contraction-induced damage (29). Moreover, inhibition of myostatin signal, a member of TGF-β superfamily, improved the success of transplantation of myoblasts in mdx mice and in immunodeficient recombination-activating gene (Rag)-/-/mdx mice (3,12). Thus, in order to verify whether the blockade of TGF-β1 signal improves the success of myoblast transplantation, we have treated Rag-/-/mdx mice with losartan and transplanted normal human myoblasts in their tibialis anterior (TA) muscles.
Materials and Methods
Isolation of Human Myoblasts
Primary myoblasts cultures were grown from muscle biopsies of a human cadaveric donor obtained following informed consent of the family and approval by the Human Ethic Committee of the Research Center Hospital of Laval University (CRCHUL). The muscle samples were dissociated with collagenase and trypsin as previously described (17) and cultured in MB-1 growth medium (Hyclone, South Logan, UT, USA) with 15% fetal bovine serum (FBS), 1% penicillin-streptomycin (Gibco, Burlington, Ontario, Canada), and 10 ng/ml basic fibroblast growth factor (Strathmann Biotec AG, Hamburg, Germany) in a humidified atmosphere with 5% CO2 at 37°C.
Cell Proliferation Assay
Cellular growth curves were measured using the CyQuant Cell Proliferation Assay Kit (Invitrogen, Burlington, ON, Canada) according to the manufacturer's instructions. Human myoblasts were seeded in 24-well plates at initial densities of 2 times; 103 cells/well in a total volume of 100 μl MB-1 supplemented with 10% FBS and two concentrations of losartan potassium (Galenova, St-Hyacinthe, QC, Canada) ranging from 0 to 100 μM. Medium was changed every 2 days. At 0, 3, and 6 days, the medium was discarded, and plates were frozen at −80°C. The day of the assay, plates were thawed and a cell lysis buffer [20 mM Tris, 1 mM dithiothreitol (DTT), 1 mM phenylmethylsulfonyl fluoride (PMSF), 1% sodium dodecyl sulfate (SDS)] was added to each well. The total cellular nucleic acid was measured using florescence at 530 ± 12.5 nm emission following excitation at 485 ± 10 nm by a Hitachi F-4500 fluorescence spectrophotometer (Hitachi Instruments, Boulder, CO).
Cell Differentiation Assay
Myoblast differentiation was evaluated by counting the percentage of nuclei inside myotubes (i.e., syncytia with three or more nuclei). For this test, myoblasts were cultured in MB-1 for 24 h and the proliferation medium was replaced for 72 h by a fusion medium (Dulbecco's modified Eagle medium) containing only 2% FBS in presence or not of losartan (10 or 100 μM). Myotubes were visualized by myosin heavy chain (MyHC) immunodetection. The cells were fixed with 95% ethanol, washed twice with phosphate-buffered saline (PBS) for 10 min, and blocked with 10% FBS in PBS for 1 h. The samples were incubated 2 h at room temperature with a mouse anti-MyHC mAb (MF20, DSHB, Iowa City, IA) (1:100) and then for 1 h with an anti-mouse Ig Alexa 546-conjugated secondary antibody (Invitrogen) diluted 1:100. Finally, the cells were washed with PBS and mounted with PBS/glycerol. 4′,6-Diamidino-2-phenylindole (DAPI) staining was done with PBS to label the nuclei. The number of nuclei in the myotubes was counted and normalized with the total number of nuclei in the culture well as a measure of terminal differentiation.
Analysis of Apoptosis and Necrosis
An apoptosis and necrosis assay was performed by flow cytometry according to the manufacturer's protocol (BD BioSciences, San Jose, CA). Briefly, myoblasts treated or not with losartan (10 μM) were exposed or not to 10 mM hydrogen peroxide (H2O2, Merck Co., Inc., NJ) for 2 h. After trypsinization, the cells were harvested, washed twice with Hank's balanced salt solution, and suspended in binding buffer (10 mmol/L HEPES, pH 7.4, 140 mmol/L NaCl, 2.5 mmol/L CaCl2). Aliquots of 100 μl suspension (1 times; 105 cells) were incubated with 5 μl Annexin V-FITC (fluorescein isothiocyanate) and 5 μl propidium iodide (PI) (50 μg/ml) for 15 min at room temperature in the dark. Binding buffer (400 μl) was added to the cell suspension. The cells were gently vortexed and analyzed within 1 h by flow cytometry (Becton-Dickinson, Oxford, UK). Apoptotic cells were quantified by percentage of Annexin V-FITC-positive cells among the total gated cells, while necrotic cells were quantified by calculating the percentage of PI-positive cells in the total gated cells.
