Abstract
Embryonic striatal graft-mediated functional recovery in the rodent lesion model of Huntington's disease (HD) has been shown to correlate with the proportion of dopamine- and adenosine 3′,5′-monophosphate-regulated phosphoprotein with a molecular weight of 32 kDa (DARPP-32)-positive neurons in the graft. The current study investigated the impact of graft distribution on the yield of DARPP-32-positive cells in the grafts following either single-tract or multitract cell delivery protocols using the microtransplantation approach. Cells derived from the whole ganglionic eminence of E15 rat embryos, ubiquitously expressing green fluorescent protein (GFP), were implanted into unilaterally QA-lesioned rat striatum either as 2 × 1.8 μl macrodeposits in a single tract, or as 18 × 0.2 μl microdeposits disseminated over six needle, multitract, penetrations. For both groups, an ultrathin glass capillary with an outer diameter of 50 μm was used. Histological assessment at 4 months after transplantation showed nearly twofold increase of DARRP-32-positive striatal-like neurons in the multitract compared to the single-tract group. However, the cellular make-up of the grafts did not translate into functional differences as tested in a basic spontaneous behavior test. Furthermore, the volumetric values for overall volume, DARPP-32-positive patches, and dopaminergic projection zones were similar between both groups. The results show that distribution of fetal striatal tissue in multiple submicroliter deposits provides for an increased yield of striatal-like neurons, potentially due to the enlargement of the graft–host border area intensifying the graft's exposure to host-derived factors. Furthermore, the use of embryonic tissue from GFP donors was validated in cell-based therapy studies in the HD model.
Keywords
Introduction
Cell transplantation as a therapeutic approach for Huntington's disease (HD), an inherited neurodegenerative disorder, has the potential to replace damaged striatal neurons, which can lead to reestablishment of dysfunctional neuronal circuits resulting in partial behavioral recovery (4,29,38). This concept has advanced beyond animal experimentation and into the clinic where trials in patients have moved forward (6,13,26). However, there are still many issues to be dealt with, not the least of which is an optimized transplantation protocol to enhance the yield of relevant cell populations within the graft.
In former studies, it has been shown that graft-induced functional recovery in animal experiments relates directly to the quantity of striatal medium spiny projection neurons stained positive for DARPP-32 (dopamine- and adenosine 3′,5′-monophosphate-regulated phosphoprotein with a molecular weight of 32 kDa) within the transplant (16,25,37). As reported by different groups for the quinolinic acid (QA)-induced excitotoxic rat model of HD, the number of DARPP-32-positive cells in the grafts derived from the lateral ganglionic eminence (LGE) is estimated to range between 10% and 40% with respect to the primary pregrafting cell population (35,37,57). This proportion is considered to be constituted by a combination of cell survival versus cell death on the one hand, and proliferation of grafted progenitor cells, which continue to divide and mature posttransplantation (25), on the other hand.
Taking into account the considerably high loss of DARPP-32-positive cells following transplantation, the correlative relationship between the quantity of this relevant cell population and behavioral recovery implies the potential and the importance of further improvement of cellular availability. Nevertheless, approaches to increase the yield of functionally important cells have rather been in the periphery of HD cell therapy research while being a central issue of interest for Parkinson's disease (PD).
With the objective to enhance survival of implanted dopaminergic cells, Nikkhah and colleagues introduced a microtransplantation protocol to the 6-OHDA animal model of PD using a glass capillary with a relatively small outer diameter of 50 μm as a novel transplantation device for cell suspension grafting (41,42,52). With this minimally traumatic and accurate technique it was possible to establish a multitract transplantation protocol, allowing for a wider dissemination of the cell suspension as multiple microdeposits. The combination of both the refinement of the injection instrument and the novel placement strategy was shown to lead to significant improvement of graft survival and a more extensive behavioral recovery in animal models of PD (41,42,52).
In the current study, hypothesizing that similar biological factors accounting for the positive outcome on key neuronal phenotype in PD may also be relevant in the HD model, we transferred the multitract microtransplantation approach to the QA-induced excitotoxic rat model. To investigate the impact of graft distribution on the yield of DARPP-32-positive neurons in the graft, unilateral striatal lesioned rats were transplanted with equal numbers of E15 cells derived from the whole ganglionic eminence (WGE) either in one injection tract (single tract or ST-group) with two large deposits or in six injection tracts (multitract or MT-group) with 18 small deposits. Histological analysis confirms that the multitract protocol can positively influence the development of DARPP-32-positive neurons within the grafts.
