Abstract
Bone morphogenetic protein-2 (BMP-2) has been commercially approved by the Food and Drug Administration for use in bone defects and diseases. BMP-2 promotes osteogenic differentiation of mesenchymal stem cells. In bone tissue engineering, BMP-2 incorporated into scaffolds can be used for stimulating bone regeneration in organoid construction, drug testing platforms, and bone transplants. However, the high dosage and uncontrollable release rate of BMP-2 challenge its clinical application, mainly due to the short circulation half-life of BMP-2, microbial contamination in bone extracellular matrix hydrogel, and the delivery method. Moreover, in clinical translation, the requirement of high doses of BMP-2 for efficacy poses challenges in cost and safety. Based on these, novel strategies should ensure that BMP-2 is delivered precisely to the desired location within the body, regulating the timing of BMP-2 release to coincide with the bone healing process, as well as release BMP-2 in a controlled manner to optimize its therapeutic effect and minimize side effects. This review highlights improvements in bone tissue engineering applying spatiotemporal and controlled BMP-2 delivery, including molecular engineering, biomaterial modification, and synergistic therapy, aiming to provide references for future research and clinical trials.
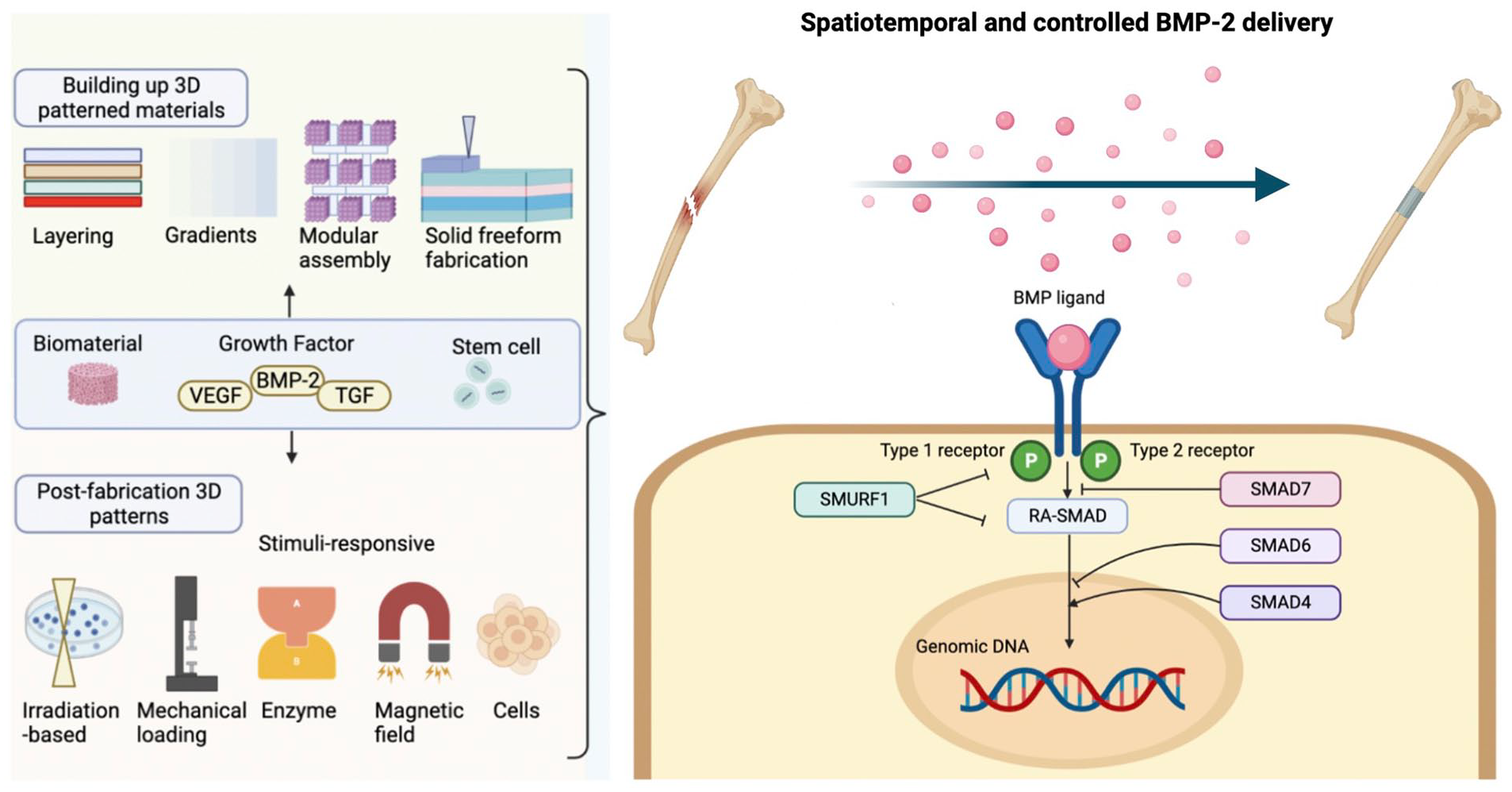
Introduction
Bone morphological protein (BMP), a cytokine of the transforming growth factor β (TGF-β) superfamily, possesses great potential to induce bone repair and regeneration. BMP has about 40 subagents that have different functions. Among them, the application of BMP-2 in clinical trials has been approved by the Food and Drug Administration (FDA) 1 . BMP-2 has been widely used in bone treatments in clinical trials, including repair of femoral head necrosis, lumbar fusion, and acute tibial fractures2,3. The function of BMP-2 is mediated by type I BMP receptor (BMPR-1), type II BMP receptors (BMPR-2), and their downstream molecules Smad1, 5, and 8 (Fig. 1) 4 . Phosphorylated Smad1, 5, and 8 proteins form complexes with Smad4, and then be transported into the nucleus to interact with BMP-2 related transcription factors, including Runt-related transcription factor 2 (Runx2) in osteoblasts 5 . During the early healing phase, BMP recruits mesenchymal stem cells (MSCs) to promote the differentiation into chondroblasts and osteoblasts. Then, continuous chondrogenesis results in a soft callus that temporarily stabilizes the fracture and promotes endochondral bone formation.