Cell Radiolabeling
Myoblasts cultured in MB-1 for 24 h were radiolabeled by further proliferation for 48 h in MB-1 containing 0.25 μCi/ml [methyl-14C]thymidine (50 μCi/mmol) before their transplantation.
Animals
All experiments were performed in 2-month-old Rag-/-/mdx mice produced in our animal facilities by crossing mdx/mdx mice (a DMD model due to a mutation leading to dystrophin deficiency) with Rag-/- mice (an immunodeficient mouse that accepts human grafts). The mice were maintained under pathogen-free conditions. All the experiments with mice were conducted in accordance with the Laboratory Animal Care and use Ethics Committee of Laval University.
Losartan Treatment and Cell Transplantation
The myoblast transplantation success was evaluated in mice previously treated or not with oral losartan [0.6 g/L (9,15,37) in drinking water; n = 5] during 15 days. Human myoblasts (5 × 105) resuspended in 10 μl of HBSS were transplanted in both TAs using a glass capillary along 12 transversal axis trajectories. Mice were euthanatized 1 month after myoblast transplantation.
For muscle regeneration and cell survival experiments, mice were treated or not with oral losartan (0.6 g/L in drinking water; n = 5) for 40 days and 5 × 105 radiolabeled cells were transplanted in both TAs.
Muscle Sampling and Processing
Mice were killed at different posttransplantation times depending on the experiment: at 1 month for experiments to evaluate the myoblast transplantation success and at days 0, 3, and 5 for experiments to evaluate the early grafted cell survival. Muscles for histological analyses were mounted in embedding medium and snap-frozen in liquid nitrogen. Serial 12-μm cross sections were obtained in a cryostat at −25°C. Muscles for DNA or RNA extraction were placed in microtubes, snap-frozen in liquid nitrogen, and stored at −80°C.
Quantification of Early Grafted Cell Death
The DNA of the TAs grafted with radiolabeled cells was extracted after 0 and 3 days using the phenol-chloroform method. Briefly, muscles were minced and incubated with 50 μl of proteinase K (10 mg/ml) at 56°C until the solution became clear. Digested muscles were then mixed with 500 μl of a solution of phenol/chloroform/isoamyl alcohol (25:24:1) and centrifuged 3 min at 13,000 rpm. The upper solution was recovered and mixed with the same volume of chloroform and centrifuged again. The upper solution was recovered and 25 μl of 3 M sodium citrate was added before the addition of 1 ml of 100% ethanol. After centrifugation 8 min at 13,000 rpm, the pellets were washed in 70% alcohol before another centrifugation. The pellets were then dried before the suspension of the DNA in 100 μl sterile water. Similar aliquots of the DNA solution were mixed with 5 ml of a liquid scintillation counting cocktail (Sigma, Oakville, ON, Canada) and radioactivity was measured in a counter system (Mod. Wallac 1409, Woodbridge, Ontario, Canada).
Histological Immunodetection
For immunohistochemistry, the TA cross sections were fixed in 4% paraformaldehyde, washed in PBS for 15 min, and blocked with FBS (10%) in PBS for 1 h. The following antibodies were used for this study: a mouse anti-human dystrophin mAb (7F7, MRIC Biochemistry Group, Wrexham, UK) (1 h, 1:300); a rabbit polyclonal antibody against pSmad2 (Cell Signal, Danvers, MA) (O/N, 1:1000); a mouse mAb against human lamin A/C (Vector laboratory, CA) (30 min, 1:300) to visualize human nuclei; and a rabbit polyclonal antibody against periostin (Abcam, Cambridge, MA) (1 h, 1:250). Depending on the primary antibody, the second reaction was done either with a biotinylated anti-mouse Ig antibody (Dako, Copenhagen, Denmark) (1:300) followed by streptavidin-Cy3 (Sigma) (1:300), Alexa 488 fluor-conjugated goat anti-rabbit Ig, or anti-mouse Ig (Invitrogen Burlington) (1:300) for 1 h at room temperature. Nuclei were stained with DAPI (1:10000) for 5 min and cryostat sections were mounted using fluoromount G (Electron Microscopy Sciences, Hatfield, PA).
