Abstract
Intratracheal transplantation of human umbilical cord blood (UCB)-derived mesenchymal stem cells (MSCs) attenuates the hyperoxia-induced neonatal lung injury. The aim of this preclinical translation study was to optimize the dose of human UCB-derived MSCs in attenuating hyperoxia-induced lung injury in newborn rats. Newborn Sprague-Dawley rats were randomly exposed to hyperoxia (95% oxygen) or normoxia after birth for 14 days. Three different doses of human UCB-derived MSCs, 5 × 103 (HT1), 5 × 104 (HT2), and 5 × 105 (HT3), were delivered intratracheally at postnatal day (P) 5. At P14, lungs were harvested for analyses including morphometry for alveolarization, terminal deoxynucleotidyl transferase-mediated deoxyuridine triphosphate nick end labeling (TUNEL) staining, myeoloperoxidase activity, mRNA level of tumor necross factor-α (TNF-α), interleukin-1β (IL-1β), IL-6, and transforming growth factor-β (TGF-β), human glyceradehyde-3-phosphate dehydrogenase (GAPDH), and p47 phox , and collagen levels. Increases in TUNEL-positive cells were attenuated in all transplantation groups. However, hyperoxia-induced lung injuries, such as reduced alveolarization, as evidenced by increased mean linear intercept and mean alveolar volume, and increased collagen levels were significantly attenuated in both HT2 and HT3, but not in HT1, with better attenuation in HT3 than in HT2. Dose-dependent human GAPDH expression, indicative of the presence of human RNA in lung tissue, was observed only in the transplantation groups, with higher expression in HT3 than in HT2, and higher expression in HT2 than in HT1. Hyperoxia-induced inflammatory responses such as increased myeloperoxidase acitivity, mRNA levels of TNF-α, IL-1β, IL-6, and TGF-β of the lung tissue, and upregulation of both cytosolic and membrane p47 phox , indicative of oxidative stress, were significantly attenuated in both HT2 and HT3 but not in HT1. These results demonstrate that intratracheal transplantation of human UCB-derived MSCs with appropriate doses may attenuate hyperoxia-induced lung injury through active involvement of these cells in modulating host inflammatory responses and oxidative stress in neonatal rats.
Introduction
Despite recent improvements in neonatal intensive care medicine, bronchopulmonary dysplasia (BPD), a chronic lung disease that follows ventilator and oxygen therapy in premature infants, continues to represent a major cause of mortality and morbidity among premature infants (5,7). At present, there are few clinically effective treatments available to prevent or ameliorate this common and serious disorder (1,9). Therefore, the development of a new therapeutic modality to improve the prognosis of this disease is an urgent big subject.
Mesenchymal stem cells (MSCs) have been shown to differentiate into various cell types such as respiratory epithelium, chondrocytes, adipocytes, neural cells, and endothelial cells both in vivo and in vitro (4,11,24,32,36,37,44). Among various sources including bone marrow, umbilical cord blood (UCB), and adipose tissue (41), UCB is considered a promising source for human MSCs due to its easy availability, high proliferation capacity (23,44), and less immunogenicity (34). The low expression of human leukocyte antigen (HLA) major histocompatibility complex (MHC) class I and lack of MHC class II molecules in UCB-derived MSCs indicate the immune evasion even in the allogenic transplantation (27). Recently, we have shown that xenotransplantation of human UCB-derived MSCs attenuates hyperoxia-induced lung injuries such as impaired alveolarization, inflammatory responses, increased apoptosis, and fibrosis in the immunocompetent neonatal rats (11). Furthermore, local intratracheal administration was more effective than systemic intraperitoneal transplantation for this protection (11). These findings suggest that human UCB-derived MSC transplantation could be a novel therapeutic modality for BPD. However, the optimal dose for their administration has not yet been determined. We thus tried to determine the optimal dose at which intratracheally delivered human UCB-derived MSCs attenuate the hyperoxia-induced lung injuries in newborn rat pups in the present preclinical translational study.
Hyperoxia-induced lung injuries were assessed by morphometric analyses such as mean linear intercept (MLI) and mean alveolar volume, and terminal deoxynucleotidyl transferase-mediated deoxyuridine triphosphate nick end labeling (TUNEL) staining. The presence of human RNA derived from donor cells in the lung was assessed by measuring human glyceradehyde-3-phosphate dehydrogenase (GAPDH) expression. The extent of inflammatory responses was evaluated by measuring myeloperoxidase (MPO) activity and the mRNA expression of tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6), IL-1β, and transforming growth factor-β (TGF-β) in lung tissue. Production of reactive oxygen species (ROS) was evaluated by measuring activation of nicotinamide adenine dinucleotide phosphate oxidase (NADPH oxidase), as evidenced by upregulation of cytosolic and membrane p47 phox (10).
