Abstract
A major limitation of neural transplantation studies is assessing the degree of host–graft interaction. In the present study, rat hippocampal/cortical embryonic neurons (E18) were infected with a lentivirus encoding enhanced green fluorescent protein (GFP) under control of the neuron-specific synapsin promoter, thus permitting robust identification of labeled neurons after in vivo grafting. Two weeks after transient forebrain ischemia or sham-surgery, GFP-expressing neurons were transplanted into CA1 hippocampal regions in immunosuppressed adult Wistar rats. The survival, distribution, phenotype, and axonal projections of GFP-immunoreactive (IR) positive transplanted neurons were evaluated in the sham-operated and ischemia-damaged CA1 hippocampal regions 4, 8, and 12 weeks after transplantation. In both experimental groups, we observed that the main phenotype of host-derived afferents projecting towards grafted GFP-IR neurons as well as transplant-derived GFP-IR efferents were glutamatergic in both animal groups. GFP axonal projections were seen in the nucleus accumbens, septal nuclei, and subiculum—known target areas of CA1 pyramidal neurons. Compared to sham-operated animals, GFP-IR neurons grafted into the ischemia-damaged CA1 migrated more extensively throughout a larger volume of host tissue, particularly in the stratum radiatum. Moreover, enhanced axonal sprouting and neuronal plasticity of grafted cells were evident in the hippocampus, subiculum, septal nuclei, and nucleus accumbens of the ischemia-damaged rats. Our study suggests that the adult rat brain retains some capacity to direct newly sprouting axons of transplanted embryonic neurons to the correct targets and that this capacity is enhanced in previously ischemia-injured forebrain.
Keywords
Introduction
Stroke is the third leading cause of death and the number one leading cause of disability in the US. Given the aging population and the correlation of age and stroke incidence, significant advancements in the treatment of stroke patients are vital. Indeed, both neurogenesis and endogenesis-mediated functional repair following injury to the adult CNS are quite limited (77), and currently only few stroke treatments exist, mostly focused on the immediate peristroke time frame (e.g., thrombolytics). As a result, many cerebral ischemia patients suffer long-term neurological deficits. For example, cells especially vulnerable to an ischemic insult are the neurons in the pyramidal cell layer of the CA1 hippocampal region, whose death leads to learning and memory deficits (6,37).
Progress in the field of neural stem cell biology/transplantation has highlighted the potential for cell replacement therapy as a treatment for brain/spinal cord injuries that are left untreated or undertreated. For example, human spinal neuroprecursors transplanted into the ischemia-injured spinal cord of rats ameliorated behavioral indices of spasticity (10). Daadi et al. (13) reported a reduction in motor deficits following the injection of rat medial ganglionic eminence cells into the ischemia-injured rat striatum/cortex. Indeed, previous studies have shown that embryonic tissues, stem cells, or neuroprecursor cells can survive after transplantation into intact or ischemia-damaged adult brains, can differentiate into neurons or glia, and improve functional recovery (2,7,8,12,13,61,62). Moreover, grafts transplanted (62) into the ischemia-vulnerable hippocampal CA1 region ameliorated impairment of neurological function (41,49,68,79). Functional improvement after such transplantation appears to be related to both reestablishment of damaged hippocampal circuitry and graft-derived trophic support (54,55,61,63,76). Integration into and reestablishment of host circuitry is also supported by studies showing that rat neuroprecursors transplanted into the ischemic brain expressed synaptic proteins, formed synapses with host circuits, expressed neurotransmitters including serotonin, γ-aminobutyric acid (GABA), or acetylcholine, and had electrophysiological properties resembling mature neurons (3,8). However, functional recovery has not always been well correlated with graft–host interaction or synapse formation, suggestive of other mechanisms mediating the behavioral benefits (7). Indeed, numerous mechanisms have been postulated to explain functional recovery following cell transplantation strategies, including: 1) integration into host circuitry (27,78); 2) reduced host cell death (33,38,44); 3) induction of host plasticity (59,63); and 4) increased neovascularization (32).
Unfortunately, a major limitation of many neural transplantation studies is assessing the degree of host–graft integration, thus limiting the ability to distinguish reestablishment versus trophic effect of the graft if/when a functional effect is noted. To overcome these limitations several techniques have been developed to identify transplanted tissue, including: 1) a variety of exogenous markers that stain cytoplasm and/or nuclei [e.g., phaseolus vulgaris leucoaglutinin, bromodeoxyuridine (BrdU)] (60,65); 2) xenotransplants, allowing the use of species-specific immunostaining (34,74); 3) transgenic animals in which donor-derived cells express human-specific or fluorescence markers (13,28,53); and 4) in vitro vector-mediated DNA labeling by using fluorescence-tagged plasmids, adeno-associated virus (AAV), or lentiviral constructs before the cells are transplanted (46,51). In the last decade viral-mediated DNA delivery has enabled significant progress in labeling techniques. Following in vitro infection with a viral vector encoding the foreign gene, expression of the new protein in the infected cells can be detected by autofluorescence, histochemistry, or immunocytochemistry. Moreover, by utilizing promoters that are selectively activated in a specific cell population [e.g., glial fibrillary associated protein (GFAP) promoter in astrocytes], the reporter protein expression can be limited to a cell group of interest (19,24).
