Abstract
Wound healing can be improved by transplanting mesenchymal stem cells (MSCs). In this study, we have demonstrated the benefits of the conditioned medium derived from human MSCs (CM-MSC) in wound healing using an excisional wound model. CM-MSC accelerated wound closure with increased reepithelialization, cell infiltration, granulation formation, and angiogenesis. Notably, CM-MSC enhanced epithelial and endothelial cell migration, suggesting the contribution of increased cell migration to wound healing enhanced by CM-MSC. Cytokine array, ELISA analysis, and quantitative RT-PCR revealed high levels of IL-6 in CM-MSC. Moreover, IL-6 added to the preconditioned medium enhanced both cell migration and wound healing, and antibodies against IL-6 blocked the increase in cell motility and wound closure by CM-MSC. The IL-6 secretory pathway of MSCs was inhibited by SB203580, an inhibitor of p38 MAPK or siRNA against p38 MAPK, suggesting IL-6 secretion by MSCs is mediated through the activation of p38 MAPK. Inactivation of p38 MAPK also reduced the expression and production of IL-8 and CXCL1 by MSCs, both of which were also demonstrated to enhance cell migration and wound closure. Thus, our data suggest MSCs promote wound healing through releasing a repertoire of paracrine factors via activation of p38 MAPK, and the CM-MSC may be applied to enhance wound healing.
Introduction
The wound-healing process requires a well-orchestrated integration of complex biological and molecular events of cell migration and proliferation, extracellular matrix deposition, angiogenesis, and remodeling (23). Impaired production of cytokines by local inflammatory cells and fibroblasts with reduced angiogenesis is one of the crucial factors contributing to nonhealing wounds (11). Bone marrow-derived stromal or mesenchymal stem cells (MSCs) are capable of self-renewal and have the potential to differentiate into mesenchymal and non-mesenchymal tissues (31). They are an abundant source of stem cells for use in regenerative medicine for diseases such as myocardial infarction (22) and spinal cord injury (5). In normal and diabetic animal models, MSC-treated wounds exhibited significantly accelerated wound closure with increased reepithelialization, cellularity, and angiogenesis (37). The therapeutic effects were partly attributed to the multilineage potential of MSCs (12,16, 25,37). We have previously demonstrated that angiogenic factors secreted by MSCs promoted angiogenesis by activating the PI3K–AKT pathway in endothelial cells (15). Further, the paracrine effects of MSCs have also been demonstrated in rodent models of the ischemia limb (18) and wound healing (3). Therefore, MSCs have been demonstrated to stimulate angiogenesis and wound healing by secreting a multitude of paracrine factors to enhance endogenous repair mechanisms following injury.
The paracrine factors secreted by cells can accumulate in the conditioned medium (CM). The conditioned medium derived from the MSC culture (CM-MSC) has been reported to serve multiple positive functions in tissue regeneration. The CM-MSC has an anti-inflammatory role in corneal wound healing following chemical injury, and this effect may be mediated in part through paracrine pathways involving several soluble factors (28). CM-MSC can inhibit apoptosis, increase survival, and enhance angiogenesis in endothelial cells cultured under hypoxic conditions (15). Furthermore, CM-MSC has also been reported to support proliferation, migration, and extracellular matrix invasion of endothelial cells in vitro (30). Although the therapeutic effects of the CM-MSC on angiogenesis and wound healing have been well documented, few, if any, of these paracrine factors in the CM-MSC have been explored. In addition, the underlying signaling pathways involved in the secretion of a repertoire of factors, which are important in stimulating wound healing still remain unknown.
Recently, some studies showed interleukin-6 (IL-6) could promote migration of Langerhans cells, bronchial epithelial cells, and peripheral lymphocytes (6,14,33), and it also protected endothelial cells from hypoxia-induced apoptosis and cell death (15). IL-6 knockout mice showed deterioration of wound healing with impaired granulation tissue formation and a decrease in the fibroblast numbers; these results suggest IL-6 influences the function of fibroblasts in wound healing (20). IL-6 could also stimulate the angiogenesis of circulating blood-derived endothelial progenitor cells in vitro (13).
