Abstract
Blood vessels obtained from cadavers and amputated limbs stored at 4°C (i.e., cold stored) potentially represent an economical and readily sourced alternative to autologous vessels and synthetic prostheses for vascular reconstructive surgery. However, cold-stored vessels would need to have a reduced antigenicity and an antithrombogenic autologous endothelial cell (EC) lining before they could function as patent vascular allografts. The aim of this study was to determine the effect of cold storage for 1–16 weeks on the morphology of the ovine carotid artery wall. Ovine carotid arteries (n = 6) were rinsed and flushed with 0.9% saline, cut into segments, wrapped in 0.9% saline-soaked gauze, and stored at 4°C for 1, 2, 4, 8, or 16 weeks. Following storage, the segments were sampled and the samples fixed and sectioned for light microscopic, immunohistochemical, or transmission electron microscopic examination. After 1 and 2 weeks the ECs were karyolitic or contained pyknotic nuclei. After 4 weeks the EC layer was depleted, the subendothelial matrix exposed, and the number of smooth muscle cells (SMCs) and fibroblasts reduced. The 8- and 16-week samples demonstrated complete loss of the EC lining and only occasional remnants of SMCs or fibroblasts. The subendothelial basement membrane appeared to undergo degradative changes as early as 1 week following cold storage. At each time point examined, the subendothelial connective tissue stroma, the internal elastic lamina (IEL), and the collagenous and elastic extracellular framework were retained. These results demonstrate that the ovine carotid artery wall progressively loses its cells but retains its extracellular components during cold storage for up to 16 weeks. They suggest that cold-stored vessels may function as allografts with a reduced antigenicity for vascular reconstructive surgery. It is conceivable that seeded autologous ECs may be used to restore the antithrombogenic EC lining prior to graft implantation.
Introduction
Reconstructive vascular surgery frequently requires the use of vessels with an internal diameter of 4 mm or more for cardiac bypass procedures and restoration of circulation to ischemic limbs. Autologous vessels including the saphenous vein are preferred for such procedures but chemically treated umbilical veins (7,8,26) or synthetic prostheses (10,18,27,30,37) may be used if the former are not available. Vessels obtained from cadavers and amputated limbs stored at 4°C potentially represent an economical and readily available alternative.
A previous morphological study demonstrated that rabbit femoral arteries wrapped in normal saline-moistened gauze without nutrient or protective media and stored at 4°C for various periods sustained a decline in cell viability (6). By 4 weeks, the EC lining of the intima, the smooth muscle cells (SMCs) of the media, and the fibroblasts of the adventitia were nonviable and necrotic, producing an essentially acellular tube with an intact extracellular matrix framework. This framework remained intact for at least 6 months. The loss of the antigenic cellular component and retention of the extracellular framework enabled 4-week cold-stored rabbit femoral arteries to function as allografts in a rabbit femoral artery interposition model (5). The acellular matrix did not initiate the cell-mediated immune rejection response observed with fresh rabbit femoral artery and vein allografts (11,24).
The internal diameter of ovine carotid arteries is larger than rabbit femoral arteries (4 vs. 1 mm) and similar in size to grafts used for macrovascular reconstruction. The effect of cold storage on the ovine carotid artery wall is unknown. This study was designed to examine the histology of the progressive cold storage-induced morphological changes in ovine carotid arteries. Histological staining, immunohistochemical and transmission electron microscopy (TEM) techniques were used to investigate the effect of cold storage at 4°C on: a) the EC lining, the SMC layer, and fibroblasts present in the arterial wall, and b) the subendothelial basement membrane and extracellular matrix framework of the artery wall.
Materials and Methods
Cold Storage and Sampling
All sheep weighed 40–45 kg at the time of surgery. Ethical approval was obtained from the Bio Nova (Melbourne, Australia) Animal Ethics Committee (protocol number 2/00) and the Howard Florey Institute (Melbourne) Animal Ethics Committee (preexisting protocol numbers).
Carotid arteries (15–20 cm length) were bilaterally excised from six sheep (sheep A, B, C, D, E, and F) of the Merino strain and flushed with 50 ml 0.9% saline containing 10 U/ml heparin sodium (porcine mucous; Multiparin, Fisons Pty Ltd) to clear the lumen of blood. Each pair of arteries was divided into six segments (5–6 cm length). Each segment, with the exception of a control segment (i.e., 0 h cold storage), was individually wrapped in 0.9% saline-moistened gauze, sealed in a plastic container (Technoplas), and stored at 4°C in the refrigerator for 1, 2, 4, 8, or 16 weeks. Aseptic technique was used throughout.
Sampling of the artery segments is summarized in Table 1 and was performed as follows. Following storage, a segment was removed from the refrigerator and equilibrated to room temperature. One end was cannulated with plastic tubing secured with surgical nylon thread and the segment flushed with 40 ml Hank's balanced salt solution (Sigma, USA) injected using a syringe attached to a luer on the tubing. Samples (0.2–0.5 cm length) were cut from the segment using a scalpel blade before and after flushing and fixed for a minimum of 24 h in 10% neutral buffered formalin (NBF) for processing, paraffin embedding, and light microscopy, or in 2.5% GA for processing, Araldite-Epon embedding, and TEM (Table 1). A control segment was also sampled and fixed.
Sampling of Sheep Carotid Artery Segments

