Abstract
In skin, the regeneration of the ontogenically distinct mesenchymal and epithelial compartments must proceed in a coordinated manner orchestrated by extracellular signaling networks. We have recently found that the switch from regeneration to remodeling during repair is modulated by chemokines that bind CXCR3 receptor. If this signaling is disrupted wounds continue to be active, resulting in a chronic hypercellular and hypertrophic state characterized by an immature matrix composition. As healing is masterminded in large part by fibroblasts and their synthesis of the extracellular matrix, the question arose as to whether this ongoing scarring can be modulated by transplanted fibroblasts. We examined wounds in the CXCR3-/-mouse scarring model. These wounds exhibited a significant delay in healing in all areas compared to young and aged wild-type mice. Full-thickness wounds were transplanted with fibroblasts derived from newborn CXCR3-/- or wild-type mice. The transplanted fibroblasts were labeled with fluorescent dye (CM-DiI) and suspended in hyaluronic acid gel; by 30 days, these transplanted cells comprised some 30% of the dermal stromal cells regardless of the host or source of transplanted cells. Wild-type fibroblasts transplanted into CXCR3-/- mice wounds reversed the delay and dysfunction previously seen in CXCR3-/- wounds; this correction was not noted with transplanted CXCR3-/- fibroblasts. Additionally, transplant of CXCR3-/-cells into wounds in wild-type animals did not adversely affect those wounds. The transplanted fibroblasts exhibited strong survival and migration patterns and led to an increase in tensile strength. Expression of matrix proteins and collagen in CXCR3-/- wounds transplanted with wild-type fibroblasts resembled normal wild-type healing, and the wound matrix in wild-type mice transplanted with CXCR3-/- cells also presented a mature matrix. These suggest that the major determinant of healing versus scarring lies with the nature of the matrix. These findings have intriguing implications for rational cellular interventions aimed at promoting wound healing via cell therapy.
Introduction
Scarring is a consequence of the imperfect wound healing response. In adult wound repair, scarring results mainly from dysregulated collagen synthesis by fibroblasts that proliferate and differentiate within the provisional matrix (9). In fact, as we age the elaborate healing process is impaired (6). Thus, in an population with more elderly individuals with underlying pathologies that contribute to wounds, the dysfunctional repair leads to an increased incidence of chronic wounds that can result in slow/delayed healing early and dysfunctional remodeling. While it is well recognized that the quality and rate of healing depends on a balance of cellular responses and signaling that is disrupted in nonhealing wounds, it remains unknown how best to improve healing, whether by cell or matrix replacement.
The process of wound healing requires a well-orchestrated series of events involving numerous extracellular components, cell types, and mediators (5). Even though inflammation, angiogenesis, and reepithelialization are important components of tissue repair, it is fibroplasia that allows the injured dermis to regain its form and function. Fibroplasia is marked by the ingrowth of fibroblasts and neomatrix deposition to form loosely woven tissue that provides support for the neoepidermis and the origin of a neodermis (21). In dermal injuries, wound repair results in a matrix (collagen I-predominant)—cell (mainly fibroblasts) composite, wherein a significant degree of disorganization leads to a tissue phenotype that is commonly described as a scar (7,26). Thus, fibroblasts guided by a panoply of signals play an important role, possibly even the initiator or pathfinder role from which the other events follow. It is now known that the dysfunction of this process results in delayed and deficient healing.
Interestingly, we have found a dynamic signaling system that modulates fibroblast migration, differentiation, and subsequent involution in the wound bed after injury. The cysteine-X amino acid—cysteine receptor CXCR3 signaling system modulates the later stages of repair to drive this towards resolution and maturation of the dermis (3,15,25). This seven-transmembrane classical G-protein-coupled receptor and its ligands [two of which are inducible protein 9 (IP-9)/CXC11 and IP-10/CXC10] mediates a paracrine epithelial—mesenchymal loop that contributes to transitioning the wound towards dermal maturity (15,23). CXCR3 activation limits the fibroblast motility induced by classical growth factors (18). In mice lacking CXCR3, fibroblasts failed to channel motility into contractility and matrix maturation (25). Additionally, we have found that the exuberant cellular responses of wound repair is reversed late in the healing process, at least in part, by this related group of chemokines that appears late in the regenerative phase and persists into the resolving phase of wound healing; this is the time at which cellularity is reversed and the wound bed matures (23,25).
Herein, we explore whether cells can correct the fibroblast-generated matrix dysfunction exhibited in both aged and CXCR3-/- mice wounds. As both geneand stem cell-based therapy approaches may result in numerous technical and safety problems, and even oncogenic potential (11,12), we have considered using mature cells. The goal of this present study was to determine if transplant of normal newborn fibroblasts into aged and/or CXCR3-/—deficient wounds could restore matrix maturation and wound resolution with minimal scarring and inflammation despite the presence of endogenous fibroblasts. Our finding provides proof of the feasibility of using transplanted fibroblasts for the development of cellular therapies for wound healing to create a more functional and regenerative healing response.
