Abstract
Cellular cardiomyoplasty (CMP) is a novel therapeutic approach to myocardial injury (MI). Post-MI remodeling of the left ventricle (LV) comprises dilatation and impairment of systolic function and gives rise to progressive hemodynamic deterioration. We aimed to investigate: a) the impact of CMP on global and regional parameters of LV remodeling (LVR) as well as contractile reserve and b) the suitability and validity of different echocardiographic methods in this scenario. Murine ventricular cardiomyocytes (E13.5–E16.5) were transplanted into cryolesioned hearts of male HIM-OF1 mice. Echocardiography was performed at rest 4 and 14 days postoperatively. For quantification of akinetic myocardial mass and contractile reserve 2 weeks postoperatively additionally low-dose dobutamine stress echocardiography was conducted. Reconstructive 3D-echocardiography (r3D-echo) was compared to “plain” echocardiographic investigations and was compared to invasive measurements with conduction catheter. CMP significantly attenuated LV dilatation and reduced LV function decline on day 14, as obtained with all echocardiographic modalities and confirmed with conduction catheter measurements. In contrast to plain echocardiography and invasive testing, r3D-echo allowed noninvasive quantification of scar size and assessment of regional contractile reserve. Cell transplanted hearts demonstrated a significant decrease of akinetic myocardial mass (-CMP: 13 ± 2%; +CMP 7 ± 1%; p < 0.001) and increased regional contractile reserve, an indirect sign of myocardial viability. The present study demonstrates beneficial effects of CMP on global and regional parameters of LVR and contractile reserve after MI. In contrast to “simple” 2D echocardiography, r3D-echo allowed the assessment of regional contractile reserve and quantification of akinetic myocardial mass as additive functional and morphological measures of LVR.
Keywords
Introduction
A loss of functional cardiomyocytes due to myocardial injury (MI) causes changes of myocardial geometry and function well known as left ventricular remodeling (LVR). LVR displays a dynamic process and comprises dilatation of the LV, decrease of systolic LV function, progression of scar size, and impairment of contractile reserve (4,15). Clinically LVR is characterized by progressive heart failure and consecutively increased incidence of clinical pathologic events and mortality (8). Cellular cardiomyoplasty (CMP) with fetal cardiomyocytes is a novel, causal therapeutic approach, which demonstrated improved survival following MI in mice (10,11). Nevertheless, until now the impact of CMP on functional key parameters of LVR was not visualized in detail was the goal of the present study employing consecutive three-dimensional (3D) echocardiography, validated by left heart catheterization.
“Basic” one- and two-dimensional echocardiography (1D-echo, 2D-echo) allows fast and noninvasive quantification of LV dimensions and global systolic function (1,3,13). So far, the assessment of fractional shortening by M-mode echocardiography (1D-echo) was the parameter of choice to assess improvement of LV function following novel therapeutic approaches. However, the plasticity of changes of regional LV function and scar size is limited with “plain” echocardiography, since particularly injury models result in asymmetrically shaped ventricles. Thus, conventional mathematical models fail to accurately assess global left ventricular function and might under- or overestimate regional changes of cardiac performance. Moreover, the underlying mathematical models rely on symmetrically shaped ventricles and do not allow the quantification of contractile reserve, a noninvasive measure of myocardial viability (4). Hence, simple echocardiographic methods are not only prone to misjudge regional functional effects of LVR, but could also lead to misinterpretation of CMP effects.
We previously demonstrated that reconstructive 3D echocardiography (r3D-echo) reliably allows quantification of a) global and regional myocardial morphology and function, b) borderzone myocardium and scar size, as well as c) global and regional contractile reserve following MI (4,5,7). The aims of this study were a) to study the impact of CMP on global and regional parameters of LVR and contractile reserve and b) to investigate the suitability and validity of r3D-echo following CMP compared with LV catheterization.
Materials and Methods
The experimental protocol was approved by the regional government's Animal Care and Use Committee, and conformed to the US National Institutes of Health Guidelines.
