Abstract
Ex vivo expanded CD4+CD25highCD127- T regulatory cells (Tregs) are recognized as a promising candidate for immunosuppressive therapy in humans. However, due to the plasticity of Tregs lineage and artificial environment present during ex vivo expansion, Tregs easily lose suppressive activity. Here, we followed expanding CD4+CD25highCD127- Tregs and their naive (CD45RA+) and memory-like (CD45RA-) subsets in order to establish the best conditions of the expansion. We found that, regardless of the phenotype sorted, expanding Tregs were undergoing changes resembling homeostatic proliferation and transformed into effector memory-like cells which produced not only suppressive interleukin-10 (IL-10) but also IL-6, IL-17, and interferon-γ (IFN-γ). With the time ex vivo, Tregs were losing the expression of FoxP3 and suppressive activity both when stimulated and when at rest. The only variable that helped preserve suppressive abilities of Tregs was the limitation of the time of ex vivo cultures to 2 weeks only. According to our study, the highest number of highly suppressive Tregs could be yielded with CD4+CD25highCD127- Tregs cultured no longer than 2 weeks. Thorough quality check, preferentially with the assessment of FoxP3 expression and IFN-γ suppression assay, should be applied to assess suppressive activity of the cells.
Keywords
Introduction
CD4+CD25+ T regulatory cells (Tregs) are considered master regulators of immune tolerance. The cells have been found to play a crucial role in the tolerance to self, acceptance of bone marrow and solid organ allotransplants, and prevention from autoimmune diseases and allergies. Extensive studies on these cells discovered their potential to modulate and silence immune responses. The promise of safe immunosuppression with Tregs resulted in multiple reports on the influence of particular immunosuppressive drugs on the level and activity of Tregs (1,2,5,12,14,16,24,26,33,35,39). Progress in this area has made Tregs “now more than just a laboratory solution” (37). Transplant laboratories have developed several protocols aimed at ex vivo expansion of Tregs for clinical use [reviewed in (27,31)]. However, only few clinical trials in humans with expanded Tregs have started until now. The major obstacles for the translation of this therapy to the human settings are low numbers of these cells available from the body, controversies on the phenotype of Tregs, problems with the purity of sorted Tregs, and decreasing immunosuppressive activity of these cells with the time of ex vivo expansion (31).
Our laboratory applied successfully ex vivo expanded Tregs in the treatment of graft-versus-host disease (GvHD) (30). While it is still an ongoing project, we can already confirm usefulness of this method in the chronic form of GvHD. Nevertheless, preserving the suppressive effect of Tregs during ex vivo expansion requires several measures to be taken. Here, we compare and discuss several approaches tested in our lab during development of the procedure of ex vivo expansion of Tregs for the clinic.
Materials and Methods
Reagents
The following monoclonal antibodies conjugated with the following fluorochromes: fluorescein isothiocyanate (FITC), phycoerythrin (PE), peridinin-chlorophyll-protein complex (PerCP), PerCP-Cy5.5, PE-Cy7, allophycocyanin (APC), APC-Cy7, APC-AlexaFluor750, and PacificBlue were used in the flow cytometric studies (clones in parentheses): anti-CD3 (UCHT1), anti-CD4 (RPA-T4), anti-CD8 (SK1), anti-CD19 (4G7), CD14 (MφP9), anti-CD16 (3G8), anti-CD25 (M-A251), anti-CD127 (hIL-7R-M21), anti-CD45RA (HI100, L48), interferon-γ (IFN-γ) (B27), and 7-aminoactinomycin D (7-AAD) Via-probe (all items purchased from BDBiosciences, Poland), anti-CD62L (3B5) (Invitrogen, USA) with appropriate isotype controls. Immunomagnetic sorting of CD4+ T cells was performed with EasySep kit (StemCell Technologies, Canada). 7-AAD Via-probe was used 1 μl/1 × 106 cells together with other antibodies only in experimental and not therapeutic sorts in our laboratory. Foxhead box P3 (FoxP3) staining kit was supplied by ebioscience (USA). Tregs labeling with carboxyfluorescein succinimidyl ester (CFSE) was performed with CFDA kit (Invitrogen, USA). The levels of interleukins (IL) IL-6, IL-17, and IL-10 from supernatants were measured with flexset array (BDBiosciences, Poland). Unless otherwise specified, the assays with particular kits were performed according to the manufacturer's instructions.
