Abstract
Recent evidence suggests mesenchymal stem cells (MSCs) can downmodulate bleomycin-induced lung injury, and umbilical cord blood (UCB) is a promising source for human MSCs. This study examined whether intratracheal or intraperitoneal transplantation of human UCB-derived MSCs can attenuate hyperoxia-induced lung injury in immunocompetent newborn rats. Wild-type Sprague-Dawley rats were randomly exposed to 95% oxygen or air from birth. In the transplantation groups, a single dose of PKH26-labeled human UCB-derived MSCs was administered either intratracheally (2 × 106 cells) or intraperitoneally (5 × 105 cells) at postnatal day (P) 5. At P14, the harvested lungs were examined for morphometric analyses of alveolarization and TUNEL staining, as well as the myeoloperoxidase activity, the level of tumor necrosis factor (TNF)-α, interleukin (IL)-6, and transforming growth factor (TGF)-β mRNA, α-smooth muscle actin (SMA) protein, and collagen levels. Differentiation of MSCs to the respiratory epithelium was also evaluated both in vitro before transplantation and in vivo after transplantation. Despite one fourth dosage of MSCs, significantly more PKH26-labeled donor cells were recovered with intratracheal administration than with intraperitoneal administration both during normoxia and hyperoxia. The hyperoxia-induced increase in the number of TUNEL-positive cells, myeloperoixdase activity, and the level of IL-6 mRNA were significantly attenuated with both intratracheal and intraperitoneal MSCs transplantation. However, the hyperoxia-induced impaired alveolarization and increased the level of TNF-α and TGF-β mRNA, α-SMA protein, and collagen were significantly attenuated only with intratracheal MSCs transplantation. MSCs differentiated into respiratory epithelium in vitro and a few PKH26-positive donor cells were colocalized with pro surfactant protein C in the damaged lungs. In conclusion, intratracheal transplantation of human UCB-derived MSCs is more effective than intraperitoneal transplantation in attenuating the hyperoxia-induced lung injury in neonatal rats.
Introduction
Bronchopulmonary dysplasia (BPD) is a chronic pulmonary disease that usually occurs in premature infants receiving prolonged ventilatory support and oxygen supplementation. It is also an important cause of mortality and long-term respiratory morbidity with few effective treatments (3,6). The histopathological characteristics of BPD include impaired alveolarization and interstitial fibrosis (30). Prolonged exposure of neonatal rodents to hyperoxia results in decreased alveolarization and increased lung fibrosis, which simulates the histopathology of human BPD (11,43). Inflammation is believed to play a key role in the lung injury process leading to the development of hyperoxic neonatal lung injury and BPD (42).
Recently, the exogenous administration of bone marrow (BM)-derived mesenchymal stem cells (MSCs) significantly attenuated the bleomycin-induced lung injury by downmodulating the inflammatory responses and ameliorating their fibrotic effects (33). In addition, the transplanted cells assumed the lung parenchymal cell phenotypes (34). The anti-inflammatory effect of MSCs has also been reported in various animal models such as an ischemia/reperfusion injury of the kidney (36) and acute myocarditis (31). Overall, MSCs transplantation might be a new therapeutic modality for the treatment of hyperoxic neonatal lung injury or BPD.
Among the various sources, including BM, umbilical cord blood (UCB), and adipose tissue (39), UCB is considered a promising source for human MSCs on account of its easy availability. Furthermore, human UCB-derived MSCs have been successfully isolated, cultivated, and shown to differentiate into various cell types such as osteoblasts, chondrocytes, adipocytes, stromal cells, skeletal cells, neural cells, and endothelial cells both in vivo and vitro (17,21,25,45), and even to lung-specific cells in vitro (4). However, it is not known if human UCB-derived MSCs can differentiate into lung specific cell types in vivo, and whether these cells are suitable exogenous stem cell sources in lung injuries of experimental or clinical settings.
The optimal route for stem cell transplantation is another issue that needs to be determined before it can have clinical applications. In contrast to a normal lung, an injured lung produces soluble factors that cause MSCs to proliferate and migrate toward an injured lung (34). Moreover, systemically administered MSCs have been shown to localize to an injured lung (33). However, it is unclear if the local, intratracheal administration of MSCs is comparable or better than systemic transplantation.
Therefore, the therapeutic efficacy of exogenous human UCB-derived MSCs transplantation, the optimal route of its administration, and its possible protective mechanism were investigated in a hyperoxic neonatal lung injury. In particular, this study examined whether intratracheal or intraperitoneal human UCB-derived MSCs transplantation could attenuate an hyperoxia-induced lung injury in immunocompetent newborn rat pups and, if so, whether its protective effect is associated with anti-inflammatory responses and/or by regeneration through their differentiation into lung specific cells.
Materials and Methods
Cell Preparation
The MSCs were isolated and cultivated from human UCB, as previously reported (18,43). The cells expressed CD105 (99.6%) and CD73 (96.3%), but not CD34 (0.1%), CD45 (0.2%), and CD14 (0.1%). They were positive for HLA-AB but generally not for HLA-DR. The cells also expressed the pluripotency markers such as Oct 4 and SSEA-4. As previously reported (18, 24,45), the human UCB-derived MSCs differentiated into various cell types such as osteoblasts, chondrocytes, and adipocytes with in vitro induction by the specific osteogenic, chondrogenic, and adipogenic differentiation stimulus, and this differentiation ability of MSCs was maintained up to the 10th passage. Human fibroblast (HS68; ATCC, CRL-1635, Rockville, MD, USA) were cultured in α-MEM supplemented with 10% fetal bovine serum. These cells were isolated from human newborn foreskin fibroblasts.