• denotes sample fixed in neutral buffered formalin for paraffin embedding and light microscopy (pre- and postflush samples). ▪ denotes sample fixed in 2.5% glutaraldehyde for transmission electron microscopy (postflush samples only).
Histology
Cross sections (5 μm) obtained from paraffin-embedded samples were stained with hematoxylin and eosin (H&E) to determine the appearance of ECs, SMCs, fibroblasts, elastin, and collagen. Additional cross sections (sheep A) were stained using orcein/van Gieson, Masson trichrome, and periodic acid-Schiff (PAS) techniques to determine the appearance of elastin, collagen, and subendothelial basement membrane, respectively. Additional cross sections (sheep A) were labeled with the antibody to von Willebrand's factor (vWF; Dako, USA), the ovine EC-binding lectin Datura stramonium lectin (DSL; Vector, USA), and antibodies to fibronectin (Dako) and laminin (Dako). Sections obtained from the araldite–epon-embedded samples were stained with uranyl acetate/lead citrate and examined by TEM to determine the ultrastructure of ECs, subendothelial basement membrane, and subendothelial connective tissue stroma.
Digital Microscopy and Assessment
All images except those of TEM sections were acquired with a Spot 2 digital camera (Diagnostic Instruments Inc., USA). In five random H&E-stained sections from each control and cold-stored segment from sheep A–D, the percentage coverage of the luminal surface with ECs was semiquantitatively scored and the results expressed as a mean. In H&E-stained sections from each control and cold-stored segment from sheep A–D, the number of ECs, SMCs, and fibroblasts was semiquantitatively graded, the appearance of collagen and elastin including the internal elastic lamina (IEL) noted, and the results summarized. In orcein/van Gieson-, Masson trichrome-, and PAS-stained sections, staining for elastin, stromatous collagen, and subendothelial basement membrane, respectively, was semiquantitatively graded. In antibody- and DSL-labeled sections, immunostaining was semiquantitatively graded.
Results
Macroscopic Appearance
Sheep carotid artery segments stored at 4°C for up to 16 weeks had a white color, a compressed appearance, and were sticky to touch. The length and diameter appeared to have undergone little, if any, change. The tubular structure was retained and the lumen readily recognized.
Histological Staining: Progressive Changes in the Artery with Increasing Cold Storage
Intima
In the control artery, the intima contained a monolayer of ECs at the luminal surface (Fig. 1A). These cells had the flattened morphology that is typical of vascular ECs. The subendothelial connective tissue stroma was of variable thickness (~1–30 μm) and contained collagen and occasional cells that are believed to be fibroblasts or SMCs. Subjacent to the stroma was the IEL, a mostly continuous band (~1–4 μm thick) that formed the boundary between the intima and media (Fig. 1A).