This study demonstrates the enhancement of wound healing and cell migration by CM-MSC. We have also identified IL-6 as the factor in the CM-MSC that played an important role in stimulating wound healing and cell migration. Moreover, we have further elucidated the p38 MAPK signaling pathway involved in the secretion of IL-6 and other factors, among which IL-8 and CXCL1 were also demonstrated to stimulate wound healing and cell migration.
Materials and Methods
Cell Culture Conditions
Primary MSCs from three normal human volunteers were obtained from the Tulane Center for Distribution of Adult Stem Cells prepared as described previously (35). Briefly, bone marrow aspirates were taken from the iliac crest of normal adult donors after informed consent and under a protocol approved by an Institutional Review Board. Nucleated cells were isolated by a density gradient (Ficoll-Paque; Pharmacia; Peapack) and resuspended in complete culture medium [CCM: α-MEM (α-minimal essential medium; Gibco-BRL, Gaithersburg, MD) supplemented with 16.6% fetal bovine serum (FBS), 100 U/ml penicillin, 100 μg/ml streptomycin, and 2 mM L-glutamine]. After 24 h, nonadherent cells were discarded, and adherent cells were thoroughly washed twice with phosphate-buffered saline (PBS) and were incubated for another 11 days with medium change twice per week. The cells were then harvested with trypsin/EDTA, suspended at 1 × 106 cells/ml in 5% dimethyl sulfoxide and 30% FBS, and frozen in 1-ml aliquots in liquid nitrogen (passage 1 cells). The cells were delivered as passage 1 cells. For expansion, the cells were seeded at 100 cells/cm2 and grown in CCM. They were recovered and used for studies after they reached approximately 80% confluency in 8–9 days. Human aortic endothelial cells (HAECs) harvested from the aorta of two normal males were obtained from a commercial source (catalog number 304–05a; HAOEC; Cell Applications Inc, Bath, UK). They were positive for factor VIII and Dil-Ac-LDL uptake. Frozen vials of the cells were plated at 5,000 cells/cm2 and cultured in endothelial growth medium (catalog number 211–500; Cell Applications, Inc.) containing 2% FBS. When the cells reached 80% confluency, they were trypsinized, diluted 1:3, and expanded to passages 3–6 under the same conditions. The human alveolar epithelial cell line A549 was obtained from American type culture collection (ATCC; Rockville, MD) and cultured in DMEM supplemented with 10% FBS. Primary human gingival epithelial cells and mouse skin epithelial cells were isolated by the method previously described (9) and cultured in serum-free EpiLife (Cascade Biologics™) and 10% FBS-CnT-07 medium (CELLNTEC), respectively. Primary mouse aorta endothelial cells were isolated using a modification of the method described previously (24) and cultured in Endothelial Cell Growth Medium (PromoCell) supplemented with 16.6% FBS.
Preparation of Conditioned Medium
MSCs, HAECs, and A549 cells were replaced at 10,000 cells/cm2 and incubated in respective growth medium for 1 day. The attached cells were washed three times with PBS, and the medium was replaced with serum-free basal medium and incubated for 48 h. The conditioned medium was further concentrated 25-fold (for in vitro experiments) and 50-fold (for in vivo experiments) by ultrafiltration using centrifugal filter units with 5 kDa cut-off (Millipore, Billerica, MA) following the manufacturer's instructions.