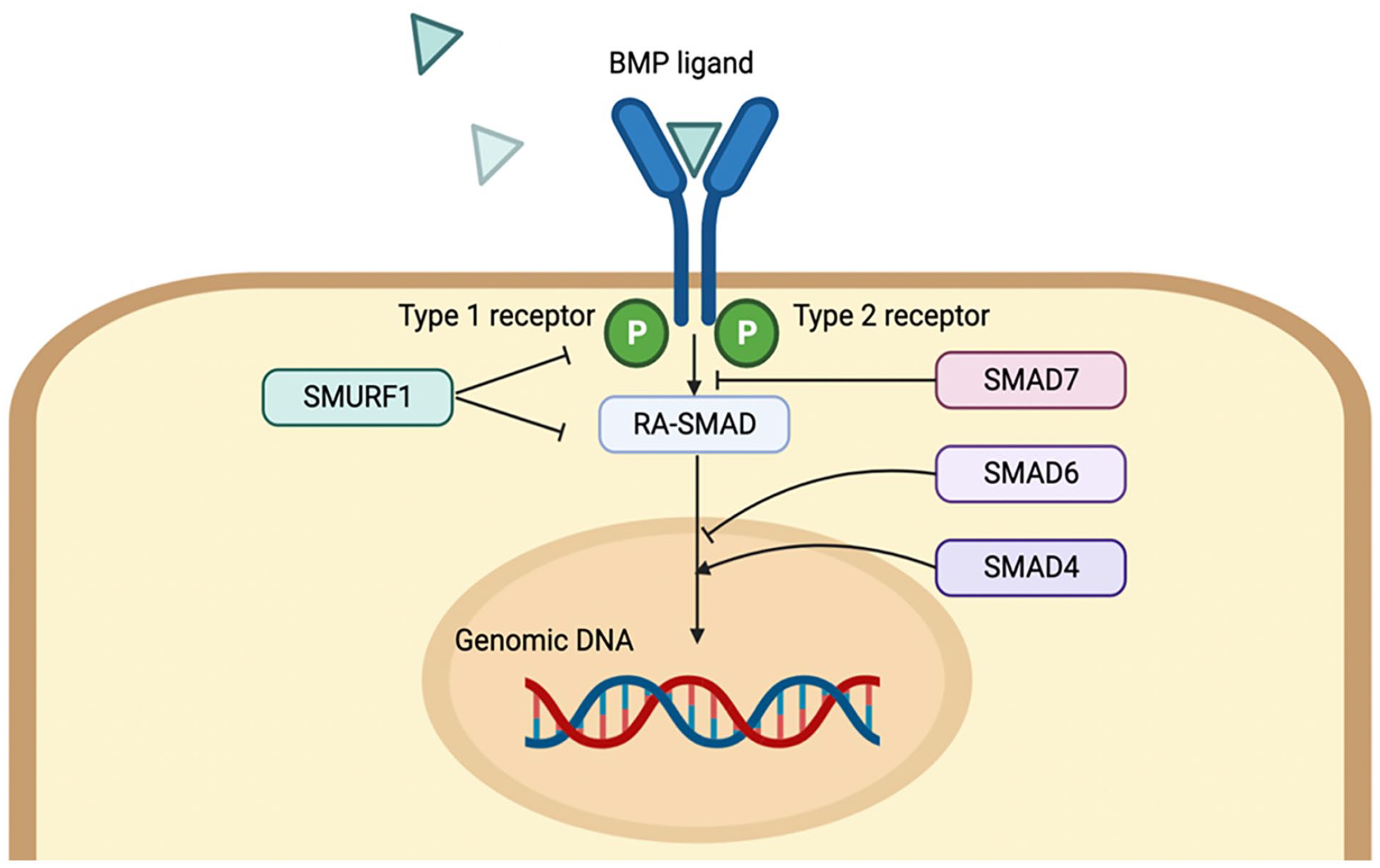
BMP signaling pathway.
Although BMP-2 has remarkable effects on osteogenesis, the application has several challenges including short circulation half-life and overdosage use. First, BMP-2 has rapid in vivo clearance because its short half-life is only 7–16 min in the physiological environment 6 . Second, the FDA-approved concentration of 1.5 mg/ml for human use was based on non-human primates (0.75–2.0 mg/ml), which may be overdosage 7 . The overdosage use of BMP-2 causes various side effects such as ectopic bone formation, tissue swelling, postoperative inflammation, osteoclast-mediated bone resorption, and improper adipogenesis1,8. For example, off-label use of recombinant human BMP-2 in posterior interbody fusion resulted in nerve root compression and postoperative radiculitis 9 . Ectopic bone formation and bone cysts in neuroforaminal and soft tissues have been observed after lumbar fusion with BMP-2 10 . Moreover, overdosage BMP-2 can induce cancers. The 40 mg or more dose of BMP-2 in anterior cervical fusion surgery was found to highly correlate with the occurrence of cancer11,12. To conclude, the short circulation half-life and overdosage use of BMP-2 need to be addressed for clinical application.
In bone tissue engineering (BTE), combining interdisciplinary knowledge of stem cell biology, biomaterials, drug delivery, and molecular engineering, has shown great potential for spatiotemporal and controlled BMP-2 delivery. First, BMP-2 activates various important signaling pathways for osteogenesis, cell survival, and apoptosis 13 . Thus, the BMP-2 signaling pathway can be manipulated to enhance its effectiveness, which involves designing biomaterials and gene therapy, to control how BMP-2 is delivered and activated in vivo. Moreover, the activation of BMP-2 should be stabilized to ensure BMP-2 remains active and effective for a long period, avoiding rapid degradation or inactivation. Finally, stabilizing the BMP-2 activity can more effectively promote the formation of new bone tissue, which is particularly beneficial in treatments involving bone fractures, spinal fusions, and other orthopedic procedures 13 . As for the modification, the BMP-2 protein and its activators and inhibitors can be targeted by molecular engineering. Second, BMP-2 can not only affect the proliferation and differentiation of progenitor cells but also show synergistic effects with other growth factors. Therefore, combining BMP-2 with different growth factors can strengthen the osteoinductive capability of BMP-2 on stem cells. Third, regulating the chemical and physical properties of extracellular scaffolds can reduce the burst release and supraphysiological levels of BMP in the microenvironment 14 . For instance, the polyetheretherketone cages and absorbable collagen sponge have been applied to block the exposure of BMP-2 to surrounding tissues, showing reduced side effects 15 . Additionally, the demineralized bone matrix around the BMP-2 sponge has been reported to reduce the complications compared to the use of the BMP-2 directly 12 . Above all, the dosage and localization of BMP-2 are closely related to the treatment results and complications. Here, cutting-edge BTE strategies to achieve spatiotemporally controlled release of BMP-2 have been discussed, aiming to provide a reference for bone repair and regeneration.
Bone Diseases and Regenerative Proposals
Bone occupies approximately 15% of human body weight. As a highly metabolic active connective tissue, bone has the capability of resorption and remodeling 16 . However, large bone defects, defined as more than 2.50 cm in critical size, cannot be healed fully and require regenerative and reconstruction treatment 17 . For example, trauma, congenital anomalies, age-related diseases, and musculoskeletal tumors are common pathotypes 18 . Nowadays, bone disease incidence is increasing mainly because of the sedentary lifestyle and rising average age 19 , which has become a significant socio-health problem.
BTE is an interdisciplinary strategy that manipulates biomaterial scaffolds, biochemical factors, and cells in vitro to make a graft for implantation. First, biomaterial scaffolds with porous microstructure possess high biocompatibility, mechanical properties, and biodegradability20,21. Thus, the scaffolds can mimic the extracellular matrix, providing a regenerative microenvironment for osteogenesis, vascularization, and nutrient transportation. Second, osteoinductive growth factors including BMP-2 regulate cell metabolism to promote the growth of bone and blood vessels20,22. The controlled delivery of growth factors in scaffolds can enhance progenitor cell proliferation, differentiation, and migration both in vitro and in vivo. Third, the BMP-2 protein and its signaling pathway can be modified to increase the stability and metabolism of BMP-2 in the process of bone regeneration. Overall, current trends of BTE are reducing morbidity on the donor site, enhancing the geometric compatibility of the graft with the recipient site, and addressing constraints on graft volume. Various BTE research explored the effects of protein modification, molecular engineering, and delivery system optimization on the BMP-2 delivery, to enhance the therapeutic effects of bone diseases. To conclude, the development of novel strategies for the spatiotemporal and controlled delivery of BMP-2 in BTE aims to improve the efficiency and safety of BMP-2 therapy. By leveraging advanced materials, nanotechnology, and innovative delivery methods, researchers hope to enhance the healing process and achieve better clinical outcomes of bone healing and regeneration.