In Vitro Differentiation to Respiratory Epithelium
As reported previously (4), when the culture of cells (MSCs and HS68) reached 70% confluence, culture medium was replaced completely with small airway growth medium (SAGM; Cambrex BioScience, Walkersville, MD, USA) to induce a respiratory epithelial differentiation. The cells were visualized by optical microscopy (Eclipse TE2000-U, Nikon, Tokyo, Japan) throughout the culture period.
Five days after the induction of the differentiation, the cells were harvested and lysed to isolate RNA. Reverse transcriptase polymerase chain reaction (RT-PCR) was performed according to previously published techniques (45). The total RNA was isolated from undifferentiated and differentiated cells using Trizol reagent (Invitrogen, La Jolla, CA, USA) according to the manufacturer's instruction. Complementary DNA (cDNA) was synthesized from 2.5 μg total RNA using SuperScript II reverse transcriptase (Invitrogen), oligo-dT primer, 5x first-strand buffer, dNTP, dTT, and RNaseOUT ribo-nuclease inhibitor. Two sequential PCR reactions were performed to test for the presence of a surfactant protein C (SP-C) gene. The glyceraldehyde-3-phosphate dehydrogenase (GAPDH) gene was tested as a control for the quality and quantity of RNA. One microliter of cDNA (200 ng/μl) was used in the first PCR amplification using the following conditions and sequence-specific primers: 5 min hot start at 95°C, 30 cycles of 95°C for 30 s, 57°C for 30 s, 72°C for 30 s, and a final extension for 5 min at 72°C; 5′-AAAGAGGTCCTGATGGA GAGC-3′ and 5′-TAGATGTAGTAGAGCGGCACCT-3′, which amplify a product of 456 base pairs. One microliter of the product from the first reaction was used as a template for the second PCR amplification step, using the same cycling conditions outlined above but with the sequence-specific primers, 5′-AACGCCTTCT TATCGTGGTG-3′ and 5′-GTGAGAGCCTCAAGAC TGG-3′, which amplify a product of 313 base pairs. The PCR products were resolved by agarose gel electrophoresis and visualized by ethidium bromide staining.
For the immunostaining procedure, the cells in SAGM by 0 and 5 days were fixed in 4% paraformaldehyde for 10 min, and incubated overnight at 4°C with pro SP-C (Chemicon, Temecula, CA, USA). The secondary Cy-3 goat anti-rabbit antibody (Jackson ImmunoResearch, West Grove, PA, USA) was incubated with the cells for 30 min at room temperature. Nuclear counterstaining was performed using 4′,6-diamidino-2-phenyl-indole (DAPI), and the images were captured on a Nikon TE 2000-U using a Nikon digital camera. For flow cytometric analysis of pro SP-C expression, undifferentiated and differentiated cells by day 0 and day 5 in SAGM, respectively, were suspended in Cytofix/Cytoperm fixation/permeabilization solution (Becton-Dickinson, San Jose, CA, USA), and were washed with Perm/Wash buffer (Becton-Dickinson). After blocking with 10% goat serum in Perm/Wash buffer for 45 min on ice, the cells were incubated with rabbit anti-human pro SPC antibody (at 1:200 dilution) (Chemicon) in the blocking solution for 45 min on ice. The cells were resuspended in Perm/Wash buffer after incubation with goat anti-rabbit IgG conjugated with fluorescein isothiocyanate (FITC) (at 1:200 dilution) (Jackson ImmunoResearch) for 45 min on ice, and analyzed by flow cytometer (Becton-Dickinson).
Animal Model
All the procedures and experimental protocols were reviewed and approved by the Institutional Animal Care and Use Committee of Samsung Biomedical Research Institute. This study also followed the institutional and National Institutes of Health guidelines for laboratory animal care. Timed-pregnant Sprague-Dawley rats were purchased from Orient Co. (Seoul, Korea), and housed in individual cages with free access to water and laboratory chow. The rats were delivered spontaneously. The experiment began within 10 h after birth and was continued until postnatal day (P) 14. One hundred fifty-eight rat pups were divided into the following six groups: normoxia control group (NC, n = 10); normoxia with intraperitoneal human UCB-derived MSCs transplantation group (NP, n = 14); normoxia with intratracheal MSCs transplantation group (NT, n = 14); hyperoxia control group (HC, n = 40); hyperoxia with intraperitoneal MSCs transplantation group (HP, n = 40); and hyperoxia with intratracheal MSCs transplantation group (HT, n = 40). The rat pups from the normoxia groups were kept in a standard cage with a nursing rat at room air, and the pups from the hyperoxia groups were kept in a standard cage with a nursing rat within a 50-L Plexiglas chamber in which hyperoxic condition (95% oxygen concentration) was maintained throughout the experiment. The humidity was maintained at 50%, and the environmental temperature was maintained at 24°C. The nursing mother rats were rotated daily between litters in the normoxia and hyperoxia groups to avoid oxygen toxicity.
We also performed an additional set of experiments comparing the effects of intratracheal administration of fibroblasts and UCB-derived MSCs to verify that the beneficial effects described in this study are specific to UCB-derived MSCs. Animals for this separate study were divided into four groups: normoxia control group (NC2, n = 4), hyperoxia control group (HC2, n = 6), hyperoxia with intratracheal fibroblasts (HS68) transplantation group (HF, n = 9), and hyperoxia with intratracheal MSCs transplantation group (HT2, n = 9).