Scratch Wound and Transwell Migration Assay
Wound assay was performed as previously described by Denker and Barber (7). HAECs and A549 alveolar epithelial cells were grown to confluence in six-well or 24-well plates and wounded with a yellow tip. Wounded monolayers were washed three times with PBS and were incubated with basal medium (0.1% FBS) in the absence (served as a control) or presence of 25× concentrated CM-MSC (to make up 1×, 2×, and 4× concentration) or recombinant human IL-6 (206-IL, R&D Systems, Hornby, Ontario, Canada), with or without neutralizing antibodies against IL-6 (MAB206; R&D Systems). For transwell migration assay, 5 × 103 cells suspended in 100 μl of serum-free α-MEM were seeded in the upper chamber of 24-well transwell plates containing 8 mm pore size filters (Corning, Costar, USA). In the lower chamber, 600 μl of α-MEM or CM-MSC was added. Cells were incubated at 37°C for 16 h. Cells retained in the upper chamber were removed by swab and those that had migrated through the filter were stained with DAPI and counted under a fluorescence microscope at 200× magnification. These experiments were done with CM-MSC prepared from MSCs of three donors with each donor more than twice.
Cytokine Array
Samples of condition medium were centrifuged at 1,500 × g for 10 min to remove cell debris and concentrated 30-fold using Amicon® Ultra Centrifugal Filter Devices (Millipore). Membranes from a human protein cytokine array kit (Proteome Profiler™ Array; R&D) were blocked with blocking buffer and incubated with 1 ml of the concentrated condition medium at room temperature for 1 h. The membranes were then washed with wash buffer and assayed by chemiluminescence.
Pathway Inhibitors and Assay for IL-6 Release
MSCs seeded at 10,000 cells/cm2 were incubated in CCM for 1 day; attached cells were washed three times with PBS, and the medium was replaced with serum-free α-MEM with vehicle (DMSO or water) or one of the inhibitors against PI3 kinase (LY294002; 50 μM), protein kinase C (Ro31-8220; 1 μM), phospholipase C (U73122; 5 μM), protein kinase A (PKI; 20 μM), JAK (JAK inhibitor; 1 μM), MEK (U0126; 10 μM or PD98059; 50 μM), JNK (SP600125; 5 μM), p38 MAPK (SB203580; 50 μM) (all were purchased from Cell Signaling Technology, Beverly, MA) for 1 h, and then were washed with PBS and incubated with serum-free α-MEM with those inhibitors. The cells were harvested 48 h later and assayed for IL-6 secretion using enzyme-linked immunosorbent assay (ELISA) kits (R&D) according to the manufacturer's instructions. These experiments were done with MSCs from three donors.
Quantitative RT-PCR
Total RNA was prepared using the TRIzol® Reagent (Invitrogen). For cDNA synthesis, random sequence primers were used for the reverse transcription reactions and synthesis was carried out with SuperScript™ III Reverse Transcriptase (Invitrogen). For real-time PCR, the amplification was carried out in a total volume of 25 μl containing 0.5 μM of each primer, 4 mM MgCl2, 12.5 μl of LightCycler™-FastStart DNA Master SYBR green I (Roche Molecular Systems, Alameda, CA), and 10 μl of 1:20 diluted cDNA. PCR reactions were prepared in duplicate and heated to 95°C for 10 min followed by 40 cycles of denaturation at 95°C for 15 s, annealing at 60°C for 1 min, and extension at 72°C for 20 s. Standard curves (cycle threshold values versus template concentration) were prepared for each target gene and for the endogenous reference (GAPDH) in each sample. The quantification of the unknown samples was performed using the LightCycler Relative Quantification Software version 3.3 (Roche).
Western Blotting
Cell extracts were prepared with M-PER (Pierce, Rockford, IL) plus protease inhibitor cocktail (Halt™; Pierce) and protein concentrations were determined using the BCA assay (Pierce). Aliquots of protein lysates were separated on 10% SDS-polyacrylamide gels and transferred onto PVDF membrane, which was blocked with 5% blotting grade milk (Bio-Rad, Hercules, CA) in TBST (20 mM Tris-HCl, pH 7.6“ 137 mM NaCl, 1% Tween 20). Membranes were then probed with the indicated primary antibodies, reacted with corresponding secondary antibodies, and detected using a chemiluminescence assay (Millipore). Membranes were exposed to X-ray film to visualize the bands (Amersham Pharmacia Biotech, Piscataway, NJ). The primary antibodies against p-p38 MAPK (1:1,000; Thr180/Thr182), p38 MAPK (1: 2,000), β-tubulin (1:5,000) were purchased from Cell Signaling Technology.