In the present study, we infected a rat neuronal culture with a lentivirus vector encoding neuron-specific enhanced green fluorescent protein (EGFP) expression using the synapsin promoter (18,19,51) and transplanted those cells into the rat hippocampus (CA1) 2 weeks after transient forebrain ischemia or sham-surgery. In our study we: i) evaluated survival and distribution of grafts 4, 8, and 12 weeks after transplantation; ii) quantitatively and qualitatively evaluated putative nerve terminals contacting grafted cells; iii) compared short and long axonal sprouting of grafted neurons in sham-operated and ischemic animals; and iv) investigated the phenotype of graft-derived nerve terminals contacting host cells.
Materials and Methods
Induction of Transient Forebrain Ischemia
All procedures were approved by the Institutional Animal Care and Use Committee of the University of California, San Diego. Male Wistar rats (Harlan Industries, Indianapolis, IN), 275–325 g, were housed in standard cages with corn cob bedding with free access to food and water. Prior to cerebral ischemia animals were fasted for 12 h with ad libitum access to water. Anesthesia was induced with 4% isoflurane in oxygen. The trachea was intubated and the lungs were mechanically ventilated to achieve a PaCO2 of 35–42 mmHg, as confirmed by arterial blood gas analysis. Anesthesia was maintained with 1.5–2.0% as required during surgical preparation. A temperature probe was placed under the temporalis muscle and temperature was servo-controlled at 38°C with a heating pad and heat lamp. The tail artery was cannulated (PE-20) to permit continuous recording of blood pressure. The common carotid arteries and the right external jugular vein were exposed and the latter was cannulated (PE-50) to permit blood removal. Heparin (300 units), was administered intravenously. After completion of the surgical preparation, the inspired isoflurane concentration was reduced to 1% and a 15-min equilibration period ensued. The mean arterial pressure (MAP) was decreased to 35 ± 3 mmHg by blood withdrawal from the jugular vein and the common carotid arteries were occluded with vascular clips for 10 min. Thereafter, the blood was returned to the animal over 2 min. Protamine (3 mg) was administered intravenously, catheters were removed, and anesthesia was discontinued. Sham-operated rats were subjected to similar surgery, including carotid isolation, but neither blood withdrawal nor carotid occlusions was performed.
Lentiviral Vector Production
To construct the HIV1-SYN-EGFP lentiviral vector, the synapsin promoter-EGFP cassette was made by replacing the human cytomegalovirus (hCMV) promoter in pEGFP-N1 (Clontech, Mountain View, CA) with human synapsin (SYN) promoter. The SYN-EGFP unit was then inserted into the HIV-1 vector backbone plasmid pHIV7 containing the HIV-1 central flap sequence (cPPT) and the woodchuck hepatitis virus posttranscriptional regulatory element (WPRE). Lentiviral vectors were produced by transient cotransfection of 293T cells maintained in Dulbecco's modified Eagle's medium (DMEM) with 10% fetal calf serum (FCS). 293T cells in 150-mm dishes were cotransfected by CaPO4-DNA coprecipitation method with each HIV1 vector plasmid, pLP1 and pLP2 (Invitrogen, Carlsbad, CA), and pCMV-G. Conditioned media at day 2 and 3 posttransfection were collected, filtered through 0.45-μm filter, and concentrated by ultracentrifugation at 25,000 rpm for 2 h at 4°C with Beckman SW28 rotor. The resulting pellets were resuspended with PBS and stored at −80°C.
Isolation, Preparation, and Labeling of Rat Hippocampal/Cortical Tissue In Vitro
The Wistar pregnant rats were terminally anesthetized with an overdose of pentobarbital (100 mg/kg, IP) and the hippocampal/cortical tissue was isolated from each rat fetus at embryonic day 18 (E18). After dissection, the hippocampal/cortical tissues were dissociated with Papain System (Worthington Biochemical Corp., Freehold, NJ), following the manufacturer's instructions. Briefly, tissue was triturated in 5 ml of papain dissociation solution, followed by agitation for 20 min at 37°C in 5% CO2. The diluted cell suspension was then centrifuged at 300 rpm for 5 min. Then the cell pellet was resuspended in Deact solution (albuminovomucoid inhibitor/DNase solution) and layered on top of a discontinuous density gradient of albumin-ovomucoid inhibitor mixture and centrifugated (70 rpm) for 5 min. The resulting cell pellet was resuspended in the SYN-EGFP lentivirus solution [300 μl; 10 muliplicity of infection (MOI)] (51) and plated at 2.5 × 104 cells/well onto Nunclon uncoated six-well plates for 16 h. Culture medium contained DMEM/F12 (11330-32, Gibco), 2 mM L-glutamine, N2 supplement (17502-048, Gibco), 100 U/ml penicillin, and 100 μg/ml streptomycin. The following day, the culture medium was replaced with a fresh medium without virus and cells were cultured for an additional 3 days before cell grafting.