Materials and Methods
Subjects
Animals housed in standard cages in groups up to five animals in a temperature-controlled room (temperature of 21 ± 1°C, 50–60% relative humidity) on a 12-h light/12-h dark schedule. Each animal had ad libitum access to laboratory chow and to water, and were weighed once per week. All animals were treated according to the ethical guidelines set by the local ethical board of the University of Freiburg and the Regierungspräsidium Freiburg, Germany.
Experimental Design
The experimental design is summarized in Table 1. A total of 59 adult female Sprague-Dawley rats (Charles River, Germany) weighing 225–250 g at the beginning of the study were used in the study, and were distributed into control (n = 15), lesion-only (n = 14), multitract (MT, n = 15), or single-tract (ST, n = 15) grafting group.
Experimental Design With Time Points of Procedures Relative to the First Behavioral Testing Session

Lesion
Two investigators carried out the lesion procedure over 1 day using one single preparation of quinolinic acid. In order to secure a stable and comparable lesion (30), the lesion surgery, as well as all other surgical procedures, was performed under inhalative gas anesthesia with isoflurane (Abbott, Germany) using O2 as carrier gas (induction: 5 vol% of isoflurane with 1.5 L O2 per minute; maintenance: 1.5–2.0 vol% isoflurane in 1.5 L O2 per minute).
Using a stereotactic frame (Stoelting, USA), animals received unilateral striatal lesions by injecting 4 × 0.2 μl of 0.12 M quinolinic acid (Sigma, USA) dissolved in 0.1 M phosphate-buffered saline (1 x PBS solution), pH 7.4, at two depths in each of the two tracks in the left neostriatum. Each injection was infused over 90 s via a 26-gauge stainless steel cannula connected to a 10-μl Hamilton syringe (Hamilton Europe, Switzerland), which was placed on a micropump (World Precision Instruments Inc., UK). In total, the animals received 0.8 μl of 0.12 M quinolinic acid solution (=96 nmol of QA in total) with an injection rate of 0.13 μl/min. Injection coordinates with measurements in mm anterior (A) in front of bregma, lateral (L) to the midline, vertical (V) below dura were: A = −0.4, L = +3.7, V = 5.2/4.2; A = +1.2, L = +2.9, V = 5.2/4.2. The inter-aural line was set at 2.3 mm. The cannula was held in place for 2 min before retraction to reduce leakage. While still under anesthesia, the animals received SC Temgesic (0.05 mg buprenorphin hydrochlorid/kg body weight; Essex Pharma, Germany) in the neck as postsurgical analgesic.
Breeding, Dissection, and Single Cell Suspension
Inbred heterozygous green fluorescent protein (GFP)-transgenic Lewis rats were used as cell donors. Kindly provided by Dr. Eiji Kobayashi, the transgenic animals express GFP under the control of a cytomegalovirus CMV/β-actin promoter (CAG promoter) (22,28). Wild-type Lewis rats were purchased from Charles River, Germany, and bred with heterozygous GFP-transgenic animals to maintain the strain. Graft tissue was obtained from the WGE of E15 heterozygous GFP-transgenic Lewis rat embryos.
Fifteen days before harvesting the embryos, one wild-type Lewis GFP-/- male with normal phenotype was bred overnight with one or two GFP (GFP+/-)-transgenic females. The following day was counted as day 0. At gestation age E15, the embryos had a crown–rump length (CRL) of 13–14 mm (45,49). The abdomens of the deeply anesthetized pregnant animals were opened, and the uterus was removed and cleaned in sterile PBS. Subsequently, the embryos were taken out of the uterus and kept in Hank's balanced salt solution (HBSS). GFP-positive embryos were distinguished from wild-type embryos under UV light (λ = 489 nm). WGEs were dissected bilaterally only from E15 GFP-transgenic embryos. Dissected tissue was collected and preserved in a 0.5-ml Eppendorf tube containing 0.1% DNase diluted in HBSS. Pooled striatal ganglionic eminences were incubated in 0.1% trypsin and 0.05% DNase for 20 min at 37°C. The suspension was then rinsed four times in 0.05% DNase diluted in HBSS. Following centrifugation at 1000 rpm for 4 min, the supernatant was removed and the tissue was mechanically triturated using flame-polished 1000-μl and 200-μl pipette tips.