The dystrophin-positive myofibers were counted in 12 cryostat sections separated by 150 μm for each of the five muscles of each group. Afterward, the five highest sections of five TAs from each group were selected and the means + SDs were calculated.
Immunodetection of inflammatory cells were performed on muscle cross sections of mice treated 45 days with oral losartan and transplanted 5 days before the end of the treatment. After fixation with 4% paraformaldehyde, nonspecific binding sites were blocked by incubating the sections with FBS (10%) in PBS for 1 h. Sections were then incubated with a rabbit anti-mouse Mac-1 polyclonal antibody (Roche Diagnostics, Mississauga, ON, Canada) or a biotinylated monoclonal anti-mouse F4–80 (Peninsula Laboratories, San Carlos, CA) for 1 h at room temperature, followed by 30 min with a goat anti-rabbit Ig secondary antibody conjugated with Alexa 546 (Invitrogen) and streptavidin-Cy3, respectively. To quantify the labeling fluorescence, we used the Image J. 1.40g (Wayne Rasband, National Institutes of Health, USA).
Quantitative RT-PCR
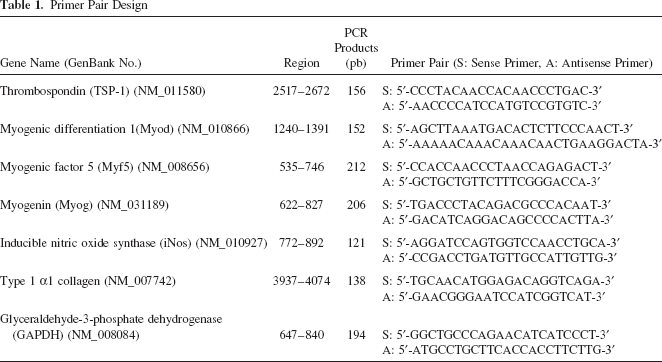
Quantitative RT-PCR (Q-RT-PCR) was used to examine the mRNA expression levels of TSP-1, myogenic regulator factors (MRFs) [myogenic differentiation (MyoD), myogenic factor 5 (Myf5), and myogenin], inducible nitric oxide synthase (iNOS), and type 1 collagen in the TAs of 45 days oral losartan-treated or untreated mice. The mRNA was extracted using an RNeasy Plus kit (Qiagen, Mississauga, ON Canada). The cDNA templates for Q-RT-PCR were synthesized by an inverse transcriptase with 1 μg RNA in superscript II RNase H-RT reaction (Invitrogen). Q-RT-PCR was carried out in a Light Cycler 480 (Roche) with SYBR Green light Cycler Fast Start DNA Master Plus (Roche) as a detector. All target gene expressions were normalized to glyceraldehyde 3-phosphate dehydrogenase (GAPDH) levels. The primer pairs were designed with GeneTools logiciels (Biotools Inc., Madrid, Spain) and are displayed in Table 1.
Primer Pair Design
Statistical Analysis
The experimental values were presented as means + SDs. Statistical analyses were performed by analysis of variance (one-way ANOVA) with Fisher's PLSD test and Tukey's test for multiple comparisons post hoc in the StatView statistical package (StatView 5, SAS Institute Inc., Cary, NC). Differences were considered statistically significant atp < 0.05.
Results
Losartan Treatment Increased Myoblast Growth In Vitro
A fluorescence-based assay that correlates with cell number was used to determine the effects of losartan on human myoblast proliferation. Myoblast proliferation was significantly increased after 6 days of treatment with 10 and 100 μM losartan (p < 0.05) (Fig. 1A).