BMP-2 Delivery System
The physiochemical and biological properties of BMP-2 motivate the need to design scaffolds that maintain BMP-2 bioactivity and enhance its retention at implant sites. The delivery system mainly includes binding and non-covalent binding 23 . First, the covalent binding of BMP-2 protein to scaffolds by coupling amine functional groups can enhance cell adhesion and differentiation in culture substrates 24 . However, this method is time-intensive and cost-intensive. Due to the difficulty in controlling the modification site, the active site of the BMP-2 may be obstructed, hindering the biological activity 25 . Second, the non-covalent incorporation of BMP-2 into scaffolds contains electrostatic interactions, hydrophobic interactions, hydrogen bonding, and Van der Waals forces 26 . The disadvantage is that it may cause an initial uncontrolled burst release, resulting in super-physiological levels of growth factor in the microenvironment and systemic abnormalities such as ectopia bone formation, osteoclast-mediated bone resorption, mineral deposition in muscle tissue, and inappropriate lipogenesis27,28. To address these problems, pretreatment with biomaterials and pharmacological agents to control the time and speed of the release of BMP-2 can improve the results of bone regeneration and repair.
Three-Dimensional (3D) Bioprinting
Three-dimensional (3D) bioprinting promoted the development of BTE by enabling the free-form fabrication of cell-laden hydrogel scaffolds with controllable features and interconnected pores. This technique uses biomaterials to produce elaborate 3D structures with specific shapes and features for clinical implantation 29 , showing great potential in the delivery of BMP-2.
Hydrogels are good candidates for bio-ink due to good biocompatibility and plasticity30,31. For example, alginate is a natural biopolymer. In the presence of divalent cations such as calcium, alginate is easily cross-linked by ions to form stable hydrogels 32 . Although alginate bio-inks lack the ability of bone conductivity and bone inductance, BMP-2 incorporation can enhance bioactivity. A novel bio-ink using BMP-2-loaded poly lactic-co-glycolic acid (PLGA) nanoparticles and alginate has been formulated 33 , showing enhanced mechanical properties and printability. Moreover, 30 mg/ml PLGA nanoparticles and 3% w/v alginate were suitable parameters for 3D bioprinting concerning printability and stability. BMP-2-loaded PLGA nanoparticles displayed sustained release in vitro for up to two weeks. As for the osteogenesis efficiency, the nanoparticles showed enhanced calcium deposition, alkaline phosphatase activity, and gene expression of osteogenic markers in MSCs. Additionally, a projection stereolithographically fabricated hydrogel scaffold has been bio-printed to encapsulate BMP-2-engineered human MSCs 34 . MSCs showed sustained expression of BMP-2 and differentiation into osteogenic lineages, without extra BMP-2 supplementation. As for in vivo evaluation, an intramuscular implantation model was utilized in severe combined immunodeficiency mice. Results revealed rapid bone formation, and mature trabecular bone structures accompanied by substantial vascularization 14 days after implantation. In a word, these findings highlight the applications of 3D bioprinting for the customized treatment of bone defects.
Scaffold Modification
In BTE, scaffolds play a crucial role by providing a 3D environment that mimics the extracellular matrix to recruit and direct osteogenic cells. The biocompatibility and biochemical properties of scaffolds significantly influence cell survival, infiltration, and osteogenesis. Various biomaterials can be utilized to incorporate BMP-2 into the scaffold, including natural polymers, synthetic polymers, bioceramics, and nanomaterials. To achieve spatiotemporally controlled release of BMP-2, variable modifications have been considered to improve the mechanical strength, osteoconductive capacity, and osteoinductive capacity of the biomaterials, such as mineralization, chemical functionalization, surface modification, and covalent functionalization via various linkers 35 .
Natural polymers
Inspired by the composition of the extracellular matrix, collagen was one of the first natural polymers applied for BMPs delivery. It has been considered as the gold standard carrier for BMPs approved by FDA to treat long bone fractures, spinal fusion and periodontal regeneration 36 . Currently, novel natural polymers have been characterized and detected in the application of BMP-2 delivery in BTE.
Recent studies reported that Heparin, a common glycosaminoglycan, is a natural polymer that can form a reversible combination by electrostatic interaction to adsorb positively charged BMP-2 37 . Heparin has a dual effect on the osteogenic activity of BMP-2. A low concentration of heparin was effective on bone regeneration, but a high concentration of heparin showed inhibiting effects 38 . Prolonged culture with heparin can stimulate BMP-2-induced osteogenesis via the downregulation of BMP-2 antagonists and inhibitory regulator Smads 39 . As for BMP-2 delivery, the heparin chains have been added into fibrin 40 , collagen 41 , alginate 42 , and PLGA scaffolds 43 . However, the noncovalent adsorption of heparin onto collagen sponge scaffolds did not enhance BMP-2–mediated bone repair in vivo 41 . Therefore, covalent heparin immobilization strategies, including 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide/N-hydroxysuccinimide coupling and photo-cross-linking were used 44 . While these strategies have improved BMP-2–mediated bone formation in vivo, the dose dependency of heparin adding to scaffold on bone formation did not be detected. Based on these, heparin has been modified with methacrylamide and turned into heparin microparticles (HMPs) to adsorb BMP-2 37 . The results showed that HMPs had a strong effect on BMP-2 adsorption and sustained release. Every 0.8 mg HMP can absorb 1 mg BMP-2. Later, BMP-2-loaded HMPs with arginine-glycine-aspartic acid-activated alginate hydrogels were generated to cure the 8-mm femur bone defect. The patients showed successful union without obvious ectopic bone formation 45 . However, heparin is an anticoagulant drug in clinical use and may cause uncontrolled bleeding and even death. Currently, the side effects of heparin microspheres on hematoma require further research, which is an unavoidable problem in clinical applications. Taken together, heparin increases BMP-2 retention in vivo, improves the spatial localization of bone formation in large bone defects, and reduces heterotopic ossification. Further study should balance the strong affinity interactions between heparin and BMP-2 to improve BMP-2 delivery.
Alginate is a polysaccharide derived from algae, which is widely used as a natural polymer for cell encapsulation in tissue engineering 46 . Alginate possesses favorable properties with cytocompatibility, adhesion, and degradability. In addition, alginate can absorb positively charged BMP-2 through electrostatic force, and then release it gradually. Alginate microbeads have demonstrated their effectiveness as carriers for BMP-2, significantly increasing ALP activity in mesenchymal stem cells in vitro over 14 days, and promoting bone formation in both ectopic and calvarial defect sites in animal models 47 . Alginate has been modified with γ-ray irradiation 48 , but about 98% of BMP was released within the first week. Additionally, the alginate has been oxidated but failed to have sustained release of BMP-2 49 . Later, BMP-2 was encapsulated with alginate in microspheres, and the rats with 5-mm skull defects recovered in 2 months 47 . Although BMP-2 was released 70% to 80% in 7 days, the loading capability was greater than in the previous two experiments. To improve the retention of BMP-2, alginate can work with other biomaterials, such as collagen 50 , calcium-based cement 51 , nano-silica 52 , nano-hydroxyapatite 53 , saponite 54 , and polycaprolactone scaffold 55 . Recently, optimization of the BMP-2-alginate delivery system has been reported. Due to poor interaction between BMP-2 and alginate, BMP-2 embedded in alginate saline gel is prone to rapid diffusion and cannot maintain long-term activity 56 . It has been reported that a mixture of alginate and heparin hydrogels could bind to BMP-2 45 . Furthermore, alginate has been chemically sulfated to produce alginate sulfate, a structural and functional mimic of heparin which can strongly interact with BMP-2 57 . As a result, the modified scaffold maintained the long-term activity of BMP-2 for BTE. The proliferation and phenotypic maintenance of bovine chondrocytes were significantly improved in hydrogels containing alginate sulfate compared to alginate saline gel controls 58 . In conclusion, alginate can be utilized as an effective BMP-2 delivery vehicle in BTE.