Protocol
Male mice (HIM:OF1-strain, 25–30 g) were anesthetized, intubated, and mechanically ventilated as previously described (9). Transmural myocardial lesions of identical size were generated at the free LV wall and 100,000 embryonic cardiomyocytes (E13.5–E16.5) were injected intramyocardially into the lesion (+CMP, treated group, n = 8) (11). In control mice the same amount of medium without cells was injected (-CMP, control group, n = 8). In addition, sham-operated mice without MI and CMP were investigated (sham, n = 6). Baseline echocardiography was performed 4 days after CMP by 1D-, 2D-, and r3D-echo. Follow-up studies were performed 14 days after CMP at rest and under inotropic stimulation with intravenous dobutamine at low dose (10 μg/kg/min) (5,7). LV catheterization was performed 1 day after stress echocardiography under general anesthesia and mice sacrificed afterwards.
Cell Isolation
Embryonic ventricular cardiomyocytes (E13.5–E16.5) of transgenic mice (HIM:OF1 strain) expressing enhanced green fluorescent protein (eGFP) under a cardiac-specific promoter were harvested as reported (9, 11). For intramyocardial injection the cells were resuspended in DMEM containing 10% fetal calf serum (FCS; 20,000 cells/μl).
Image Acquisition
High-resolution echocardiography was performed using a commercially available ultrasound system (HDI-5000, Philips Medical Systems, The Netherlands), equipped with a 15 MHz linear array transducer in harmonic imaging mode (3,4). Mice were anesthetized with isoflurane (2% for induction and 0.8–1.2% isoflurane in O2 for maintenance) (12). Heart rate was monitored continuously by use of a 6-lead surface electrocardiography (ECG) to minimize cardiodepressant effects (ADInstruments, Castle Hill, Australia). Chest fur was carefully depilated and a layer of centrifuged contact gel served as coupling medium, allowing data acquisition without thoracic compression (4). 1D- and 2D-echo were performed as described previously (3). Briefly, 2D-guided M-mode data were acquired in the parasternal short-axis view at the level of the papillary muscle. For 2D-echo, two positions of the short-axis scan plane were selected and compared: a) at the midpapillary imaging plane (AL-35, 35 mm apical of the aortic valve level), and b) at the basis of the papillary muscles (AL-45, 45 mm apical of the aortic valve level) (Fig. 1A, B). For r3D-echo, the transducer was positioned perpendicular to the long-axis of the left ventricle and the heart was scanned by consecutive short-axis views (0.5 mm intervals) starting at the aortic valve level as depicted in Figure 1A (4,5,7). Cineloops of 2 s duration were stored digitally for each short-axis view and analyzed offline.
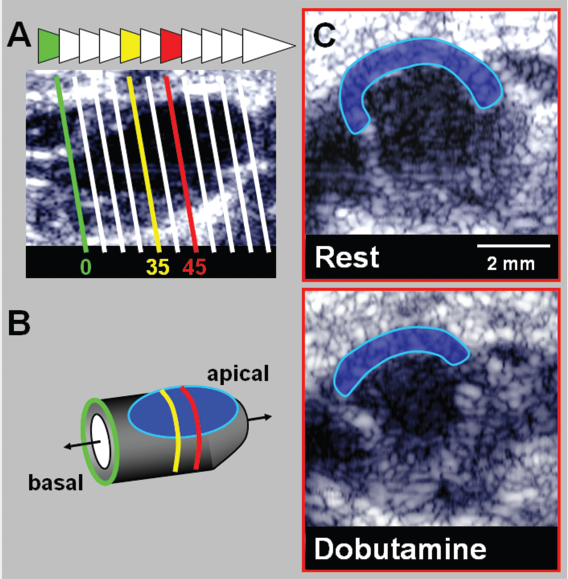
Noninvasive quantification of contractile reserve with reconstructive three-dimensional echocardiography. (A) Movement (arrowheads) of scanhead allows acquisition of parallel scan planes perpendicular to parasternal long axis (below) for calculation of full volume datasets. Selected imaging planes are displayed as white lines. Mathematical approximations were performed via area–length method highlighted in (B) at the corresponding levels [35 mm (35) and 45 mm (45) apical of the aortic valve level]. (C) B-mode imaging positioned 45 mm below aortic valve plane at the base of the papillary muscles during baseline conditions and inotropic stimulation. Representative end-systolic B-mode images demonstrate anteroseptal dysfunctional myocardium (blue), characterized by wall motion abnormality and wall thinning. Note its decrease during inotropic stimulation.