Materials and Methods
Cell Preparation
This study was approved by Institutional Review Board of Samsung Medical Center and by Medipost, Co., Ltd. (Seoul, Korea). MSCs were isolated and cultivated from human UCB as previously reported (21,44). UCB was collected from umbilical veins after neonatal delivery with informed consent from pregnant mothers. The cells expressed CD105 (99.6%) and CD73 (96.3%), but not CD34 (0.1%), CD45 (0.2%), and CD14 (0.1%). They were positive for HLA-AB (96.8%), but generally not for HLA-DR (0.1%). The cells also expressed pluripotency markers such as octamer-binding transcription factor 4 (Oct 4; 30.5%) (8) and stage-specific embryonic antigen 4 (SSEA-4; 67.7%) (17). Human UCB-derived MSCs differentiated into various cell types such as respiratory epithelium, osteoblasts, chondrocytes, and adipocytes with specific in vitro induction stimuli (11,21,28,44) (Fig. 1). We confirmed the differentiation potential and karyotypic stability of the human UCB-derived MSCs up to the 11th passage.
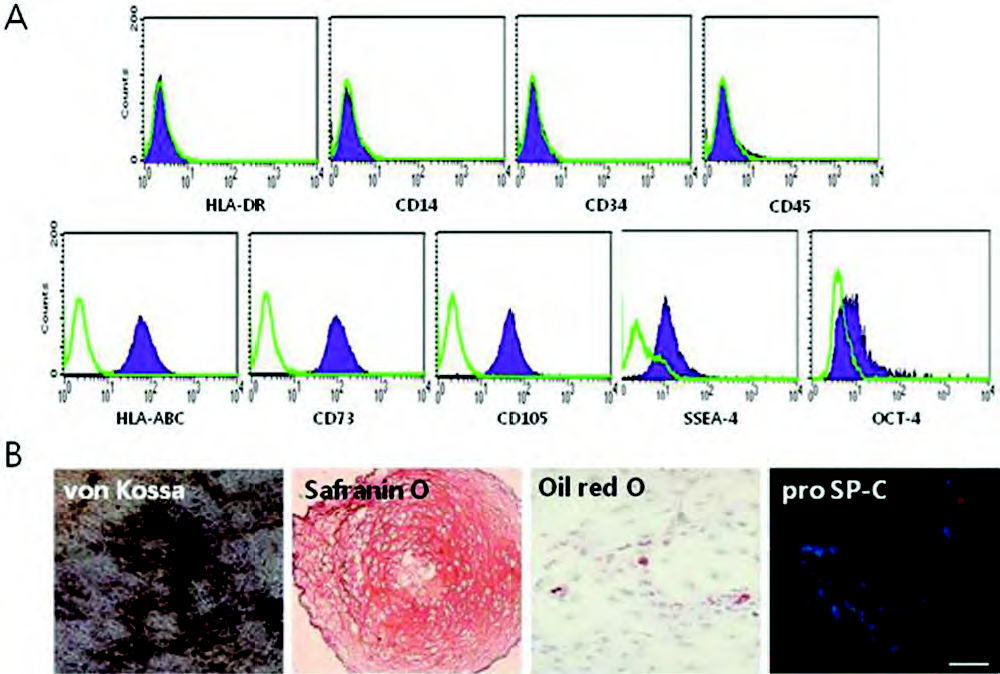
Immunophenotype and multilineage differentiation of umbilical cord blood (UCB)-derived mesenchymal stem cells (MSCs). (A) Cells were positive for human leukocyte antigen (HLA)-ABC, CD73, CD105, stage-specific embryonic antigen 4 (SSEA-4), and octamer-binding transcription factor-4 (Oct-4), and were negative for HLA-DR, CD14, CD34, and CD45. (B) Cells were induced to differentiate into osteoblast, chondroblast, adipoblast, and respiratory epithelial cells, and stained by von Kossa, safranin O, oil red O, and pro SP (surfactant protein)-C, respectively. Scale bar: 50 μm.
Animal Model
The experimental protocols described herein were reviewed and approved by the Animal Care and Use Committee of Samsung Biomedical Research Institute (Seoul, Korea). This study was also performed in accordance with the institutional and National Institutes of Health guidelines for laboratory animal care. Timed pregnant Sprague-Dawley rats (Orient Co., Seoul, Korea) were housed in individual cages with free access to water and laboratory chow. Rat pups were delivered spontaneously and reared with their dams. The experiment began within 10 h after birth and continued through postnatal day (P) 14. Rat pups were randomly assigned to five groups: normoxia control group (NC), hyperoxia control group (HC), hyperoxia with 5 × 103 (HT1), 5 × 104 (HT2), or 5 × 105 (HT3) human UCB-derived MSC transplantation group. Rat pups of the NC group were kept with a nursing mother rat in standard cage at room air throughout the experiment, and pups of hyperoxia groups were maintained with a nursing mother in standard cage within 50 L Plexiglas chambers in which the hyperoxic (oxygen concentration of 95%) condition was maintained. Humidity and environmental temperature were maintained at 50% and 24°C, respectively. Nursing mother rats were rotated daily between litters in the normoxia and hyperxoxia groups to avoid oxygen toxicity. Survival and body weights of rat pups in each group were checked daily throughout the experiment. The rat pups were sacrificed at P14 under deep pentobarbital anesthesia (60 mg/kg, IP), and whole lung tissue was obtained for morphometric and biochemical analyses. Six to eight animals were used in each subgroup of analysis.