Transplantation of Donor Cells
The human UCB-derived MSCs from the 5th passage from a single donor were labeled using a PKH26GL Red Fluorescent Cell Membrane Labeling Kit (Sigma-Aldrich, St. Louis, MO, USA) for transplantation according to the manufacturer's protocol. The donor cells were transplanted at P5. For the donor cell transplantation, 2 × 106 MSCs in 0.2 ml phosphate-buffered saline (PBS, pH 7.4) were administered intraperitoneally (NP and HP) and 5 × 105 cells (MSCs or fiborblasts) in 0.05 ml PBS were administered intratracheally (NT, HT, HT2, and HF). As we expected better delivery of donor cells with direct local administration than with indirect systemic transplantation, one fourth dosage of cells compared to that of systemic intraperitoneal administration was given for local intratracheal MSCs transplantation.
For intratracheal transplantation, the rats were anesthetized with an intraperitoneal injection of a ketamine and xylazine mixture (45 mg/kg and 8 mg/kg, respectively), and restrained on a board at a fixed angle. The MSCs or fibroblasts were administered into the trachea through a 26-gauge needle syringe. After the procedure, the animals were allowed to recover from the anesthesia, and were returned to their dam. There was no mortality associated with the transplantation procedure.
Tissue Preparation
All tissue preparation procedures for the various analyses were performed only in the surviving animals at P14. The animals were anesthetized with sodium pentobarbital (100 mg/kg). The lungs were exposed by a thoracotomy and transcardiac perfusion with ice-cold PBS. The lungs were fixed by the tracheal instillation of a 4% formaldehyde solution at a constant inflation pressure of 25 cm H2O (n = 6, 4, 8, 8, 11, 4, 13, 6, 10, and 7 in the NC, NC2, NP, NT, HC, HC2, HP, HF, HT, and HT2 groups, respectively). The trachea was then ligated and the lungs were removed and immersed in the same fixative overnight at room temperature. The fixed right lungs were embedded in paraffin, and the left lungs were embedded in an optimal cutting temperature (OCT) compound (SAKURA 4583, Sakura, Torrance, CA, USA). Blocks of the OCT compound were sectioned at 10 μm on a cryostat (Shandon Cryotome, Thermo Electron Co., Waltham, MA, USA) and stored in a deep freezer until analyzed by immunohistochemistry. Transverse serial sections (4 μm thick) were taken from the paraffin blocks of the right middle and lower lobe for morphometric and some immunohistochemistry analyses. For the scanning electron microscopy (SEM) examination, paraffin blocks (6 × 6 × 3 mm) of the right upper lobe of the lung (n = 3 in each group) were dehydrated with ethanol and isoamyl acetate, further processed using the critical point method, coated with gold (100 A), and examined with a examined by SEM (JSM-5300, Jeol, Tokyo, Japan). Particular attention was paid to the expansion pattern of the terminal airspaces and to the surface of conducting airways.
The lungs were excised, frozen in the liquid nitrogen, homogenized, and the myeloperoxidase activity, α-smooth muscle actin (SMA) protein, collagen levels as well as RT-PCR of the tissue cytokines such as tumor necrosis factor (TNF)-a, interleukin (IL)-6, and transforming growth factor (TGF)-β were measured (n = 4, 6, 6, 6, 8, and 6 in the NC, NP, NT, HC, HP, and HT group, respectively).
Morphometric Analysis
Sections (4 μm thick) from the paraffin blocks were stained with hematoxylin and eosin. Two sections per rat were chosen randomly, and three microscopic random fields of the distal lung were analyzed in each section. The level of alveolarization was determined by measuring the mean linear intercept (MLI) and mean alveolar volume. Briefly, images of each field were captured using a pixera 600CL-CU digital camera (Pixera Corporation, Los Gatos, CA, USA) through an Olympus BX50 microscope (Olympus, Tokyo, Japan) at 10x or 20x objectives and saved as JPEG files. The mean inter-alveolar distance was measured as the mean linear intercept (MLI), by dividing the total length of the lines drawn across the lung section by the number of intercepts encountered, as described by Cooney and Thurlbeck (10). The mean alveolar volume was calculated using the method reported by Snyder et al. (8,38). Briefly, a grid containing equally spaced crosses was placed on a uniformly enlarged photomicrograph of each lung field. The diameters (R) of the alveoli containing a cross were measured along the horizontal axis of the cross. The cube of the alveolar diameter times % and divided by 3 (ℓ3π/3) was used to estimate the mean alveolar volume. The analysis of each section was carried out in a blinded manner.
Quantification of the PKH26-Positive Cells
Cryosections (10 μm thick) were mounted with a Vector shield mounting solution containing DAPI (H-1200, Vector, Burlingame, CA, USA). The cell counts for the transplanted or donor-derived cells were measured using PKH26 red fluorescence, as described above after combining the 20x objective images of the DAPI-stained nuclei signals. Five fields per section were selected randomly, focused, and counted with the naked eye under a fluorescence microscope (Nikon E600, Nikon, Tokyo, Japan) using a filter to detect the PKH26 red fluorescence. The PKH26 red fluorescence was counted manually and averaged per high-power field (HPF) in a single animal. Two random sections per animal were evaluated in a blinded manner.
TUNEL Staining
The immunofluorescent terminal deoxynucleotidyl transferase-mediated deoxyuridine triphosphate nick end labeling (TUNEL) technique (kit S7110 ApopTag, Chemicon, Temecula, CA, USA) was used to determine the level of apoptosis in the 4-μm-thick paraffin sections of the lung according to the manufacturer's protocol. The slides were mounted with a vector shield mounting solution containing DAPI (H-1200, Vector) and visualized by fluorescent microscopy (Nikon E600, Nikon) at 20x objective. The number of TUNEL-positive cells was determined in 10 nonoverlapping random fields per animal in a blinded manner.
Myeloperoxidase Activity (MPO)
The MPO activity in the homogenized lung tissues, which is an indicator of neutrophil accumulation or activity, was determined using a modification of the method reported by Gray et al. (14). One unit of MPO activity was defined as the quantity of enzyme degrading 1 μmol of peroxide/min.