Chitosan is a biocompatible and biodegradable polysaccharide derived from chitin, which has been considered a good candidate for bone grafts 59 . The presence of amino and hydroxyl groups of chitosan improves the absorption ability of many materials such as drugs, ions, and growth factors 60 . These groups are also modifiable positions to link extra functional groups to further enhance the covalent adsorption. For example, chitosan has been modified with methacrylate, thus, it can be transformed into hydrogel under the reaction of visible light. The methacrylate-modified chitosan was applied to deliver BMP-2 61 . Results showed that about 30% of BMP-2 was released on the first day, and 70% in the later week. As a result, the 3-mm defect of the rat tibia could be recovered in 4 weeks, suggesting the potential role of chitosan delivery of BMP-2 in bone repair. Moreover, 2-N,6-O-sulfated chitosan (26SCS) showed an immunomodulatory role in the BMP-2-induced osteogenesis. The modified chitosan induced beneficial immune microenvironments, enhanced the relationship between immune cells and stem cells, and promoted the osteogenic differentiation of stem cells 62 . As for the mechanism, the 26SCS-triggered immune microenvironment promoted the chemotaxis of bone marrow stromal cells through upregulated BMPR-IA expression and amplified BMP-2/Smad signaling. Furthermore, the immune microenvironment had a positive effect on the pro-angiogenic effect of bone marrow-derived mesenchymal stem cells (BMSCs). Therefore, modifications of chitosan are significant for constructing a profitable microenvironment to enhance BMP-2-triggered osteogenesis.
Synthetic polymers
Synthetic polymers have received great attention in the last decades because they can combine various properties of individual polymers, including stability, biocompatibility, and mechanical strength 63 . Compared to natural polymers, synthetic polymers can avoid the potential risks of transferring diseases from animals, immunogenicity, or inflammation. For example, PLGA has good biocompatibility and biodegradation and has been approved by the FDA 64 . PLGA possesses great potential to deliver BMP-2 in BTE. For example, BMP-2 can be absorbed in the pores between PLGA and polyelectrolyte multilayer fabricated by 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide and N-hydroxysulfosuccinimide reaction 65 . Results showed that the PLGA/PEG scaffold loading BMP-2 reduced the hematoma complication compared to the direct use of BMP-2. Besides, PLGA-multistage vector composite microspheres (PLGA-MSV) were applied to transport BMP-2, showing enhanced osteoinduction in BMSCs in rats 66 . However, the manufacturing operation of the PLGA-MSV may be toxic in vivo due to the toxic solution. To overcome this limitation, the electrospinning technique provides a safe opportunity to produce PLGA nanofibers. For instance, electrospinning can be used to combine PLGA nanofibers with BMP-2, achieving the sustainable release of BMP-2. However, the osteoinduction level in vivo needs to be further verified 67 . Besides, high voltage during electrospinning could damage the BMP-2 and PLGA scaffold and then decrease the absorption dosage of BMP-2. These results suggested that PLGA can be applied to construct BMP-2-loaded composite scaffolds that had the potential to be used as a bone implant, as osteogenic ability was promoted by the prolonged release of BMP-2.
Bioceramics
The carrier of BMP-2 with calcium-phosphate-based materials was inspired by the natural system of bone. Hydroxyapatite occupies around 70% mineral component of teeth and bones and has been widely used as bone scaffolds, implant coatings, and drug delivery in bone regeneration 68 . Because of its osteoconductive capacity, and physicochemical and biological properties, hydroxyapatite induces the migration of host bone-forming cells into porous scaffolds, enhancing the osteogenesis process63,69. In the application of BMP-2 delivery, the micro- or nanostructure of hydroxyapatite has shown improved performance in delivering BMP-2. For instance, 100-µm nanostructured microspheres have been applied in controlled novel delivery of rhBMP-2, which showed significantly enhanced capacity to reconstitute radial bone defects in rabbits and quick and complete new bone formation within 12 weeks compared to hollow hydroxyapatite microspheres without BMP-2 or soluble BMP-2 without a carrier 70 . Thus, the hollow hydroxyapatite microspheres could be a potential carrier for BMP-2.
Besides hydroxyapatite, mesoporous bioactive glass (MBG) possesses promising biocompatibility, osteo-conductivity, osteo-inductivity, osteogenesis, biodegradability, and excellent mechanical strength, which are essential to be selected as bone substitutes 71 . Due to the structure, MBG can mimic the native bone pore size and interconnectivity to promote bone regeneration. Besides, it is a good candidate to deliver drugs or growth factors 72 . The carboxyl bond of BMP-2 has been connected to the amine bond of MBG nanoparticles through 1-ethyl-3-(-3-dimethyl aminopropyl) carbodiimide hydrochloride-N-hydroxy succinimide reaction 73 . In vitro, MBG was found to be cytocompatible and induce a pro-osteogenic response in primary human MSCs. In preclinical rodent models, BMP-2-loaded MBG significantly enhanced bone formation and microarchitecture of bone. The release of BMP-2 in MBG was about 70% within 1 month, which could recover a 5-mm skull defect. Lin et al. immobilized BMP-2 on the MBG scaffold surface via electrostatic interaction 74 . Although 25% of BMP-2 was released in 7 days, MBG-loaded BMP-2 still showed great regeneration in 16-mm bone defect in rabbits. In a word, the application of MBG is limited by the low encapsulation efficiency of BMP-2 and the burst release. Additionally, the harmful solvents used in scaffold fabrication can influence the bioactivity of BMP-2 and threaten the health of patients. MBGs produced by an aerosol-assisted spray-drying scalable process showed prolonged release of low-dose BMP-2 without affecting material properties 75 . The use of MBG as a biocompatible carrier for localized and sustained low-dose BMP-2 release could limit BMP-2-derived side effects, allowing for the safe use of BMP-2.