Grafting of Labeled Neurons Into the CA1 Region of Hippocampus
Two weeks after the surgery described above, sham (n = 9) and ischemic (n = 9) animals were anesthetized and secured into a stereotaxic apparatus (Stoelting). A 1-cm midline incision was made in the scalp and, using a dental drill burr, holes were made in the bone bilaterally using the following coordinates (in millimeters measured from bregma): −3.3 AP, ±2 L; −4.3 AP, ±3 L; and −5.3 AP, ±3.5 L. The GFP-labeled cell suspension was injected through each burr hole to a depth of 3.5 mm (from the skull surface) into the hippocampal CA1 region with a 30-gauge needle connected to a stereotaxic manipulator and a pressure-controlled microinjector (Harvard Apparatus, Holliston, MA). At each of the six sites 1 μl of cell suspension was injected (each 1 μl containing 10,000 cells). The viability of cells was tested using trypan blue before and after implantation and was routinely 75–90%. After cell grafting, the incision was cleaned with 3% H2O2 and sutured closed. Rats were then allowed to recover and survived for 4, 8, or 12 weeks. To label mitotically active cells after grafting, BrdU (B5002, Sigma; 50 mg/kg/day, IP) was injected for 4 days after transplantation. Grafted animals were immunosuppressed daily with FK-506 (Prograf; Astellas Pharma, Deerfield, IL; 1 mg/kg/day, IP) during the entire survival period.
Tissue Preparation and Immunohistochemical Staining
After the various survival periods, the animals were deeply anesthetized with pentobarbital (100 mg/kg) and transcardially perfused with 0.1 M PBS, followed by 4% paraformaldehyde (pH 7.4). The brain was removed and postfixed in buffered 4% paraformaldehyde for 24 h at 4°C. The tissue was then cryoprotected in ascending concentrations of sucrose (15–30%) with the 0.1 M phosphate buffer over 24 h. Thereafter, 30-μm-thick transverse sections were cut on a cryostat at −16°C and collected free-floating in 0.1 M PBS with 0.01% thimerosal. Free-floating sections were first incubated with a solution containing 5% normal donkey serum and 0.2% Triton X-100 in 0.1 M PBS for 60 min at room temperature. The sections were then incubated separately with each primary antibody in PBS containing 0.2% Triton X-100, 0.01% thimerosal, and 5% normal donkey serum overnight at 4°C. The following primary antibodies were used: 1) mouse anti-neuron-specific nuclear protein (NeuN or biotin-conjugated NeuN; MAB377 or MAB377B, respectively; Chemicon, 1:1000); 2) rabbit or chicken anti-green fluorescent protein (GFP; ab6556, Abcam; or AB1 6901, Chemicon, 1:5000); 3) rat anti-BrdU (OBT0030, Serotec, 1:50); 4) mouse anti-synaptophysin (Syn; MA B5258, Chemicon, 1:1000); 5) guinea pig anti-vesicular glutamate transporter 1 (VGLuT 1; AB5905, Chemicon, 1:4000); 6) guinea pig anti-VGLuT 2 (AB5907, Chemicon, 1:3000); 7) guinea pig anti-VGLuT 3 (AB5421, Chemicon, 1:10,000); 8) a mixture of guinea pig VGLuT 1-3 antibodies (at the corresponding concentrations), which visualized all glutamatergic presynaptic terminals; 9) mouse or rabbit anti-glutamate decarboxylase, 65 kDa isoform, clone GAD-6 (GAD 65; Hybridoma Bank, University of Iowa or Sigma G4913, 1:500); 10) mouse anti-glutamate decarboxylase 67 (GAD 67; MAB5406, Chemicon, 1:2000); and 11) a mixture of mouse GAD 65 and 67 antibodies (at the corresponding concentrations, which visualized all GABAergic presynaptic terminals). After washing three times for 10 min with PBS, the sections were incubated in appropriate combinations of secondary donkey anti-mouse (Alexa 647; A31571, Invitrogen), anti-rabbit (Alexa 488 or 555; A21206 or A31572, Invitrogen), anti-guinea pig (Cy3; 706-165-148, Jackson ImmunoResearch), anti-chicken IgY (Cy2, 703-226-155, Jackson ImmunoResearch), and/or donkey anti-rat (Cy3, 712-166-153, Jackson ImmunoResearch) antibodies diluted 1:250 in the same solution as the primary antiserum. The biotin conjugated NeuN antibody was visualized with avidin-Cy3 conjugate (E4142, Sigma). For visualization of BrdU, the sections were incubated in 50% formamide-2× SSC for 2 h at 65°C, 2N HCl for 30 min at 37°C, boric acid (pH 8.5) for 10 min, and then processed with immunostaining protocol as described above. 4′,6-Diamidino-2-phenylindole (DAPI; 1:1000) was used for general nuclear staining of all sections. After washing three times for 10 min with PBS, the sections were mounted on slides, air dried, and then cover-slipped with ProLong Antifade Kit (P7481, Molecular Probes).
Quantitative Assessment
Coronal brain sections (30 μm thick) from −2.8 to −5.8 mm from bregma were examined under light microscope to identify those with a clear injection tract in the pyramidal cell layer. Since the density of grafted cells in each section typically declines with distance away from the injection site, we chose to use the injection tract as a reliable landmark in selecting sections for quantification, making it possible to compare cell survival between groups. Those selected sections (3–4 per injection tract) were then immunostained for GFP (as described above) and imaged (MicroFire camera model S99809, Olympus DMLB microscope). Images of the entire section were analyzed with UTHSCSA Image Tool 3 (University of Texas, Health Science Center) using the same detection parameters (e.g., pixel threshold) for all measurements. The number of GFP-immunoreactive (IR) somata per section ± SD were compared between sham-operated and transient forebrain ischemia-operated rats surviving for 4, 8, or 12 weeks.