Image Analysis
For the assessment of global LV geometry and function, epi- and endocardial regions of interest (ROIs) were traced in end-systole and end-diastole, allowing calculation of left-ventricular end-diastolic volume (LVEDV) and left-ventricular ejection fraction (EF) applying the area–length model (3,13). LV mass was calculated by multiplication of LV myocardial volume and myocardial density (1.05 g/cm3) during end-diastole. End-diastolic measurements were obtained at the peak of the R-wave. End-systolic measurements were obtained at the time of minimum internal chamber dimensions. ROIs were multiplied with the intersection interval (0.5 mm) and summed up, as validated previously (4). To quantify regional LV function, all imaging planes from basal to apical were visually assessed by a single blinded experienced investigator for 1) wall motion, 2) wall thickness and systolic wall thickening, and 3) contractile reserve. Myocardial compartments were differentiated by visual assessment following established echocardiographic criteria according the recommendations of the American Society of Echocardiography (2). Briefly, for calculation of wall motion scores (WMS), 16 myocardial segments were assigned to normokinetic, hypokinetic, akinetic, or dyskinetic wall motion scored 1 to 4 points, respectively. Further, for quantitative segmental analysis the myocardium was subdivided in remote and borderzone myocardium, as well as collagenous scar tissue: normokinetic wall motion (remote myocardium)—normal myocardial thickness, good systolic wall thickening, good inotropic response to dobutamine; hypokinetic wall motion (peri-infarction borderzone myocardium)—normal myocardial thickness, markedly reduced systolic wall thickening during baseline, reduced inotropic response to dobutamine; akinetic/dyskinetic wall motion (collagenous scar tissue)—thinned, echodense wall, no systolic wall thickening, no inotropic response to dobutamine.
In addition, quantitative assessment of akinetic myocardial mass was performed during end-diastole based on visual judgment. Global contractile reserve was calculated as ratio of ejection fraction during dobutamine and baseline, respectively (see Fig. 1C). Regional contractile reserve was calculated as ratio of akinetic myocardial mass at rest and during pharmacologic stimulation (5).
Hemodynamic Measurements
Mice were anesthetized by isoflurane (1.0 vol% isoflurane in O2 for maintenance), intubated, and ventilated. A 1.4F microconductance pressure catheter (ARIA SPR-719, Millar Instruments Inc., Houston, TX) was positioned in the LV via the right carotid artery for continuous registration of LV pressure–volume loops. Systolic function and myocardial contractility were quantified by LV end-systolic pressure, peak rate of rise in LV pressure (dP/dtmax), ejection fraction, cardiac output, end-systolic volume, and stroke volume. Diastolic performance was measured by LV end-diastolic pressure, peak dP/dtmin, end-diastolic volume, and time constant of iso-volumetric pressure relaxation (10).
Histology
The animals were sacrificed after the hemodynamic investigations. Hearts were scanned under a stereomicroscope equipped with a fluorescent light source (SMZ 1000, Nikon Corp., Tokyo, Japan) for engrafted EGFP positive cardiomyocytes (Fig. 2), perfusion fixed with paraformaldehyde (4%), and incubated in sucrose solution (18%). The samples were frozen at −80°C in Tissue-Tek® (Sakura Fintek, Zoeterwoude, Netherlands) and cut into 8-μm-thick tissue slices. Engraftment and morphology of fetal cardiomyocytes were investigated by fluorescence microscopy as described previously (11).

Epifluorescence microscopy. (A) Pale cryolesion can be depicted on the anterior left ventricular wall 14 days after myocardial injury (8x). (B) Fluorescence microscopy reveals enhanced green fluorescent protein (eGFP)-positive depositions of intramyocardially transplanted embryonic cardiomyocytes (15x). ∗Left atrium. Scale bars: 0.5 mm.
Statistical Analyses
The statistical significance between treatment groups was tested by means of one-way ANOVA and post hoc Tukey test. Differences during time course, as well as between rest and stress, were tested with paired Student's t-test. A value of p < 0.05 was considered significant. Data are indicated as mean ± SD.