Nanomaterials
Nanomaterials possess high surface area, tunable size, and functionalization capabilities, making them ideal candidates for delivering BMP-2 in a controlled and efficient manner. For instance, carbon quantum dots (CDs) with ultra-small sizes are emerging stars in drug delivery because of their biocompatibility, high water dispersibility, and remarkable photoluminescence properties76,77. CDs can be conjugated with drugs through surface modification78,79. Based on these, BMP-2-conjugated CDs (BMP-2-CDs) can be used for bioimaging and tracking of growth factor delivery that can meet the requirements of therapeutic applications. Besides, sustained release and long persistence of BMP-2 undergoing the treatment are important to prevent side effects 80 . A composite scaffold containing pectin particles loaded with BMP-2-CDs has been constructed for BMP-2-CDs release systems 77 . Using the microfluidic method, BMP-2-CDs were encapsulated in pectin particles with narrow particle distribution and then loaded into a composite scaffold composed of gelatin, elastin, and hyaluronic acid. BMP-2-CDs were continuously released from the composite scaffold for 21 days, showing high controllable drug release ability. In vitro tests of MG-63 cells demonstrated its biological properties of intercellular absorption and enhancement of BMP-2-CDs and their bone-promoting effects. This platform showed great potential in bone tissue regeneration by utilizing pectin particles carrying BMP-2-CDs as a growth factor delivery biological imaging agent and adjusting the composition of scaffolds.
Codelivery of BMP-2 with Other Growth Factors
Bone regeneration is under the control of the interaction of osteoblast-derived bone formation and osteoclast-derived bone resorption, which can be regulated by various growth factors. BMP-2 possesses synergistic effects with other growth factors, including antiadipogenic, anti-inflammatory, pro-vascularization, and osteogenesis. Thus, an appropriate combination of growth factors is significant to improve the orthopedic surgeon for osteo-angiogenesis.
Vascular Endothelial Growth Factor
Vascular endothelial growth factor (VEGF), one of the strongest angiogenic factors, regulates the proliferation and migration of endothelial cells to promote angiogenesis. VEGF also involves in osteogenesis by influencing osteoblast differentiation and cartilage absorption 81 . Therefore, the combination of VEGF and BMP-2 can promote vessel invasion and blood supply to facilitate new bone formation. In the process of osteogenesis, osteoblasts produce VEGF, stimulating osteogenesis and inhibiting lipogenesis through Runx2 and PPARY2 82 (Fig. 2). Meanwhile, VEGF can activate the Akt/β-catenin signaling pathway, rising BMP-2 levels in the vascular-associated MSCs 83 . As a result, the increased BMP-2 mediated by osteoblasts stimulates osteoblast differentiation as well as VEGF production in a positive feedback loop 84 . Therefore, angiogenesis and bone regeneration are closely coupled, which can mutually promote osteogenesis (Fig. 3).
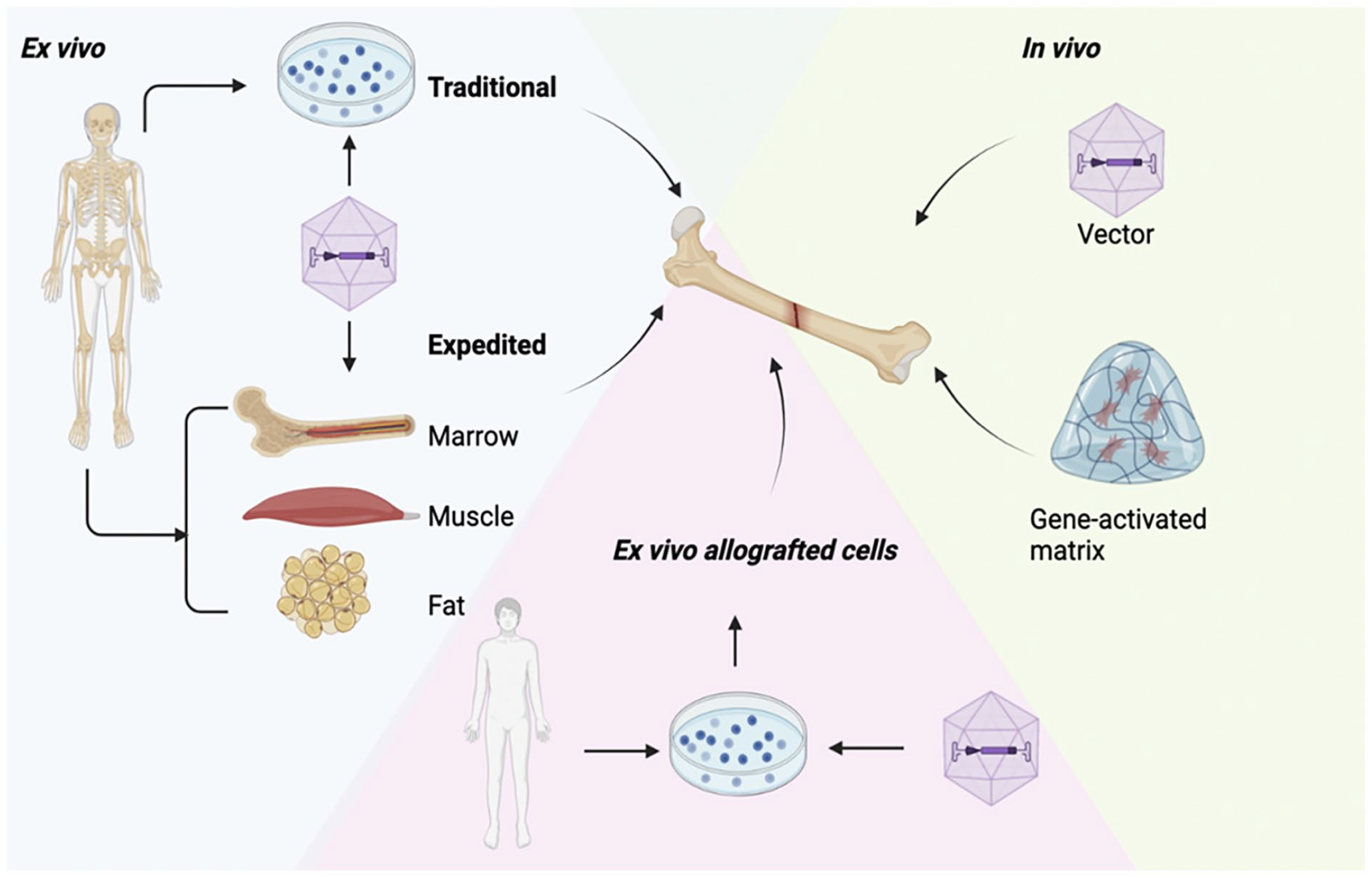
Strategies for BMP-2 gene therapy in bone treatment.