Transplantation of Human UCB-Derived MSCs
The human UCB-derived MSCs from the 5th passage from a single donor were transplanted at P5. For donor cell transplantation, 5 × 103 (HT1), 5 × 104 (HT2), or 5 × 105 (HT3) cells in 0.05 ml phosphate-buffered saline (PBS, pH 7.4) were administered intratracheally. For NC and HC, equal volume of PBS was given intratracheally. For intratracheal transplantation, the rats were anesthetized with an IP injection of ketamine and xylazine mixture (45 and 8 mg/kg, respectively), and restricted on a board at a fixed angle. MSCs were administered into the trachea through a 30-gauge needle syringe. After the procedure, the animals were allowed to recover from anesthesia, and were returned to their dams.
Tissue Preparation
Lungs were resected after transcardiac perfusion with ice-cold PBS, snap-frozen in liquid nitrogen, and stored at −80°C for later biochemical analyses.
For morphometric analyses, lungs were fixed in situ by tracheal instillation of 10% buffered formalin at a constant inflation pressure of 20 cm H2O, and then fixed overnight at room temperature in the same fixative. The fixed lung tissue was embedded in paraffin wax after tissue processing. Sections (4 μm thick) were cut from the paraffin blocks, and stained with hematoxylin and eosin. Images of each section were captured with a magnifier digital camera through an Olympus BX40 microscope (Olympus Optical Co. Ltd., Tokyo, Japan), and were saved as JPEG files.
Morphometry
Level of alveolarization was determined by measuring MLI and mean alveolar volume. The mean interalveolar distance was measured as MLI, by dividing the total length of lines drawn across the lung section by the number of intercepts encountered, as described by Cooney and Thurlbeck (15). The mean alveolar volume was calculated using the method reported by Snyder and colleagues (12,40). Briefly, a grid containing equally spaced crosses was placed on a uniformly enlarged photomicrograph of each lung field. The diameters (ℓ) of the alveoli containing a cross were measured along the horizontal axis of the cross. The cube of the alveolar diameter times τ and divided by 3 (ℓ3τ/3) was used to estimate mean alveolar volume. A minimum of two sections per rat and six fields per each section were randomly examined for each analysis.
TUNEL Assay
Immunofluorescent TUNEL staining with an in situ cell death detection kit (S7110 ApopTag, Chemicon, Temecula, CA, USA) was done to measure the extent of apoptosis in the lung. Paraffin section slides were deparaffinized, rehydrated, and digested with Proteinase K (20 μg/ml in PBS) (Sigma Co., St. Louis, MO, USA) at room temperature for 15 min and then washed in PBS for 10 min. Sections were then incubated with equilibration buffer for 1 min and immediately incubated with working strength TdT enzyme in a humidified chamber at 37°C for 1 h. Each section was immersed in a stop/wash buffer and gently rinsed with PBS. Fluorescein isothiocyanate (FITC)-labeled anti-digoxigenin conjugate was applied to the sections, which were then incubated at room temperature for 30 min in the dark. Nuclear counterstaining was performed with propidium iodide (0.5 μg/ml, Sigma). Slides were washed again in PBS, mounted with Vectashield mounting solution (Vector Laboratories, Burlingame, CA, USA), and visualized with a fluorescent microscope (Nikon E600 fluorescence microscope, Tokyo, Japan) using an excitation wavelength of 460–490 nM. Ten nonoverlapping fields were examined at a magnification of x200 to count TUNEL-positive cells.
Human GAPDH
Total RNA in the sample was extracted using RNA Trizol according to the manufacturer's protocol (Invitrogen Corporation, Carlsbad, CA, USA). Total RNA concentration was measured by spectrophotometry (Nanodrop Wilmington, DE, USA) at 260 nm. One microgram of RNA was used to produce cDNA with a Protoscript® II RT-PCR kit (New England Biolabs, Ipswich, MA, USA). PCR primers for rat actin and human glyceraldehyde-3-phosphate dehydrogenase (GAPDH) were designed with Primer 3 (Whitehead Institute, Cambridge, MA, USA) and synthesized by Bioneer Inc. (Bioneer, Daejeon, Korea). The sequence of primers used was as follows: rat actin (sense -ATGCCAACACAGTGCTGTCT-, antisense -CCATGGTGTCTGAGCGATGT-), human GAPDH (sense -CGCTCTCTGCTCCTCCTGTT-, antisense -CTGCTTGCTGATCCACATCT-). For each reaction, 1 μl of cDNA was placed in a 19-μl reaction mixture of PCR Master Mix (Bioneer) and 0.5 μM of each primer (Bioneer). All amplification reactions were performed in a T1 thermocycler (Biometra, Goettingen, Niedersachsen, Germany). The following cycle profile was used: 45 s at 94°C, 45 s at X°C, 45 s at 72°C for 33 cycles, where X is the annealing temperature for each pair of cytokine primers. PCR products were run on a 1.2% agarose gel electrophoresis, visualized by ethidium bromide, and scanned by a Gel Doc 2000 analyzer (Bio-Rad). The expression levels for each gene were semi-quantified by densitometric analysis using software (Quantity One, Bio-Rad). Relative expression levels were estimated by the density ratio of human GAPDH to rat actin.