Cross sections showing the intima of the sheep carotid artery following cold storage for (A) 0 h (control), (B) 1 week, (C) 2 weeks, (D) 4 weeks, (E) 8 weeks, and (F) 16 weeks. Note the progressive loss of endothelial cells (ECs) and the exposure of the subendothelium. L, lumen; M, media; SECTS, subendothelial connective tissue stroma; IEL, internal elastic lamina. Hematoxylin and eosin staining.
The first indications of damage to the ECs included rounding or contraction of nuclei and detachment of the cells from the vessel wall and from other ECs (Fig. 1B, C). In the ECs that remained attached to the wall, damage was reflected by reduced nuclear mass (pyknosis, Fig. 1B) and dissolution of the nucleus (karyolysis, Fig. 1D) as indicated by weakly staining nuclei. The EC coverage on the wall at each time point was semiquantitatively assessed and demonstrated a decrease over time (Tables 2 and 3). After 4 weeks, the luminal surface appeared to be devoid of intact ECs. As a result of the loss of ECs, the subendothelium was exposed (Fig. 1D–F).
Summary of the Morphological Appearance of the Sheep Carotid Artery Before and After Cold Storage

Semiquantitative grading range; normal denotes no change from control, ↓ denotes number of cells reduced from number in control, ↓↓ denotes number of cells reduced from number represented by single arrow, ↓↓↓ denotes number of cells reduced from number represented by double arrow, with few if any cells present. Grades assessed in hematoxylin and eosin-stained cross sections from the artery segments of four sheep (i.e., sheep A, B, C, D). EXF, extracellular framework (collagen and elastin); EC, endothelial cell; SMC, smooth muscle cell; IEL, internal elastic lamina.
Semiquantitative Grade of Periodic Acid-Schiff Staining for Subendothelial Basement Membrane, of Masson Trichrome Staining for Stromatous Collagen, and of Orcein/ Van Gieson Staining for Elastin [Including the Internal Elastic Lamina (IEL)] in the Sheep Carotid Artery Before and After Cold Storage

Semiquantitative grading range: +++ marked staining, ++ moderate staining, + mild staining. Grades assessed in cross sections from the artery segments of one sheep (i.e., sheep A).
Media
In the control artery, the media contained more than 30 layers of concentrically arranged SMCs. The SMC nuclei had an elongated morphology typical of contractile muscle cells (Fig. 2A). The SMCs were embedded in extracellular material that consisted of bands or fibers of collagen and elastin.

Cross sections showing the media of the sheep carotid artery following cold storage for (A) 0 h (control), (B) 2 weeks, (C) 4 weeks, (D) 8 weeks, and (E) 16 weeks. Note the progressive loss of smooth muscle cells (SMCs). L, lumen; M, media; Ad, adventitia. Hematoxylin and eosin staining.
Damage to the SMCs was reflected by karyolysis (Figs. 1C, 2C, D). Damage to SMCs appeared to take longer to develop than in ECs. The number of SMCs at each time point was semiquantitatively assessed and demonstrated a decrease over time (Table 2). After 8 and 16 weeks, few, if any, intact SMCs were present (Fig. 2D, E).
Adventitia
In the control artery, the adventitia contained scattered fibroblasts. The fibroblast nuclei had a round, elongated, or stellate morphology typical of fibroblasts (Fig. 3A). The fibroblasts were embedded in extracellular material containing collagen and elastin.