Proliferating cells were labeled in vivo by BrdU administration and subsequent cellular incorporation. From each animal, three sections from each graft site were selected and scanned with a confocal laser microscope (Leica Microsystems, Bannockburn, IL). Sections were scanned sequentially with each laser line through a 20× lens to produce z-series of 20 optical sections separated by 0.6 μm, starting at least 2 μm below the section surface. The percentage of BrdU/NeuN double-positive cells was calculated.
Putative presynaptic axonal terminals were visualized with synaptophysin and/or markers specific for glutamatergic and GABAergic neurons. At least five sections from each animal were scanned sequentially with each laser line through a 100× lens to produce z-series of 10 optical sections separated by 0.3-μm intervals, starting at least 2 μm below the section surface. The percentage of the cell body membrane adjacent to the bouton-like terminals was calculated in the pyramidal cell layer and graft core of sham- and ischemia-injured rats. The quantitative analysis of Syn-IR, VGluT1-3, or GAD65 and GAD67 afferents in all animal groups was compared to the occurrence of the corresponding marker around host pyramidal CA1 neurons in sham-operated rats (i.e., afferentation of host pyramidal neurons in sham-operated animals was used as a standard for comparison). The number of putative GFP-IR boutons (i.e., derived from grafted neurons) expressing glutamatergic or GABA ergic markers of presynaptic axonal terminals was analyzed in the nucleus accumbens, septal nuclei, and subiculum—target areas of CA1 pyramidal neurons. The number of putative GFP-IR axonal terminals in a volume of 60,000 μm3 was counted and the number of putative GFP-IR glutamatergic or GABAergic presynaptic axonal terminals of grafted neurons in the same axonal target areas were compared between sham-operated and ischemia-damaged rats 4, 8, or 12 weeks after cell transplantation.
Statistical Analysis
Two-way comparisons were analyzed by an unpaired t -test. Multiple comparisons were performed by one-way analysis of variance (ANOVA) and Student Newman-Keuls test when needed. Values of p < 0.05 were considered statistically significant.
Results
Characterization of In Vitro Differentiated Rat Primary Culture
The rat primary culture, infected previously with HIV1-SYN-EGFP lentivirus, progressively showed extensive axodendritic branching and the development of neuron-like morphology by the seventh day in vitro. Immunostaining cultured cells for the neuron-specific cytoskeletal protein microtubule-associated protein 2 (MAP-2) confirmed that at least 60% of the cell population was neurons. Double-labeling the cell culture for GFP and MAP-2 revealed that over 90% of infected GFP-IR neurons were also MAP-2 positive (Fig. 1A–C).

Expression of enhanced green fluorescent protein (EGFP, green) in embryonic neurons infected with synapsin (SYN)-EGFP lentivirus immediately after harvesting (E18) and then cultured for 3 days. Neurospheres (A, B) were dissociated into single cells (C) before transplantation. The GFP-expressing neurons were also positive for microtubule-associated protein (MAP-2; B, C; red). Nuclei were stained with 4′,6-diamidino-2-phenylindole (DAPI; C, blue). Scale bar: 100 μm.
Forebrain Ischemia
Transient forebrain ischemia consistently resulted in a dramatic cell loss in the CA1 region of the hippocampus. Indeed, compared to sham-operated animals, only 8 ± 3% of NeuN-IR CA1 pyramidal neurons were observed 2 weeks after transient forebrain ischemia (time of cell transplantation, see below). Sham-surgery did not result in any cell loss compared to intact animals (data not shown). Following cell transplantation, no further changes in the host CA1 pyramidal cell population were noted at any survival time after cell transplantation (i.e., 4, 8, 12 weeks after grafting; data not shown).
Cell Grafts
After transplantation into ischemic and sham-operated animals, GFP-expressing grafts were detected in all three injection sites of CA1 region in each animal. The average number of GFP-IR neurons per section in the sham-operated hippocampal CA1 regions was 600 ± 55; that is comparable to 586 ± 42 counted in the ischemia-damaged CA1. The soma of grafted neurons was quite comparable in both shape and size with host hippocampal CA1 pyramidal neurons, regardless of the experimental group. Host CA1 neurons had an average cross-sectional area (cell soma) of 123.6 ± 30.3 μm2, while that for grafted neurons in the sham and ischemic groups was 126.3 ± 28.4 and 124.1 ± 31.4 μm2, respectively. The cross-sectional area occupied by the graft core did not vary between experimental groups: 1.028 ± 0.28 and 1.171 ± 0.38 mm2 in sham-operated and ischemic animals, respectively.