Viability of the embryonic cells was assessed by trypan blue exclusion just prior to grafting using a hemocytometer and was always more than 97% for these striatal cell suspensions. The resulting single cell suspension was adjusted to a concentration of 130,000 cells/μl. Cell viability was again monitored at the end of the transplantation procedure and was more than 90%.
Transplantation Procedure
Transplantation surgery was carried by two people during 1 day, with animals from the two grafted groups equally matched between both investigators. Transplantation in both grafted groups was performed using a glass capillary with a diameter of 50 μm connected via an adaptive polyethylene tube to the cannula of a 2-μl Hamilton syringe (41,42,44) (Fig. 1A, B). Host animals were anesthetized using isoflurane as described above and positioned in the stereotactic frame. After rinsing the capillary with cell-free medium, the single cell suspension was aspirated into the glass capillary and placed into the left neostriatum according to the group protocol. After each tract, the capillary was kept in place for 2 min, then slowly retracted and rinsed with medium. The animals underwent the same postoperative treatment as described for the lesion. In the present study, two different transplantation protocols were used constituting the two transplantation groups (the distribution of transplantation deposits for both transplantation groups is illustrated in Fig. 1C, D).
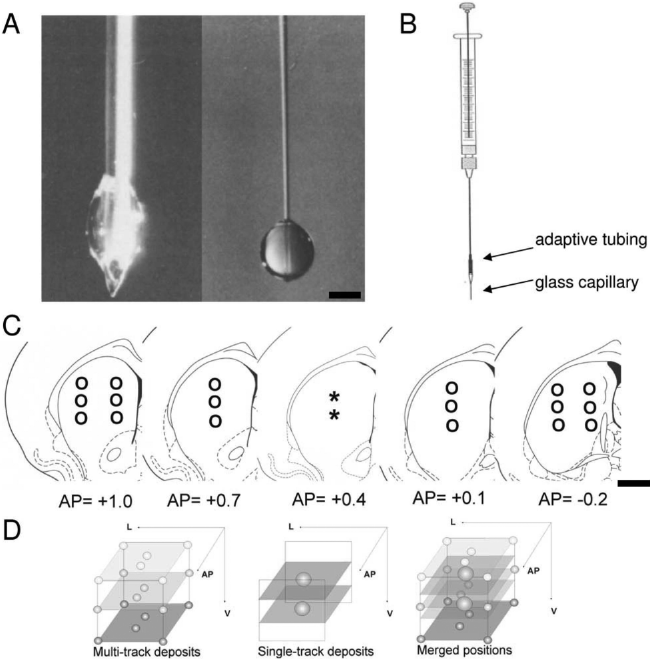
Photograph illustrating the tips of the regular metal Hamilton cannula (left side, 500 μm outer diameter) and the used glass capillary (right side, 50–70 μm), which are shown with 1.0 μl of culture medium (DMEM) being extruded by either instrument. A clear difference can be seen in the shape of the drop and the ratio between tip size and injected volume (A). The microtransplantation instrument consists of a 2-μl Hamilton microsyringe fitted with the glass capillary using a cuff of polyethylene tubing as an adapter (B). Distribution and anterior–posterior coordinates of deposits for the single-tract (asterisks) and multitract group (circles) (C). Three-dimensional illustration of deposit distribution for both groups (D). Scale bar: (A) 500 μm; (C) 1 mm.
Single-Tract Transplantation Group
Two macrodeposits of 1.8 μl each (total of 3.6 μl) were implanted in the lesion area using one tract. Stereotactic implantation coordinates in mm were: A = +0.4, L = +3.3, V = 5.0/ 4.4. The interaural line was set at 2.3 mm.
Multitract Transplantation Group
Eighteen microdeposits of 0.2 μl each (total of 3.6 μl) were implanted in the lesion area using six tracts (three deposits per tract). Transplantation coordinates exhibited in mm were: A = +1.0, L = +2.6/+3.7, V = 5.1/4.7/4.3; A = −0.2, L = +2.6/+3.7, V = 5.1/4.7/4.3; A = +0.7, L = +3.1, V = 5.1/ 4.7/4.3; A = +0.1, L = +3.5, V = 5.1/4.7/4.3. The interaural line was set at 2.3 mm.