Semiquantitative RT-PCR
The total RNA in the sample was extracted using a RNA Trizol kit (Invitrogen Corporation, Carlsbad, CA, USA) according to the manufacturer's protocol. One mi-crogram of RNA was used to produce the cDNA using a Protoscript® II RT-PCR kit (New England Biolabs, Ipswich, MA, USA). The PCR primers for the rat TNF-α, IL-6, and TGF-β, and GAPDH were designed with Primer3 (Whitehead Institude, Cambridge, MA, USA), and synthesized from Bioneer Inc. (Bioneer, Daejeon, Korea). The following primers sequences were used: TNF-α (sense 5′-TACTGAACTTCGGGGTGATCGG TCC-3′, antisense 5′-CAGCCTTGTCCCTTGAAGA GAACC-3′); TGF-β (sense 5′-CAACTGTGGAGCAA CACGTAGA-3′, antisense 5′-CAACCCAGGTCCTTC CTAAAGT-3′; IL-6 (sense 5′-CACCAGGAACGAAA GTCAACTC-3′, antisense 5′-GGAAGCATCCATCAT TTCTTTG-3′); GAPDH (sense 5′-CTCTACCCACGG CAAGTTCAA-3′, antisense 5′-GGGATGACCTTGCC CACAGC-3′).
For each reaction, 1 μl of cDNA was placed in a 19-μl reaction mixture PCR Master Mix (Bioneer), and 0.5 μM of each primer (Bioneer). All amplification reactions were carried out in a T1 thermocycler (Biometra, Goettingen, Niedersachsen, Germany). The cycle profile involved 33 cycles of the following: 60 s at 95°C, 60 s at X°C, where X is the annealing temperature for each pair of cytokine primers (60°C for TNF-α, 56°C for TGF-β, 55°C for IL-6, and 57°C for GAPDH), and 60 s at 68°C. The PCR products were separated by 1.2% agarose gel electrophoresis, visualized by ethidium bromide, and scanned using a Gel Doc 2000 analyzer (Bio-Rad, Hercules, CA, USA). The expression level of each gene was semiquantified by densitometric analysis using Quantity One software (Bio-Rad). The relative expression levels were estimated from the density ratio of the cytokines to GAPDH (control).
Western Blot for α-SMA
The frozen lung samples were homogenized in a cold buffer (50 mM Tris-HCl, pH 7.4), EDTA (1 mM), EGTA (1 mM), PMSF (1 mom), KCl (42 mM), and MgCl2 (5 mM). The samples were centrifuged at 7000 rpm for 20 min at 4°C to remove the cellular debris. The protein content in the supernatant was determined using the Bradford method, with bovine serum albumin (Sigma-Aldrich) as the standard. Western blot analysis was carried out using the standard method and enhanced chemiluminescence detection reagents (ECL- kit, Amersham Pharmacia Biotech, Piscataway, NJ, USA). Monoclonal α-SMA (1:500, Sigma-Aldrich) and anti-α-tubulin (1:1000, Calbiochem, Darmstadt, Hessen, Germany) antibodies, and the anti-mouse IgG horseradish peroxidase (HRP) antibody (1:1,000, Dako, Carpinteria, CA, USA) were used. The α-SMA expression levels were estimated from the density ratio of α-tubulin. Densitometry was performed using Image J (NIH, Bethesda, MA, USA).
Measurement of Collagen
The lung collagen level was determined from the total soluble collagen using a Sircol collagen assay kit (Bio-color Ltd., Newton Abbey, UK) according to the manufacturer's instructions. The lungs were homogenized in 5 ml of a 0.5 M acetic acid solution containing 1 mg pepsin (Sigma Chemical Co., St. Louis, MO, USA) per 10 mg tissue. Each sample was incubated for 24 h at 4°C with constant shaking. After centrifugation, 100 μl of each supernatant was assayed. Sircol dye reagent (1 ml) that specifically binds to collagen was then added to each sample and mixed for 30 min. After centrifugation, the pellet was suspended in 1 ml of an alkali reagent (0.5 M NaOH) included in the kit, and the OD was evaluated with a spectrophotometer at a wavelength of 540 nm.
The values in the test samples were compared with those obtained with the standard collagen solutions provided by the manufacturer, which were used to construct a standard curve.
Immunohistochemistry
SMA expression in the lung tissue was evaluated from eparaffinized 4-μm-thick lung sections using the avidin-biotin-peroxidase method. After quenching of the endogenous peroxidase activity with 3% H2O2 for 20 min, the sections were incubated with the mouse anti-SMA antibody (1:200 DAKO, Carpinteria, CA, USA). The immune complexes were visualized using the relevant peroxidase-coupled secondary antibody provided in the LSAB™2 Kit (DAKO).