Losartan-stimulated human myoblast proliferation and differentiation. Human myoblasts were cultured in MB-1 alone or with losartan potassium at 10 and 100 μM for 6 days. The number of cells was evaluated with the CyQuant Cell Proliferation Assay Kit at time 0 and after 3 and 6 days (A). The results are expressed as arbitrary fluorescent units measured at 530 nm (means + SD, N = 5). *Significantly greater values at 6 days in losartan-treated cultures than untreated cultures due to increased proliferation. (B) Myoblasts were transferred to differentiation medium [Dulbecco's modified Eagle medium (DMEM), 2% fetal bovine serum (FBS)] for 3 days in the presence of losartan at 10 and 100 μM or without losartan. The cultures were immunostained with anti-MyHC (myosin heavy chain) and with 4′,6-diamidino-2-phenylindole (DAPI) to visualize the nuclei. Representative pictures are presented in (B). Scale bar: 0.2 mm. (C) The mean fusion indexes (+SD, N = 3) are illustrated. *Significantly higher results in losartan-treated myoblasts than control myoblasts. **Significantly higher in 100 μM losartan-treated myoblasts than 10 μM losartan-treated myoblasts.
Considering that TGF-β1 is a myoblast differentiation inhibitor, we wanted to evaluate in vitro the effect of losartan on the myogenic differentiation of human myoblasts. After switching confluent myoblasts to a differentiation medium, a losartan treatment during 3 days increased significantly the number of myotubes immunostained for MyHC (Fig. 1B). Moreover, the fusion index was significantly increased by 10% with 10 μM losartan and by 15% at 100 μM (Fig. 1C). Thus, the increased differentiation of human myoblasts by losartan was dose dependent.
Losartan Prevented H2O2-Induced Apoptosis and Necrosis in Myoblasts
Myoblast apoptosis was induced with H2O2 in the presence or absence of losartan. The percent of apoptotic and necrotic cells was determined by flow cytometry following staining with Annexin V-FITC and PI (Fig. 2A–D). The treatment with 10 μM losartan did not change the percentage of viable cells when they were not exposed to H2O2, but reverted apoptosis and necrosis induced by H2O2 (Fig. 2E).

Losartan prevents myoblast apoptosis and necrosis induced by H2O2. Myoblasts were treated with or without losartan potassium (10 μM) in vitro for 2 days. They were then incubated with hydrogen peroxide for 2 h and stained with Annexin V-FITC (fluoroscein isothiocyanate) and PI (propidium iodide) for flow cytometry analysis. The downright quadrants of (A) to (D) represent early apoptosis (Annexin V-FITC-positive cells and PI-negative cells). The top right quadrants represent late apoptosis and necrosis (Annexin V-FITC-positive cells and PI-positive cells). (A) Untreated control cells. (B) Cells treated with 10 μM losartan potassium. (C) Cells treated only with hydrogen peroxide. (D) Cells treated with both 10 μM losartan potassium and hydrogen peroxide. (E) Cells were counted in quadrant scatter plot. The percentage of apoptotic cells was calculated by dividing the Annexin V-FITC-positive and PI-negative cells (AV+/PI-) by the total number of gated cells and multiplying by 100. The percentage of necrotic cells was calculated as the number of the Annexin V-FITC-negative and PI-positive cells (AV-/PI+) divided by the total number of gated cells and multiplying by 100. Representative pictures are presented. The mean expressions (+SD, N = 3) are illustrated. *Significantly higher levels of apoptosis in H2O2-treated myoblasts than in other treatments. ¶Significantly higher levels of necrosis in H2O2-treated myoblasts than other treatments.
Improved Success of Human Myoblast Transplantation in Mice Treated with Losartan
To investigate the effect of blocking the TGF-β1 signal on the long-term success of the transplantation of human myoblasts in dystrophic mice, human dystrophin was detected by immunohistochemistry in muscles transplanted with myoblasts in mice treated or not with losartan during 45 days (Fig. 3A, B). About 50% more dystrophin-positive myofibers were present in the muscle cross sections of mice treated with losartan (191 ± 40 myofibers per muscle cross section) than in control mice (126 ± 29) (Fig. 3C).