Sorting and Cultures of Tregs
Thirty donations from either anonymous volunteers (n = 20) or peripheral blood stem cells donors (n = 10) were sorted to CD4+CD25highCD127- Tregs as previously described (30,31). Briefly, health status of all the donors was verified based on medical record, physical examination, and laboratory tests (18). One fourth to one half liter of peripheral blood was preserved during standard donation in the blood bank. Leukocyte buffy coats from the donations were presorted to CD4+ cells with negative immunomagnetic sorting, then stained with fluorochrome-conjugated mAbs and sorted with FACSARIA sorter (BDBiosciences, Poland) to CD3+CD4+CD25high CD127-doubletlineage-dead- “all Tregs (CD127-)” or subdivided into CD3+CD4+CD25highCD127-doublet-line age-dead-CD45RA+ “naive Tregs (CD45RA+)” and CD3+ CD4+CD25highCD127-doublet-lineage-dead-CD45RA- “memory Tregs (CD45RA-).” In addition, CD3+CD4+CD127+ doublet-lineage-dead- T effectors were harvested for control cultures and assays.
Sorted Tregs (1 × 105 per well) were then cultured under GMP conditions for maximum 3 weeks in 96-well U-bottom plates in RPMI-1640 medium supplemented with 10% of complement-inactivated autologous fresh plasma from donation with high concentration of interleukin 2 (1,000 U/ml, Proleukin, Chiron, USA) and anti-CD3/anti-CD28 beads (T cell Expander, Invitrogen, USA) in a 1:1 ratio. Passages of expanding Tregs were made every 2 days during the first week and every day during the second and third weeks of the culture. In addition, at the day +3, a small proportion of Tregs was washed out from the beads and left in RPMI-1640 with 10% of inactivated autologous plasma and 15 U/ml of IL-2 in order to follow Tregs in the resting conditions.
Quality Check of the Cultures
The quality of the cells was checked every 7 days using the samples of cells and supernatants taken from the cultures. The quality check consisted of FoxP3 staining (Fig. 1) suppression assays (Figs. 2 and 3), phenotype check (Figs. 4 and 5), and assessment of the cytokines (Fig. 6). Tregs were left for 3 days in medium without expander beads and IL-2 prior to setting up the assay in order to make Tregs quiescent. Two kinds of suppression assays were performed. In both assays samples of Tregs were mixed in various proportions with autologous effectors and stimulated.

The level of FoxP3 in the cultures of Tregs subsets. Levels of FoxP3-positive T cells in all Tregs (CD127-), naive Tregs (CD45RA+), memory Tregs (CD45RA-), and CD4+ T effectors (CD127+) are presented. The left column presents the levels immediately after sorting and in the cultures undergoing expansion and the right column in those at rest. All the cultures were tested every 7 days up to 3 weeks. The results throughout the figure are presented as medians (symbols inside the boxes), 20–80% percentiles (boundaries of the boxes) and minimum–maximum (error bars outside the boxes). Asterisks show the points that differ significantly from the postsort results in particular cultures (Friedman's ANOVA, p < 0.05).

IFN-γ suppression assay. Samples of Tregs from the cultures were cocultured with autologous effectors in different concentrations of effectors/Tregs (x axis) and then the cocultures were stimulated to produce IFN-γ. Immediately prior to the assay Tregs were stained with cell tracer CFSE in order to distinguish them from effectors and therefore it was possible to give separately the proportions of IFN-γ-positive Tregs and effectors at the end of the assay (examples of the dot-plots in the very left column; fluorescence of anti-IFN-γ mAb on the y axis with the cut-offs of positive signal based on isotype controls; CFSE signal on the x axis with effectors as CFSE-low cells and Tregs as CFSE-high cells; proportions of effectors/Tregs are given with numbers). The percentages of IFN-γ-positive effectors cocultured with Tregs from the cultures of all Tregs (CD127-), naive Tregs (CD45RA+), and memory Tregs (CD45RA-) are shown (y axis). The middle column presents the results of the assay with Tregs from the cultures undergoing expansion and the very right column with Tregs from resting cultures. All the cultures were tested every 7 days up to 3 weeks. The results are presented after subtraction of the values of the background from nonstimulated cultures. The results throughout the figure are presented as medians (symbols inside the boxes), 20-80% percentiles (boundaries of the boxes), and minimum–maximum (error bars outside the boxes). Lines with asterisks cover the cultures that differ significantly from the cultures of stimulators only (labeled 0:1; i.e., second column) (Kruskal-Wallis ANOVA, p < 0.05).