Cross sections showing the adventitia of the sheep carotid artery following cold storage for (A) 0 h (control), (B) 4 weeks, and (C) 8 weeks. Note the progressive loss of fibroblasts. Ad, adventitia; M, media. Hematoxylin and eosin staining.
Damage to fibroblasts was reflected by karyolysis (Fig. 3B, C). Damage to fibroblasts appeared to occur at a similar rate as in SMCs. The number of fibroblasts at each time point was semiquantitatively assessed and demonstrated a decrease over time (Table 2). After 8 weeks, few, if any, intact fibroblasts were present.
Basement Membrane and Extracellular Framework
In the control artery, the EC monolayer rested on a continuous basement membrane (Fig. 4A). The basement membrane overlay the subendothelial connective tissue stroma.

Cross sections showing the intensity of staining for the subendothelial basement membrane (arrowhead) of the sheep carotid artery following cold storage for (A) 0 h (control), (B) 1 week, (C) 2 weeks, (D) 4 weeks, and (E) 8 weeks. Note the reduction in the intensity of staining from that seen in the control. L, lumen; M, media; IEL, internal elastic lamina. Periodic acid-Schiff staining.
Following cold storage, the subendothelial basement membrane appeared to be partly degraded (Table 3, Fig. 4B–E). The subendothelial connective tissue stroma appeared to be intact and remained attached to the IEL (Fig. 1B–F). The IEL also appeared intact (Tables 3 and 4 Fig. 1B–F). Collagen in the media and adventitia remained intact (Table 2). Elastin in the media and adventitia also remained intact (Tables 3 and 4). Spaces formerly occupied by SMC were noticeable in the media.
Semiquantitative Grade of Immunostaining for Von Willebrand's Factor (vWF) and Datura stramonium Lectin (DSL)-Labeled Glycoproteins in Endothelial Cells and for Fibronectin and Laminin in the Subendothelial Basement Membrane of the Sheep Carotid Artery Before and After Cold Storage

Semiquantitative grading range: +++ marked staining, ++ moderate staining, + mild staining, +/- possible staining. Grades assessed in cross sections from the artery segments of one sheep (i.e., sheep A).
Immunohistochemistry
von Willebrand's Factor
The progressive changes in immunostaining for vWF expressed by ECs are summarized in Table 4, and demonstrated in Figure 5. In the control artery, intense staining appeared in the ECs (Fig. 5A), which were present as a monolayer at the luminal surface. The subendothelial connective tissue stroma did not stain. Following 1 and 2 weeks of cold storage, the ECs continued to stain intensely although their numbers were reduced (Fig. 5B, C). The subendothelial connective tissue stroma also stained, suggesting release of vWF into the stroma as a result of EC breakdown during cold storage. Following 4 and 8 of weeks cold storage, no intact ECs were present and the subendothelium was exposed (Fig. 5D, E). The subendothelial connective tissue stroma continued to stain throughout. There was no difference between pre- and postflush samples at each time point.

Cross sections showing immunostaining for von Willebrand's factor (vWF) in endothelial cells (ECs) of the sheep carotid artery following cold storage for (A) 0 h (control), (B) 1 week, (C) 2 weeks, (D) 4 weeks, and (E) 8 weeks. Note there is no reduction in intensity of immunostaining from that seen in the control but that the ECs are progressively lost. L, lumen; M, media; SECTS, subendothelial connective tissue stroma; IEL, internal elastic lamina. Stain: anti-vWF antibody/biotinylated immunoglobulin/peroxidase-conjugated streptavidin/diamino benzidine; hematoxylin counterstaining.
DSL-Labeled Glycoproteins
The progressive changes in immunostaining for DSL-labeled glycoproteins expressed by ECs are summarized in Table 4 and demonstrated in Figure 6. In the control artery, intense staining with a fine linear distribution appeared to be confined to the EC membranes (Fig. 6A). The subendothelial connective tissue stroma did not stain. Following 1 and 2 weeks of cold storage, the ECs stained less intensely and their numbers were reduced (Fig. 6B, C). The reduction in staining intensity suggests that the sugar moieties in the EC glycoproteins, which bind to DSL, were less resistant to cold storage than the antigenic determinant in vWF. The subendothelial connective tissue stroma stained lightly, suggesting release of glycoproteins into the stroma as a result of EC breakdown during cold storage. Following 4 and 8 weeks of cold storage, no intact ECs were present and the subendothelium was exposed (Fig. 6D, E). The subendothelial connective tissue stroma continued to stain throughout. There was no difference between pre- and postflush samples at each time point.