Materials and Methods
Animals
C57BL/6J mice, wild-type mice, and CXCR3 expression-abrogated mice (obtained from Dr. Wayne Hancock and Dr. Bao Lu; Millennium Pharmaceutical Incorporated, Cambridge, MA and Perlmutter Laboratory, Children's Hospital, Boston, MA) (8) were bred in house. All of the offspring were genotyped by PCR before use. Control, age-matched C57Bl6/J mice were obtained from Jackson Laboratories (Bar Harbor, ME). Serological analyses did not detect blood-borne pathogens or evidence of infection. Mice were housed in individual cages after wounding and maintained under a 12-h light/dark cycle and temperature. All studies on these animals were performed after approval by, and in compliance with, the Institutional Animal Care and Use Committees of the Veteran's Administration and University of Pittsburgh; these animals were housed in a facility of the Veteran's Affair Medical Center (Pittsburgh, PA) accredited by the Association for the Assessment and Accreditation of Laboratory Animal Care.
Wounding
Male and female mice [7—8 weeks (hereafter referred to as young) or 12—14 months of age (aged) weighing approximately 25—35 g] were anesthetized with an intraperitoneal injection containing ketamine (75 mg/kg) and xylazine (5 mg/kg). No less the eight mice from each group were used in each experiment. The backs were cleaned, shaved, and sterilized with betadine solution. For full-thickness wounds, a 6-mm-diameter punch biopsy through the epidermis and dermis was made on one side of the dorsal midline (to minimize wound contraction), and the contralateral uninjured skin served as a control. The wounds were covered with liquid occlusive dressing (New-Skin; Medtech, Jackson, WY) to prevent infection and desiccation.
Cultured Skin Fibroblasts
The donor cells were cultured according to existing protocols (24). These were derived from neonatal (2—4 days old) and adult (12 months old) mice. Full-thickness biopsy specimens (2×0.5 cm) were obtained from the mouse skin after the skin was prepared with 10% povidone-iodine and 70% alcohol swabs. Briefly, primary fibroblasts were isolated from the skin by standard procedures. Skin from the torso of mice was exposed to 0.25% trypsin in saline overnight at 4°C. The epidermis was removed as a sheet, and the dermis was minced and then agitated at 37°C for 45 min in 5 ml of fibroblast complete growth media. The minced dermis was filtered through nylon screens (Sefar Filtration Inc., Depew, NY) to remove the stratum corneum and remaining epidermal layers. Cells were pelleted by centrifugation and resuspended in fresh complete growth media for experiments. Fibroblasts were harvested from primary culture after 2 days and either subcultured immediately or cryopreserved and stored in liquid nitrogen for future experiments.
Cell Labeling and Gel Suspension
Carbocyanine dye (CellTracker CM-DiI; Molecular Probes Inc., Eugene, OR) was used to label the cultured fibroblasts according to the manufacturer's standard protocol. The cells were counted and suspended in Hanks balanced salt solution (Life Technologies Inc, Rockville, MD) followed by mixing using extrusion from a syringe with hyaluronic acid (HA) non-cross-linked polymer (Hylan A gel; Genzyme Biosurgery, Cambridge, MA) until a homogeneous mixture was obtained containing approximately 15 × 106 cells/ml (13).
Cell Transplantation
A minimum of eight mice in each group were used in the experiments based on statistical considerations to provide power to detect a 30% difference. Full-thickness incisional wounds were created in the dorsal skin of the animals. A grid was made using a permanent marker. A 1-ml syringe with a 24-gauge needle was then used to place approximately 25 μl of the hyaluronic acid—fibroblast mixture to each wound. Control wounds received HA only. The wounds were left untouched for 5—10 min after which a coating of liquid bandage (NewSkin; Medtech Products Inc.) was applied to every treated and control wound individually and allowed to dry for 5 min. The entire experimental area was then covered with a sterile transparent occlusive dressing (Tegaderm; 3M, Minneapolis, MN) and secured with a nontoxic degradable adhesive (Mastisol; Ferndale Laboratories Inc., Ferndale, MI).
Histological Analysis
Wound bed biopsies surrounded by a margin of non-wounded skin were collected after euthanasia at 7, 14, and 30 days postwounding. Wound biopsies were fixed in 10% buffered formalin, processed, and embedded in paraffin blocks using standard protocols. Tissue sections (4 μm) were stained with hematoxylin and eosin (H&E) and analyzed for general tissue and cellular morphology. Collagen deposits were evaluated by Masson's trichrome staining and picrosirius red staining using MetaMorph software analysis (25).