Immunofluorescence histochemistry for donor cell localization was performed on the 10-μm-thick cryostat sections. The following primary antibody dilutions were used: 1:500 for pro SP-C (rabbit polyclonal; Sterologicals Co. Lake Placid, NY, USA), 1:100 for aquaporin 5 (rabbit polyclonal; Chemicon,), 1:50 for vimentin (mouse monoclonal; Chemicon), 1:100 for SMA (Spring Bioscience, Fremont, CA, USA), and 1:100 for monocyte/macrophages (CD68) (ED-1, mouse monoclonal, Chemicon). Alexa Fluor 488 goat anti-mouse IgG (1:500, Molecular Probes, Eugene, OR, USA) and Cascade Blue goat anti-mouse IgG (1:200, Molecular Probes) were used as the secondary antibodies for immunofluorescence. The frozen lung sections were fixed with 4% paraformaldehyde for 30 min and treated with 1% bovine serum albumin containing 0.1% Triton for a further 30 min. The sections were then blocked with 3% normal goat serum containing 0.1% Triton X-100 in PBS for 1 h at room temperature. The sections were incubated with the primary antibodies overnight at 4°C, rinsed in PBS, and incubated with the secondary antibodies in a blocking solution for 1 h. The confocal microscopy examination was carried out using a Biorad Radiance 2100 (Bio-Rad) with a krypton/argon laser and the images were generated using lasershop 2000 software (Bio-Rad,). The cells were considered to be double-labeled if colabeling with the relevant morphology had occurred when viewed in the x–z cross-sections, which was produced by an orthogonal reconstruction from the z-series (z-step, 1 or 2 μm) taken with 60x or 80x objectives. The images were pseudocolored using image processing software (Adobe Photoshop, San Jose, CA, USA).
Statistical Analysis
The data are expressed as the mean ± SEM. Survival curve comparisons were performed using Kaplan-Meier analysis followed by a log rank test. For continuous variables with a normal distribution, multiple group comparisons were performed using t-test with a Bonferroni correction. Variables not showing a normal distribution, such as the cell counts, were analyzed using a Wilcoxon Rank test with Bonferroni correction for the multigroup comparison. All data were analyzed using the SAS software program, SAS Enterprise Guide 3.0 (SAS Institute Inc., Cary, NC, USA). Values of p < 0.05 were considered significant.
Results
In Vitro Differentiation of MSCs to Respiratory Epithelium
While the undifferentiated cells maintained a fibroblast-like morphology (Fig. 1A: a), after 5 days of induction in SAGM, the human UCB-derived MSCs differentiated and showed an epitheloid morphology (Fig. 1A: b). In contrast, HS68 showed no morphological changes after 5 days of induction in SAGM (Fig. 1A: c, d). The presence of SP-C, a specific marker of type II pneumocytes, was confirmed to be in the differentiated MSCs by both RT-PCR (Fig. 1B: a) and immunofluorescence staining (Fig. 1C: b, c) at day 5, but not in the HS68 (Fig. 1B: b, 1C: e, f). Specific pro SP-C protein expression examined by flow cytometry was observed only in the differentiated MSCs (Fig. 1D: a), but not in the HS68 fibroblasts (Fig. 1D: b).

In vitro differentiation of human umbilical cord blood (UCB)-derived mesenchymal stem cells (MSCs) to respiratory epithelium. (A) Morphology of MSCs and HS68 with small airway growth media (SAGM) induction observed by light microscope; undifferentiated MSCs appeared fibroblast like cells that have a spindle and bipolar shape (a), differentiated MSCs induced in SAGM showed epitheloid morphological changes by day 5 (b), HS68 without SAGM induction (c) and with SAGM induction by day 5 (d) showed no morphological changes (scale bars: 100 μm). (B) mRNA of surfactant protein C (SP-C) was expressed in differentiated MSCs only (a), but not in HS68 (b) with SAGM induction by day 5 (M, marker; d, day). (C) Immunofluorescence images revealed pro SP-C-positive red cells only in differentiated MSCs (b, c) (scale bars: 25 μm), not in HS68 (e, f) (scale bars: 50 μm) with SAGM induction by day 5. Nuclei were stained with DAPI (blue; a, c, d, f). (D) Flow cytometric analysis showed pro SP-C expression in differentiated MSCs (a), but not in HS68 (b). Black line: isotype control; red line: SAGM induction day 0; green line: SAGM induction day 5.
Histological Evaluation by Optical Microscopy and SEM
Although the hyperoxia-induced decrease in the survival rate was not significantly improved (43% in HC, 53% in HP, 40% in HT, and 100% in NC, NP, and NT), intratracheal MSCs transplantation significantly attenuated the hyperoxia-induced histopathological lung injuries in the surviving rat pups at P14. Figure 2 presents typical photomicrographs showing the histopathological differences observed by optical microscopy (Fig. 2A) and SEM (Fig. 2B) in each experimental group. Both the optical microscopy and SEM examination revealed uniform and small alveoli in the normoxia control group (Fig 2A, B in NC), and the human UCB-derived MSCs transplantation to the normoxic newborn rats did not cause any significant histological abnormalities in the P14 lungs (Fig. 2A, B in NP, NT). In HC, impaired alveolar growth, as evidenced by the fewer and larger alveoli (Fig. 2A in HC), focal airspace enlargement, a heterogeneous alveolar size, and more corrugated alveolar surface of the lung (Fig. 2B in HC) were observed. After MSCs transplantation, the hyperoxia-induced morphological changes and impaired alveolar growth were attenuated, particularly in HT (Fig. 2A, B in HT) when compared to HP (Fig. 2A, B in HP).