While the overall area of the graft core was similar in each animal group, distribution of the GFP-IR somata was different in the sham-operated and ischemia-injured environments (Fig. 2). In sham-operated animals the majority of GFP-IR somata and their fibers were distributed in the alveolus and the superficial part of the oriens layer (Fig. 2G, J), and along the needle tract in the pyramidal layer and stratum radiatum. However, in the ischemia-damaged CA1 region, lateral spreading of GFP-IR somata was observed also in the stratum radiatum (Fig. 2H, K). In the pyramidal layer of both animal groups, GFP-IR somata and their fibers were distributed along the needle tract with minimal occupation of the pyramidal layer itself. Quantification of the grafted cell distribution in the CA1 region showed that while more than 90% of the total GFP-IR cell population was located in the alveolus and oriens layer of sham-operated animals, less than 70% were found in the same locations in ischemia-injured rats. The remaining GFP-IR cells were seen mainly in the stratum radiatum of both experimental groups (Fig. 2I).

Distribution of grafted neurons in the sham-operated (A–C, G, I, J) and ischemic-damaged (D–F, H, I, K) CA1 regions. Embryonic cells were injected bilaterally into CA1 region 3.3, 4.3, and 5.3 mm posterior from bregma. GFP-immunoreactive (IR) neurons were readily identified in each position (A–F). (A–F) Grafts from animals 4 weeks after transplantation (6 weeks after ischemia). Schematic drawings show the distribution of transplanted neurons and their fibers in the sham-operated (G) and ischemia-damaged (H) CA1 region. The majority of grafted neurons and their fibers were observed in the alveolus and oriens layer (A+Or) of the sham-operated CA1 region. In contrast to the sham-operated CNS, significantly more grafted neurons were seen in the stratum radiatum (Rad) of ischemic injured animals (I). In the pyramidal cell layer (Pyr), grafted neurons (GFP, green) were observed only along the needle track in both sham-operated (J) and damaged (K) environment. Following ischemia we observed over 90% reduction of pyramidal neurons in the CA1 region. GFP-IR neurons and their fibers omitted the areas with neuronal loss (K, NeuN, red). Colocalization of GFP and NeuN in the graft core (J, K, yellow) was noted in less than 20% of transplanted neurons. Statistical significance is indicated by **p < 0.01; ***p < 0.001 (I). Scale bars: 1 mm (A–F, G–H); 80 μm (J–K).
In order to evaluate proliferation of grafted cells, animals were given a daily injection of BrdU for 4 days immediately after transplantation. A similar number of BrdU-labeled cells were found at each of the survival times (i.e., 4, 8, or 12 weeks after transplantation). For example, in the graft core at 4 weeks postgrafting double immunolabeling showed that 21.21 ± 5.9% and 18.31 ± 4.6% of GFP-IR neurons were BrdU positive in sham-operated and ischemic animals, respectively (Fig. 3).

Proliferation of grafted cells was assessed by detecting bromodeoxyuridine (BrdU; red) incorporated in GFP-IR neurons (A, B; green). Proliferation levels were similar in sham-operated as well as ischemic animals in all survival periods. These images were captured from the graft core, within the ischemic lesion site, 4 weeks after transplantation. Scale bar: 60 μm.
Integration of Transplanted Neurons Into Host Circuitry
Afferentation of Grafted Neurons
Putative afferent contact with neurons was assessed in four groups of cells: 1) host CA1 neurons in sham-operated animals; 2) surviving host CA1 neurons in ischemia-injured animals; 3) transplanted GFP-IR cells in sham-operated animals; 4) transplanted GFP-IR cells in ischemia-injured animals. For the purposes of examination, putative synaptic contact was defined as the percentage of the cell body membrane contacted by adjacent synaptophysin-IR terminals derived from both the host (GFP-negative) and grafted (GFP-IR) neurons.
In sham-operated animals, host pyramidal neurons had 50.2 ± 5.3% of their cell body surface contacted by synaptophysin-IR terminal, compared to 52.72 ± 3.2% for similarly located surviving CA1 neurons in ischemia-injured animals. Examination of grafted cells in sham-operated animals revealed that 45.8 ± 2.3% of their cell body surface was contacted by synaptophysin-IR terminals; grafted cells in ischemia-injured animals had an average of 52.4 ± 4.4% of their cell body contacted (Fig. 4A–D). A subpopulation of synaptophysin-IR terminals found around either host CA1 or grafted cell bodies were derived from grafted neurons (GFP-IR).

Afferentation of grafted neurons 12 weeks after transplantation (14 weeks after ischemia/sham-surgery). The percentage of the cell body membrane apposing each specific presynaptic axonal terminal marker [i.e., synaptophysin (Syn), vesicular glutamate transporters 1-3 (VGLuTs), and glutamate decarboxylase 65, 67 (GADs)] was analyzed for host pyramidal neurons of the CA1 region (pyr) and grafted neurons (graft) in sham-operated (sham) and ischemic (isch) animals (A). Compared to host pyramidal neurons of sham-operated animals, an increased number of putative glutamatergic presynaptic axonal terminals (VGLuTs) were observed around grafted neurons of all animals. In contrast, the putative GABAergic presynaptic axonal terminal population (GADs) was decreased around grafted neurons of sham-operated animals (B–D; NeuN, red; Syn, cyan; GFP, green). (E–G) Putative gluta-matergic (VGLuTs, red) and GABAergic (GADs, cyan) presynaptic axonal terminals around GFP-IR grafted neurons. Statistical significance is indicated by *p < 0.05; **p < 0.01 (I). Scale bars: 8 μm.