Myeloperoxidase Activity (MPO)
The activity of MPO, an indicator of neutrophil accumulation, was determined by modification of the method by Gray et al. (18). The lung tissues were homogenized in a phosphate buffer (pH 7.4) and centrifuged at 30,000 x g for 30 min. The pellet was resuspended in fresh phosphate buffer (50 mM, pH 6.0) containing 0.5% hexadecyltrimethyl ammonium bromide. MPO activity in the resuspended pellet was assayed by measuring absorbance changes spectrophotometrically at 460 nm, using 0.167 mg/ml of O-dianisidine ihydrochloride and 0.0005% hydrogen peroxide. One unit of MPO activity was defined as the quantity of enzyme degrading 1 μM of peroxide/min.
Measurement of Collagen
The lung collagen level was determined from the total soluble collagen using a Sircol collagen assay kit (Biocolor Ltd., Newton Abbey, UK) according to the manufacturer's instructions. Lungs were homogenized in 5 ml of 0.5 M acetic acid solution containing 1 mg pepsin (Sigma) per 10 mg tissue. Each sample was incubated for 24 h at 4°C with constant shaking. After centrifugation, 100 μl of each supernatant was assayed. Then Sircol dye reagent (1 ml) specifically binding to collagen was added to each sample and mixed for 30 min. After centrifugation, the pellet was suspended in 1 ml of an alkali reagent (0.5 M NaOH) included in the assay kit, and the optical density was measured with a spectrophotometer at 540 nm. Values for the test samples were compared with values obtained using standard collagen solutions provided by the manufacturer, which were used to construct a standard curve.
Semiquantitative Reverse Transcriptase Polymerase Chain Reaction (RT-PCR) of Cytokines
The total RNA in the sample was extracted using a RNA Trizol kit (Invitrogen Corporation, Carlsbad, CA, USA) according to the manufacturer's protocol. One microgram of RNA was used to produce cDNA with a Protoscript® II RT-PCR kit (New England Biolabs). PCR primers for rat TNF-α, IL-1β, IL-6, TGF-β, and GAPDH were designed with Primer 3 (Whitehead Institute) and were synthesized by Bioneer Inc. The sequence of primers used was as follows: (TNF-α) sense 5-TAC TGAACTTCGGGGTGATCGGTCC-3, antisense 5-CAGCCTTGTCCCTTGAAGAGAACC-3; (IL-1β) sense CA CAGTTCCCCAACTGGTACAT, antisense CACAGTTCCCCAACTGGTACAT; (IL-6) sense 5-CACCAGGAACGAAAGTCAACTC-3, antisense 5-GGAAGCATCCATCATTTCTTTG-3; (TGF-β) sense 5-CAACTGTGGAGCAACACGTAGA-3, antisense 5-CAACCCAGGTCCTTCCTAAAGT-3; (GAPDH) sense 5-CTCTACCCACGGCAAGTTCAA-3, antisense 5-GGGATGACCTTGCCCACAGC-3.
For each reaction, 1 μl of cDNA was placed in a 19-μl reaction mixture PCR Master Mix (Bioneer), to which 0.5 μM of each primer (Bioneer) was then added. All amplification reactions were carried out in a T1 thermocycler (Biometra). The following cycle profile was used: 60 s at 95°, 60 s at X°C, where X is the annealing temperature for each pair of cytokine primers (60° for TNF-α, 56°C for TGF-β, 55°C for IL-6, and 57°C for GAPDH), and 60 s at 68°C. The PCR products were separated by 1.2% agarose gel electrophoresis, visualized by ethidium bromide, and scanned by using a Gel Doc 2000 analyzer (Bio-Rad Laboratories, Hercules, CA, USA). The expression level of each gene was semi-quantified by densitometric analysis using Quantity One software (Bio-Rad Laboratories). The relative expression levels were estimated from the density ratio of cytokines to GAPDH (control).
Western Blot for p47phox
The upregulation of p47 phox , a subunit of NADPH oxidase, both in the cytplasmic and plasma membrane portion serves as an indicator of NADPH oxidase activation that is responsible for generating reactive oxygen species (2,16). For this purpose, tissue sample from each animal were separated into membrane and cytosolic components for Western blot examination. Tissues were homogenized in ice-cold hypertonic solution and centrifuged at 600 x g for 10 min. The supernatant was ultracentrifuged at 100,000 x g for 1.5 h. The supernatant contained the cytosolic fraction and the membrane-particulate pellet was resuspended in hypotonic solution containing 1% Triton X-100. Samples were analyzed by Western blot 5 ng using antibodies against the NADPH oxidase cytosolic subunit, p47 phox (1:500, BD Biosciences, San Diego, CA, USA). The bands were recognized by horseradish peroxidase-conjugated anti-mouse secondary antibody (1:1,000, DAKO, Glostrup, Denmark), and then Western blots were developed with enhanced chemiluminescence detection reagents (Amersham Pharmacia, Uppsala, Sweden), and exposed to X-ray film (Fuji Photo Film, Tokyo, Japan). The blots were reprobed with antibodies against GAPDH (1:1,000, Santa Cruz Biotechnology Inc., Santa Cruz, CA, USA). To determine the relative degree of membrane purification, the membrane fraction was subjected to immunoblotting for calnexin (1:500, Santa Cruz Biotechnology Inc.), a membrane marker.