Results
Echocardiographic imaging protocol was feasible in all animals. Adequate image quality was obtained at all time points with all modalities. In all, 720 of 768 segments were visualized adequately and were used for qualitative and quantitative analyses. All groups (+CMP, -CMP, and sham) demonstrated comparable heart rates without significant differences at day 4 (-CMP: 458 ± 42/min, +CMP: 450 ± 55/min, sham: 451 ± 40/min; p = 0.9) and day 14 (-CMP: 450 ± 44/min, +CMP: 455 ± 38/min, sham: 460 ± 50/min; p = 0.91). Heart rates increased significantly during pharmacologic stimulation in control (-CMP: 500 ± 55/min) and cell-injected mice (+CMP: 495 ± 45/min) as well as in sham-operated animals (498 ± 55/min), without a significant difference between the groups.
Left Ventricular Remodeling
The initial r3D-echo on day 4 revealed a minor increase of LVEDV in both -CMP and +CMP mice compared to sham-operated animals (Fig. 3). In the further course -CMP mice developed a prominent dilatation (+55%) of the left ventricle (p < 0.001). In contrast, in cell-transplanted hearts further dilatation of the left ventricular cavity was significantly reduced compared to -CMP mice but still significantly higher than in sham-operated mice (Fig. 3).

Left ventricular end-diastolic volume (LVEDV) increases within 4 days after myocardial infarction compared to animals without (sham). LVEDV of mice receiving cardiomyoplasty (+CMP) and control animals (-CMP) is not significantly different between both groups at baseline. In contrast, 14 days after therapeutic intervention (white columns) progress of LV dilatation is significantly attenuated in mice receiving CMP. ∗p < 0.05, ∗∗p < 0.01, ∗∗∗p < 0.001.
r3D-echo demonstrated significantly impaired systolic LV function following myocardial injury at both time points compared to sham-operated mice (Fig. 4). However, r3D-echo ejection fraction (EF) was slightly lower in treated mice (+CMP) than in controls (-CMP) 4 days after MI. In contrast, at day 14 cell transplanted animals demonstrated a significantly higher EF at rest and under pharmacological stress (+CMP: 38 ± 8% and 44 ± 4% vs. -CMP: 26 ± 8% and 35 ± 8%; p < 0.05). Compared to sham-operated animals, LV ejection fraction remained significantly reduced in both groups under all conditions. No significant differences were obtained in total LV mass 4 days (-CMP: 112 ± 24 mg, +CMP: 124 ± 19 mg; p = 0.62) and 14 days (-CMP: 121 ± 23 mg, +CMP: 129 ± 13 mg; p = 0.81) after CMP.

Compared to mice without myocardial infarction (sham) left ventricular ejection fraction (EF) of treated (+CMP) and control (-CMP) animals is severely reduced, without significant difference at baseline. Reduced left ventricular systolic function as sequel of ischemic cardiomyopathy can be demonstrated again 14 days after therapeutic intervention. However, significantly higher EF of CMP mice is obtained (p < 0.05). In spite of inotropic stimulation EF remains significantly lower in control mice compared to treated mice (p < 0.05). ∗p < 0.05, ∗∗∗p < 0.001.
Regional systolic function was assessed qualitatively by use of the wall motion score (WMS) system and quantitatively by r3D-echo. Four days postoperatively, WMS was not different between the groups (+CMP: 2.3 ± 0.3 vs. -CMP: 2.3 ± 0.4; p = 0.94). In the further course +CMP mice demonstrated a significant improvement of regional LV function 2 weeks after MI at rest (WMS 2.0 ± 0.3 vs. 2.4 ± 0.6; p < 0.05) and under dobutamine stress (WMS 1.8 ± 0.4 vs. 2.4 ± 0.6; p < 0.05) indicated by the lower scores. These data were corroborated by the quantification of the akinetic mass using r3D-echo. Especially under inotropic stimulation the amount of akinetic myocardium was significantly smaller in the +CMP group as shown in detail in Figure 5.