Cross sections showing immunostaining for glycoproteins on endothelial cells (ECs) of the sheep carotid artery following cold storage for (A) 0 h (control), (B) 1 week, (C) 2 weeks, (D) 4 weeks, and (E) 8 weeks. Note the reduction in intensity of immunostaining from that seen in the control and the progressive loss of ECs. L, lumen; M, media; SECTS, subendothelial connective tissue stroma; IEL, internal elastic lamina. Stain: biotinylated Datura stramonium lectin/peroxidase-conjugated streptavidin/diaminobenzidine; hematoxylin counterstaining.
Fibronectin
The progressive changes in immunostaining for fibronectin are summarized in Table 4 and demonstrated in Figure 7. In the control artery, fibronectin had an intensely staining, continuous linear distribution immediately subjacent to the EC monolayer where the basement membrane resides (Fig. 7A). The subendothelial connective tissue stroma did not stain. Following 1 and 2 weeks of cold storage, the linear staining was less readily distinguished due to: a) a reduction in staining intensity, b) a discontinuous distribution of staining, and c) the staining of the subendothelial connective tissue stroma (Fig. 7B, C). Following 4 and 8 weeks of cold storage, no further changes appeared to occur (Fig. 7D, E). There was no difference between pre- and postflush samples at each time point.

Cross sections showing the intensity and distribution of immunostaining for fibronectin in the subendothelial basement membrane of the sheep carotid artery following cold storage for (A) 0 h (control), (B) 1 week, (C) 2 weeks, (D) 4 weeks, and (E) 8 weeks. Note the reduction in both the intensity and continuity of immunostaining from that seen in the control. L, lumen; M, media; SECTS, subendothelial connective tissue stroma; IEL, internal elastic lamina. Stain: anti-fibronectin-horseradish peroxidase conjugated antibody/diaminobenzidine; hematoxylin counterstaining.
Laminin
The progressive changes in immunostaining for laminin are summarized in Table 4 and demonstrated in Figure 8. In the control artery, laminin had an intensely staining, continuous linear distribution immediately subjacent to the EC monolayer where the basement membrane resides (Fig. 8A). The subendothelial connective tissue stroma did not stain. Following 1 and 2 weeks of cold storage, the linear staining was less readily distinguished due to: a) a reduction in staining intensity, b) a discontinuous distribution of staining, and c) the staining of the subendothelial connective tissue stroma (Fig. 8B, C). Following 4 and 8 weeks of cold storage, no further changes appeared to occur (Fig. 8D, E). There was no difference between pre- and postflush samples at each time point.

Cross sections showing the intensity and distribution of immunostaining for laminin in the subendothelial basement membrane of the sheep carotid artery following cold storage for (A) 0 h (control), (B) 1 week, (C) 2 weeks, (D) 4 weeks, and (E) 8 weeks. Note the reduction in both the intensity and continuity of immunostaining from that seen in the control. L, lumen; M, media; SECTS, subendothelial connective tissue stroma; IEL, internal elastic lamina. Stain: anti-laminin antibody/biotinylated immunoglobulin/peroxidase-conjugated streptavidin/diaminobenzidine; hematoxylin counterstaining.
Transmission Electron Microscopy
In the intima of the control artery, the luminal lining consisted of a monolayer of cells with morphologic features that are consistent with ECs (Fig. 9A). These included a flat shape, attenuated peripheral cytoplasm, intercellular contacts, Weibel-Palade bodies, pinocytotic vesicles, glycogen granules, and a large nuclear to cytoplasmic ratio (Fig. 9B). The EC monolayer sat on the subendothelial basement membrane (Fig. 9B). Subjacent to the basement membrane was the subendothelial connective tissue stroma (Fig. 9A). The tissue stroma contained: a) collagen fibrils organized into bundles that appeared to run approximately at right angles to one another, b) a nonstaining gel-like substance composed of proteoglycans and glycosaminoglycans, and c) cells that may include fibroblasts and SMCs. Subjacent to the stroma was the IEL, a mostly continuous band that formed the boundary between the intima and media.