Focusing on the two main neurotransmitters of the hippocampus, we visualized glutamatergic axonal terminals by immunostaining vesicular glutamate transporters (1, 2, and 3; VGLuTs) and GABAergic terminals by staining GAD65/67 (GADs) (Figs. 4E–H). Quantitative confocal analyses of synaptophysin-IR, glutamatergic, and GABAergic axonal terminals around surviving host pyramidal neurons and grafted neurons in sham-operated and ischemia-injured animals are summarized in Figure 4I. Several differences were noted between grafted cells and host CA1 neurons; the ratios between each VGLuT and GAD subtype are shown in Table 1. While cerebral ischemia reduced the occurrence of VGLuT2-positive terminals around surviving host pyramidal cells, more VGLuT1-IR terminals were found around the same cells. The majority of glutamatergic afferents detected within the graft core of both animal groups were VGLuT1-IR. Triple labeling of GFP, VGluTs, and GADs within the graft core and CA1 region revealed examples of GFP-IR terminals positive for either phenotype (Figs. 4E–G).
Ratios Between VGLuT and GAD Subtypes

Ratio between each vesicular glutamate transporter (VGLuT) subtype as well as between each glutamate decarboxylase (GAD) isoform found adjacent to the cytoplasmic membrane of pyramidal and grafted green fluorescent protein-immuno-reactive (GFP-IR; Graft) neurons in the sham-operated and ischemic animals 14 weeks after ischemia/sham surgery (12 weeks after transplantation).
Statistical significance of injured and grafted neurons versus sham-operated pyramidal neurons: p < 0.001.
Projections of Grafted Neurons
Axodendritic sprouting of grafted neurons (GFP-IR fibers) was observed in all transplanted animals, but the volume per section occupied with GFP-IR fibers in the CA1 region was over threefold larger in the ischemic compared with the sham-operated animals at each time point. In fact, the GFP-IR volume increased with survival periods in the ischemic groups but not in the sham-operated animals (Fig. 5A). The axodendritic sprouting spread laterally from the injection site in the alveolus, oriens layer, and stratum radiatum of all ischemic animals, but as described above for the GFP-IR somata, the fibers were typically absent from the pyramidal cell layer (Fig. 5B).

Axodendritic sprouting in the CA1 region. Comparison of GFP-IR fiber distribution in the CA1 region of sham-operated (sham) and ischemic (isch) animals revealed a greater than threefold increase in fiber sprouting of grafted neurons in the damaged areas. Moreover, the volume of tissue containing GFP-IR fibers in the damaged CA1 region increased with survival periods (A). GFP-IR fibers in ischemic animals 12 weeks after transplantation appear to be a mix of axons and dendrites (d) with dendritic spines (B, arrow). Statistical significance between sham and ischemic animals is indicated by ***p < 0.001 (A). Scale bar: 160 μm (16 μm for the inset).
In the rostrocaudal axis (Fig. 6A–C), GFP-IR fibers grew from the injection sites caudally in the subiculum (Fig. 6D), and rostrally into the septal nuclei (Fig. 6E) and the nucleus accumbens (Fig. 6F). After 4-week survival, GFP-IR axons were observed in the subiculum and the septal nuclei. In contrast, only after 12 weeks were GFP-IR fibers found in the nucleus accumbens. Analysis of GFP-IR volume in the selected target regions of animals with the longest survival times showed more than fourfold more GFP-IR fibers in the subiculum and the nucleus accumbens of ischemic animals as compared to sham-control (Fig. 6C).

Projection of GFP-IR neurons into specific target areas. GFP-IR axons grew caudally from the injection sites into the subiculum (S) and rostrally into the septic nuclei (Sep) and nucleus accumbens (NA; A). The schematic drawing (B) shows injection sites (from 3.3 to 5.3 mm posterior from bregma) and the axonal termination areas (green-shaded areas) in the subiculum (–5.6 mm posterior from bregma), septum and nucleus accumbens (+0.2 and +2.2 mm rostrally from bregma). Projection of GFP-IR axons was analyzed in the animals with the longest survival period. Per volume of tissue (60,000 μm3), a significantly larger GFP-IR area was found in the subiculum and nucleus accumbens of the ischemic-damaged animals (C). Microphotographs (D–F) show the localization of GFP-IR axons in the subiculum (D), septal nuclei (E), and nucleus accumbens (F). Statistical significance between sham and ischemic animals is indicated by ***p < 0.001 (C). Scale bars: 500 μm and 60 μm (A and enlarged inset), 3 mm (B), 150 μm (D), 60 μm (E, F).