Lentiviral-Mediated RNAi
The expression plasmids for lentiviral vectors and the bacterial clone for p38 MAPK shRNA (TRCN0000010051) were provided by the National RNAi Core Facility in Taiwan. Lentiviral production was performed by transfecting expression and packaging plasmids into 293T cells using Lipofectamine 2000 (LF2000; Invitrogen, Carlsbad, CA). Supernatants were collected 48 h after transfection, filtered, and the viral titers were determined by FACS. Subconfluent cells were infected with lentivirus in the presence of 8 μg/ml polybrene (Sigma-Aldrich, St. Louis, MO). At 24 h postinfection, culture medium was replaced with fresh growth medium containing puromycin (1 μg/ml) and incubated for 48 h for selecting infected cells. The infected cells were assayed with Western blotting for p38 MAPK expression and ELISA for IL-6 secretion.
Wound Healing Model
All procedures involving animals were in accordance with the institutional animal welfare guideline of Taipei Veterans General Hospital. BALB/c mice (8 weeks old; female) were obtained from BioLASCO Taiwan Co., Ltd. The excisional wound splinting model was generated as described previously (37). In brief, after hair removal from the dorsal surface and anesthesia, two 6-mm full-thickness excisional skin wounds were created on each side of the midline. The animals were randomly divided into four groups and each group had eight wounds, which received one of the four injections including MEM (α-MEM), CM (50× CM-MSC), MEM + IL-6 (α-MEM and IL-6; 10 ng/ml), and CM + anti-IL-6 (50× CM-MSC and anti-IL-6 antibodies; 3 μg/ml) with 80 μl for subcutaneous injection around the wound and 20 μl for topical application on the wound bed. A donut-shaped-like splint was placed so that the wound was centered within the splint. An immediate-bonding adhesive was used to fix the splint to the skin followed by interrupted sutures to stabilize its position and Tegaderm (3M) was placed over the wound. The animals were housed individually. The adhesive on the skin in mice were tested prior to the experiment and no skin irritation or allergic reaction was observed.
Wound Closure Analysis
Digital photographs of wounds were taken at days 0, 4, 7, 11, and 14. Complete wound closure was defined when the wound bed was completely filled with new tissue. Wound margin was traced in the photographs and wound area was calculated using an image analysis program. The splinted hole was used to represent the original wound area since the inside edge of the splint exactly matched the edge of the original wound. The percentage of wound closure was calculated as: (original wound area – wound area)/original wound area × 100.
Histologic Examination
Tissue specimens were fixed in 4% paraformaldehyde for 24 h and embedded in OCT. Sections (10 μm thick) were stained with H&E for light microscopy. Histological scoring was performed in a blinded fashion. Each slide was given a histological score (healing index) ranging from 3 to 10 according to the following parameters (Table 1), modified from previous reports (37): reepithelialization, cell infiltration, and granulation tissue formation. Angiogenesis was demonstrated by capillary density, the density of capillary per mm2, as assessed morphometrically by examining three fields per section of the wound between the edges after immunohistochemistry for endothelial cells with an anti-CD31 antibody (2). The criteria used for histological scores of wound healing are summarized in Table 1.
Criteria for Histological Scores

Statistical Analysis
All values are expressed as mean ± SD. Comparisons between two groups were analyzed by Student t-test, and comparisons between more than two groups were analyzed by ANOVA followed by Bonferroni post hoc test. A value of p < 0.05 was considered statistically significant.