Histology and Image Analysis
Sixteen weeks following the intrastriatal grafting, the animals were terminally anesthetized by intraperitoneal (IP) injection of an overdose of ketamine (Essex Pharma, Germany) and rompun (Bayer, Germany) and then transcardially perfused with 300 ml 0.1 M PBS followed by 300 ml 4% paraformaldehyde in 0.1 M PBS. The brains were removed and postfixed in 4% paraformaldehyde for 1.5 h, then immersed in 30% sucrose in 0.1 M PBS for 24 h until they had sunk. Tissue was serially sectioned on a freezing microtome at a thickness of 40 μm through both the striatum and substantia nigra and collected as six series.
Free-floating sections were washed three times with PBS, preincubated for 2 h in 3% bovine serum albumin (BSA; Sigma, Germany)/0.3% Triton X-100 (Sigma)/ PBS, and incubated overnight at room temperature with the primary antibodies: GFP (mouse anti-GFP, 1:500; Invitrogen, Germany), NeuN (mouse anti-NeuN, 1:1000; Chemicon, Germany), a marker of mature neurons, DARPP-32 (rabbit anti-DARPP-32, 1:500; Chemicon), a marker of striatal tissue (17–19,31,46), and tyrosine hydroxylase (mouse anti-TH, 1:500; Sigma), a marker used to identify dopaminergic neurons and axonal input. After three washes with PBS, sections were incubated for 2 h with biotinylated secondary antibody (1:200; Dako, Germany) for DAB staining or with an Alexa secondary antibody (Alexa Fluor, 1:200; Invitrogen) for fluorescence staining. Visualization for the biotinylated antibody was carried out with the ABC kit (Vector, Germany) and DAB (Merck, Germany). Sections were mounted on slides, dehydrated, and cover slipped, and subjected to image analysis (Olympus Ax-70; “Analysis,” Soft Imaging Systems, Germany). Fluorescence staining was used for confocal microscopy pictures illustrating DARPP-32 GFP coexpression; all quantitative measurements were carried out with DAB-stained sections. The extent of the lesion in the dorsal striatum was estimated by tracing the outlines of each section throughout the anterior–posterior axis of the striatum on the DARPP-32-stained sections giving the cross section areas. The volumes for the various parameters measured were calculated by taking into account the sum of the areas and correcting for section thickness and sample frequency [volume (mm3) = sum of areas (mm2) x 40 μm x 6). Confocal microscopy pictures were taken from immunofluorescence-stained sections using a Leica TCS SP2 AOBS spectral confocal microscope.
DARPP-32 Cell Count
Cell counting was performed using a Leica DMRB microscope with the Stereo Investigator software. DARPP-32-positive cells were counted from all mounted sections containing a graft in a 1:6 series from all transplanted animals. The Abercrombie formula for correction of cell counts was used to obtain estimates of total cell numbers from the raw cell counts. The corrected cell number P was calculated by the formula: P = 1/f∗A∗M/(D + M), where A is the cell count from the entire graft, f is the frequency of sections, M is the section thickness, and D is the average cell diameter. The proportion of cells coexpressing DARPP-32 and GFP was evaluated during counting on DARPP-32-stained sections, as the GFP signal could be clearly detected using autofluorescence.
Table Lift Behavior Test
Table lift is a spontaneous behavior task looking at the rats' ability to make adjustment movements with its forepaws (14). The animal's hindlimbs and one of the forepaws are held by the investigator with the test paw remaining unrestrained. Starting from approximately 20 cm below the table level, in a continuous motion, the animal is moved upwards, towards, and beyond the edge of the table. With this stimulation, the rat tried to grasp the table. The animal's attempt with the test paw was scored according to a system ranging from 0 to 3 according to the following: (0) no reaction—the animal ignores the stimulation and makes no attempt to make contact with the table using its free forelimb; (1) muscle reaction without reaching—the animal turns its head towards the table or shows movement in the free paw without successfully reaching the table; (2) delayed reaction with the free limb making successful contact with the table—the animal reaches with a temporal delay to the table, but only in the upper part of the edge; (3) prompt reaction with the free limb making successful contact with the table—the animal reaches immediately to the table and tries to hold it.