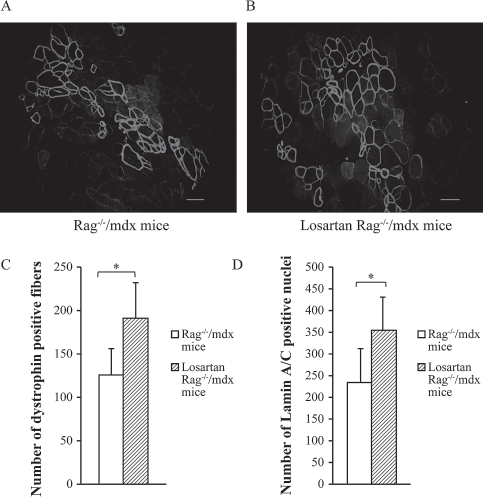
Increased success of human myoblast transplantation in mice treated with losartan. Immunohistochemical detection of human dystrophin (red fluorescence) in tibialis anterior (TA) sections of untreated Rag-/-/mdx (recombination-activating gene/mutated dystrophin) mice (A) and losartan-treated Rag-/-/mdx mice (B) 1 month after the transplantation of human myoblasts. (C) Graphical representation of the mean number (+SD, N = 5) of dystrophin-positive myofibers per cross section of TAs transplanted with human myoblasts in Rag-/-/mdx mice treated or not with losartan during 45 days (15 days before and 30 days after the transplantation). There are, on average, 51% more dystrophin-positive myofibers in TA of mice treated with losartan. (D) The mean numbers (+SD, N = 5) of lamin A/C-positive nuclei per cross section of TA muscles transplanted with human myoblasts and treated or not with losartan during 45 days. There are, on average, 50% more human lamin A/C-positive nuclei in the TA of mice treated with losartan. Representative pictures are presented in (A) and (B). Scale bar: 50 μm. *Significantly higher result in losartan-treated Rag-/-/mdx mice than control Rag-/-/mdx mice.
Losartan Enhanced the Survival of Engrafted Human Myoblasts
To identify the cause of the increased success of human myoblast transplantation in mice treated with losartan, we evaluated the survival of the grafted cells. We first quantified the human nuclei (identified by human lamin A/C immunodetection) present in the cell-grafted TAs. The number of human nuclei was significantly higher in the TAs of losartan-treated mice compared to the controls (Fig. 3D).
In a second experiment, the early posttransplantation survival of human myoblasts was evaluated by first radiolabeling them with [14C]thymidine before their transplantation and then quantifying the radiolabel in the cell-grafted muscles at the time of transplantation and 3 days later. Approximately 6% of the radiolabel present in the cell-grafted muscles at time 0 was detected 72 h posttransplantation in mice untreated with losartan; in contrast, 10% of the radiolabel was still present in losartan-treated mice (Fig. 4). Thus, there was a significant increase of myoblast survival in mice treated with losartan.

Losartan treatment increased the early survival of transplanted myoblasts. Radiolabeled cells (5 × 105) were transplanted in the TA of Rag-/-/mdx mice treated or not with oral losartan. The muscles were removed at day 3 after transplantation and the DNA was extracted to quantify the radioactivity. There is 40% more radiolabel in the DNA extracts of losartan-treated mice, indicating a better survival of the radiolabeled human myoblasts. The mean radioactivitys (+SD, N = 5) are illustrated. *Significantly higher result in losartan-treated Rag-/-/mdx mice than in control Rag-/-/mdx mice.
Increased Expression of pSmad2 and Periostin in Losartan-Treated Mice
To evidence a cause-and-effect relationship between losartan treatment and inhibition of TGF-β1 signaling, we detected periostin, pSmad2, and TSP-1 in skeletal muscles of mice treated or not with losartan during 45 days (Fig. 5). Periostin, a protein induced by TGF-β1 and expressed in regenerating skeletal muscle, was detected by immunoshistochemistry in the sarcolemma and endomysium of control mice, but was almost absent in losartan-treated mice (Fig. 5A). pSmad2, the mammalian homolog of the Drosophila mothers against decapentaplegic (Mad) implicated as downstream effectors of TGF-β/BMP (bone morphogenetic protein) signaling, was detected in myofiber nuclei of control mice, but not in mice treated with losartan (Fig. 5B). Expression of TSP-1 mRNA, a potent mediator of angiotensin II-induced TGF-β1 activation via AT-1 was greatly reduced in losartan-treated mice compared with control mice (Fig. 5C).