Suppression of proliferation assay. Samples of Tregs from the cultures were cocultured with autologous effectors in different concentrations of effectors/Tregs (x axis) and then the cocultures were stimulated to proliferate and the proliferation was measured with [3H]thymidine incorporation test. The intensity of proliferation (y axis) was assessed in the cocultures with Tregs from the cultures of all Tregs (CD127-), naive Tregs (CD45RA+), and memory Tregs (CD45RA-). The left column presents the results of the assay with Tregs from the cultures undergoing expansion and the right column with Tregs from resting cultures. All the cultures were tested every 7 days up to 3 weeks. The results are presented after subtraction of the values of the background from nonstimulated cultures. The results throughout the figure are presented as medians (symbols inside the boxes), 20–80% percentiles (boundaries of the boxes), and minimum–maximum (error bars outside the boxes). Lines with asterisks cover the cultures that differ significantly from the cultures of stimulators only (0:1) (Kruskal-Wallis ANOVA, p < 0.05).

The percentage of naive and memory cells in the cultures of Tregs subsets. Percentages of naive CD62L+CD45RA+ (very left column), central memory CD62L+CD45RA- (middle column), and effector memory CD62L-CD45RA- (very right column) cells in the cultures of all Tregs (CD127-), naive Tregs (CD45RA+), memory Tregs (CD45RA-), and CD4+ T effectors (CD127+) are shown. Corresponding example dot-plots are shown in Figure 5. The percentages were assessed immediately after the sorting, in the cultures undergoing expansion (open boxes) and in those at rest (shaded boxes). All the cultures were tested every 7 days up to 3 weeks. The results throughout the figure are presented as medians (symbols inside the boxes), 20–80% percentiles (boundaries of the boxes), and minimum–maximum (error bars outside the boxes). Lines with asterisks cover the cultures that differ significantly from postsort results (Friedman's ANOVA, p < 0.05).

The percentage of naive and memory cells in the cultures of Tregs subsets. Representative examples of the dot-plots used to assess the percentages of naive CD62L+CD45RA+, central memory CD62L+CD45RA-, and effector memory CD62L+CD45RA- cells from Figure 4 are shown. The cultures of all Tregs (CD127-), naive Tregs (CD45RA+), memory Tregs (CD45RA-), and CD4+ T effectors (CD127+) were assessed. The dot-plots immediately after the sorting, at +7 days, at +14 days, and at +21 days are shown from the cultures undergoing expansion. For comparison, the dot-plots from +21 days from the resting cultures are shown in the very right column. Numbers in the dot-plots represent the percentages of cells in particular quadrants.