Statistical Analysis
Data are expressed as mean ± SEM. Survival curve comparisons were performed using Kaplan-Meier analysis followed by a log rank test. For continuous variables with a normal distribution, ANOVA test with Bonferroni's correction was performed. For variables without normal distribution, Wiloxon signed-rank tests with Bonferroni's correction were done. Values of p < 0.05 were considered significant. Stata software (ver. 11.0, StataCorp LP, College Station, TX, USA) was used for all analyses.
Results
Survival Rate and Body Weight Gain
Exposure to oxygen (HC) significantly reduced survival rate by the end of experiment (P14) compared to the 100% survival rate of NC. However, the reduced survival rate observed in HC (p < 0.05 vs. NC) was improved with MSC treatment in both HT2 (p > 0.05 vs. NC) and HT3 (p > 0.05 vs. NC) but not in HT1 (p < 0.05 vs. NC) (Fig. 2A). The seeming lower survival rate in HT1 and HT2 compared to NC from P5 to P10 did not reach a statistical significance.

Kaplan-Meier survival curve (A) and body weights at birth and postnatal day 14 (P14) (B). Data are mean ± SEM for n = 24, 40, 38, 38, and 38 in normoxia control (NC), hyperoxia control (HC), hyperoxia exposed rats with 5 × 103 MSC intratracheal transplantation (HT1), with 5 × 104 MSC intratracheal transplantation (HT2), and 5 × 105 MSC intratracheal transplantation (HT3). ∗∗∗p < 0.001 versus NC; #p < 0.05 versus HC; †† p < 0.01 versus HT1; ‡‡p < 0.01 versus HT2.
Although birth weights were not significantly different between the five experimental groups (7.1 ± 0.03, 7.1 ± 0.04, 7.2 ± 0.03, 7.2 ± 0.03, and 7.2 ± 0.02 g in NC, HC, HT1, HT2, and HT3, respectively), body weight at P14 in HC (21.6 ± 0.9 g) was significantly lower compared to NC (29.4 ± 0.3 g) (p < 0.001 vs. NC). The decreased body weight gain observed in HC was significantly improved in HT3 (25.3 ± 0.6 g, p < 0.01 vs. HC, vs. HT1, and vs. HT2), but not in HT1 and in HT2 (21.7 ± 1.3 and 22.1 ± 1.7 g, p > 0.05 vs. HC, respectively) (Fig. 2B).
Lung Histopathology
Impaired alveolar growth, as evidenced by fewer and larger alveoli and heterogenous alveolar sizes, was observed in HC compared to NC. After MSC transplantation, hyperoxia-induced impairments in alveolar growth and morphological changes were attenuated in a dose-dependent manner, showing significant attenuation in both HT2 and HT3, but not in HT1, with better attenuation in HT3 than in HT2. In morphometric analyses, the MLI and mean alveolar volume, indicating the size and volume of the alveoli, respectively (Fig. 3), were significantly higher in HC (75.5 ± 3.5 μm in MLI and 22.7 ± 1.8 × 104 μm3 in mean alveolar volume, p < 0.001) than in NC (46.6 ± 0.7 μm in MLI and 4.9 ± 0.3 × 104 μm3 in mean alveolar volume). Our data demonstrate significant attenuation of increased MLI in HT3 (60.7 ± 1.3 μm with 19% attenuation relative to HC, p < 0.001 vs. HC, p < 0.01 vs. HT1 and vs. HT2) and in HT2 (68.3 ± 3.0 μm with 10% attenuation relative to HC, p < 0.05 vs. HC), and mean alveolar volume in HT3 (13.2 ± 1.6 × 104 μm3 with 42% attenuation relative to HC, p < 0.001 vs. HC, p < 0.05 vs. HT1 and vs. HT2) and in HT2 (18.2 ± 1.7 × 104 μm3 with 19% attenuation relative to HC, p < 0.05 vs. HC) but not in HT1 (68.9 ± 1.5 μm in MLI and 18.5 ± 1.6 × 104 μm3 in mean alveolar volume, p > 0.05 vs. HC). These findings support the dose-dependent protection of MSCs transplantation.

Morphometric analysis of the P14 rat lungs: degree of alveolarization measured by the mean linear intercept (A) and mean alveolar volume (B). Data are mean ± SEM for n = 7, 18, 13, 18, and 20 in NC, HC, HT1, HT2, and HT3. ∗∗∗p < 0.001 versus NC; #p < 0.05 versus HC; ###p < 0.001 versus HC; † p < 0.05 versus HT1; †† p < 0.01 versus HT1; ‡p < 0.05 versus HT2; ‡‡p < 0.01 versus HT2.
The number of TUNEL-positive cells in the lung of P14 rats per high-power field was significantly increased in HC (3.1 ± 1.2, p < 0.05), but not in transplantation groups (1.7 ± 0.5 in HT1, 2.4 ± 0.9 in HT2, and 1.4 ± 0.4 in HT3, p > 0.05) compared to NC (1.0 ± 0.2) (Fig. 4).