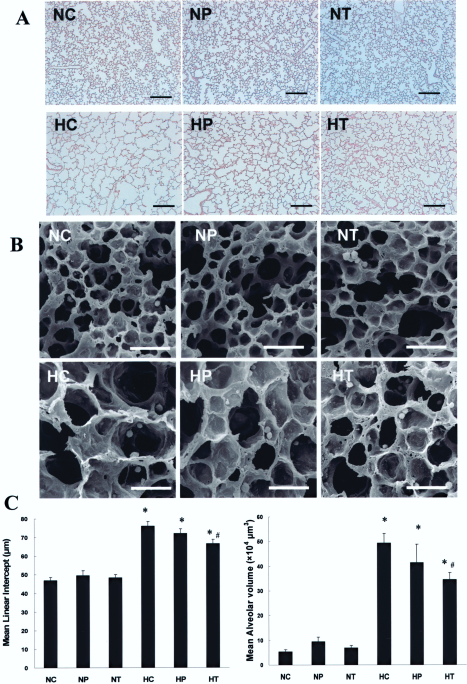
Histology and morphometric analysis of the surviving P14 rat lung. (A) Representative optical microscopy photomicrographs of the lungs stained with hematoxylin and eosin (scale bars: 200 μm). (B) Scanning electron microscopy image of the distal lungs (scale bars: 50 μm). (C) Degree of alveolarization measured by the mean linear intercept (left) and mean alveolar volume (right). Data are mean ± SEM; n = 6, 8, 8, 11, 13, and 10 in NC, NP, NT, HC, HP, and HT. *p < 0.01 versus NC; #p < 0.05 versus HC. NC, normoxic control rats; NP, normoxic rats with intraperitoneal human UCB-derived MSCs transplantation; NT, normoxic rats with intratracheal MSCs transplantation; HC, 95% hyperoxia-exposed control rats; HP, hyperoxia-exposed rats with intraperitoneal MSCs transplantation; HT, hyperoxia-exposed rats with intratracheal MSCs transplantation.
In morphometric analyses, the mean linear intercept (MLI) and mean alveolar volume, indicating the size and volume of the alveoli, respectively, were significantly higher in HC (p < 0.01) than those in the normoxia groups. These hyperoxia-induced abnormalities in morphometry were significantly attenuated in HT (p < 0.05 vs. HC), but not in HP (Fig. 2C). Furthermore, the significant attenuation of the hyperoxia-induced morphometric abnormalities with the intratracheal MSCs transplantation (HT2; p < 0.01 vs. HC2, p < 0.01 vs. HF) was not observed with the intratracheal fibroblast transplantation. These findings indicate that these beneficial effects are specific to UCB-derived MSCs (Fig. 3).

Comparisons of degree of alveolarization measured by the mean linear intercept (upper) and mean alveolar volume (lower) between P14 rat lungs exposed in hyperoxia with intratracheal fibroblast transplantation and those with intratracheal MSCs transplantation. Data are mean ± SEM; n = 4, 4, 6, and 7 in NC2, HC2, HF, and HT2. *p < 0.01 versus NC2; **p < 0.001 versus NC2; #p <0.01 versus HC2; † p <0.01 versus HF. NC2, normoxic control rats; HC2, hyperoxic control rats; HF, hyperoxic rats with intratracheal fibroblast transplantation; HT2, hyperoxic rats with intratracheal MSCs transplantation.
The deposition of PKH26 red fluorescence-positive donor cells was observed only in the MSCs transplantation groups, but not in NC and HC (Fig. 4). The number of donor cells identified per lung field was significantly larger in the intratracheal MSCs transplantation groups (NT and HT) (p < 0.05) than in the intraperitoneal transplantation groups (NP and HP). However, there were no significant differences in the donor cells between the hyperoxia and normoxia groups (NP vs. HP, NT vs. HT).

Donor cell localization in the lung of the P14 rats. Top panels: fluorescent microscopic observation of the PKH26-labeled human UCB-derived MSCs (donor cells, arrow, red) localized in the lungs of the P14 newborn rats and the nuclei labeled with DAPI (blue) (scale bars: 25 μm). Bottom: number of PKH26-positive cells in the lung per high-power field. Data are mean ± SEM; n = 6, 8, 8, 11, 13, and 10 in NC, NP, NT, HC, HP, and HT. *p < 0.05 versus NP.
TUNEL Staining
The number of TUNEL-positive cells in the lung of the P14 rats per high-power field was significantly higher in HC (p < 0.001) than in the normoxia groups (Fig. 5). This hyperoxia-induced increase in the number of TUNEL-positive cells was significantly attenuated after both intraperitoneal and intratracheal MSCs transplantation (p < 0.05 vs. HC).

TUNEL-positive cells in the distal lungs of the P14 rat pups. Top panels: TUNEL-positive cells were labeled with FITC (green, arrow) and the nuclei were labeled with DAPI (blue) (scale bars: 25 μm). Bottom: number of observed TUNEL-positive cells per high-power field. Data are mean ± SEM; n = 6, 8, 8, 11, 13, and 10 in NC, NP, NT, HC, HP, and HT. *p < 0.01 versus NC; **p < 0.001 versus NC; #p < 0.05 versus HC.
Myeloperoxidase Activity
In HC, the MPO activity was significantly higher (p < 0.001) than in the normoxia groups (NC, NT, and NP) (Fig. 6A). This hyperoxia-induced increase in MPO activity was significantly attenuated in both HT (p < 0.001 vs. HC) and HP (p < 0.01 vs. HC), and this attenuation was more significant in HT (p < 0.01) than in HP.

Myeolperoxidase activity and cytokine expressions in the P14 rat lungs. (A) Activity of myeloperoxidase (MPO) and (B) representative RT-PCR blots (at top) and densitometric histograms (below) for TNF-α, IL-6, and TGF-β in the P14 rat lungs. Data are mean ± SEM; n = 4, 6, 6, 6, 8, and 6 in NC, NP, NT, HC, HP, and HT. *p < 0.001 versus NC; #p < 0.05 versus HC; ##p < 0.01 versus HC; ###p < 0.001 versus HC; †p < 0.05 versus HP; ††p < 0.01 versus HP.
mRNA Expression of TNF-α, IL-6, and TGF-β
The mRNA levels of TNF-α, IL-6, and TGF-β, which were analyzed by semiquantitative RT-PCR, were significantly higher in HC (p < 0.001) than in the normoxia groups (NC, NT, and NP) (Fig. 6B). The increased IL-6 levels observed in HC were significantly attenuated in both HT (p < 0.01 vs. HC) and HP (p < 0.05 vs. HC), and this attenuation was more profound in HT (p < 0.05) than in HP. The hyperoxia-induced increase in TNF-α and TGF-β (p < 0.001 vs. NC) was significantly attenuated only in HT (p < 0.01 vs. HC, p < 0.05 vs. HP), but not in HP.