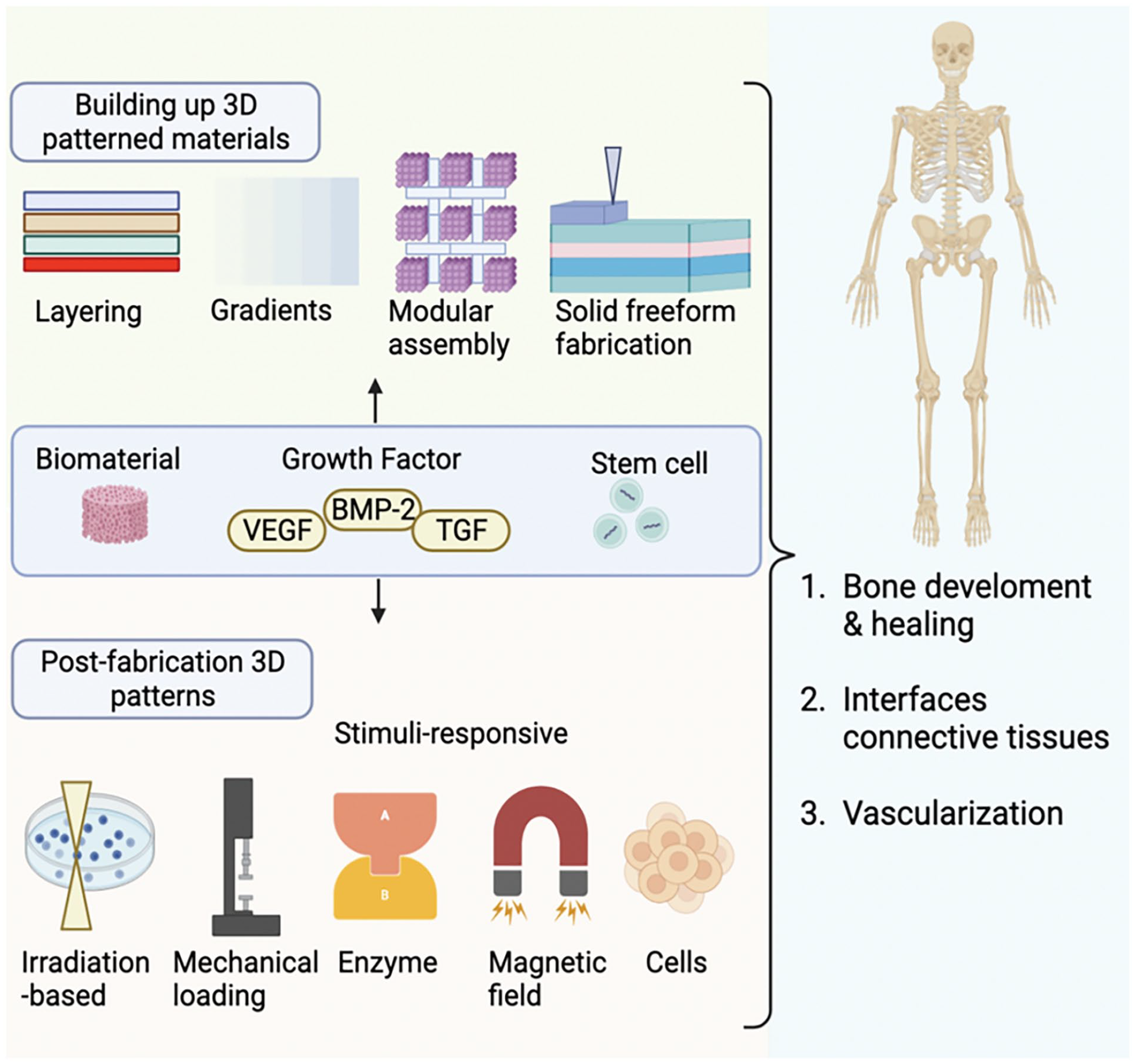
Various and complex combinations of bioactive factors with delivery systems for bone tissue engineering applications being developed recently.
As for applications, the joint treatment of VEGF and BMP-2 effectively promoted the osteogenic induction of mouse osteoblasts mainly through the activation of the p38 mitogen-activated protein kinase (MAPK) pathway to enhance the nuclear translocation of osterix protein
85
. Consistent with this study, the combination therapy in rabbit mandibular bone defect also showed enhanced osteogenesis and angiogenesis than those therapies that used BMP-2 alone
86
. To achieve the controlled release of BMP-2 and VEGF, polydopamine coating was applied to load the BMP-2 and VEGF easily and safely. The polydopamine possesses high hydrophilicity and bioactive functional groups to facilitate cell attachment and expansion on the surface of materials
87
. In vitro experiments showed high osteogenesis expression of runt-related transcription factor 2, osteopontin, and alkaline phosphatase induced by BMP-2 immobilized on nanofibers
88
. Similarly, VEGF sustained release showed angiogenic differentiation of BMSCs with the increased angiogenesis expression of fetal liver kinase-1 and vascular endothelial cadherin
88
. Moreover, using an electrospinning poly-
Basic Fibroblast Growth Factor
Similar to VEGF, basic fibroblast growth factor (bFGF) is an angiogenic growth factor that promotes the migration and proliferation of various cells including vascular cells and osteoblasts. In the early stage of wound healing, angiogenesis is necessary for the transport of MSCs and the supply of oxygen and nutrients 93 . Combined treatment with BMP-2 and bFGF has been reported to synergistically enhance bone regeneration compared with using BMP-2 or bFGF alone 94 . For example, a composite nanofiber scaffold composed of poly(l-lactic-co-glycolic acid), hydroxyapatite, and graphene oxide has been fabricated to deliver bFGF and BMP-2 simultaneously. The result showed that bFGF and BMP-2 loaded in the composite nanofiber scaffolds have a synergistic differentiation effect on the cell adhesion, proliferation, and osteogenesis differentiation of MC3T3-E1 cells. More importantly, the release kinetics and doses are vital to determine the synergistic effect of BMP-2 and bFGF. A low dose of bFGF combined with a high dose of BMP-2 has been found to enhance bone formation, while a high dose of bFGF combined with a low dose of BMP-2 impeded the process 95 . Moreover, a study using rat models with femoral condyle defects showed that the rapid release of bFGF combined with the sustained release of BMP-2 caused an inhibitory effect on osteogenesis 86 . The reason mainly lies in the high-dose bFGF-induced BMP-2 inhibition signaling Smad-6, enhancing osteoclast formation, and finally, the bone density was decreased 96 . In addition to the dose effect, the release kinetics of growth factors can influence the process of osteogenesis. bFGF has mitotic activity and can reduce the expression of osteoblastic markers in less mature cells 95 . Rapid release of bFGF may reduce the osteogenic potential of osteoprogenitor cells, thus delaying bone regeneration. Therefore, exploring a standard application of the relative doses and release kinetics of BMP-2 and bFGF is significant to achieve a greater synergistic effect on bone healing. For instance, bFGF can be released more intelligently at the early stage of wound healing, and BMP-2 can be released later to enhance the osteogenic differentiation of stem cells to promote bone regeneration. In general, bFGF combined with BMP-2 has a wide application prospect in bone regeneration.
NELL-1
Neural EGFL-Like 1 (NELL-1) is an osteoinductive cytokine that can induce bone and cartilage regeneration. Additionally, NELL-1 inhibits osteoclast metabolism and adipogenesis process 97 . As for applications, NELL-1 has been used to induce bone healing of femoral segments and skull defects in rats and promote lumbar fusion in rats, sheep, and non-human primates 98 . Based on these, the synergistic effect of NELL-1 and BMP-2 has great potential to enhance the osteogenic response and significantly reduce the side effects of BMP-2 99 . First, through regulating the lipid metabolism, NELL-1 is antiadipogenic which can deal with the BMP-2-induced adipose tissue, thus improving the quality of regenerated bone99–101. Second, NELL-1 is anti-inflammatory by inhibiting the immune response induced by BMP-2 because of the single nucleotide polymorphisms in the NELL-1 gene 99 . Third, NELL-1 can also stimulate the generation of blood vessels in the early stage of bone healing and strengthen the migration of MSC 102 . In the rat spinal posterolateral fusion experiments, low doses of BMP-2 and NELL-1 synergy in the osteogenesis of MSC differentiation showed great mineralization with negligible heterotopic bone and adipose tissue 103 . However, in vivo research showed that NELL-1 failed to enhance osteoblastic differentiation of BMSCs or induce spinal fusion because the ability of NELL-1 to promote osteogenesis and suppress adipogenesis needs Wnt signaling 104 . Moreover, the short half-life of NELL-1 in vivo may limit its clinical use in systemic therapy. To overcome these shortcomings, structural modifications such as PEGylation have been used to improve the pharmacokinetics of NELL-1 to prolong its circulation time in vivo 105 . In conclusion, NELL-1 can enhance BMP-2-induced osteogenesis which is prospective to be translated into clinical.