This test was repeated five times per day during the testing sessions on each forelimb for 1 day and the final results are expressed as the sum of 1 day. Table lift test was performed prelesion, pregrafting, and 8 weeks following transplantation.
Statistical Analysis
Statistical analyses was carried out with StatView software (Abacus, USA) using the ANOVA test. A multiple ANOVA test was applied for the Table lift test, using as main effects the Groups (control, lesion, MT, ST) and the Sessions (baseline, postlesion, and posttransplantation 1 and 2). In case of main effects, the Newman-Keuls post hoc analysis was used to identify the significant differences. Level of significance was set at p < 0.05 for all tests. Results are expressed as mean ± SEM.
Results
Histology
Photomicrographs of the control, the lesioned, and the grafted animals are shown in Figure 2. The volumetric measurements are summarized in Table 2.

Photomicrographs of coronal sections showing representative examples of control (A–C), lesion (D–F), and the two graft groups (MT, G–I; ST, J–L) using NeuN (A, D, G, J), DARPP-32 (B, E, H, K), and TH (C, F, I, L) stainings. Increased magnification reveals the microstructure of patch (P) and nonpatch (NP) zones within the graft (M, N, O). MT, multitract; ST, single tract. Scale bar: (A–L) 1 mm; (N) 50 μm; (O) 250 μm.
Volume (mm3) Measurements on NeuN-Stained Sections (Unless Otherwise Indicated) of the Graft, the Intact, and the Lesioned Striatum

Lesion Effect
Quinolinic acid injections resulted in a marked unilateral cell loss and local atrophy in the striatum, which could be visualized immunohistochemically with the adult neuronal marker NeuN and the striatal marker DARPP-32 (Fig. 2D, E).
The volumetric measurements (mm3) were carried out using NeuN-stained sections quantifying the extent of the lesion by subtracting the absolute volume of the surviving ipsilateral striatum from the contralateral unaffected one serving as control.
In both the lesion and the treatment groups, the toxin equally reduced the ipsilateral striatal volume to 31–35% of the contralateral side [Groups, F(1, 22) = 39.24, p < 0.0001] being in line with the behavioral findings (see below) and implying comparable pregrafting conditions for both transplantation groups. The same applies for the calculated absolute volumes of the lesion [Groups, F(1, 22) = 16.03, p < 0.05]. As expected, there was no difference in the volume of the intact contralateral striatum across the lesioned groups as this side was not affected by the lesion [Groups, F(1, 22) = 0.06, p > 0.05, n.s.).
Graft
NeuN-stained sections were used to measure the total volume of the graft because it allowed unambiguous identification of the boundary between the graft, the neuron-depleted lesioned area, and the remaining host tissue (Fig. 2G, J). The grafts survived in the host striatum, were rich in cells of neuronal morphology, as seen in the NeuN sections, and exhibited a characteristic patchy staining with DARPP-32, a selective marker for the striatal-like neurons within the grafts (Fig. 2I, L, O).
Transplants of both groups exhibited a regular droplike configuration (Fig. 2G–L), which suggests that during maturation the 18 microdeposits of the MT grafts merged into a unit with a continuous graft–host border making it impossible to distinguish between individual deposits.
MT and ST grafts also had similar overall graft volumes, as measured by NeuN staining, as well as comparable DARPP-32 positive patch volumes [Table 2: Groups graft volume, F(1, 24) = 0.25, p > 0.05, n.s.; Groups DARPP-32-patch/nonpatch volume, F(1, 24) = 0.9, p > 0.05, n.s.]. The pattern of dopaminergic input to the graft, identified by the TH-positive fiber staining, coincided with the DARPP-32-positive patches and exhibited comparable mean values in both transplanted groups [Table 2: Groups TH patch/nonpatch volume, F(1, 24) = 1.44, p > 0.05, n.s.].