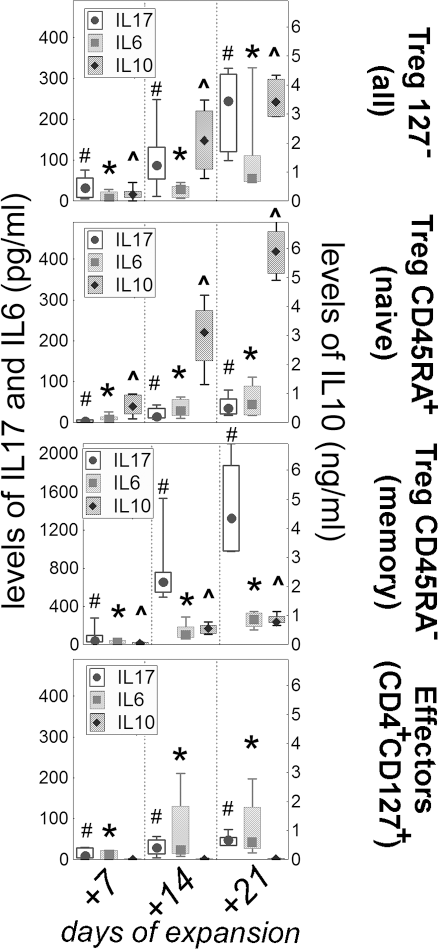
The levels of cytokines in the cultures of Tregs subsets. Levels of IL-17, IL-6 (left y axis) and IL-10 (right y axis) in the supernatants from the expanding cultures of all Tregs (CD127-), naive Tregs (CD45RA+), memory Tregs (CD45RA-), and CD4+ T effectors (CD127+) are presented. All the cultures were tested every 7 days up to 3 weeks. The results for IL-17 (circles), IL-6 (boxes) are shown on the left y axis (note different values for memory Tregs) and those for IL-10 (diamonds) are shown on the right y axis. The results throughout the figure are presented as medians (symbols inside the boxes), 20–80% percentiles (boundaries of the boxes), and minimum–maximum (error bars outside the boxes). Significant increase in the level of cytokines with the time of cultures is shown with symbols: # for IL-17, ∗ for IL-6, and ^ for IL-10 (Friedman's ANOVA, p < 0.05).
In the first assay (Fig. 2) stimulation consisted of 50 ng/ml of phorbol 12-myristate 13-acetate, 500 ng/ml of ionomycin (Sigma, Poland) and 2 μl/ml of cytokine leakage inhibitor GolgiPlug (BDBiosciences, Poland) for 5 h. Prior to the test Tregs were labeled with 5 μM CFSE (10 min in 37°C followed by extensive washing) in order to distinguish Tregs from effectors. After stimulation the cells were stained to CD3+CD4+IFN-γ+ phenotype with intracellular staining kit (BDBiosciences, Poland). The readout of the assay was the suppression of IFN-γ production by effectors.
In the second type of suppression assay (Fig. 3) the stimulation consisted of anti-CD3/anti-CD28 beads in 1: 1 ratio with effectors for 4 days. Readout of the test was the suppression of proliferation measured after incubation with [3H]thymidine (0.5 μCi/well) for the last 16 h.
Phenotype and cytokine checks consisted of the staining of CD3/CD4/CD45RA/CD62L receptors on the cells (Figs. 4 and 5) and assessment of the concentration of cytokines in the supernatants (Fig. 6) taken every 7 days from the cultures.
Ethics
All the procedures were approved by the Ethics Committee of the Medical University of Gdansk (approval No. NKEBN/458/2007 with amendment NKEBN/458-310/2008). Blood samples were only taken after informed consent from the patients was received.
Statistics
Data were computed using Statistica 8.0 (Statsoft, Poland). The analysis was based on nonparametric tests as indicated by data distribution. Dependent variables were assessed with Friedman's ANOVA with Wilcoxon test correction for multiple comparisons, and Kruskal-Wallis ANOVA was used for independent variables. A value of p < 0.05 was recognized as significant. For clarity of the text, the most crucial statistics only are presented.
Results
Tregs Lose FoxP3 Expression with the Time of Culture
In vitro culturing of Tregs was associated with time-dependent decrease in the level of FoxP3 (Fig. 1). The decrease was noted for all three phenotypes of Tregs examined in the study, both in expanded and resting cultures. The fastest decrease in the level of FoxP3 expression was noted in the cultures of memory Tregs (CD45RA), in which more than half of the cells were FoxP3- 1 week after the sorting. Compared with memory Tregs, the rate of decrease in the cultures of all Tregs (CD127-) and naive Tregs (CD45RA+) was slower. The decrease was comparable between these cultures and around 75% of the cells were still FoxP3+ 2 weeks after the sorting in both all Tregs (CD127-) and naive Tregs (CD45RA+). Importantly, the length of the cultures seemed to be the most important factor that contributed to the decrease in the level of FoxP3. Neither the age of donors nor the stimulation status affected the decrease in a significant way.