Results
CM-MSC Promotes Wound Healing and Angiogenesis
To investigate whether MSCs promote wound healing through the paracrine effect, we prepared CM-MSC from 2-day culture of MSCs and examined its effects on wound healing in a mouse model (Fig. 1A). A single dose of CM-MSC, administered through healthy skin around the periphery of the wound (80 μl) and topically applied in the central wound bed (20 μl) of 8-week-old BALB/c mice, induced 91.5% and 99.8% of complete wound closure at 11 and 14 days, respectively, while control preconditioned medium caused only 61.2% and 80.2% of complete closure at 11 and 14 days, respectively (Fig. 1B). The histology at 7 and 14 days further showed increased in reepitheliation, cell infiltration, and granulation formation in wounds treated with CM-MSC. The healing index (score parameters are shown in Table 1) of wounds treated with CM-MSC was significantly greater than those treated with preconditioned medium (Fig. 1C). Immunohistochemistry also demonstrated an increase of CD31+ capillary density in wounds treated with CM-MSC compared to wounds treated with preconditioned medium (Fig. 1D). These data suggest CM-MSC promotes wound healing and angiogenesis.

Conditioned medium derived from human mesenchymal stem cells (CM-MSC) enhances wound closure in vivo. Two excision wounds were made in the dorsal skin of each mouse, which received 100 μl of 50× concentrated CM-MSC (CM) with or without 3 μg/ml of anti-interleukin-6 (CM + αIL-6), or preconditioned medium (α-minimal essential medium; MEM) with or without 10 ng/ml of interleukin-6 (MEM + IL-6). (A) Representative photographs of wound receiving CM treatment were taken 0, 7, and 14 days after surgery. Scale bar: 6 mm. (B) The percentage of wound closure was measured up to 14 days after treatment. (C) Healing index of wound was measured 7 and 14 days after treatment. (D) Immunohistochemistry for CD31 and capillary density in sections of wounds were assessed 14 days after treatment. CM versus MEM, p < 0.05; MEM versus MEM + IL-6, #p < 0.05; or CM versus CM + αIL-6, ^p < 0.05. Scale bars: 50 μm.
CM-MSC Enhances Migration of Alveolar Epithelial Cells and Aortic Endothelial Cells
The wound-healing process entails the migration of epithelial cells and the recruitment of endothelial lineage cells to lesion sites. Because epithelial migration has been well studied using human alveolar epithelial cells (A549) (38) and migration of aortic endothelial cells toward mechanical wound has also been clearly demonstrated (21), we therefore investigated the effects of CM-MSC on the migration of A549 cells and HAECs using an in vitro scratch wound assay. To exclude the effect of cell proliferation on scratch wound healing, we prepared the conditioned medium under serum-free conditions and confirmed no significant cell growth was observed during the whole experimental period. After 24 h, A549 epithelial cells treated with control preconditioned medium or 293-fibroblast conditioned medium (data not shown) failed to migrate significantly toward the gap (Fig. 2A). Interestingly, CM-MSC significantly increased the migration rate of A549 epithelial cells in a dose-dependent manner compared to the control medium, and the effects were noted in CM-MSC from all three donors (Fig. 2A, B). Similarly, CM-MSC from three individuals also significantly increased the migration rate of HAEC in a dose-dependent manner (Fig. 2C). These data suggest CM-MSC stimulated epithelial and endothelial cell migration, which in turn promoted wound healing.

Conditioned medium derived from human mesenchymal stem cells (CM-MSC) enhances migration of human alveolar epithelial cells and human aortic endothelial cells. (A) A549 epithelial cells were seeded at 10,000 cells/cm2 and cultured for 48 h. A scratch was introduced and the cells were then cultured in indicated medium (0.1% FBS), and digital pictures were taken at indicated time after scratching. Scale bar: 500 μm. (B, C) The migration of A549 cells (B) and human aortic endothelial cells (HAECs) (C) was quantitatively assessed 24 h after the introduction of a scratch in monolayer cells using an image analysis program. The results showed that cells treated with 1× to 4× concentrated CM-MSC from three donors (D1, D2, D3) demonstrated higher motility compared with the cells treated with control preconditioned medium (CTR). Experiments were performed twice (e.g., D1–1 and D1–2, etc.). ∗p < 0.05 versus CTR.