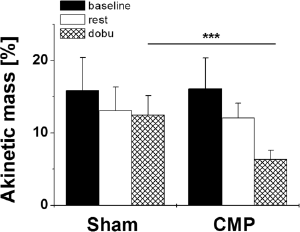
Regional contractile reserve assessed with r3D-echocardiography. Dysfunctional myocardial mass assessed with reconstructive three-dimensional echocardiography 4 days after myocardial infarction is almost equal in animals receiving cardiomyoplasty (+CMP) and control injections (-CMP). Also 14 days after myocardial infarction akinetic mass was comparable during resting conditions. However, inotropic stimulation with low-dose dobutamine triggered recruitment of dysfunctional but viable myocardium and resulted in significantly lower akinetic myocardium in CMP, but not in control animals. ∗∗∗p < 0.001.
All treatment groups (+CMP, -CMP, and sham) revealed significant increase of EF during inotropic stimulation with dobutamine (see Fig. 4), indicating preserved global inotropic response to dobutamine. To investigate regional inotropic recruitment, as a measure for viability, the akinetic myocardial mass was quantified at rest and during pharmacologic stimulation. Interestingly, only +CMP mice demonstrated a significant decrease of dysfunctional LV mass (13% to 7%, p < 0.001) during dobutamine infusion (see Fig. 5). Especially within the borderzone, functional recruitment of viable myocardium could be visualized under inotropic stimulation (see Fig. 1C), as a morphologic correlate of viability. This phenomenon was not obtained in -CMP mice. In conclusion, CMP not only reduced LV dilatation and improved global and regional systolic LV function, but also decreased scar size and improved regional inotropic response following MI.
Comparison of Echocardiographic Modalities
Four and 14 days after MI and CMP M-mode imaging (1D-echo) revealed slight, but not significant, progress of LV dilatation. Further, systolic function demonstrated lower fractional shortening in control mice compared to treated mice (Table 1). However, M-mode imaging was not able to measure functional impact of CMP on key mechanisms of LVR, such as scar size and regional inotropic recruitment. 2D-echo demonstrated increase of LV dimensions using both investigated positions (AL-35, AL-45; see Table 1). In parallel with r3D-echo, 2D-echo was able to discriminate amelioration of progressive LV dilatation 2 weeks, but not 4 days, after MI. However, 2D-echo overestimated LVEDV slightly, but not significantly, at days 4 and 14 compared to r3D-echo (data not shown).
Global Left Ventricular Systolic Function in M- and B-Mode
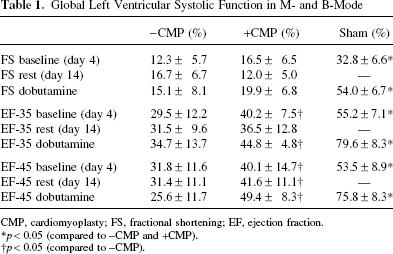
CMP, cardiomyoplasty; FS, fractional shortening; EF, ejection fraction.
p < 0.05 (compared to -CMP and +CMP).
p < 0.05 (compared to -CMP).
Quantification of global systolic LV function with 2D-echo demonstrated significant improvement of EF following CMP utilizing the AL-45 protocol (see Table 1). Hence, 2D-echo allows evaluation of functional CMP impact on global LV function following MI, when applied at the basis of the papillary muscles. However, mathematical approximations of ‘plain” echocardiography do not allow comprehensive investigation of LVR mechanisms, such as quantification of akinetic myocardial mass and regional contractile reserve. Sensitivity and specificity for the identification of a reduced LV function compared with conduction catheter measurements as “golden standard” yield a sensitivity and specificity of 100%. Correlation of global systolic function as assessed with r3D-echo and conduction catheter was close (y = 0.77x + 0.2; r = 0.91, p < 0.0001). Further, conduction catheter measurements confirmed the functional echocardiographic data and demonstrated significant improvement of systolic and diastolic function related to CMP 14 days after MI (Table 2).
Hemodynamic Parameters

CMP, cardiomyoplasty.
Histology
Van Gieson staining evidenced that cryoinjury resulted in reproducible transmural lesions. EGFP-positive cardiomyocytes were observed within cryolesioned areas of all animals displaying successful engraftment of the transplanted cells. Further, cross-striation as prove of differentiation of the transplanted cardiomyocytes was observed in all animals.