Tregs Lose Suppressive Capabilities with the Time of Culture
The decrease in the percentage of FoxP3+ cells was parallel to the suppressive capabilities of Tregs revealed in the functional assays performed as the quality checks of the cultures. Interestingly, the suppressive effects were clearer in the IFN-γ secretion assay (Fig. 2) than in the proliferation assay (Fig. 3). The suppression of IFN-γ production was noted for all three examined phenotypes of the Tregs in the assays performed 1 and 2 weeks after the sorting (Fig. 2). Tregs from the cultures of all Tregs (CD127-) and naive Tregs (CD45RA+) revealed comparable suppressive effect, while the effect of memory Tregs (CD45RA) was weaker. The suppressive effect disappeared in the majority of the cultures 3 weeks after the sorting. The loss of suppression was more prominent in the cultures undergoing expansion than in those in the resting state and the most significant decrease was noted for memory Tregs (CD45RA-).
In general, similar results were obtained in the proliferation assay (Fig. 3). Nevertheless, the suppression of proliferation was not revealed for Tregs from the cultures of some donors. The lack of suppression was associated with particular donors rather than with the phenotype of Tregs or the length of the cultures. In 6 out of 30 apparently healthy donors we could not see clear suppression in the proliferation assay. However, none of the available clinical features of these donors could be linked to the lack of suppression.
Tregs Change the Phenotype with the Time of Expansion
The majority of all Tregs (CD127-) immediately after the sorting were CD62L+ and belonged mainly to CD45RA+CD62L+ naive and CD45RA-CD62L- central memory subsets (Figs. 4 and 5). Additional sorting into naive Tregs (CD45RA+) and memory Tregs (CD45RA-) resulted immediately after the sorting in mainly CD45RA+CD62L+ naive phenotype in the former and CD45RA-CD62L+ central memory phenotype in the latter. In addition, small but significant proportion of the cells sorted as memory Tregs (CD45RA-) expressed CD45RA-CD62L- effector memory phenotype after the sorting (Figs. 4 and 5).
With the time of expansion the phenotype was evolving from naive, through central memory, into effector memory cells. It was the most prominent in naive Tregs (CD45RA+) in which a significant proportion of cells transferred into CD45RA-CD62L+ central memory compartment as early as within 1 week of expansion. Within the same time, around half of the memory Tregs (CD45RA-) transformed from mainly CD45RA-CD62L+ central memory cells into CD45RA-CD62L- effector memory cells. CD62L+ phenotype (naive or central memory) was relatively stable in the cultures of all Tregs (CD127-) and naive Tregs (CD45RA+) up to 2 weeks of expansion. At 3 weeks, in all examined cultures, the majority of the cells became CD45RA-CD62L- effector memory cells (Figs. 4 and 5).
Tregs Change the Pattern of Secreted Cytokines with the Time of Expansion
The levels of examined cytokines were gradually increasing with the time of expansion in all cultures (Fig. 6). Interestingly, both inflammatory IL-17 and IL-6 and anti-inflammatory IL-10 were increasing in the cultures of all Tregs (CD127-). Of note, the fold increase in the level of IL-10 was significantly higher than the fold increase in the levels of IL-6 and IL-17. At the same time, the cultures of naive Tregs (CD45RA+) were characterized mainly by the increase in IL-10, while those of memory Tregs (CD45RA-) by the increase in IL-17. No such trends were noted in any of the cultures of resting cells, where the levels of the cytokines were comparable to the first week of expansion during the entire experiment (not shown).
Initial Number of Sorted Tregs Is Crucial for the Final Yield
In all the cultures of Tregs, the mode of proliferation was exponential and fold increase in the numbers was comparable between the cultures (Fig. 7). Nevertheless, absolute numbers of Tregs in particular points of expansion differed due to the fact that the yield was highly correlated with the numbers of cells in the starting population, immediately after the sorting. When half of CD4+ T cells from the buffy coat was sorted to all Tregs (CD127-) and the other half to naive (CD45RA+) and memory (CD45RA-) subsets of all Tregs, significant differences in the number of expanded Tregs were noted. At 2 weeks after the sorting, the very last moment when the quality of Tregs was relatively preserved, all Tregs (CD127-) were the only culture in which the number of Tregs was sufficient to be used in the therapy. Similar yield in the subsets of naive Tregs (CD45RA-) and memory Tregs (CD45RA-) were achieved as late as at 3 weeks, when the suppressive potential of these cells was already deteriorated substantially.