IL-6 Is Involved in Cell Migration Enhanced by CM-MSC
To explore the factors involved in the enhancement of cell migration by CM-MSC, the cytokine profile of CM-MSC was analyzed using a cytokine array. CM-MSC has abundant levels of cytokines such as IL-6 and IL-8 (Fig. 3A) and IL-6 is well known as an important component of the wound healing process (20). The ELISA result revealed the CM-MSC had 38.6-fold and 55.7-fold increase in IL-6 level compared to CM-A549 and CM-HAEC, respectively (Fig. 3B). Quantitative RT-PCR further demonstrated increase in the IL-6 mRNA levels of MSCs (Fig. 3C). IL-6 stimulates brain or aortic endothelial cell migration (32) and is also involved in CM-MSC-induced angiogenesis by endothelial cells (15). We therefore examined if the stimulatory effect of IL-6 on endothelial cell migration is also observed in A549 epithelial cells. As expected, IL-6 stimulated the migration of A549 in a dose-dependent manner and the increase attained a significant difference at 10 ng/ml, corresponding to the IL-6 level in 4× CM-MSC (Fig. 4A). Because A549 is a lung adenocarcinoma cell line, we further examined whether the migration of primary normal noncancerous cells can also be stimulated by IL-6. Similarly, IL-6 stimulated the migration of primary human gingival epithelial cells (Fig. 4B). Furthermore, IL-6 also stimulated transwell migration of mouse-derived primary skin epithelial cells (Fig. 4C) and aorta endothelial cells (Fig. 4D). On the contrary, the anti-IL-6 antibodies added into CM-MSC abrogated the stimulatory effect of CM-MSC on the migration of A549 cells (Fig. 4E) and primary human gingival epithelial cells (Fig. 4F) compared to control isotype IgG. Similarly, blocking of IL-6 activity by antibodies inhibited the stimulatory effect of CM-MSC on transwell migration of both the primary mouse skin epithelial cells (Fig. 4G) and aorta endothelial cells (Fig. 4H). Taken together, these data suggest IL-6 plays an important role in CM-MSC-mediated enhancement of epithelial and endothelial cell migration.

Release of interleukin-6 (IL-6) by human mesenchymal stem cells (MSC). (A) Cytokine profile of conditioned medium derived from human mesenchymal stem cells (CM-MSC) was analyzed by human protein cytokine array. 25× CM-MSC was blotted onto membranes, which were arrayed in duplicate with 36 human cytokines. Each measurement was performed in duplicate with accepted reproducibility. The results were normalized to the positive controls. (B) Enzyme-linked immunosorbent assays for IL-6 in preconditioned medium (α-minimal essential medium; MEM), conditioned medium from MSCs (CM-MSC), A549 (CM-A549), and human aortic endothelial cells (CM-HAEC). ∗∗p < 0.01 versus CM-MSC. (C) Quantitative RT-PCR of IL-6 mRNA in MSCs, A549, and HAECs.

Involvement of interleukin-6 (IL-6) in cell migration enhancement by conditioned medium derived from human mesenchymal stem cells (CM-MSC). Scratch wound assay for (A) A549 and (B) human gingival epithelial cells in the preconditioned medium with or without (CTR) indicated concentration of IL-6. Digital pictures were taken at indicated time after scratching. The migration rate was quantitatively assessed 24 h later. Transwell migration assay for (C) mouse-derived primary skin epithelial cells and (D) aorta endothelial cells in the preconditioned medium with or without (CTR) indicated concentration of IL-6. The migration rate was quantitatively assessed 16 h later. Scratch wound assay for (E) A549 and (F) human gingival epithelial cells in CM-MSC with or without (CTR) indicated concentration of anti-IL-6. Digital pictures were taken at indicated time after scratching. The migration rate was quantitatively assessed 24 h later. Transwell migration assay for (G) mouse-derived primary skin epithelial cells and (H) aorta endothelial cells in CM-MSC with or without (CTR) indicated concentration of anti-IL-6. The migration rate was quantitatively assessed 16 h later. Values are the ratio of CTR (mean ± SD). ∗p < 0.05 versus CTR. Scale bars: 500 μm.