Discussion
This follow-up study was performed to determine the time course of global and regional functional effects following intramyocardial transplantation of embryonic cardiomyocytes. For this, different echocardiographic methods were applied and results compared to LV catheter measurements. Our data demonstrate that CMP attenuates LV dilatation within the first 2 weeks after MI. Furthermore, transplantation of embryonic cardiomyocytes augmented systolic function as well as contractile reserve. Additionally, this investigation demonstrated that r3D-echo, but not 1D- or 2D-echo, allows comprehensive noninvasive characterization of LVR following MI and should therefore be the preferred echocardiographic modality in this scenario.
Functional Impact of CMP on Left Ventricular Remodeling
The irreversible loss of contractile tissue after MI induces LVR, which comprises LV dilatation, impairment of systolic LV function, progress of scar size, and decrease of contractile reserve. In the present study, LV dilatation reached level of significance 14 days after MI in untreated controls. Similarly, Yang and colleagues described markedly increased LV dimensions at 2 weeks following infarction and unchanged LV dimensions thereafter (15). This component of LVR was attenuated by intramyocardial transplantation of syngeneic embryonic cardiomyocytes. In line with invasive investigations, LV function was not significantly different at day 4 (11). While control mice demonstrated deterioration of systolic LV function in the further course, +CMP mice showed a significant increase of LV ejection fraction proved by echocardiography as well as invasive catheterization. The relative confined lesion (<20%) could be an explanation for the lack of further LVM increase within 2 weeks (6). r3D-echo during dobutamine infusion allowed valid quantification of nonviable myocardium (4,5). Inotropic stimulation with systemic dobutamine increased global systolic LV function in all mice (controls, treated, and shams). Therefore, this parameter cannot serve as an indicator for regional myocardial viability as already shown also in a clinical setting after transplantation of autologous bone marrow cells (14). r3D-echo, but not 1D- and 2D-echo, allowed detailed classification of dysfunctional and functional myocardium within the lesion and quantification of the functional impact of CMP under rest and inotropic stimulation. Employing r3D-echo, we could visualize that the transplantation of embryonic cardiomyocytes caused, especially under inotropic stimulation, a significant decrease of dysfunctional tissue within the infarcted region of the heart.
Comparison of Echocardiographic Modalities
To elucidate the effects of cell replacement therapies, echocardiography seems to be a suitable diagnostic tool, because repetitive scans can be performed that are not stressful or harmful for the animal. To investigate beyond the global LV performance, also regional effects of the transplanted cells within the lesion or the borderzone scan, modes with high spatiotemporal resolution and discriminatory power are mandatory. An important limitation is the accuracy in the wall thickness measurement by M-mode echocardiography, which could be explained with wall thinning after cryoinfarction and consecutively impaired endocardial border delineation. ‘Simple” 1D-echo failed to demonstrate the functional impact of CMP statistically in this sample size, in contrast to r3D-echocardiography and invasive left ventricular catheterization, because this method calculates three-dimensional parameters with mathematical approximations assuming symmetrical LV dimensions (3,13). Our data confirmed that r3D-echo, but not 1D- or 2D-echo, allows comprehensive noninvasive characterization of LVR following MI and should therefore be the preferred echocardiographic modality in this scenario.
Limitations
Myocardial cryoinjury does not resemble pathophysiology of acute myocardial infarction, but is an established method to investigate morphological and functional response to myocardial injury. We chose this established experimental protocol due to its (a) reproducibility of infarct size and localization, (b) minimal circulatory side effects because of the rapid application, and (c) low peri-operative mortality and high success rates of intramural injection performed in the beating heart.
Conclusions
CMP reduces LV dilatation, impairment of global and regional systolic function, and scar size, as well as contractile reserve following MI. In contrast to “simple” echocardiography, only r3D-echo allows the noninvasive assessment of regional contractile reserve and quantification of akinetic myocardial mass after CMP and MI.
Footnotes
Acknowledgments
A.G. was supported by BONFOR (O-109.0027) and the German Heart Foundation (F-20/08). K.T. was supported by a research grant of the German Research Foundation (DFG-SFB 656, C3) and the IZKF Core-Unit Echo. We thank Pascal Paschenda and Rafael Struck for their excellent technical assistance. The authors declare no conflicts of interest.