Dermal Maturation Assessment
Histopathological examination of the murine tissues was performed in a blinded manner as previously described (24). Qualitative assessments were made concerning aspects of dermal and epidermal maturation, inflammation, and granulation tissue. The samples were scored on a scale of 0 to 4 for epidermal healing (0, no migration; 1, partial migration; 2, complete migration with partial keratinization; 3, complete keratinization; and 4, normal epidermis) and dermal healing (0, no healing; 1, inflammatory infiltrate; 2, granulation tissue present—fibroplasias and angiogenesis; 3, collagen deposition replacing about half of granulation tissue; and 4, complete healing). Quantification of fibroblastic hypercellularity was performed using Meta-Morph software (Molecular Devices, Downingtown, PA). The cell counting function allowed for the direct enumeration of dermal nuclei per high-powered field (23,24).
Inflammation
Histopathological examination of mouse tissues was performed in a blinded manner. Acute inflammation was defined as the presence of neutrophils, and chronic inflammation by the presence of plasma and monocytic cells (0, none; 1, slight; 2, moderate; 3, abundant). In both situations the scale was relative level of cells per high-power field as previously described (23).
Tensile Strength
Biopsies were bisected, wrapped flat in foil, snap-frozen in liquid nitrogen, and then stored at −80°C. For the tensile strength measurements, the frozen specimens were divided into two samples, the cross-sectional area measured with calipers, and then the samples were clamped in a tensiometer and force was exerted until wound disruption. Measurements were recorded by a customized computer software program and tensile strength calculated using the formula: maximum tensiometer reading (converted to g) divided by cross-sectional area (mm2) = tensile strength (g/mm2). The results for individual specimens from one wound were combined to determine an average tensile strength per wound. The average tensile strength per wound was tabulated for each group.
Quantitative Real-Time RT-PCR
RNA was isolated and reverse transcribed with a High Capacity cDNA Reverse Transcription Kit (Applied Biosystems, Rotkreuz, Switzerland). TaqMan real-time PCR primer/probe mixtures for mouse fibronectin (Mm01256734_m1), mouse tenascin-C (Mm00495662_m1), mouse type I collagen (α-1 chain; COL1A1; Mm00801666_g1), and mouse glyceraldehyde 3-phosphate dehydrogenase (GAPDH; Mm99999915_g1) as well as TaqMan Universal Master Mix were purchased from Applied Biosystems. The primers and Taqman probes were designed using Primer Express software (Applied Biosystems, Foster City, CA) (14). Forward and reverse primers were purchased from Integrated DNA Technologies (Coralville, IA) and fluoro-coupled Taqman probes were purchased from Applied Biosystems. The reverse transcription (RT) reaction (using reverse primer) and subsequent real-time PCR assays were performed using the same conditions as previously described (14). For every experimental condition, reverse transcribed cDNA (6 ng) was amplified for each of the three genes on an ABI Prism 7900 real-time PCR cycler (Applied Biosystems). Using the comparative critical cycle (Ct) method and using GAPDH as the endogenous control, the expression levels of the target genes were normalized and the relative abundance was calculated. Results shown are the synthesis of at least three independent determinations. Data were analyzed using the 7900 HT SDS software version 2.1 provided by Applied Biosystems.
Tunel Assay
Frozen sections (5 μm thickness) or cells from each case and time point under investigation were examined for the presence of fragmented DNA in apoptotic cells by the terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) technique using the Roche TUNEL in situ staining kit (Roche Molecular Biochemicals, Basel, Switzerland), according to the manufacturer's instructions. To detect DNA fragmentation associated with apoptosis, we used a fluorescence-based TUNEL (false colored red) followed by counterstaining with 4,6-diamidino-2-phenylindole (blue) (Vector Laboratories, Burlingame, CA). Staining was quantified using MetaMorph software analysis (24).
Statistical Analyses
Results are expressed as mean ± SEM with all individual experiments performed in triplicate. Statistical differences between groups were determined by the Student's t-test. Paired analyses were performed between all groups; number of animals per group was determined a priori based on a 90% confidence of finding a 30% difference between the groups. Comparisons over time were performed by analysis of variance. Significance was claimed for p < 0.05.
Results
Aging and Lack of CXCR3 Independently Both Diminish Dermal Wound Repair
Our earlier work had established that the absence of CXCR3, which is present on most formed elements of the skin, leads to a delay in dermal wound healing (24). Further, full-thickness wounds in mice lacking this receptor presented abnormal healing, hypercellularity, and ultimately a hypertrophic scar (23). In vitro analysis revealed that this signaling system is necessary for the inhibition of fibroblast locomotion by preventing cellular deadhesion or detachment at the trailing edge and thus channeling the transcellular contractility into matrix/substratum contraction (20). A histological evaluation of wounds even after 30 days in CXCR3-/- young mice (8—10 weeks old) demonstrated dermal immaturity shown by cellularity (Fig. 1A). Measurement of overall wound maturation included examination for epidermal thickness, hyperkeratinization, chronic inflammation, overall cellularity, fibroblast, cellularity, vascularity, and collagen modeling of wound bed; these analyses confirmed this wound immaturity in the absence of CXCR3 (Fig. 1B) (23).