GFP Expression of the Graft
In unstained sections using GFP autofluorescence as well as in immunohistochemically enhanced ones using primary GFP antibodies, the expression of the GFP remained stable (Fig. 3). Coexpression analysis revealed that 84% of the counted DARPP-32-positive cells within the graft were also GFP positive 16 weeks after grafting, independent of the respective implantation protocol. This means that, in the process of graft maturation, only 16% of the striatal cells lost their GFP expression, which is comparable to unpublished results from our lab where GFP-positive dopamine-rich single cell suspensions were transplanted in the 6-OHDA lesion model of PD. In this latter study, 80% of the grafted dopaminergic TH-positive cells maintained a stable GFP expression, which was independent of immunosuppressive treatment with cyclosporine, even though, as in the present experiment, GFP-transgenic Lewis rat tissue was allografted in Sprague-Dawley rats (Krause et al., manuscript in preparation). With the current experiment we validated the use of embryonic tissue from GFP donors in cell-based therapy studies in the HD lesion model and showed the potential of this cell source as a new and valuable means to stably visualize and identify most of the transplanted cells.
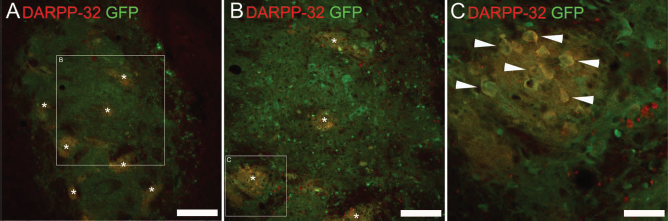
Immunofluorescence staining for GFP (green) and DARPP-32 (red) demonstrates stable GFP expression in the graft (A) that exhibits characteristic DARPP-32-positive patches (A–C, asterisks). Increased magnification reveals the coexpression of DARPP-32 and GFP of some transplanted cells (C, arrowheads). Scale bar: (A) 500 μm; (B) 250 μm; (C) 50 μm.
Sections stained for GFP as a transplant-specific marker were used to assess the dissemination and axonal projection profile of the implanted cells (Fig. 4). All grafts exhibited a homogenous GFP staining and no apparent cell migration was found (Fig. 4B). Furthermore, analyzing the local extension of GFP-positive axonal fibers, a vast innervation of the striatum throughout the whole anterior–posterior axis and even adjacent areas by grafts of both transplantation groups was noted (Fig. 4A).

Schematic illustration of the de facto distribution of the grafts and their axonal fiber outgrowth along the anterior–posterior axis in the MT and ST groups using the transplant-specific marker GFP. The y-axis gives the percentage of animals per group where a GFP-positive signal could be found in the respective coronal section (x-axis) (A). GFP immunohistochemistry illustrating stable GFP expression in the graft at 4 months after transplantation. As the axonal fibers exhibited stable GFP immunoreactivity, the staining could be used to assess the extent of graft-derived striatal reinnervation (B). MT, multitract; ST, single tract. Scale bar: (B) 1 mm.
Cell Counting of DARPP-32-Positive Neurons
Using the Abercrombie correction formula, the total number of DARPP-32-positive cells was calculated for both therapeutic groups. As shown in Figure 5, the results reveal a significant 1.8-fold higher number of DARPP-32-positive cells in the MT compared to the ST group [Groups, F(1, 24) = 8.62, p < 0.05]. Given similar graft volumes, the significantly higher DARPP-32-positive cell count implies that this cell type benefited particularly and more than others from the MT protocol. This corresponds with the observation that P zones in the MT grafts exhibited a significantly higher density of DARPP-32-positive cells/mm3 patch volume [Groups, F(1, 24) = 8.14, p < 0.05] (Fig. 5B).

Number of DARPP-32-positive neurons following transplantation comparing the multitract (MT) with the single-tract (ST) group (A). Density of DARPP-32-positive neurons within DARPP-32-positive patches expressed in cells per mm3 (B). Data represent group means ± SEM. MT, multitract; ST, single tract. ∗p < 0.05.
Behavioral Evaluation: Table Lift Test
The results of the table lift test are shown in Figure 6. At baseline, all animals reached the maximum of 15 points. The lesion surgery induced a significant decrease in the contralateral paw performance in all of the experimental groups compared to the controls; this functional deficit was subsequently reversed to control levels in the grafted groups in both posttransplantation sessions, with no functional differences between the MT and the ST groups [Groups x Sessions, F(9, 147) = 8.88, p < 0.001; post hoc: Baseline, no significant differences; Post-les, control > lesion/MT/ST, p < 0.001; Post-tx1 and Post-tx2, control/MT/ST > lesion, p < 0.001].