Terminal deoxynucleotidyl transferase-mediated deoxyuridine triphosphate nick end labeling (TUNEL)-positive cells in the distal lungs of the P14 rat pups. (A) TUNEL-positive cells were labeled with fluorescein isothiocyanate (FITC; green, arrow) and the nuclei were labeled with propidium iodide (red). Scale bar: 25 μm. (B) Number of observed TUNEL-positive cells per high-power field (HPF) in rat lungs. Data are mean ± SEM for n = 10, 14, 13, 17, and 17 in NC, HC, HT1, HT2, and HT3. ∗p < 0.05 versus NC.
Human GAPDH Expression
Human GAPDH expression, indicative of the presence of human RNA in the lung tissue, was observed only in the MSC transplantation groups, with 0.07 ± 0.01, 0.28 ± 0.05, and 0.46 ± 0.06 of human GAPDH relative to rat actin expression in HT1, HT2 (p < 0.01 vs. HT1), and HT3 (p < 0.001 vs. HT1, p < 0.01 vs. HT2), respectively, in a dose-dependent manner (Fig. 5).

Human glyceraldehyde-3-phosphate dehydrogenase (GAPDH) expression, indicative of the presence of human RNA derived from donor cells, in the lungs of P14 rats. Representative RT-PCR blots (upper panel) and densitometric histograms (lower panel). Data are mean ± SEM for n = 4, 5, 5, 5, and 5 in NC, HC, HT1, HT2, and HT3, †† p < 0.01 versus HT1; †††p < 0.001 versus HT1; ‡‡p < 0.01 versus HT2.
Lung Myeloperoxidase Activity and Collagen
MPO activity was significantly higher in HC (7.3 ± 0.4 U, p < 0.001) than in NC (1.8 ± 0.1 U). This hyperoxia-induced increase in MPO activity was significantly attenuated in both HT2 (4.7 ± 0.7 U) and HT3 (4.3 ± 0.2 U) (p < 0.001 vs. HC, p < 0.001 vs. HT1) but not in HT1 (7.3 ± 0.3 U, p < 0.05 vs. HC) (Fig. 6A).
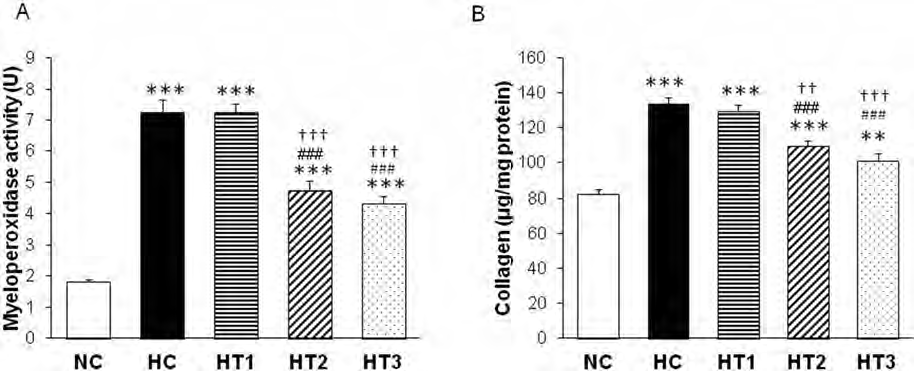
Myeloperoxidase activity (A) and levels of collagen (B) in P14 rat lungs. Data are mean ± SEM for n = 6, 5, 6, 6, and 6 in in NC, HC, HT1, HT2, and HT3. ∗∗p < 0.01 versus NC; ∗∗∗p < 0.001 versus NC; ###p < 0.001 versus HC; †† p < 0.01 versus HT1; †††p < 0.001 versus HT1.
The lung collagen levels at P14 were significantly higher in HC (134 ± 3 μg/mg protein, p < 0.001) than in NC (82 ± 3 μg/mg protein). This hyperoxia-induced increase in the lung collagen levels was significantly attenuated in HT2 (109 ± 3 μg/mg protein) and HT3 (101 ± 4 μg/mg protein) (p < 0.001 vs. HC) but not in HT1 (129 ± 4 μg/mg protein, p > 0.05 vs. HC). This attenuation was more profound in HT3 (p < 0.001 vs. HT1) than in HT2 (p < 0.01 vs. HT1) (Fig. 6B).
mRNA Expressions of TNF-α, IL-1β, IL-6, and TGF-β
In semiquantitative RT-PCR, significantly increased mRNA levels of TNF-α, IL-1β, IL-6, and TGF-β were observed in HC (p < 0.001) compared to NC. This hyperoxia-induced increase in mRNA expression of these cytokines was significantly attenuated in both HT2 and HT3 (TNF-α, IL-1β, and TGF-β, p < 0.001 vs. HC; IL-6, p < 0.01 vs. HC) but not in HT1, and the attenuation of TGF-β mRNA levels was more profound in HT3 (p < 0.05 vs. HT2) than in HT2 (Fig. 7).