Projection Areas of Grafted Neurons
In both ischemia-injured and sham-operated animals, GFP-IR terminals expressed specific markers for glutamate as well as GABA in the subiculum and septal nuclei; however, no appreciable population of GABAergic GFP-IR terminals was found in the nucleus accumbens (Fig. 7A, B). The majority of putative glutamatergic axonal terminals of grafted neurons in the target areas were VGLuT1-IR and the least were VGLuT2-IR. GABAergic axonal terminals of grafted neurons were mainly GAD65-IR rather than GAD67-IR. Analysis of putative GFP-IR presynaptic terminals in the selected target regions showed a significantly higher number of glutamatergic terminals in ischemic animals compared to sham-operated (Fig. 7A, B). In addition to GFP-IR terminals apparently making direct contact with host cell bodies, we also observed examples of host GABAergic terminals making contact with GFP/VGLuT-IR terminals, suggesting presynaptic inhibition (Fig. 8).
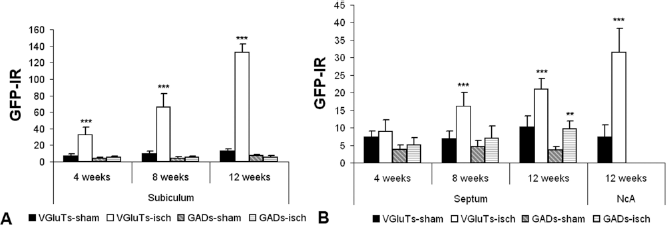
Efferentation of grafted neurons. Glutamatergic (VGLuTs) and GABAergic (GADs) axonal terminals of grafted neurons (GFP-IR) were analyzed in the subiculum (A) and septum and nucleus accumbens (B) for each survival period. In contrast to sham-operated animals, those with ischemic damage showed significantly more putative glutamatergic GFP-IR axonal terminals in each target area and survival period. GABAergic GFP-IR terminals were more abundant in the septal nuclei of ischemic animals 12 weeks after transplantation. Statistical significance is indicated by *p < 0.01; ***p < 0.001 compared to corresponding sham values.

GFP-IR axonal projections in the subiculum. Panels show GFP (A, E; GFP, green), glutamatergic (B, F; VGLuTs, red), and GABAergic (C, G; GADs, cyan) putative presynaptic terminals in the subiculum of an ischemic animal 12 weeks after transplantation. In some cases putative presynaptic inhibitory GABAergic terminals can be identified in contact with GFP-IR glutamatergic terminals (D, H; arrows). Asterisks in (D) indicate putative neuronal soma. Scale bar: 10 μm (A–D), 4 μm (E–H).
Discussion
The present results demonstrate that embryonic neurons transplanted into the CA1 region of the adult hippocampus can survive and form appropriate connections with the host neural network. By expressing GFP in grafted cells under control of the synapsin promoter, neuron-specific expression of GFP in the graft readily allowed: 1) clear, long-term identification of grafted neurons; 2) visualization of fiber sprouting throughout the graft area; 3) identification of short- and long-distance axonal projections; 4) quantitative evaluation of the phenotype of putative graft-derived presynaptic axonal terminals.
Numerous transplantation studies have illustrated the importance of the host environment regarding migration of transplanted cells (4,23,61); for example, Daadi et al. (13) reported that rat medial eminence ganglionic neurons transplanted into the ischemia-damaged rat cortex assumed the columnar cytoarchitecture of an intact cortex by 4 weeks after grafting. Indeed, results from the present study support this concept in that pronounced migration of transplanted cells was noted in the CA1 stratum radiatum of the ischemic animals, while in the sham-operated hippocampus grafted neurons spread laterally only in the alveus and the superficial part of the oriens layer. Interestingly, these differences in the distribution of the GFP-IR somata in the sham-operated versus ischemic hippocampus paralleled the differences in axodendritic sprouting in both groups. It may be that similar factors influence migration of grafted neurons as well as the pattern of axodendritic sprouting. Partial support for this hypothesis comes from the work of Fournier and colleagues (15), which demonstrated that the axonal growth inhibitor Nogo-A also affects fibroblast migration. The observation of limited migration and axodendritic sprouting of transplanted cells locally within the damaged pyramidal cell layer raises the question of what might mediate this apparent inhibition. It is possible that activated glial cells, which partly form scar tissue in the ischemic damaged areas (26,57,70), drastically limit the migration of grafted neurons into the most damaged area of the hippocampus.
It should be noted, however, that the current study used cell grafts that expressed GFP under neuronal-specific control and while about 60% of the culture expressed neuronal markers, more than 90% of those neurons in culture expressed GFP. Thus, a limitation of neuron-specific GFP expression is the inability to assess nonneuronal transplants. On the other hand, neuron-specific GFP expression can be utilized in fluorescence-based cell sorting to select and enrich cell populations with neurons prior to transplantation. As well, because not all cells in the graft core expressed GFP (e.g., glia were negative), neuronal GFP was much more clearly imaged and allowed detection of fine fiber sprouting, formation of short axonal projections, and phenotype evaluation of putative graft-derived presynaptic axonal terminals around GFP-positive neurons.
It has been well demonstrated in many species that axons of embryonic neurons can grow in the adult CNS (9,47,48,73,74). In our study, fibers derived from grafted neurons showed a wider area of distribution in the ischemia-injured CA1 region when compared to grafts in sham-operated animals. It is possible that this pronounced fiber sprouting in the ischemia-damaged environment is a result of increased growth factor levels in the damaged region (14,50,72) (e.g., those produced by activated astrocytes and/or microglial cells) (1,39,40,42).