The Table Lift test scores reveal an equal baseline, a significant reduction of performance following lesioning in the lesioned groups, and a graft-related recovery in both transplantation groups. No significant protocol-specific difference regarding Table Lift performance was detected, meaning that the increased DARPP-32-positive cell yield in the MT group did not translate into a functional improvement. Data represent group means ± SEM. MT, multitract; ST, single tract. ∗∗p < 0.001.
Discussion
The present study, using a rodent model of Huntington's disease (HD), demonstrates that a multiple placement strategy of striatal neural progenitor cells distributed over 18 implantation sites as small submicroliter deposits leads to a higher total number of DARPP-32-positive cells when compared to a standard single-tract macrodeposit protocol with only two implantation sites. This strategy is already applied in some current clinical trials on fetal cell transplantation for HD patients using four or five trajectories (for eight deposits each) per side (5,13). As indicated by the literature, there is evidence for a correlative relationship between the amount of DARPP-32-positive cells within a transplant and the functional recovery mediated by the grafts (16,24,37). These data emphasize the importance of further optimizing the yield of functionally relevant cell populations for the clinical context and underlines the significance of the present study, even though a protocol-specific behavioral impact was not observed in the table lift test. One possible explanation could be the low sensitivity of this test that is based on simple spontaneous behavior. Furthermore, the use of embryonic tissue from GFP donors was validated in cell-based therapy studies in the HD model.
The novel implantation strategy protocol, as well as the microtransplantation device were adopted from preclinical studies in Parkinson's disease (PD) where the combination of both was shown to lead to significant improvement of dopaminergic graft survival (41,42,52). The similar findings converging from both the HD and PD animal models suggest that the increase in the functionally relevant grafted neuronal phenotype could arise out of basic and common biological principles affecting the transplanted embryonic tissue in the host brain. In the case of PD, consensus has emerged that specific conditions associated with the transplantation procedure and posttransplantation interval including hypoxia/ischemia, trophic factor withdrawal, and oxidative stress render grafted cells susceptible for early cell death (1). Neuroprotective treatment strategies were developed and successfully tested that aim to reduce these detrimental triggers, which led to higher survival rates [for review see (10,51)]. In this context, Nikkhah and coworkers identified the modification of the grafting procedure as one of these strategies, demonstrating that using the multitract microtransplantation, as opposed to the single-tract macrotransplantation protocol, positively impacts on dopamine neuron survival, producing more consistent and complete reinnervation of the denervated striatum, while reducing the overall host tissue trauma and subsequent MHC and GFAP expression (9,41,43). For HD cell therapy, only few factors were identified to significantly increase DARPP-32-positive cell availability, reflecting the lack of emphasis given to this question to date. The age of donor tissue (23,54) and the grafting medium (55) were proven to influence the DARRP-32-positive cell yield; also, the extent of the QA lesion providing the environment in which the graft is placed into was positively correlated with the number of striatal neurons (56).
Applying a more active neuroprotective approach, Mundt-Petersen and colleagues (35) showed a significantly positive effect when pretreating the graft tissue with the caspase inhibitor acetyl-tyrosinyl-valyl-alanyl-aspartyl-chloromethyl-ketone (Ac-YVAD-cmk). Comparable experiments with the potentially neuroprotective substance ciliary neurotrophic factor (CNTF) (48), the spin-trapping agent α-phenyl-tert-butyl nitrone (PBN) (37), the NMDA receptor blocker MK-801, or the lazaroid U-83836E (34) have failed to produce evidence of neuroprotection of the striatal cells under the applied conditions.
As an easily applicable alternative avoiding the use of additional substances, the multitract microtransplantation protocol is a modification of the grafting procedure, based on the principle that a defined amount of cells is more widely distributed and organized in smaller sub-units. This, per definition, leads to an enlargement of the graft–host border area (mathematically, given identical total volumes, the surface area of 18 spherical bodies is more than twofold larger than of two spherical bodies). Consequently, the proportion of cells at or in close proximity to the enlarged graft–host border increases, intensifying the graft's exposure to factors provided by the host. A time course analysis looking at when the MT grafted tissue merges is required to confirm the suggestion that an increased graft–host border can be a factor in the graft development.