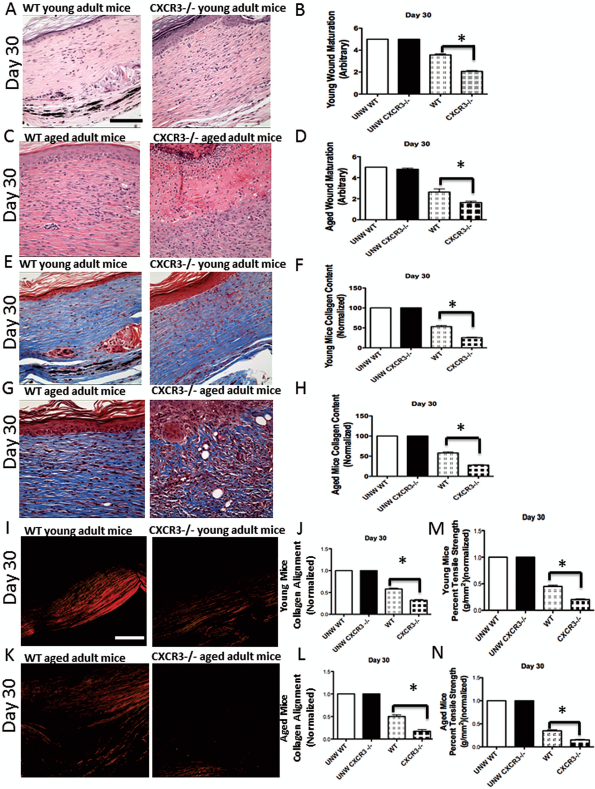
Age, in addition to cysteine-X amino acid—cysteine receptor CXCR3 presence, impacts skin wound healing. (A) Histological analysis of H&E-stained sections of wounds in the CXCR3-/- young mice presented impaired maturation compared to wounds in the wild-type (WT) mice. (B) Quantification of these wounds revealed that wound maturation in CXCR3-/- mice was significantly impaired in comparison to that in the wild-type mice. (C) Histological analysis of wounds in aged mice showed further impairment and delay healing in both CXCR3-/- and wild-type mice. (D) Quantification revealed that the wound maturation impairment was greater in the aged CXCR3-/- wounds compared to the aged wild-type mice. (E) Masson's trichrome staining for collagen showed similar patterns of collagen between young CXCR3-/- and wild-type mice. (F) MetaMorph analysis of the wound collagen confirmed that the CXCR3-/- wounds had significantly less collagen than wild-type wounds at day 30. (G) In aged mice the Masson's trichrome microscopic images showed that CXCR3-/- wound collagen was less organized than that of wild-type wounds. (H) Quantitative analysis confirmed that aged CXCR3-/- mice presented significantly less collagen. (I) Picrosirius red staining revealed poorly aligned short collagen fibrils in CXCR3-/- mice even at a young age. (J) Quantitative analysis of total integrated birefringence compared with contralateral unwounded (UNW) skin shows a significant difference between the wild-type and CXCR3-/- mice at day 30. (K) Picrosirius red staining showed that the healing of both the aged wild-type and CXCR3-/ − wounds were less organized with shorter collagen fibers and an immature scar. (L) Quantitative analysis confirms that both the aged wild-type and CXCR3-/- wounds were less organized with aged CXCR3-/-, demonstrating an even greater deficit. (M) The tensile strength of the healing wounds in the young CXCR3-/- mice was less than that of young wild-type mice. (N) The strength of the wounds of the aged CXCR3-/- mice was reduced compare to the wild-type mice and compared to the younger mice. Shown are representative of nine wounds in each mouse variant. The images measure 300 μm on each side. Original magnifications 400x. The white and black bar in the photomicrographs measures 50 μm. For the graphs, shown are mean ± SEM, n = 9 (*p < 0.05).
Healing is slower in older animals but is still effective in most situations. Healing in older mice results in a number of morphological changes such as increased dermal thickness, deceased absolute cell number, and a disorganized microcirculation, all of which lead to a modified cellular response and matrix remodeling (2). We postulated that the dual insults of aging and absent CXCR3 signaling would further aggravate this deficit. Analysis of full thickness wounds of 12-month-old CXCR3-/- mice demonstrated many physiological differences compared with the healing of wild-type aged mice (Fig. 1C). Additionally, it is noted that the wild-type aged wound had a thicker and immature epidermis; this is usually an indication of delayed or incomplete healing. In fact, histological assessment show a significant difference in overall wound maturation (Fig. 1D). These CXCR3-/- wounds failed to reepithelialize in a timely manner, and showed a defective extracellular matrix. This deficit was larger than that noted in wounds in 6—8-week-old CXCR3-/- mice.