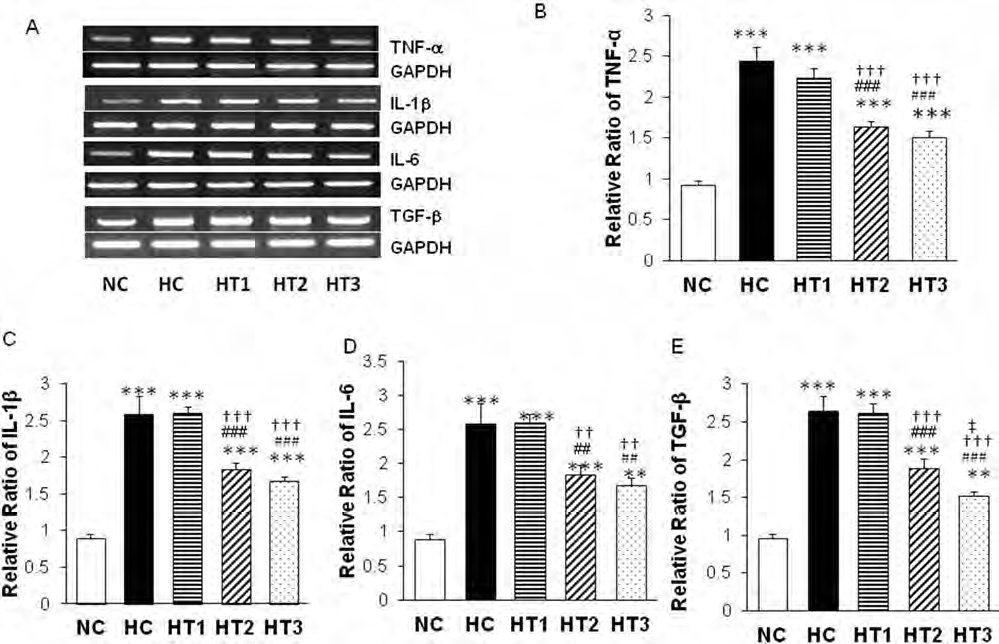
Cytokine expressions in P14 rat lungs. Representative RT-PCR blots (A) and densitometric histograms for tumor necrosis factor-α (TNF-α) (B), interleukin-1β (IL-1β) (C), IL-6 (D), and transforming growth factor-β (TGF-β) (E) in P14 rat lungs. Data are mean ± SEM for n = 6, 5, 6, 6 and 6 in in NC, HC, HT1, HT2, and HT3. ∗∗p < 0.01 versus NC; ∗∗∗p < 0.001 versus NC; ##p < 0.01 versus HC; ###p < 0.001 versus HC; †† p < 0.01 versus HT1; †††p < 0.001 versus HT1; ‡p < 0.05 versus HT2.
Cytosolic and Membrane Expressions of p47phox
Since NADPH oxidase produces oxygen free radicals in both phagocytic (33) and nonphagocytic cells (20,29), hyperoxia-induced production of ROS was evaluated by NADPH oxidase activation, as evidenced by increased cytosolic and membrane expression of a cytosolic subunit of NADPH oxidase p47 phox . In Western blot analyses, significantly higher levels of p47 phox were observed in HC both in the cytosolic (p < 0.01) and membrane (p < 0.001) fractions than in NC, indicating activation of NADPH oxidase. This hyperoxia-induced increase in both cytosolic and membrane expression of p47 phox was significantly attenuated both in HT2 (p > 0.05 vs. NC in cytosolic fraction, p < 0.01 vs. HC in membrane fraction of p47 phox ) and HT3 (p > 0.05 vs. NC in cytosolic fraction, p < 0.001 vs. HC in membrane fraction of p47 phox ) but not in HT1 (Fig. 8).

Western blot analysis of p47 phox , a cytosolic subunit of nicotinamide adenine dinucleotide phosphate oxidase (NADPH oxidase), in the cytosol and the membrane fractions of P14 rat lung homogenates. Upper panel: representative Western blots of the protein extracts. Lower panel: densitometric histograms. Data are mean ± SEM for n = 4, 5, 6, 6, and 6 in NC, HC, HT1, HT2, and HT3. ∗∗p < 0.01 versus NC; ∗∗∗p < 0.001 versus NC; ##p < 0.01 versus HC; ###p < 0.001 versus HC; †p < 0.05 versus HT1; ††† p < 0.001 versus HT1.
Discussion
In the present study, prolonged exposure of newborn rat pups to hyperoxia for 2 weeks increased mortality, retarded growth, and developed lung injuries similar to those seen in the premature human infants with BPD (14,22), exhibiting decreased alveolarization as evidenced by increased MLI and alveolar volume (25), and significantly increased TUNEL-positive cells (31). After human UCB-derived MSC transplantation, hyperoxia-induced damage was attenuated in a dose-dependent manner, except TUNEL-positive cells, showing significant attenuation in both HT2 and HT3, but not in HT1, with better attenuation in HT3 than in HT2. Dose-dependent human GAPDH expression, indicative of the presence of human RNA in the lung tissue, was observed only in the transplantation groups, with higher levels of expression in HT3 than in HT2, and higher levels of expression in HT2 than in HT1. Due to technical limitations, a maximum of 5 × 105 cells were transplanted in the present study, the same amount as in our previous study (10). The maximum concentration of MSCs without adhesion is 1 × 107 cells/ml, and 50 μl is the maximum volume that could be given intratracheally to newborn rat pups at P5 that weighed an average of 8 g. The maximum number of cells feasible for intratacheal transplantation in a newborn rat pups was 5 × 105 cells. In our previous study (11), the number of donor cells localized in the lung tissue was positively associated with the protective effects of stem cell transplantation against hyperoxia-induced lung injuries. Taken together, these findings suggest that mitigation of hyperoxia-induced neonatal lung injuries depends on the number of human UCB-derived MSCs administered, and that intratracheal delivery of 5 × 105 donor cells seems to be the optimal to maximize protective effects in this experimental model and setting.