α-SMA and Collagen
The α-SMA and collagen levels were measured to assess the degree of lung fibrosis. The level of the α-SMA protein was significantly higher in HC (p < 0.001) than in the normoxia groups (NC, NT, and NP). This hyperoxia-induced increase in α-SMA was significantly attenuated only in HT (p < 0.01 vs. HC, p < 0.01 vs. HP), but not in HP (Fig. 7A).

Expression of α-smooth muscle actin (SMA) and level of collagen in the P14 rat lungs. (A) Representative Western blots of the protein extracts (top), densitometric histograms (middle), and avidin-biotin-peroxidase immunostaining (brown, bottom) for SMA in the P14 rat lung (scale bars: 100 μm). (B) Collagen level of the P14 rat lung tissue. Data are mean ± SEM; n = 4, 6, 6, 6, 8, and 6 in NC, NP, NT, HC, HP, and HT. *p < 0.001 versus NC; #p < 0.01 versus HC; †p < 0.01 versus HP.
The lung collagen levels at P14 were significantly higher in HC (p < 0.001) than in the normoxia groups (NC, NP, and NT). This hyperoxia-induced increase in the lung collagen levels was significantly attenuated only in HT (p < 0.01 vs. HC, p < 0.01 vs. HP), but not in HP (Fig. 7B).
Colabeling of Localized Donor Cells In Vivo
In order to determine the differentiation of the transplanted MSCs into the lung tissue in vivo, the colocalization of PKH26 red fluorescence with various lung cell markers was examined by immunofluorescence staining. A few definite colabelings of PKH26 with pro SP-C, a type II alveolar epithelial cell-specific marker, were observed (Fig. 8A). There was no colocalization of ED-1, a specific marker of alveolar macrophages, with this double labeling with PHK26 and pro SP-C, thus excluding the possibility that this colocalization was an incidental finding due to phagocytosis by the alveolar macrophages, rather than a true in vivo differentiation (Fig. 8B). Colocalization of the PKH26 red fluorescence with Aquaporin A, an indicator of the type I alveolar epithelial cell-specific marker, vimentin, an indicator of fibroblast, or α-SMA, an indicator of myofibroblasts was not observed in the present study.

Intratracheally transplanted human UCB-derived MSCs localizing to the hyperoxia-induced damaged lung of the P14 rat express the marker for type II pneumocytes. (A) Confocal 3D reconstruction and six consecutive sections of the z-series through the same cell or a transplanted donor cell showing PKH26 (a, d) and Pro SP-C (b, e) immunoreactivity separately or as a merged image (c, f) (scale bar: 15 μm, z-series distance: 1 μm). (B) Confocal 3D reconstruction and z-series consecutive sections of a transplanted donor cell (arrow) showing PKH26 (a, e) and pro SP-C (b, f), but not ED-1 (c, g) immunoreactivity separately or as a merged image (d, h) (scale bars: 25 μm, z-series distance: 2 μm). Bottom values: relative distance in the z-direction from the reference confocal plane.
Discussion
In this study, although the survival rate was not significantly improved, the transplantation of human UCB-derived MSCs to the wild-type newborn rat pups significantly attenuated the hyperoxia-induced lung injury in the surviving animals. This protective effect was associated and probably mediated by the downmodulation of the pulmonary inflammatory and the ensuing fibrotic responses. The engraftment of donor cells and attenuation of the hyperoxia-induced lung injury were more prominent with the intratracheal administration of MSCs than with intraperitoneal transplantation. Furthermore, the human UCB-derived MSCs had differentiated successfully into type II pnumocytes in vitro. Moreover, a few transplanted MSCs localized and assumed the lung cell phenotypic characteristics in the in vivo damaged lung. Taken together, these findings suggest that the intratracheal administration of human UCB-derived MSCs might be a possible candidate for the new therapeutic modality for the hyperoxia-induced neonatal lung diseases, such as clinical BPD.
Although BM-derived MSCs and UCB-derived MSCs have a similar morphology and immuno phenotype (20), they have distinct characteristics including different differentiation tendencies (7) and unique cytokine production properties in vitro (18,27). However, it is unclear if there are any in vivo differences in the various characteristics between BM- and UCB-derived MSCs. Human UCB-derived MSCs are primitive in nature and can differentiate into various tissues (17,21,25,45), including lung cells in vitro (4). Furthermore, human UCB-derived MSCs can be obtained easily and readily expanded ex vivo to a concentration sufficient for cell replacement therapy on account of their higher proliferation capacity compared with adult BM-derived MSCs (20,45). Moreover, UCB-derived MSCs can be transplanted between HLA-incompatible individuals because they barely elicit alloreactive lymphocyte proliferate responses and modulate the immune responses (19,23,41). In this study, the xenotransplantation of MSCs to wild-type immunocompetent newborn rats was accomplished successfully, no effect was seen when the lung was normal, and beneficial effects on hyperoxic lung injury were demonstrated. Overall, these findings suggest that human UCB-derived MSCs might be a useful alternative source for MSCs-based therapies.
Although UCB-derived MSCs look similar to fibroblasts in morphology, our in vitro data of multipotentiality could easily differentiate them from fibroblasts. Furthermore, in our additional in vivo experiments, we have also confirmed that the protective effects observed in the present study are specific to UCB-derived MSCs, but not to fibroblasts.