Transmission electron micrograph showing (A) the intima of the sheep carotid artery following 0 h cold storage (control) and (B) an endothelial cell (EC) of the intima of the sheep carotid artery following 0 h cold storage (control). L, lumen; SECTS, subendothelial connective tissue stroma. Uranyl acetate and lead citrate staining. Original magnification: (A) x5,000, (B) x10,000.
Following 4 weeks of cold storage, the intima was devoid of intact ECs and the luminal surface consisted of an exposed subendothelium containing occasional cellular debris. In parts of the surface, the subendothelial basement membrane or its remains were present as a layer of fine fibrils (Fig. 10). In other parts of the surface, the subendothelial membrane or its remains were absent (Fig. 11). In the parts of the surface that were devoid of the basement membrane or its remains, the subendothelial connective tissue stroma was exposed (Fig. 11). Bundles of collagen fibrils and the gel-like substance consisting of proteoglycans and glycosaminoglycans persisted within the stroma (Figs. 10, 11). Cells in the stroma had a damaged appearance. The IEL was intact (Figs. 10, 11A).

Transmission electron micrograph showing the intima of the sheep carotid artery following 4 weeks of cold storage. L, lumen; M, media; IEL, internal elastic lamina; SECTS, subendothelial connective tissue stroma. Uranyl acetate and lead citrate staining. Original magnification: x5,000.
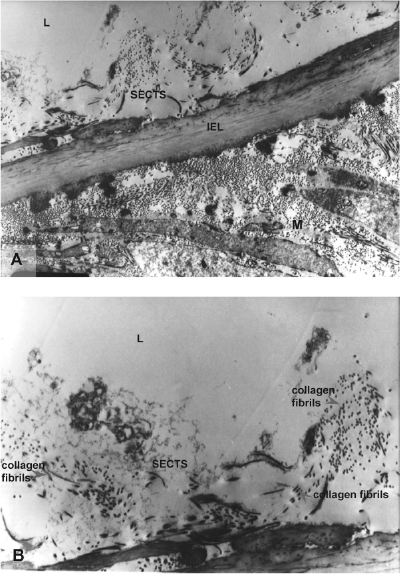
Transmission electron micrograph showing the intima of the sheep carotid artery following 4 weeks of cold storage. L, lumen; M, media; IEL, internal elastic lamina; SECTS, subendothelial connective tissue stroma. Uranyl acetate and lead citrate staining. Original magnification: (A) x4,000, (B) x8,000.
Figure 12 shows the appearance of the intima following 16 weeks of cold storage. At this time period, uranyl acetate and lead citrate staining intensity in the intima was generally reduced compared to that observed following 0 h and 4 weeks cold storage. No intact ECs were present and the luminal surface consisted of an exposed subendothelium. The subendothelial basement membrane or its remains were present in parts along the surface but absent in others. Bundles of collagen fibrils persisted within the subendothelial connective tissue stroma as did the gel-like substance consisting of proteoglycans and glycosaminoglycans. Cellular debris was occasionally seen in the stroma. The IEL was intact.