Despite the comparable data of NC in the present and previous studies (11), increase in mortality, MLI, mean alveolar volume, TUNEL-positive cells, and the expression of cytokines observed in both HC and HT groups of the present study were significantly less than those of our previous study (11). These findings suggest that although we tried to conduct the experiments under the same conditions, the hyperoxic lung injuries observed in the present study were less intense than those in the previous study (11) for some unknown reasons. Nonetheless, these results do not contradict the beneficial effects of UCB-derived MSC transplantation, including improved mortality observed in the present study.
Our data of significant increases in MPO activity and mRNA of TNF-α, IL-1β, IL-6, and TGF-β observed in HC support the assumption that inflammatory responses mediated by neutrophils (42) and proinflammatory cytokines (6,13,30) play a pivotal role in the development of BPD (42). In our previous study (11), we have shown that the protective effects of MSC therapy against hyperoxia-induced lung injuries are mediated primarily by their anti-inflammatory effects rather than by their regenerating capacity. Although the precise mechanisms of this anti-inflammatory effect still remain to be elucidated, Sadan et al. (35) have suggested that the beneficial outcome observed after MSC transplantation into neurodegenerating tissues might be attributable to their paracrine effects, including trophic factor secretion and immunomodulation by cytokines. In addition, a growing body of evidence highlights the fact that MSCs can modulate T-cell-mediated immunologic responses (43). In the present study, hyperoxia-induced increase in MPO activity and proinflammatory cytokines was significantly attenuated both in HT2 and HT3 but not in HT1 after MSC transplantation. These findings suggest that the inratracheal delivery of at least 5 × 104 donor cells is necessary to induce effective paracrine anti-inflammatory effects in neonatal hyperoxic lung injury of newborn rats. Further studies will be necessary to identify the humoral substances secreted from the engrafted MSCs mediating this anti-inflammatory effect.
TGF-β1 is a key fibrogenic cytokine, and activates fibroblast differentiation into myofibroblasts (19,39). Kwong et al. (26) reported that lung inflammatory cells, such as neutrophils and macrophages, are the main sources of TGF-β1 in preterm lung tissue. In the present study, the attenuation of hyperoxia-induced increase in collagen levels was positively correlated with the extent of attenuation of TGF-β expression after MSC transplantation. Overall, the antifibrotic effects of MSC transplantation as evidenced by reduced collagen levels might be attributable to their anti-inflammatory effects as evidenced by reduced TGF-β mRNA levels.
Oxidative stress to the immature lung is a well-known risk factor for the development of BPD (38). NADPH oxidase is a multicomponent enzyme complex responsible for production of superoxide anion (O2-), which generates other ROS such as hydrogenperoxide, hydroxyl radical, and hypochlorous acid (33). Upon its activation, the cytoplasmic subunits p47 phox , p67 phox , p40 phox , and Rac upregulate their expressions and translocate to membrane bound cytochrome (3). The upregulation of both cytosolic and membrane portions of p47 phox and p67 phox can thus be served as in vivo indicators of NADPH oxidase activation in the lung tissue (10). In the present study, increased expression of the p47 phxo protein was observed both in the cytosolic and membrane fractions in HC. After MSC transplantation, significant attenuation of hyperoxia-induced NADPH oxidase activation, as evidenced by significant down modulation of upregulation in both cytosolic and membrane portions of the p47 phox was observed both in HT2 and HT3 but not in HT1. These findings suggest that the intratracheal delivery of at least 5 × 104 donor cells is necessary to achieve effective antioxidative effects in this model.
In summary, intratracheal transplantation of human UCB-derived MSCs significantly attenuated hyperoxia-induced lung pathology such as decreased alveolarization in both HT2 and HT3, but not in HT1, with better protection in HT3 than in HT2. The intratracheal delivery of at least 5 × 104 cells was necessary to achieve effective anti-inflammatory, antifibrotic, and antioxidative effects. Although this dose might be optimal for newborn rats, it might not be appropriate to extrapolate these results on a kilogram basis to humans. Further studies will be necessary to determine the optimal dose of human UCB-derived MSCs for potential clinical benefit in human infants.
Footnotes
Acknowledgments
This work was supported by a grant of the Korea Healthcare technology R&D Project, Ministry for Health, Welfare & Family Affairs, Republic of Korea (A084036) and IN-SUNG Foundation of Medical Research (CB08141). The authors declare no conflict of interest.