NGF
Nerve growth factor (NGF) promotes osteoblast mitosis, skeletal formation, and bone ossification via BMP-2/Smad signaling. NGF can be widely found in skeletal components, including bone marrow mesenchymal stem cells, regional callus tissue, osteoclasts, osteocytes, chondrocytes, and synovial fibroblasts 106 . Local administration of NGF enhanced bone cell metabolism and osteogenic capacity, and promoted the synthesis of VEGF during the healing of fractures 107 . Exogenous NGF could facilitate innervation and angiogenesis, ultimately improving the mechanical properties of bones 108 . Furthermore, high expression of NGF and its high-affinity receptor P140TrkA has been observed in human osteoarthritic cartilage, where they stimulate chondrocyte metabolism, cartilage matrix formation, and collagenase production 109 . Notably, NGF effectively reduces inflammatory reactions during the healing of fractures. Consequently, applying NGF in BMP-2-driven BTE holds significant importance.
BMP-2 Signaling Pathway Modification
In BTE, modifying the BMP-2 signaling pathway represents great potential to enhance bone regeneration. BMP-2 is a key growth factor that regulates osteoblast differentiation and bone formation. By altering its signaling pathway, the BMP-2 activity can be stabilized to improve outcomes in bone repair and regeneration. Techniques for modifying the BMP-2 pathway include protein engineering of BMP-2 and applying gene therapy or small molecules to affect BMP-2 receptor interactions and downstream signaling cascades. These modifications can enhance BMP-2 effectiveness in stimulating osteogenesis and angiogenesis. Advances in this area hold promise for developing effective bone grafts, implants, and other therapeutic strategies for bone defects and injuries (Table 1).
The Example, Pros, and Cons of In Vivo and Ex Vivo Gene Delivery, Viral and Non-Viral Vectors.

Protein Modification of BMP-2
Currently, BMP-2-driven BTE is widely used in clinical trials, but it is concerned with dosage efficacy and short circulation half-life. To solve these problems, protein modification has the potential to realize site-specific covalent immobilization of BMP-2 to scaffolds. The BMP-2 can be infused with a second protein which has been utilized in enzyme immobilization. For example, T4 lysozyme, a soluble and small enzyme, functions as an inert protein spacer to attach to the carriers to enhance the rescued activity of target proteins110,111. Based on these, polyhistidine tag-T4 Lysozyme was genetically fused at the N-terminus of BMP-2 and used as a protein spacer to enhance the BMP-2 stability and solubility 112 . Additionally, the fused protein could mediate the site-specific covalent anchoring of BMP-2 upon binding to nickel-chelated nitrilotriacetic acid microparticles 112 . As for the mechanism of BMP-2 modification, the modified BMP-2 featured an N-terminal His6 tag as a location for the targeted covalent integration of BMP-2 onto the solid carrier. Moreover, the T4L domain played a role in improving solubility and stability. This domain also created a separation between BMP-2 and the scaffold, thereby promoting its bioactivity. Meanwhile, the C-terminal BMP-2 domain contributed to the osteogenic activity. In the future, the positions of domains can be applied for BMP-2 engineering. For example, the T4 Lysozyme domain can be placed at the C-terminus of BMP-2. Additionally, the length and composition of the protein linkers need to be further explored for an optimal active BMP-2 construct.
Besides protein modification, the metabolism of BMP-2 can be improved. BMP-2 shows rapid metabolic adaption including glycolysis and oxidative phosphorylation. Bone remodeling is a biosynthesis process that requires a large amount of energy 113 . Therefore, the energy metabolism of BMP-2 may affect the bone repair and regeneration of BTE. The inadequate activation of the metabolic state may cause the ineffectiveness of low-dose BMP-2. Magnesium, a main component of bone, is critical to balance the metabolism and biomineralization in osteogenesis 114 . In cellular physiology, magnesium regulates the protein and ATP synthesis of mitochondria directly 115 . Inspired by the dual functions of magnesium, a magnesium-based bioenergetic-driven system was developed to improve low-dose BMP-2-driven regeneration 33 . In this system, magnesium upregulated the expression and activity of metabolic enzymes and the glycolysis and oxidative phosphorylation signaling pathways. Through enhancing the metabolism level, the osteo-inductivity of BMP-2 can be increased to fuel osteogenesis.
Molecular-Engineered BMP-2 Signaling Pathway
BMP-2 is involved in many important signaling pathways for osteogenesis, cell survival, and apoptosis, thus, BMP-2 signaling cascades can be regulated by various proteins 116 . In nature, BMP-2 induces the differentiation of stem cells into osteogenic lineage by interacting with surface receptors, activating subsequent signaling pathways, and increasing the expression of osteogenic genes (Fig. 1).
BMP-2 is regulated by several secreted antagonists intracellularly and extracellularly, such as noggin, sclerostin, and follistatin. The antagonists bind to BMP-2 and hinder its attachment to surface receptors, thereby reducing the activity of BMP-2 and impeding the osteogenic differentiation of osteoprogenitor cells 117 . In BTE, BMP-2 antagonists can be repressed to increase the activity of BMP-2, reducing the dosage use of BMP-2. When osteoprogenitor cells are stimulated by BMP-2, they produce a BMP antagonist Noggin 118 . The Noggin driven negative feedback loop may elucidate the need for higher-than-normal doses of BMP-2 to address bone defects in clinical applications. Based on this mechanism, a study co-delivered the BMP-2 gene and CRISPR interference (CRISPRi) targeting Nog into adipose-derived stem cells (ASCs) by hybrid baculovirus (BV) system 119 . This system showed prolonged BMP-2 expression upregulated the Noggin level as well as the CRISPRi suppressed the expression of Noggin for at least 14 days. The result showed enhanced osteogenesis in ASCs and calvarial bone healing in CRISPRi-engineered ASC-implanted rats. Therefore, Noggin inhibition alleviated its antagonistic effects on BMP-2 and enhanced BMP2-induced osteogenesis. Here, the CRISPRi system is introduced by baculovirus which effectively transports large genetic payloads into stem cells with high safety 120 . BV can accommodate genetic cargoes of at least 38 kb and transduce ASC and BMSCs with efficiencies exceeding 95%121,122. Concerning safety, the BV genome does not integrate into the host chromosome and naturally degrades over time 121 . For example, a Cre/loxP-based hybrid baculovirus vector facilitates the formation of DNA minicircles in over 90% of ASC123,124. These minicircles exist as extrachromosomal episomes and eventually degrade. To conclude, the hybrid BV has great potential to engineer the BMP-2 signaling pathway in BTE because of its high trans-genetic efficiency and biocompatibility.