The dynamics of expansion in different subsets of Tregs. CD4+ T cells were divided into two halves and one was sorted to CD3+CD4+CD25highCD127-doublet-lineage-dead- all Tregs (CD127-) and the other half to the naive (CD45RA+) and memory (CD45RA-) subsets of all Tregs. After the sorts, Tregs subsets were undergoing expansion and the yield of the cultures (number of the cells—log scale on y axis) was measured every 7 days. The results throughout the figure are presented as medians (symbols inside the boxes, and numbers above the error bars), 20–80% percentiles (boundaries of the boxes), and minimum–maximum (error bars outside the boxes). The differences between particular points of the three cultures were confirmed (Kruskal-Wallis ANOVA, p < 0.05). Compared to naive Tregs (CD45RA+) and memory Tregs (CD45RA-) cultures, the number of cells was significantly higher in the cultures of all Tregs (CD127-) in each of the four time points analyzed: ∗ immediately postsort, # +7 days, $ +14 days, and ^ +21 days.
Discussion
Ex vivo expansion of Tregs with preserved suppressive abilities is a “sine qua non” requirement to apply these cells in the therapy. According to our results, both suppressive phenotype, represented by FoxP3 expression, as well as suppressive function, confirmed with functional assays, decrease with the time of expansion. The length of the culture was found to be crucial variable to keep the cells “regulatory.” The timing around 2 weeks of expansion was confirmed to be the best to receive good yield of the cells and not to spoil their suppressive abilities. Manipulations with the phenotype or other interventions during the expansion could not improve the suppression exerted by cultured Tregs.
The most important and new message from our study is the fact that suppressive activity of Tregs decreases with the time of ex vivo expansion regardless of the starting phenotype sorted. The majority of studies on expansion of Tregs for the clinic look for the most suppressive phenotype to sort, which is then believed to be preserved during expansion (10). There is currently consensus that the best candidates for expansion are naive Tregs (CD45RA+), which have been described as apoptosis resistant, proliferating vigorously after stimulation of their TCR, and, most importantly, giving stable FoxP3+ lineage after expansion. By contrast, CD45RA-Tregs, referred to as “memory” or “effector” cells, are hyporesponsive and apoptotic prone after stimulation (4,6,10,13,17). The term “memory Tregs” has been recently questioned in animal models in favor of “effector Tregs” as the turnover of CD45RA- Tregs is rapid and there is no clear evidence of recall and memory responses in these cells (21). Nevertheless, the naive, central, and memory approach (22) adapted in our study seems to be valid for expanded Tregs is some aspects. The analysis of expression of CD62L together with CD45RA revealed that Tregs had been switching the phenotype from “CD45RA+CD62L+ naïve” through “CD45RA-CD62L+ central memory” to “CD45RA-CD62L- effector memory” like other kinds of lymphocytes when stimulated (34). Importantly, suppression in the functional tests was fading with more advanced phenotype of Tregs in the cultures, mainly in the cultures of memory Tregs (CD45RA-) and, regardless of the starting population, in all cultures beyond 2 weeks of expansion (i.e., when the memory phenotype had been prevalent). This is of special interest for “CD45RA-CD62L- effector memory Tregs,” which are believed to contain nonsuppressive cells (17). Indeed, it seems that no expression of CD62L may easily indicate the presence/contamination or de novo generation of effectors in memory Tregs. The correlation between expression of CD62L and suppression has been already described (6), but our study put it in the context of Treg subsets under expansion. It may have additional consequences in vivo as CD62L is a homing receptor and its application in phenotypic studies of memory cells was primary intended to show different homing potential of central and effector memory subsets (22).