One host-derived neuroprotective key factor could be a protocol-related acceleration and enhancement of graft vascularization, as the predominantly host-derived vessels (7) penetrate and supply the primarily avascular and critically hypoxic cell suspension graft from the periphery towards the center of each deposit (15). Therefore, more cells being in close proximity to the host–transplant border could be rescued from ischemic cell death by receiving vascular connections at an earlier posttransplantation time point. That vascularization is essentially important for graft development is supported by studies in PD showing improved survival following enhancement of blood supply to the implanted cells (21,32).
Besides the obvious relevance of host-derived vasculature, it is clear that the surrounding host tissue substantially influences the graft's development. This is implied by several experiments showing a positive correlation between the intensity of the QA lesion and the DARPP-32-positive cell yield, which may be based on enhanced neurotrophic activity following brain injury (35,56). There is a marked accumulation of macrophages, activated microglia, and astrocytes beyond 1 week after injection of QA in the striatum (20), which have been shown to express growth factors like BDNF and GDNF (8), CNTF (3), and bFGF (27). Specifically, following QA injection, increased expression of NGF (12) in the striatum and BDNF (11) in the cortex was measured. Many publications report neuroprotective effects of these substances on striatal neurons in vitro and in vivo: CNTF (47) and BDNF (36,39) protect striatal neurons from NMDA-mediated toxicity; bFGF promotes the survival of DARPP-32-positive neurons exposed to serum withdrawal and NMDA (40); and delivery of the factors prior to QA lesioning can lead to reduced excitotoxic neuronal damage [for review see (2)].
The influence of growth factors may be particularly important for striatal grafts where, in contrast to E14 dopaminergic transplants (50), a subgroup of the transplanted progenitor cells continues to divide in the host brain (25). There, the proliferating cells need to differentiate and to undergo an appropriate neuronal maturation program, which may be influenced or controlled by host factors like BDGF that has been identified to stimulate the morphological differentiation of striatal neurons (33,53).
As the described growth factor-mediated neuroprotective effects are linked to the quinolinic acid lesion, the question arises to what extent the assumed multitract protocol-related increase of mechanical trauma contributes to the improved DARPP-32-positive cell availability. As concluded by several studies for the animal model of PD, there is an inverse relationship between the size of the outer diameter of the implantation instrument and cell survival (41,42,52), suggesting that the traumatic host response related to the implantation process itself is predominantly detrimental for the graft. This is supported by the observation that increased dopaminergic cell survival following microtransplantation is paralleled by a more discrete GFAP expression, both around the implantation tract and around the implants, when compared to grafts where the standard metal cannula was used (42). However, we can only speculate about the influence of the grafting device on the outcome of the present experiment, as the study design did not include the comparison between glass capillary and metal cannula. Nevertheless, we hypothesize that the microtransplantation technique may be a necessary prerequisite for the positive outcome of the multitract protocol as it might partially attenuate possible detrimental effects related to multiple insertion traumas.
Overall, clinical and preclinical experience suggests that there are three important factors influencing the outcome of clinical transplantation protocols: i) graft size—it appears that multiple smaller deposits give better outcome then fewer larger ones, perhaps due to the better vascularization that leads to increased access to nutrients and trophic elements; ii) graft distribution—the grafted tissue needs to be placed over a sufficient area, and for this reason single deposits are not clinically relevant; and iii) instrument size—the instruments used have been adopted and reduced in diameter in the clinic over time since the early clinical studies and are constantly being refined based on knowledge gained form basic research and from clinical work.
In conclusion, the current study demonstrates that by varying the distribution of graft deposit sites, and the number of embryonic striatal cells deposited per site, the transplantation protocol can have a significant impact on the yield of functionally important striatal-like neurons that develop within the transplant. The biological mechanisms supporting the finding remain to be elucidated, but the understanding of this phenomenon could have important translational value in refining transplantation protocols in the clinical application of cell based therapies for HD.
Footnotes
Acknowledgments
We thank Joanna Garcia and Beate Schmitt for their technical support and Eiji Kobayashi for providing us with the GFP transgenic animals. Wei Jiang was financially supported by the Stereotactic Neurosurgery Department, University Hospital Freiburg, Germany.