Besides noggin, Casein Kinase 2 (CK2), a key inhibitor of the BMP-2 signaling pathway, has been modified to increase the activity of BMP-2. CK2 binds to the phosphorylation sites of BMPRIa to inhibit the BMP-2-triggered downstream activation of the Smad pathway or non-Smad pathway 1 . Based on this, the deletion of BMPRIa gene showed enhanced osteogenesis in mice 125 . Referring to the three phosphorylation sites of BMPRIa, three peptides were designed to bind and prevent the interaction between CK2 with BMPRIa. The results showed increased osteogenesis, and chondrogenesis which is similar to the experiments of BMPRIa mutants 126 . Additionally, a BMPRIa mutant lacking a serine to prevent the binding of CK2 was overexpressed, showing increased mineralization 127 . All of these indicate that the presence of CK2 inhibitors may activate the BMP-2 signaling pathway. Although increasing researchers have shown different regulatory methods of BMP-2 signaling pathways, the clinical translation of the engineered BMP-2 signaling pathway still needs further exploration.
Discussion
BTE based on bone structure, bone mechanics, and tissue formation aims to induce new functional bone tissues. The compositions of BTE include a biocompatible scaffold mimicking the microenvironment of bone extracellular matrix, osteogenic cells which will proliferate and differentiate into the bone matrix, and growth factors that direct the cells to specific phenotypes. BMP-2, one of the strongest osteogenesis factors, plays a significant role in BTE. BMP-2 recruits progenitor cells on the local site and induce the osteogenic process. However, its side effects are still a concern, including osteolysis 128 , retrograde ejaculation 129 , immune response 130 , and heterotopic bone formation 131 . Most importantly, BMPs may induce cancer 132 . Therefore, appropriate BMP-2 treatment in BTE is urgent to eliminate complex bone graft surgery and long-term healing effects in the clinic.
Among the side effects of BMP-2, the oncogenicity of overdosage BMP-2 has been considered. The BMP antagonist DAN domain BMP antagonist family member 5 (DAND5) has been found to reactivate dormant metastatic breast cancer cells in the lung 133 , and BMP signaling is tumor suppressive in studies overexpressing BMP-2 receptors 134 . These conflicting functions of BMPs in cancer are further complicated by data showing that BMP-2 inhibits tumor cell lines from prostate, ovarian, and breast cancers. Currently, the FDA has ruled out the use of BMP in patients with a history of cancer. Most of the reported complications are related to supraphysiological doses administered clinically 10 . Although initial clinical studies used recombinant human BMP-2 doses in the range of 10–12 mg in the treatment of long bone defects, the optimal dose of BMP-2 was currently considered to be 1–2 μg/kg 135 . The dose can be further minimized if the release and kinetic of BMP-2 can be controlled.
Besides the reduction of the dosage use of BMP-2, novel strategies have been applied to increase the efficiency of BMP-2 by maintaining its bioactivity throughout the healing process. Current delivery systems often face difficulties in preserving the BMP-2 structure and function, leading to potential off-target effects. Efficient BMP-2 delivery requires precise targeting and controlled release, such as the development of smart nanoparticles, injectable hydrogels, or bioresponsive scaffolds. These techniques allow for localized delivery in response to specific biological signals, ensuring that BMP-2 is released exactly where and when it is needed. Future research should focus on developing stabilization techniques that protect BMP-2 from degradation and denaturation.
In BTE, BMP-2 potency can be increased through delivery systems and combination with growth factors. First, the scaffolds can control stable and sustained BMP-2 release through covalent or noncovalent binding. Moreover, the scaffolds should meet the requirement of cell metabolism, proliferation, differentiation, and attachment. Thus, easily fabricated, injectable, and transplantable biomaterials need to be considered as delivery systems of BTE. Various techniques including gene modification and 3D bioprinting have been applied to improve the functions of BMP-2. By engineering physical and chemical properties and microstructure of composite biomaterials, the loading and releasing amount as well as bonding strength of BMP-2 can be improved. For example, collagen–hydroxyapatite composite scaffolds were modified with BMP-2 and loaded with biodegradable microspheres containing encapsulated alendronate 135 . The findings revealed an initial release of BMP-2 over a few days, followed by the sequential release of alendronate after 2 weeks. These composite scaffolds notably enhanced osteogenic activity due to the synergistic effect of BMP-2 and alendronate. Improved bone regeneration was observed 8 weeks after implantation in a rat model with an 8-mm critical-sized defect.
Moreover, understanding the molecular mechanisms of crosstalk pathways can reduce the dosage of BMP for future BTE applications. Therefore, the BMP-2-induced mechanistic cascade needs to be examined. First, various growth factors can regulate the BMP-2 signaling pathway to affect bone healing. That’s mainly because bone healing is a complex process involving cell interactions, extracellular matrix production, mineralization, and vascularization which contains various growth factors as players. Properly combining growth factors with BMP-2 to modulate the BMP-2 signaling pathway has shown great potential to increase the efficiency of bone repair and regeneration. Second, molecular engineering of BMP-2 protein and it signaling pathway show great potential to increase the results the BMP-2 BTE. First, stable half-life protein structure of BMP-2 can be predicted by computational methods including Rosetta, Homology Modeling, and Ab Initio (De Novo) Structure Prediction. After constructing the stable BMP-2 protein, researchers can add them into the microenvironment of scaffold. Then, the activity and degradation of BMP-2 can be tracked. Biocompatibility testing, histological analysis, immunohistochemistry and immunofluorescence and micro-computed tomography can be applied for verification. Second, BMP-2 signaling inhibitors and activators can be targeted to modulate the efficiency of BMP-2 on osteogenesis. Characterizing the chemical and biological properties of the modulators is crucial for further development and potential clinical applications. As for experiments, we can utilize cell lines in responsive to BMP-2 signaling to measure BMP-2-induced responses including phosphorylation of SMAD proteins, gene expression, or alkaline phosphatase activity. Then, researchers can assess the impact of scaffolds containing potential inhibitors and activators on these BMP-2-induced responses. In animal models, the effects of BMP-2 signaling inhibitors in bone regeneration or disease models can provide insights into the therapeutic potential and safety of the inhibitors and activators.
In clinical trials, a combination of delivery systems including stem cells, growth factors, and biomaterials could provide optimized BMP-2 therapy to address the dual challenge of maintaining BMP-2 activity while achieving targeted delivery. Minimizing dosing requires understanding the individual patient’s perception of BMP-2 and tailoring treatment to individual needs. Controlled release is essential to stimulate the endogenous healing cascade. Existing knowledge must be combined and passed through the approval process to optimize the use of BMP-2 in BTE.
Conclusion
Globally, BMP-2 has shown great potential in bone regeneration, but the side effects caused by overdosage and short half-life have limited its application. This review analyzes historical aspects and clinical issues, and cutting-edge findings on BMP-2 efficacy and functions. The synergistic effects of BMP-2 with various biomaterials, growth factors, and molecular engineering may provide strategies for future bone regeneration and repair. In conclusion, continuing basic research is needed to elucidate the details of its function and the underlying signaling pathways involved to ensure the efficacy and safety of BMP-2 in clinical use.
Footnotes
Acknowledgements
Not applicable.
Author Contributions
J.Q. contributed to writing—original draft preparation; G.L. and H.W. contributed to review and editing. All authors have read and agreed to the published version of the manuscript.
Availability of Data and Material
Not applicable.
Ethical Approval
This study was approved by our institutional review board.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Consent for Publication
Not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work is supported by The Nature Science Foundation of Hunan Province, China (No.2023JJ30829).