Lost abilities of suppression in memory Tregs are also associated with unstable expression of FoxP3. Compared to naive Tregs, memory Tregs were proved to be inefficient due to low and quickly fading expression of Fox3 factor. This protein is crucial for the suppressive activity of Tregs and currently the most reliable phenotypic marker of these cells (11). It has been proposed that the difference in expression of FoxP3 lies in varying demethylation level of CpG motifs within the foxp3 gene locus, the so-called “Treg-specific demethylated region” (TSDR) (19). Naive Tregs were found to keep this region demethylated and therefore expressing FoxP3 in a sustained way, while memory Tregs were characterized by gradual methylation and therefore lost suppressive abilities (9). Indeed, our study confirmed fast deterioration of suppressive activity in memory Tregs defined as decreasing expression of FoxP3 and lost suppression exerted in functional assays; however, similar process was seen also in the cultures of naive Tregs, which is in contrast with previous studies (8,9,20). It seems that beyond 2 weeks of expansion none of the phenotypes guaranteed sufficient suppressive activity of expanded Tregs. Interestingly, repetitive stimulation necessary to expand Tregs revealed only slight effect on suppressive abilities of these cells as both stimulated and resting Tregs lost the expression of FoxP3 with almost the same dynamics. This finding confirms that the environment of the body (lymphatic tissue?) is necessary in the homeostasis of Tregs and in vitro conditions are not sufficient to keep them stable. From the practical point of view, the limit of 2 weeks in the culture is important as beyond this time the cells, regardless of the starting phenotype, not only lost suppressive abilities but also tended to be proinflammatory cells with expression of IL-6, IL-17, and IFN-γ (still expressed high levels of IL-10). In fact, it should not be surprising as Tregs are not exceptional and like other CD4+ T cells may transform to other phenotypes, the effect called plasticity (36). The proof of principle of this statement comes also from our attempts to save the patient with acute GvHD, who responded well to Tregs expanded for 1 and 2 weeks and revealed almost no response to Tregs expanded for 3 weeks (30).
In our opinion, the studies on expansion of Tregs for the clinic overlook also another important biological effect, which is homeostatic-like proliferation of expanding Tregs. Although homeostatic proliferation is referred to in vivo regulation of the pool of lymphocytes, mechanisms governing it in the body may be valid also in vitro and may explain rapid transition of the phenotype of Tregs in cultures. At first, Tregs were recognized mainly as guardians against homeostatic proliferation of effector cells (25) and initial studies have denied that Tregs are able to proliferate homeostatically on their own due to low/no expression of IL-7 receptor (CD127) (15,23). Nevertheless, it is rather unlikely as Tregs still respond to IL-15 and some of them express CCR7 receptor for CCL19, CCL21 chemokines (3,28). Indeed, robust expansion of Tregs after nonspecific activation of costimulatory receptors resembles homeostatic proliferation in lymphopoenia in vivo (7). The most important feature making ex vivo expansion similar to homeostatic expansion is—revealed in our study—a switch from naive to more differentiated memory-like subsets in the absence of antigenic signals, notably in the cultures of naive Tregs. The switch was significantly higher in expanding cultures than in those resting, which additionally highlights the role of proliferation stimulated with nonantigenic stimuli. In addition, despite regular passages of proliferating cells with fresh media, we detected increasing concentration of cytokines with the time of the cultures. Such increased production of cytokines has been also attributed to homeostatic proliferation (7). Again, Tregs appeared to be not exceptional and underwent homeostatic-like proliferation like other T cells. This finding prompted us to reduce the ratio of stimulating beads to Tregs in the cultures to 1:1, in order to reduce the risk of fast conversion of Tregs to memory-like cells which probably secrete IL-6 or IL-17. Previously, we were using 2:1 ratio (31) and there was even 4:1 ratio reported by other groups (10). In our hands, reduced number of the beads made all presented cultures of CD127- and CD45RA+ Tregs more stable for the first 2 weeks of expansion. In theory, the price for that is the reduced yield from the cultures but there is no question that it is better to gain somehow reduced number of suppressive cells than high number of useless effectors. The advantage of controlled proliferation of Tregs has been also found by researchers using rapamycin during expansion. This drug is much more suppressive for effectors than for Tregs, which results in selective promotion of Tregs under expansion (20). Again, at least in our hands, the number of Tregs after expansion with rapamycin was reduced as compared to those expanded without this drug (our unpublished data).