A major aspect of wound healing is fibroplasia, which functions to regenerate and organize the collagen matrix of the dermis. After robust immigration, fibroblasts become synthetic before differentiating into the contractile cell required for the proper alignment and bundling of collagen. Dysfunction in this process is noted in the young CXCR3-/- mice. Masson's trichrome staining, to detect collagen content, showed that the amount of collagen was less in the CXCR3-/- mice (Fig. 1E, F). This difference in collagen content is greater in CXCR3-/- aged mice compared to wild-type aged mice (Fig. 1G, H). Interestingly, the wounds in the aged and young CXCR3-/- mice displayed less dense and less organized collagen fibrils tissue at 30 days postwounding (Fig. 1I—L). Picrosirius red staining showed that in the CXCR3-/- aged and young mice, the collagen fibers were short, thin, and the repair remained immature compared to the wild-type mice (Fig. 1I—L).
Ultimately it is (mainly) the collagen and its alignment that are important elements that determine skin integrity. Tensiometry was performed on wounds in both young and aged mice to determine tensile strength, which serves as an end goal of the dermal matrix regeneration. Wounds in both CXCR3-/- young and aged mice required significantly less force to break (Fig. 1M, N). In fact, the CXCR3-/- aged wounds demonstrated an 80% reduction in strength compared to unwounded skin and 30% compared to wounds in wild-type mice (Fig. 1N). Additionally, aged mice presented overall weaker tensile strength in comparison to the young mice.
Taken together these data demonstrate that both young and aged CXCR3-/- mice displayed dysfunctional wound dermal maturation. It further revealed in combination with our previous studies work (23,25) that the significant dermal immaturity is a result of fibroblast failure to synthesize, bundle, and align collagen to regain dermal strength.
Fibroblast Cell Transplantation Partially Corrects the CXCR3 Signaling Milieu
The CXCR3 signaling axis functions late in the regenerative phase and into the remodeling phase. At this time IP-10/CXCL10 is produced in the dermis (25). Thus, we determined the transcript levels of this ligand and its receptor in a 30-day maturing wound. IP-10 levels were found to be elevated in the absence of CXCR3 in both young and old mice, though to a greater degree in the young mice (Fig. 2A, B). This increase in ligand in the absence of receptor-mediated downregulation has been seen previously in young mice (24). We now found that in aged mice IP-10 is protein expression is elevated as well (Fig. 2C). This is to be expected as ligands accumulate in the absence of cognate receptors serving to feedback suppress ligand production. Of interest, when cells were transplanted into wounds, the levels of IP-10 were intermediary, suggesting that CXCR3-expressing wild-type cells could act as a sink in the CXCR3-/-background, but that the presence of cells lacking CXCR3 promoted a build-up of IP-10. The CXCR3 receptor presence was as expected by the nature of host background and cells transplanted (Fig. 2D).
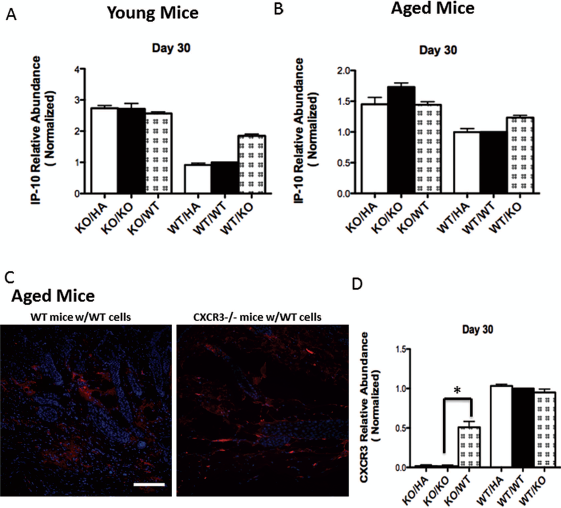
Expression of CXCR3 and it major dermal ligand-induced protein-10 (IP-10)/CXCL10 in the wounds. IP-10 and CXCR3 mRNA levels at day 30 were assayed by quantitative real time (qRT-PCR). Using the comparative critical cycle (Ct) method with glyceraldehyde 3-phosphate dehydrogenase (GAPDH) as the endogenous control, the expression levels of the target genes were normalized and the relative abundance was calculated. (A) Expression of IP-10 in young adult CXCR3-/- mice is elevated when transplanting either wild-type cells (knock out wild-type: KO/WT) or CXCR3-/- cells (KO/KO) compared to wild-type mice with wild-type cells (WT/WT) and wild-type mice with CXCR3-/- cells (WT/KO). Hyaluronic acid carrier alone (HA) severed as the cell delivery control. (B) In aged CXCR3-/- mice IP-10 levels overall were much lower, but were found to be elevated in the absence of CXCR3. (C) In aged mice both CXCR3-/- (KO/HA) and CXCR3-/- transplanted with CXCR3-/- transplanted cells (KO/KO) lack expression of CXCR3. (D) Aged wild-type mice under all conditions (WT/HA, WT/WT, and WT/KO) retained some CXCR3 receptor expression due to the wild-type cell shown by RT-PCR analysis. Data are shown as mean ± SEM of six independent studies performed in triplicate. Shown are representative of nine wounds in each mouse variant. The images measure 300 μm on each side. Original magnifications 400x. The white bar in the photomicrographs measures 50 μm. Statistical analysis was performed by Student's t-test (*p < 0.05).