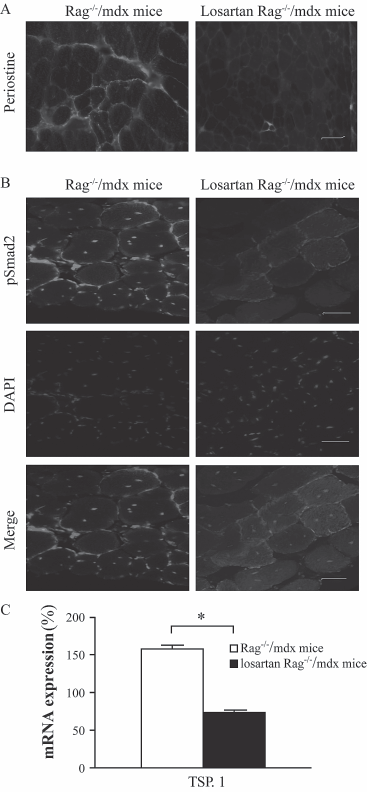
Losartan decreases angiotensin-II-mediated TGF-β (transforming growth factor-β) signaling in Rag-/-/mdx mice. (A, B) Immunofluorescent detection of target proteins downstream of angiotensin-II receptor. The TAs of control nontreated Rag-/-/mdx mice show periostin expression in the sarcolemma and extracellular matrix (A) and nuclear expression of pSmad2 (the phosphorylated mammalian homolog of the Drosophila mothers against decapentaplegic) (B) on the contrary of losartan-treated Rag-/-/mdx mice. (C) Real-time PCR to quantify the expression of throbospondin-1 (TSP-1) mRNA. The graphic shows a significant decrease of TSP-1 expression in losartan-treated Rag-/-/mdx mice compared to untreated Rag-/-/mdx. Scale bar: 50 (im. The mean expressions are represented (+SD, N = 5). *Significantly higher in losartan-treated Rag-/-/mdx mice than control Rag-/-/mdx mice.
Expression of MyoD Family mRNA in Mice Treated with Losartan
Q-RT-PCR analyses of Myf5, myogenin, and MyoD showed significant increases in the expression of Myf5 and myogenin in mice treated 45 days with losartan compared with control mice (Fig. 6).

Expression of myogenic differentiation (MyoD) family mRNAs in muscles of Rag-/-/mdx mice treated or not with losartan. The mRNA expression of the myogenic regulator factors (MRFs) was quantified by Q-RT-PCR. The expression of Myf5 and myogenin was significantly increased by the treatment with losartan in Rag-/-/mdx mice. The expression of myogenic factor 5 (Myf5), the earliest marker of myogenic commitment, was the most upregulated (more than 50%). The mean expressions are represented (+SD, N = 5). *Significantly higher in losartan-treated Rag-/-/mdx mice than in control Rag-/-/mdx mice.
Reduction of Macrophage Infiltration in Losartan-Treated Mice
Immunodetection of Mac-1, a cell surface marker for macrophages but also for bone marrow, spleen, and natural killer cells, granulocytes, and blood monocytes, in the TAs of Rag-/-/mdx mice 5 days after myoblast transplantation demonstrated the presence of inflammatory cells, but no difference was observed between losartan-treated or untreated mice (Fig. 7A). However, immunodetection of F4/80, an antigen present on major subpopulations of resident tissue macrophages (31), in the same muscles demonstrated a lower amount of macrophages in losartan-treated mice (Fig. 7B). Since iNOS is a main mediator of macrophage cytotoxicity, we wanted to verify whether the decrease in the number of macrophages was accompanied by a decrease in iNOS expression. Using real-time PCR, we observed a decrease of >60% in iNOS expression in the TAs of losartan-treated mice compared with control mice (Fig. 7C).