IL-6 Is Involved in Wound Healing Enhanced by CM-MSC
To examine if IL-6 also plays an important role in the enhancement of wound healing by CM-MSC, wound closure between wounds treated with preconditioned medium with or without IL-6, and wounds treated with CM-MSC with or without pretreatment with anti-IL-6 antibodies was compared. The addition of IL-6 to the preconditioned medium enhanced wound closure compared to wounds treated with preconditioned medium alone (Fig. 1B). IL-6 also caused increase in the Healing index (Fig. 1C) and the CD31+ capillary density (Fig. 1D). In contrast, pretreatment of the CM-MSC with anti-IL-6 antibodies reduced the wound closure rate (Fig. 1B) and decreased the Healing index (Fig. 1C) and the CD31+ capillary density (Fig. 1D) compared to wounds treated with CM-MSC without pretreatment with anti-IL-6. Thus, these data suggest IL-6 plays an important role in the stimulatory effect of CM-MSC on in vivo wound healing and angiogenesis.
Secretion of IL-6 via Constitutive Activation of p38 MAPK
To elucidate the IL-6 secretory pathways in human MSCs, the IL-6 levels of the CM-MSC derived from MSCs treated with or without a specific inhibitor of several signaling pathways were measured. Compared with the vehicle, a significant decrease in IL-6 level was noted only in the CM-MSC derived from cells treated with SB203580 (a specific inhibitor of p38 MAPK), though a slight decrease was observed in the IL-6 level of cells treated with U0126 (a specific inhibitor of MEK) (Fig. 5A). Further, the blocking effect of SB203580 on IL-6 secretion was noted in MSCs from three donors. Because CM-MSC was prepared under serum-free conditions, we wanted to know if this pathway was constitutively activated in MSCs in the basal medium. Interestingly, an apparent level of p38 MAPK activation was noted in MSCs cultured in the basal medium; however, the p38 MAPK activation was completely blocked by SB203580 (Fig. 5B). Moreover, the MSCs infected with lentivirus carrying shRNA against p38 MAPK showed a decrease in total p38 MAPK and the phosphorylation ratio of p38 MAPK compared with the cells infected with lentivirus carrying control scrambled siRNA (Fig. 5C). As expected, a decrease in IL-6 level was noted in CM-MSC derived from the p38 MAPK knockdown cells (Fig. 5D). The supernatant from the MSCs with inhibitor or shRNA blocking p38 MAPK signaling showed a significant decrease in enhancing wound closure (Fig. 5E), or in increasing the Healing index (Fig. 5F) and the capillary density (Fig. 5G) when compared to the corresponding control. Thus, the secretion of IL-6 and wound healing stimulated by MSCs are mediated through the constitutive activation of the p38 MAPK pathway.
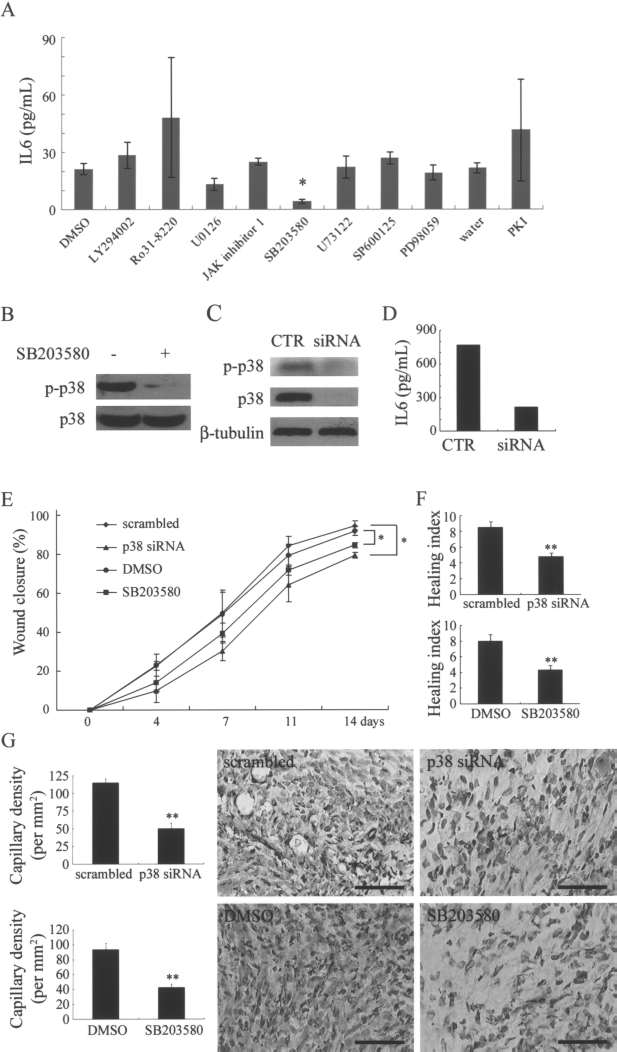