Fibroblast Cell Transplantation Prevents the Phenotypic Deficits in the Wounds
Full-thickness 6-mm punch biopsies wounds were created in both young and aged wild-type and CXCR3-/- mice. These wounds were then treated with: (a) HA alone, (b) wild-type newborn fibroblast in HA, or (c) CXCR3-/- fibroblast in the HA gel. Approximately 15 million cells in 0.5 ml of HA were injected in the 6-mm punch biopsy wound bed (Fig. 3). At days 7 and 14, wounds were biopsied. These biopsies were assessed for the success rate of cell uptake and maintenance in the wound by the presence of fluorescent cells. Donor cells retained the fluorescent labeling, exhibiting strong survival, and possibly proliferation up to day 30 when tissue biopsies were harvested (Fig. 3). Labeled fibroblasts were found in all vertical layers of the dermis. Fibroblasts show proliferation, as demonstrated by Ki67 staining in the dermis, a marker of proliferation, in the first 14 days (Fig. 3B). However, apoptosis gradually increased as observed by TUNEL staining at day 30 (Fig. 3C). Skin tissue samples, stained with TUNEL and α-smooth muscle actin (αSMA), the latter to identify myofibroblasts, reflected the total number of viable and transplanted cells throughout the study (Fig. 3D—F). The transplanted cell survival, proliferation, and apoptosis were similar for both the wild-type and CXCR3-/-cells. These labeled cells constituted about 3 in 10 stromal cells in the dermis when analyzed using the Meta-Morph imaging software. The rough equivalencies of cell uptake allowed us to determine the contribution to healing without the confounding variable of differential transplantation.

Transplanted cells are present in the skin wounds. (A) The cells were imaged after 7 and 14 days; the presence of fluorescence indicated viable cells. (B) Ki67 staining in the dermis, a marker of proliferation, at day 7 and 14 days under all conditions. (C) Apoptosis was assessed in the transplanted mice by in situ terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) labeling [red; blue is 4′,6-diamidino-2-phenylindole (DAPI) for nuclei]. (D) α-Smooth muscle actin (αSMA) staining to verify fibroblast. (E) Quantitative analysis confirmed that aged CXCR3-/- mice transplanted with wild-type fibroblast cells presented significantly increase in cell death at day 30 similar to wild-type aged mice. (F) Quantitative analysis of αSMA staining confirmed the apoptotic cells were fibroblasts. Data are shown as mean ± SEM of six independent studies performed in triplicate. Shown are representative of nine wounds in each mouse variant. The images measure 300 μm on each side. The white bar in the photomicrographs measures 50 μm. Original magnifications 400x (B) and 200x (D). Representative micrographs (n = 6 for each mouse genotype per time point). Student's t-test (*p < 0.05).
After determining that the transplanted fibroblasts became resident within the wounds, we tested whether these cells could prevent (or correct) the deficits caused by age and a lack of CXCR3 signaling. As shown above, young wild-type mice at day 30 are characterized by normal epidermal and dermal layers with a well-remodeled matrix that coincides with relatively efficient wound healing. Yet, in the CXCR3-/- mice, the wound environment results in deficient repair characterized by the poorly defined matrix, hypercellularity, and weaken skin. Additionally, this pathology was noted to an even greater extent in the wounds of the aged mice (Fig. 1).
In the young mice transplantation of wild-type fibroblasts into the CXCR3-/- mice prevented dysfunctional wound repair. Histological analysis revealed the CXCR3-/- mice injected with wild-type cells do not present the whirling appearance of collagen noted in the nontransplanted CXCR3-/- mice, a quantitative 50% difference in maturation (Fig. 4A, B). Of equal importance, the aged mice showed a significant improvement of 40% in maturation in both the wild-type and CXCR3-/- mice that were transplanted with wild-type cells (Fig. 4C, D). Additionally, in contrast to the nontransplanted CXCR3-/- young and aged wounds the collagen content of the CXCR3-/- wounds with wild-type cells transplanted showed a 30—40% increase by quantification of Masson's trichrome staining (Fig. 4E—H). Interestingly, quantitative data show that in aged CXCR3-/- mice, wild-type transplanted cells were able to restore collagen content by a remarkable 40% (Fig. 4G, H). Of note, transplantation of CXCR3-/- cells into both wild-type and CXCR3-/- mice wounds did not alter healing, neither improving nor harming, when compared to no transplanted cells in both young and aged mice.