The optimal route for stem cell transplantation in a lung injury has not been determined yet. In a bleomycin-induced lung injury, some soluble factors are produced from injured lungs but not from normal lungs, which allows systemically administered MSCs to be homed to an injured lung (33,34). In the present study, the MSCs were given intraperitoneally instead of by the intravenous or intra-arterial route for systemic cell administration due to the small size of the newborn P5 rat pups weighing only 7–9 g. This intraperitoneal route was also used in the earlier studies in the newborn mice (13,44), and P7 rats (29), and the intraperitoneally injected substances has been known to be absorbed into both systemic and portal circulations (12). Further studies will be necessary to confirm the efficacy of intraperitoneal route for systemic cell administration.
Although one fourth dosage of the MSCs was given intratracheally than those given intraperitoneally, significantly increased numbers of donor cells were localized in the intratracheal administration groups (NT and HT) than in the intperitoneal transplantation groups (NP and HP) regardless of the lung injury state in this study. Furthermore, the hyperoxia-induced decrease in alveolarization was improved more prominently in HT than in HP, and the hyperoxia-induced inflammatory responses and the ensuing fibrotic reaction, such as the increased MPO activity and TNF-α, IL-6, TGF-β, α-SMA, and collagen, were also attenuated more markedly in HT than in HP. Taken together, our data suggest that the local intratracheal administration of MSCs is a more effective transplantation method than the systemic intraperitoneal administration for the treatment of hyperoxia-induced neonatal lung disease.
The protective mechanism of human UCB-derived MSCs against a hyperoxia-induced newborn lung injury remains to be determined. In animal studies, the hyperoxia-induced lung injury was mediated by inflammatory cells, such as neutrophils and macrophages (43), as well as by proinflammatory cytokines, such as TNF-α and IL-6 (5,9,26,28,43). The hyperoxia-induced increase in mortality and the number of TUNEL-positive cells were observed in newborn mice overexpressing IL-6 compared with wild-type mice (9). Oritz et al. (32) reported that the BM-derived MSCs protected the lung tissue from bleomycin-induced lung injury by blocking TNF-α and IL-1. In the present study, the hyperoxia-induced lung injury such as decreased alveolarization and an increase in the number of TUNEL-positive cells was accompanied by pulmonary inflammatory responses, as evidenced by the increase in MPO activity and the levels of TNF-α and IL-6 mRNA. Human UCB-derived MSCs transplantation significantly attenuated the hyperoxia-induced lung injury and the inflammatory responses. Moreover, this attenuation was more prominent in HT than in HP. These findings indicate that the protective effects of MSCs therapy against a hyperoxia-induced lung injury are associated or mediated mainly by its anti-inflammatory effects.
TGF-β1 is a key fibrogenic cytokine, and activates fibroblasts differentiation into myofibroblasts (16,37). The activated myofibroblasts play an important role in the process of lung fibrosis with α-SMA being an important key fibroblast myogenic marker (35). Kwong et al. (22) reported that lung inflammatory cells, such as neutrophils and macrophages, are a source of TGF-β1 in the lung of preterm infants. Therefore, the antifibrotic effects of intratracheal human UCB-derived MSCs observed in this study, as evidenced by significant attenuation of the hyperoxia-induced increase in TGF-β, α-SMA, and collagen, might be due to their anti-inflammatory effects.
Although the anti-inflammatory action of MSCs has been reported in renal (36), myocardiac (31), and bleomycin-induced pulmonary injuries (33), the mechanism of this anti-inflammatory action is unclear. Aggarwal and Pittenger (2) suggested that human BM-derived MSCs in an inflammatory environment might alter the cytokine secretion profile of dendritic cells and T-cell subsets, resulting in a shift from a proinflammatory environment toward an anti-inflammatory or a tolerant cell environment. Torrente and Polli (40) also suggested that possible interpretations for beneficial outcome observed after MSC transplantation into neurodegenerating tissues might include trophic factor delivery and immunomodulation. Further studies will be needed to determine if there are some humoral substances secreted from the engrafted stem cells that might mediate this anti-inflammatory effect.
Several studies have suggested that BM-derived MSCs reconstitute the lung parenchymal cells such as type I pneumocytes, type II pneumocytes, or fibroblasts both in vitro and in vivo (1,15,33). In concordance with Berger et al. (4), we have demonstrated the successful in vitro differentiation of human UCB-derived MSCs to type II alveolar cells. The colocalization of PKH26 donor cells with pro SP-C, a type II pneumocyte-specific marker, was also observed in the hyperoxia-induced lung tissue, suggesting that human UCB-derived MSCs can differentiate into the lung parenchymal cells in vivo. However, this in vivo colocalization was observed in a few donor cells, and the mere colocalization does not warrant the protective effects of MSCs transplantation. Therefore, it still remains to be elucidated whether the engrafted donor cells exert their therapeutic effects by inducing the direct tissue repair and regeneration of damaged cells. Further studies will be necessary to clarify this.
In conclusion, our data indicate that the intratracheal rather than the intraperitoneal transplantation of human UCB-derived MSCs significantly attenuated the hyperoxia-induced lung injury such as decreased alveolarization and increased fibrosis. This protective effect appears to be mediated primarily by their anti-inflammatory effects and less possibly by their differentiation into type II pneumocytes. These findings are expected to have important therapeutic potential for the currently untreatable hyperoxic neonatal lung disease or BPD in preterm infants.
Footnotes
Acknowledgments
The authors wish to thank to Dr. Su Jin Cho for her advice on the morphometric analysis of the lung. This work was supported by the Korea Research Foundation Grant funded by the Korean Government (MOEHRD) (KRF-2005-041-E00219) and a grant funded by Medipost Research Institute.