Transmission electron micrograph showing the intima of the sheep carotid artery following 16 weeks of cold storage. Note the generally weak intensity of the staining. Basement membrane remnants and collagen fibrils are visible. L, lumen; M, media; IEL, internal elastic lamina; SECTS, subendothelial connective tissue stroma. Uranyl acetate and lead citrate staining. Original magnification: (A, B) x3,000.
Discussion
The effect of storage at 4°C on the morphology of small diameter (1 mm) rabbit femoral arteries wrapped in normal saline-moistened gauze has previously been described as a progressive loss or death of the constituent cells and retention of the extracellular framework (6). Until now, it was not known if the effect on sheep carotid arteries stored at the same temperature using the same protocol was similar. Using histologic, immunohistochemical, and TEM techniques, the present study found that the effect of cold storage on sheep carotid arteries was similar. These morphological methods are generally unable to identify dead cells but can detect the necrotic changes such as pyknosis and karyolysis that occur after cell death (22,36).
Up to 4 weeks of cold storage the ECs of the sheep carotid artery were progressively lost from the luminal surface. This exposed the subjacent extracellular substrate or subendothelium. The exact time point of EC death was not determined but the presence of pyknotic ECs as early as 1 week after cold storage indicates that some ECs were dead before that time. This observation is consistent with the time frame of EC death in cold-stored rabbit femoral arteries (6). Although SMCs and fibroblasts were also progressively lost from the sheep carotid artery during the initial 4 weeks of cold storage, they appeared to be somewhat more resistant than the ECs and required a further period of storage before breakdown of all cells occurred. This suggests that SMCs and fibroblasts take longer to die and is consistent with observations in cold-stored rabbit femoral arteries (6).
The overall changes in the rabbit femoral artery and now the sheep carotid artery were broadly similar to those reported for human saphenous veins stored in normal saline at 4°C (9), rat femoral veins stored in lactated Ringer's solution at 4°C (32), and rabbit aorta stored in Krebs solution at 4°C (20).
The reasons for the disparate rates of EC and SMC/fibroblast breakdown in the sheep carotid artery following cold storage are not fully understood. It is suspected that the flat morphology and monolayer nature of the ECs presents a high surface area to volume ratio that predisposes these cells to the damaging effects of the cold. The SMCs and fibroblasts, unlike the ECs, are embedded within an extracellular matrix, which may act to insulate these cells. The relatively rapid decline in viability of ECs has also been observed within 24–48 h in organs perfused with cold solutions (25) and in aortic rings stored in preservation solutions in the cold (31). A possible contributing factor to the detachment of ECs is cold storage-induced changes to the subendothelial basement membrane and its constituent adhesive proteins including fibronectin and laminin. It is thought that these changes, which appear to commence within 1 week of cold storage, may reduce the adhesiveness of the substrate and cause the ECs to become less firmly adhered and eventually detach.
The extracellular framework of a blood vessel, which consists largely of collagen and elastin, is responsible for its tensile strength (12,33,35,40). The retention of the collagenous and elastic extracellular framework in sheep carotid arteries following cold storage suggests that the vessel retains sufficient strength to sustain physiological blood pressure as a graft. Rabbit femoral arteries stored in the cold for a minimum of 4 weeks, which also retain their extracellular frameworks, function as patent, albeit noncontractile, conduits of blood in the rabbit femoral artery interposition model for 12 weeks (5,16) or 6 months (4) without aneurysm formation.
Immunohistochemistry demonstrated that two of the proteins that constitute the subendothelial basement membrane, namely fibronectin and laminin, appeared to be partly degraded during cold storage. These glycoproteins are known to contain short peptide sequences such as RGD (arginine-glycine-aspartic acid) that bind to EC integrins (1,14,15,28,29,39). It is speculated that if the RGD sequences persist in remnant fibronectin or laminin following cold storage, then the substrate may retain some of the adhesive properties of the basement membrane of the normal vessel (29). The persistence of the basement membrane or its remains in parts of the exposed luminal surface of the sheep carotid artery following cold storage for 4 weeks or greater, as determined by TEM, is consistent with observations in the 4-week cold-stored rabbit femoral artery (5).