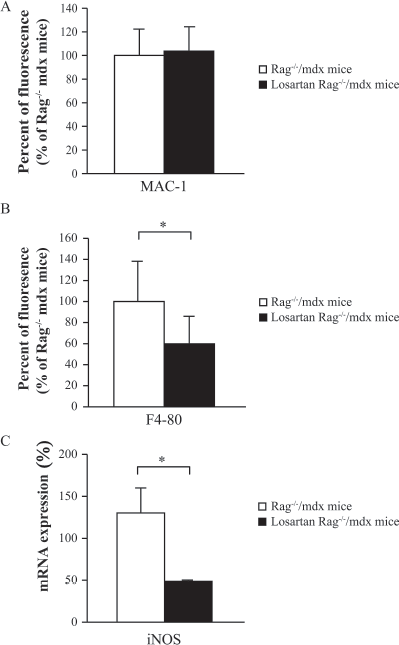
Fewer macrophages are found in muscles of losartan-treated Rag-/-/mdx mice grafted with myoblasts. (A, B) The fluorescent immunodetection of Mac-1 and F4/80 (macrophage markers) was quantified in TAs of Rag-/-/mdx mice treated or not with oral losartan during 45 days. The myoblasts were transplanted after 40 days of the treatment. (C) The mRNA expression of induced nitric oxide synthase (iNOS) was quantified by Q-RT-PCR. The expression of iNOS was significantly decreased in losartan treated Rag-/-/mdx mice compared with untreated Rag-/-/mdx mice. The mean expressions are represented (+SD, N = 5). *Significantly lower expression in losartan-treated Rag-/-/mdx mice than control Rag-/-/mdx mice.
Dystrophin-Positive Myofibers Size and Collagen Content
No difference in cross-sectional area of human dystrophin-positive myofibers was observed between losartan-treated and untreated mice (Fig. 8A). On the other hand, the losartan-treated mice had significantly lower concentrations of type I collagen mRNA than untreated mice (Fig. 8B).

Inhibition of TGF-β signaling does not induce hypertrophy but reduces expression of collagen type I. (A) Cross-sectional area of human dystrophin-positive myofibers, measured in the TAs of Rag-/-/mdx mice treated or not with oral losartan for 45 days and transplanted with human myoblasts at day 15. There is no significant difference between both groups. (B) The mRNA of collagen type I was quantified by Q-RT-PCR. The expression of collagen type I was significantly decreased by the treatment with losartan in Rag-/-/mdx mice. The mean expressions are represented (+SD, N = 5). *Significantly lower expression in losartan-treated Rag-/-/mdx mice than control Rag-/-/mdx mice.
Discussion
TGF-β1 is a member of the TGF-β family and is an endogenous negative regulator of muscle growth. It is homologous to myostatin and induces several similar effects in muscles, including the inhibition of proliferation and differentiation of muscle precursor cells, and stimulation of fibrosis (1,18). The inhibition of TGF-β1 is a potential therapeutic strategy for DMD (4,8). One study showed that losartan, an angiotensin II-type I receptor blocker usually used for the treatment of hypertension, was well tolerated in mdx mice, and that the administration of this drug for 6–9 months led to muscle improvements such as less fibrosis and increased absolute force (9). Many DMD patients in advanced stages of the disease are currently being treated with losartan for the presumptive cardiac benefits of angiotensin II-type I receptor blockers.
Considering the potential benefits of losartan for the treatment of muscle diseases, we decided to test its effects in the context of myoblast transplantation, another potential therapeutic tool for DMD (34). Indeed, our study is the first to demonstrate a potentially beneficial role of TGF-β 1 inhibition for human myoblast transplantation in DMD. We used a Rag-/-/mdx mouse model, which accommodates xenotransplantation, hence human myoblast implantation.
Despite the absence of T cells, important producers of TGF-β, the biological effect of TGF-β was present because this cytokine was secreted by other cells such as myeloid and dendritic cells (23,35). Thus, we observed that mice treated with oral losartan presented 50% more dystrophin-positive myofibers and human nuclei than untreated mice after human myoblast transplantation. Considering the other results of the present study, this improvement could be explained by factors such as an increased early survival, proliferation, and/or differentiation of the transplanted myoblasts, as discussed below.