The problem of the yield presents another important question—which phenotype should be sorted? As already mentioned, naive Tregs are considered the best candidates. However, it might be impractical to expand them for several reasons. First, naive CD45RA+CD62L+ Tregs constitute only around one third of all Tregs, which substantially reduces initial yield from the sort. Two weeks of expansion, which has been proved as limiting factor for the cultures, did not guarantee that the number of expanded naive Tregs reached plateau of the exponential growth shown in Figure 7. Hence, it may be problematic to obtain enough naive Tregs (CD45RA+) for the adoptive transfer to the patient at the end of expansion. It might be especially important in the therapy of children (e.g., in diabetes type 1) where the volume of blood available is often very low. At the same time sorting of all Tregs (CD127) guaranteed good yield at 2 weeks of expansion (naive Tregs could reach this number only at 3 weeks, when their suppressive ability was already diminished significantly). It has to be highlighted at this point that, compared to this report, our real therapeutic sorts with all Tregs (CD127) phenotype give the yields comparable to other reports, which is enough to administer up to 5 × 106 Tregs per kg body weight of the patient (30,31). Lower numbers in this report come from the fact that for the purpose of this study CD4+ T cells were split and sorted to several subsets which decreased initial numbers for expansion. In addition, at day +3 a portion of the cells was taken away from the cultures to set up resting conditions. Then, all cultures were extensively sampled and checked for quality during expansion, which also reduced the numbers. Even without these maneuvers, robust expansion of sorted naive Tregs (CD45RA+) was problematic, mainly due to low initial numbers related to their proportion in peripheral blood. Interestingly, the majority of sorted all Tregs (CD127) belonged to CD45RA+ CD62L+ naive and CD45RA-CD62L+ central memory subsets, which seemed to slow down further switch to effector memory compartment. In contrast, naive Tregs (CD45RA+) very quickly transformed from exclusively naive phenotype to the mixed naive/central memory phenotype within 7 days of expansion. Importantly, from the day +7, the percentage of naive and central memory cells were comparable between the cultures of naive Tregs (CD45RA+) and all Tregs (CD127-); however, one has to remember that central memory cells in the cultures from naive Tregs were in fact memory-like cells generated, the most likely, in a homeostatic-like manner. Such a notorious memory-like phenotype developed as a result of homeostatic proliferation was described as a barrier to tolerance in the case of effector T cells (38). Efficient expansion of small subsets should be investigated further as a frequency of antigen-specific Tregs, which are a very promising alternative to polyclonal Tregs, is even smaller than that of naive Tregs (29).
Finally, we would like to highlight the necessity of adequate quality check of the cultures. According to our results, classical check based on the suppression of proliferation may not be useful in the surveillance of Tregs under expansion as in some donors 2-3 days of the rest prior to commencing the suppression assay was not enough to completely stop the proliferation of Tregs tested. If this happened, the test was not reliable due to the contribution of proliferating Tregs supposed to inhibit effectors. Hence, we came back to our earlier works in which the readout was the inhibition of IFN-γ production (32). This test was much more practical as it did not require complete quiescence of the tested Tregs and the readout was hours and not days after commencing. Improving the test even further, we distinguished Tregs by staining them with CFSE which allowed us to assess separately production of IFN-γ by effectors and Tregs. It is easy to make an error without this step as adding of IFN-γ- Tregs to effectors poses a threat of underestimation of the percentage of effector cells producing IFN-γ in the test. Simply, IFN-γ- Tregs increases a proportion of negative cells, even if the production of the cytokine by effectors is not inhibited.
Our study highlighted the limitations of artificial environment that has to be taken into account when ex vivo expanded Tregs are prepared for cellular therapy. The cells seemed to be not exceptional and were undergoing homeostatic-like changes, in a way similar to other T cells. The phenotype of Tregs in the culture in vitro was a subject to plasticity and therefore careful monitoring was necessary to follow their suppressive function. The only variable which helped us to prepare high number of highly suppressive Tregs for the therapy was the time of expansion limited to around 2 weeks only.
Footnotes
Acknowledgments
This work was supported by the Ministry of Science and Higher Education, Poland (grant No. NN402 353038 to PT). The authors declare no conflicts of interest.