In the present investigation, efferents of the grafted neurons were found in the subiculum, septal nuclei, and nucleus accumbens, which are the normal targets of hippocampal efferents (30,56,66,67,69). It is noteworthy that the adult CNS appears to retain at least some capacity to direct newly sprouting axons to the correct targets. During CNS development, growing axons reach and then arborize in their target areas by following a set of extracellular and intracellular signals, many of which are yet to be understood (31,35,58). Our findings suggest that the mature host tissue of both the sham-operated and ischemia-injured environments presents factors important for axonal guidance; indeed, graft-derived axons reached the nucleus accumbens within 12 weeks.
Comparison of axonal growth of grafted neurons in the sham-operated versus ischemia-injured CNS showed that while GFP-IR axons were found in each target region at similar times in both environments, ischemia-injured animals had a dramatically higher number of GFP-IR fibers reaching those targets. Once a developing axon reaches a target area, synaptogenesis begins when the axonal growth cone comes in contact with an appropriate postsynaptic cell and continues as the growth cone differentiates into a presynaptic terminal (58). We identified numerous examples of putative presynaptic axonal terminals derived from grafted neurons (coexpressing GFP, synaptophysin) (75), and one of several specific phenotype markers (e.g., VGLuT1). It should be noted though, that while the putative synapses identified in the current study expressed common presynaptic proteins, had a physical appearance similar to terminals in previous studies (10,25) and in most cases were adjacent to a cell membrane, those putative presynaptic structures were visualized by light, fluorescent, and confocal microscopes only. Indeed, to unequivocally demonstrate the identified structures are anatomically correct and functional synapses, further analyses require electro-physiology and electron microscopy. That particular data would be essential to future behavioral studies employing cell replacement therapy.
In line with the pronounced axodendritic sprouting observed, more axonal terminals derived from grafted neurons were found in the ischemic animals. The principal neurons of the hippocampus and cortex (source of embryonic tissue used in our study) are glutamatergic (~90%) and GABAergic (~10%) neurons (71), and in our study these neurotransmitters were identified with specific markers for their presynaptic axonal terminals. Under normal circumstances, the majority of VGLuT1-IR glutamatergic afferent terminals from the hippocampal CA3 pyramidal cell layer, the Schaffer collaterals, make synaptic contact with the dendrites of CA1 pyramidal cells (64). In the present study, the glutamatergic afferents terminating on the cell bodies of host pyramidal neurons of sham-operated rats were mainly positive for VGLuT2. The potential source of these afferents can be the medial septum, where the glutamatergic hippocampal afferents originate (11), and the septal neurons which are positive for VGLuT2 mRNA (43). Our results are in line with those of Kim et al. (36), who reported that in the gerbil hippocampus, VGLuT2-IR was decreased in the pyramidal cell layer following transient ischemia. Compared to host pyramidal cells of sham-operated grafted animals, more VGLuT1-IR terminals were seen around the surviving pyramidal cells of the ischemic animals and the GFP grafted neurons. Identification of graft-derived (i.e., GFP-IR) axonal terminals showed that most were glutamatergic, with apparent VGLuT1-IR contacts on other grafted cells as well as GFP-negative surviving pyramidal neurons. This is in contrast to a recent report where nearly 70% of surviving transplanted rat medial eminence ganglionic cells found in the ischemia-damaged cortex/striatum expressed GABAergic markers (13). We speculate that this phenotypic difference, when the majority of grafted neuronal cells became GABAergic in the striatum and glutamatergic in the CA1 region of hippocampus, may mimic the neuronal population in the striatum (45,52) and the hippocampus (71) under physiological conditions, perhaps illustrating the importance of the environment that surrounds the graft. Interestingly however, of the GABAergic cells identified in that study, the GAD65:GAD67 ratio was remarkably similar to that found in the current study.
Host pyramidal neurons of the uninjured CA1 appeared to be heavily laden with inhibitory GAD65-IR terminals; however, our grafted cells in the same animals did not appear to receive the same level of GAD65 contact. In ischemic animals, both surviving pyramidal neurons and grafted GFP-IR neurons had slightly less GAD65-IR afferent contacts, when compared to sham-operated animals. The source of GABAergic afferents in the CA1 region is local GABAergic interneurons (21, 22,29) and projecting septal neurons (11,17,20). That GABAergic interneurons survived in all hippocampal layers after experimental focal ischemic injury (16) suggests the altered GABAergic terminal population in the ischemic CA1 region and in the graft cores may be due to a reduction in septal afferents, since the septum is similarly sensitive to ischemia as the hippocampus (5).
Our study demonstrated that embryonic neurons can survive in the adult CNS after grafting and that transplanted neurons can acquire a region-specific neuro-transmitter phenotype. In addition, there is considerable capacity to direct newly sprouting axons derived from transplanted neurons to anatomically appropriate targets. Furthermore, even greater axonal growth and putative synapse formation was apparent in ischemia-injured brains than in noninjured animals.
Footnotes
Acknowledgments
The authors are grateful to Carol Novosad for her assistance with transplanted animals. This work was supported by: the Office of the Dean, UCSD School of Medicine (J.C.D.), CIRM grant #RC1-00131 (M.M.), and NIH grant #GM52098 (P.P.).