Some of our experiments support the potential role of the increased early survival of the transplanted myoblasts in the improved myoblast transplantation success due to the losartan treatment. In vivo, we observed a 40% improvement of the early survival of the implanted myoblasts in the recipient muscle, measured by quantifying the radiolabeled cells grafted in losartan-treated mice. Indeed, one of the problems of a therapy based on myoblast transplantation is the transplanted myoblasts acute death within the first 3 days posttransplantation (13,14,33). This phenomenon was attributed by some researchers to a cell killing by the inflammatory cells that infiltrate the myoblasts grafts, such as neutrophils and monocytes/macrophages. If this is the case, the improvement of the early survival of the implanted myoblasts in losartan-treated mice could be explained by the reduction of the macrophage infiltration and iNOS expression evidenced after myoblast transplantation in the present study. Our in vitro experiment showing a protective effect of losartan against oxidative-induced cell death may further explain the increased early survival of the transplanted myoblasts and the increased myogenic differentiation (38). An attenuation expression of molecules involved in macrophage recruitment and the associated reduction of macrophage recruitment was observed in renal allograft (39).
Other causes of the myoblast transplantation improvement observed are the beneficial effects of losartan on the proliferation and/or differentiation of human myoblasts. In our study, the proliferation and differentiation of human myoblasts was significantly increased after inhibition of TGF-β1 in vitro. This positive effect was not observed in C2C12 cells, an immortal mouse myoblast line (24). Moreover, the estimation of these development stages was problematic in vivo. MRFs, which are key orchestrators of muscle gene expression, are targeted by TGF-β1 signaling. Thus, our results demonstrated the effects of losartan treatment on the mRNA expression of these genes. MyoD family (i.e., the basic helix–loop–helix transcription factors: Myf5, MyoD, and myogenin) plays an essential role as central regulators of myogenesis (26). Myf5 and MyoD are expressed in myoblasts and myotubes, and are required for myogenesis. Myogenin is critical for myotubes formation and terminal myogenic differentiation events (26), since it is highly expressed when myoblasts commit to differentiation state to form myotubes. Our result confirmed that the expression of MRFs in muscle treated with losartan was increased. Thus, inhibition of TGF-β1 reprograms gene expression in muscle cells, promoting the proliferative capacity and myogenic differentiation of myoblasts. Brennan and colleagues (7) showed that TGF-β1 inhibited the transcriptional activity of myogenin without affecting its DNA binding affinity. Further, it has been shown that TGF-β1 targets the basic helix–loop–helix region of all MRFs, decreasing their DNA transcriptional activity without affecting their binding properties (20,36). Among a plethora of secreted soluble growth factors affecting muscle differentiation, TGF-β1 has been implicated as a potent repressor of the myogenic gene expression program as it initiates an intracellular cascade that prevents activation of genes associated with terminal differentiation in muscle cells (10,22).
Collaterally, we also observed that losartan-treated mice had lower amounts of type I collagen mRNA than nontreated mice. Since fibrosis is a major factor in muscle function impairment in DMD patients, our preliminary observation implies that losartan should be a promising agent to avoid or reduce the development of fibrosis in the skeletal muscles of these patients. More studies are needed to evaluate the actual clinical relevance of this first observation in dystrophic mice. However, the effect of losartan as an antifibrotic therapy to promote optimal skeletal muscle healing has already been demonstrated (2). Similar success in reducing residual fibrosis and enhancing myofiber regeneration, as well as success in improving function, has been observed in animal models, albeit after treatment with potentially harmful compounds (19,28). Another study demonstrated a lower amount of connective tissue following islet grafts with a chronic losartan treatment was due to the inhibition of the profibrotic properties of angiotensin II mediated through the AT1 receptor (30).
In conclusion, losartan was demonstrated in this study to have many potential beneficial effects on myoblast transplantation in dystrophic mice.
Losartan is approved for clinical use and is presently being used in DMD patients, including a clinical trial to evaluate its effects on muscle strength and fibrosis that was anticipated to begin in 2010 (MDA DMD Clinical Research Network). Thus, we can envisage a potential clinical combination of losartan and myoblast transplantation for the treatment of DMD in the future, aiming to improve the quality of life of these patients.
Footnotes
Acknowledgments
We thank Glenn E. Morris and Le Thanh Lam (MRIC Biochemistry Group) for providing the MANDYS104 (7F7) antibody. This work was supported by grants from the Muscular Dystrophy Canada, Amyotrophic Lateral Sclerosis Foundation, and the Canadian Institute for Health Research, and also by a grant from the Jesse's Journey Foundation for Gene and Cell Therapy of Canada.