Normalization of wound healing in wounds transplanted with wild-type cells. (A) Histological examination and analysis by H&E staining of these 30-day wounds of young (Y) adult CXCR3-/- mice with wild-type cells (KO/WT) resemble the features of mature scarring converging on that of wild-type mice. (B) Quantification of these wounds revealed that wound maturation in CXCR3-/- mice was significantly improved with wild-type fibroblast treatment and was comparable with that of the wild-type (WT) mice wounds. (C, D) Wound maturation of aged (A) CXCR3-/- mice improved only after the addition of wild-type fibroblasts. (E-H) Wounds in both CXCR3-/- young and aged mice that were transplanted with wild-type cells (KO/WT) presented well-organized collagen remodeling when stained with Masson's trichrome and quantified. (I—N) In both young and aged mice picrosirius red staining highlighted that the collagen in wounds in CXCR3-/- mice transplanted with wild-type cells (KO/WT) was more organized and had longer fibrils, denoting a mature scar. (O, P) Hydroxyproline levels in wounds in CXCR3-/- mice were significantly increased in both young and aged CXCR3-/- transplanted with wild-type cells (KO/WT) compared with those in mice that were transplanted with CXCR3-/- (KO/HA) cells or control (KO/HA). (Q, R) mRNA expression analysis of collagen type I confirmed that transplanted wild-type cells contributed to increase in dermal strength in both young and aged mice. Data are shown as mean ± SEM of six independent studies performed in triplicate. Shown are representative of nine wounds in each mouse variant. The images measure 300 μm on each side. The white and black bar in the photomicrographs measure 50 μm. Original magnifications 400x. Student's t-test (*p < 0.05).
At the end of the fibroplastic phase, the fibroblasts are channeled toward a differentiated state as they contract the matrix in a process required for collagen bundling and alignment (1). We have previously shown that during wound healing of CXCR3-/- mice fibroblasts are delayed in this differentiation, resulting in disorganized collagen bundling (19). We now show that transplantation of wild-type cells into the CXCR3-/-wounds can correct the disorganization to result in a denser and more organized granulation tissue (Fig. 4I—M). In young and aged mice picrosirius red staining showed that in the wounds with transplanted wild-type cells collagen fibers were longer, thicker, and the scar was more mature (Fig. 4I—M).
Next, we determined if this difference in collagen content and organization was reflected in strengthened skin. Tensiometry demonstrated increased tensile strength in wounds transplanted with wild-type cells (Fig. 4K, N). We noted an impressive doubling of strength in young CXCR3-/- mice and 20—30% increase in aged CXCR3-/-mice after transplantation with wild-type cells but no change with CXCR3-/- cells (Fig. 4K, N). We confirmed that the improved tensile strength correlated with increased collagen production. Hydroxyproline levels were compared in all groups, and mice transplanted with wild-type cells resulted in at least a 45% increase (Fig. 4O, P). Further determination by qRT-PCR showed increased mRNA expression of type I collagen in all groups that were transplanted with wild-type cells (Fig. 4Q, R). We attribute the overall improvement in collagen production, betterment in alignment, and correction in composition of the matrix to the increased number of synthetic fibroblast contributed by the transplantation of wild-type fibroblasts.
Wild-Type Cells Restore a Mature Matrix
Previously, we have seen the persistence of the matrix proteins fibronectin and tenascin C in wounds in the CXCR3-/- mice that ultimately resulted in a hypertrophic and hypercellular scar (23). As such, we used RT-PCR and immunohistochemistry to analyze the transplanted wounds for gene expression and production of fibronectin and tenascin C (10). Tenascin C is an extracellular matrix glycoprotein whose presence is limited in uninjured tissues yet is expressed at high levels during wound healing and in response to invasive wounds (21,22,24). In normal mature wounds, tenascin is nearly absent. We observed a decrease in the gene expression levels of tenascin C in the wounds transplanted with wild-type cells (Fig. 5A); this decrease was also observed in protein expression by immunohistochemistry (Fig. 5B). Additionally, fibronectin show similar correction of gene expression in wounds in CXCR3-/- mice transplanted with wild-type cells (Fig. 5C). These data suggest that the mechanism for differentiation and synthetic state of the fibroblasts can at least in part regulate production and turnover of matrix components during wound repair.

Transplanted wild-type fibroblasts prevent the development of an immature matrix. (A, B) CXCR3-/- mice transplanted with wild-type cells (KO/WT). RT-PCR analysis of both fibronectin and tenascin C in aged mice revealed that transplanted wild-type fibroblast of CXCR3-/-wounds resembles levels in a mature scar not that of immaturity, as seen in wild-type mice and wild-type mice transplanted with wild-type cells (WT/WT). (C) As the scar develops and matures, tenascin C expression decreases in the wild-type wound. Section showing decrease in staining of tenascin C shows a decreases in tenascin C, comparable to normal wild-type mice, in CXCR3-/-aged mice transplanted with wild-type cells (KO/WT) compared to normal wild-type mice. Data are shown as mean ± SEM of six independent studies performed in triplicate. Shown are representative of nine wounds in each mouse variant. The images measure 300 μm on each side. The black bar in the photomicrographs measures 50 μm. Original magnifications 400x. Student's t-test (*p < 0.05).