There are several possible functional implications arising from the cold storage-induced changes to the sheep carotid artery, as indicated below.
The retention of the elastic and collagenous extracellular framework of the sheep carotid artery following cold storage suggests that these vessels may retain the strength to sustain arterial pressure when implanted as arterial grafts. It has been demonstrated that rabbit femoral arteries stored in the cold for 4 weeks, and which consequently lack viable cells, sustain arterial pressure in the rabbit femoral artery interposition model due to their intact extracellular frameworks (5,6,16).
Vascular ECs are known to be prime stimuli for immunologic rejection of fresh vascular allografts (13,34,41). These cells are situated at the blood/vessel interface and contain antigens that are readily recognized by the host as foreign. Deendothelialization of the sheep carotid artery after 4 weeks of cold storage presumably removes the antigens that initiate the cell-mediated rejection response associated with fresh vascular allograft implantation. If the SMCs and fibroblasts are damaged to an extent that reduces their antigenicity, then 4-week cold-stored sheep carotid arteries may not induce a cell-mediated rejection response when implanted as allografts. For precedent, rabbit femoral arteries stored in the cold for 4 weeks did not induce a cell-mediated rejection response (5) when implanted as allografts, the reason being that the constituent cells had been lost or reduced to structureless debris during the storage process (6). Rat aortas that had their cells removed by chemical treatment did not induce a rejection response following implantation as allografts (2). Canine jugular veins that were also chemically decellularized demonstrated little, if any, evidence of rejection following their implantation as allografts (23). If cold-stored macrovascular allografts do not induce a rejection response then they may be appropriate for vascular replacement in a clinical setting.
The subendothelium of the sheep carotid artery that is exposed following a minimum of 4 weeks of cold storage may retain some adhesive properties. If binding sites for ECs are present, it may be possible to reendothelialize the surface through the technique of seeding using ECs or stem cells isolated from blood vessels and fat (3,38). Following seeding, the ECs might be capable of producing a new, more complete basement membrane on which to spread and replicate (3,17,19,21).
Conclusion
Using histologic, immunohistochemical, and TEM techniques, it was found that increasing storage at 4°C induced morphological changes in the sheep carotid artery in the form of changes to the cells and the subendothelial basement membrane:
Cellular changes: Following 1 and 2 weeks of cold storage, many ECs were necrotic and in the process of detaching. Some of the SMCs and fibroblasts were also necrotic. Following 4 weeks of cold storage, the ECs were depleted and the subendothelium exposed. Many, if not most, of the SMCs and fibroblasts were necrotic. Following 8 and 16 weeks of cold storage, most, if not all, SMCs and fibroblasts had completely broken down.
Basement membrane changes: Following 1 and 2 weeks of cold storage, fibronectin and laminin appeared to be partly degraded. Following 4 and 8 weeks of cold storage, no further changes appeared to occur. Following 4 and 16 weeks of cold storage, the basement membrane or its remains were present in parts of the surface but absent in others when examined by TEM.
At each time point examined, the subendothelial connective tissue stroma was retained. Additionally, the IEL was retained as was the collagenous and elastic framework of the media and adventitia. The cold storage-induced decellularization of the sheep carotid artery and the retention of the extracellular framework is consistent with previous observations in rabbit femoral arteries stored at 4°C using the same storage protocol (6).
Footnotes
Acknowledgments
Hugh McCormack and Nathan Ketharanathan (Bio Nova, West Melbourne, Victoria, Australia) helped to provide the facilities for the experiments. Kim McKellar (Bio Nova, West Melbourne, Victoria, Australia) and Alan McDonald (Florey Neuroscience Institutes, Parkville, Victoria, Australia) performed the surgical excisions regarding the arteries. Stephen Cordner (Department of Forensic Medicine, Monash University, Victoria, Australia) and Lynn Ireland (Head of Donor Tissue Bank, Victoria, Australia) contributed to A.S.'s Ph.D. scholarship. The authors declare no conflict of interest.