Discussion
Molecular therapies provide an option to correct dermal matrix dysfunctions and prevent scar formation. To date clinical approaches range from mechanical and drug-based therapies through engraftment of keratinocytes and stem cells to engineered approaches such as skin substitutes and scaffolds. Unfortunately, these therapies are suboptimal at best, with many questions remaining as to how these types of therapies direct cell behavior such as migration, proliferation, differentiation, maintenance of phenotype, and apoptosis. Exquisite regulation of these cellular processes is critical to proper healing and minimization of scar formation.
One approach to regenerative therapies is to replace or supplement deficient cellular elements through transplantation (12). As dermal matrix dysplasia is central is to scarring and fibrosis, we sought to determine whether normal fibroblasts could overcome endogenous deficiencies and generate a physiological dermal tissue (26). Key to correcting any pathologies is an appreciation of the rich paracrine signaling milieu in which wound healing occurs. We posited that viable cells were best equipped to integrate these signals, as topical applications of factors and matrix have been shown wanting.
Cell survival with some degree of persistence but without eliciting an inflammatory response is likely important for the success of such transplantation. Using elevated T-cell infiltrate as a marker we found importantly, wounds with transplanted fibroblasts showed no viable sign of abnormal inflammatory cell infiltrate (data not shown). Additionally, there was no immune response to the hyaluronic acid use as vehicle for fibroblast delivery. The inoculated cells were demonstrable at up to 30 days, with evidence of expansion and dissemination.
In order to test the success of cell transplantation, a model of scarring was needed. We had earlier found that mice lacking CXCR3-/- exhibited a progressive hyper-tropic and hypercellular scar after wounding (23,25). Herein, we found that this was even more pronounced in aged mice, with postwounding matrix dysfunction resulting in excessive collagen deposition and poor alignment. The transplanted fibroblasts impacted the host wound environment even in the face of endogenous fibroblasts. In fact, it seems that transplanting fibroblasts into the wound can circumvent fibroplasia and allow earlier wound repopulation and matrix production. The decreased cell number and vessel count was curious as CXCR3 signaling predisposes fibroblasts to induced cell death (16,17) and drives endothelial apoptosis and vascular regression (4); thus, redundant signals related to a mature dermis likely subsumes these functions. The notable difference in matrix remodeling, the collagen content, and alignment that lead to an increase in regaining tensile strength suggest that the transplanted fibroblasts exhibit strong survival patterns and are able to maintain their intrinsic properties in the host environment. Furthermore, these transplanted fibroblasts contributed to the production and turnover of matrix proteins tenascin C and fibronectin. Thus, normal fibroblasts appear to be dominant over the endogenous deficient fibroblasts in maturing the dermis to minimize scarring.
Despite these promising initial findings, there are several issues that remain to be address in future studies. These include operational aspects such as optimizing the precise cell dose that results in the greatest response and matrix maturation and the optimal cell delivery scaffold or matrix that will allow for better cell contraction and migration into the wound. On the investigative side, the ability for transplanted cells to penetrate thicker dermal matrices and distribute throughout a complex wound needs to be assessed. Lastly, as most wounds heal, these approaches need to be applied to wounds that are established and only subsequently retarded or stuck in the nonhealing state. This next step of actually “healing” a “stalled” wound, and preventing scarring, is the focus of ongoing experimentation. Additionally, when translated to people, it is most likely that the cellular therapy would be allogenic (rather than the syngeneic approach used herein) in order to circumvent limited cell number or proliferative and functional capacities of the patient. Thus, other ongoing studies will examine the effect of cellular rejection upon these wounds. One would consider that if the matrix composition was key, removal of the cells after laying down a maturation matrix would not undermine the reparative effects, as long as the immune reaction did not in itself introduce new tissue wounds. This must be examined experimentally.
Finally, these findings have important implications for the genesis of fibrosis and chronic wound inflammation. The dominance of the transplanted wild-type cells to correct the dysfunctional healing in CXCR3-/- mice while the CXCR3-/- cells had minimal impact on the wounds in wild-type mice argues strongly that the product of the wild-type cells exerts the greatest effect, rather than the cells themselves. Thus, the matrix becomes not just the result of scarring but takes a prime place in regulating the scarring cascade. The data suggest that a more mature matrix, characterized by aligned and strengthened collagen I fibers with low levels of fibronectin and tenascin-C, promotes a physiological dermal condition of paucicellularity and relative avascularity. Thus, future studies would examine the role of matrix composition on cellular behaviors, bearing in mind that externally applied matrices and components have failed to functionally incorporate within the healing wound. Future work will determine whether transplanted fibroblasts can regulate the qualitative outcome, but this study reveals the feasibility of such an approach and that the fibroblast posses the ability to progress the wound repair towards optimum tissue regeneration rather than scar formation. These finding have intriguing implications for rational cellular interventions aimed at promoting wound healing via cell therapy.
Footnotes
Acknowledgments
We thank members of the Wells laboratory and Dr. Latha Satish and Dr. Patricia Hebda for suggestions and discussions. These studies were supported by grants from the National Institute of General Medical Science of the National Institutes of Health (USA) (GM063569). Services in kind were provided by the Pittsburgh VA Medical Center. The authors declare no conflicts of interest.