Secretion of IL-6 by MSCs depends on constitutive activation of p38 MAPK in serum-free medium. (A) Cells were seeded at the density of 104/cm2 and treated with the indicated specific inhibitor or vehicle for each pathway in 2 ml serum-free basal medium, and the conditioned medium was harvested 48 h later for the analysis of IL-6 level by ELISA. A significant decrease in IL-6 level was observed in cells treated with 50 μM SB203580. (B) Cells were treated with the SB203580 or vehicle (CTR) in serum-free basal medium. Western blotting of cell lysates was performed for the detection of phospho-p38 MAPK and total p38 MAPK. (C, D) Cells were treated with siRNA against p38 MAPK (siRNA) or control scrambled siRNA (CTR). Western blotting of cell lysates was performed to detect phospho-p38 MAPK and total p38 MAPK. (D) Conditioned medium was harvested for the analysis of IL-6 level by enzyme-linked immunosorbent assays. (E) Each wound received 100 μl of CM-MSC prepared from cells transduced with scrambled or p38 siRNA or cells treated with DMSO (vehicle) or SB203580. The percentage of wound closure was measured from day 0 to day 14. (F) Healing index and (G) capillary density assessed by immunohistochemistry for CD31 were measured 14 days after treatment. Scrambled versus p38 siRNA or DMSO versus SB203580, ∗p < 0.05. Scale bars: 50 μm. LY294002, inhibitor of PI3K; Ro31-8220, inhibitor of protein kinase C; U0126 or PD98059, inhibitor of MEK; JAK-inhibitor, inhibitor of JAK, U73122, inhibitor of phosphlipase C; SP600125, inhibitor of JNK; PKI, inhibitor of protein kinase A.
Constitutive Activation of p38 MAPK Mediates the Secretion of IL-8 and CXCL1
To identify other factors that might contribute to the CM-MSC-induced wound healing, global profiling and comparing of the secretory factors of MSCs and MSCs with inactivation of p38 MAPK were carried out using a cytokine array (Fig. 6A). Inactivation of p38 MAPK not only reduced IL-6 secretion but also reduced the production of IL-8 and CXCL1 by MSCs. ELISA further confirmed the reduction in IL-8 and CXCL1 levels in CM-MSC after p38 MAPK inactivation to 0.62-fold and 0.61-fold of that before inactivation, respectively (Fig. 6B). Quantitative RT-PCR further demonstrated decreased in IL-8 and CXCL1 mRNA levels of MSCs with p38 MAPK inactivation (Fig. 6C).

Release of interleukin-8 (IL-8) and CXCL1 by human mesenchymal stem cells (MSCs). (A) Cytokine profile of conditioned medium derived from human mesenchymal stem cells (CM-MSC) prepared from MSCs treated with 50 μM SB203580 (SB) or vehicle (DMSO) was analyzed by human protein cytokine array. 25× CM-MSC was blotted onto membranes, which were arrayed in duplicate with 36 human cytokines. Each measurement was performed in triplicate with accepted reproducibility. The results were normalized to the positive controls. (B) Enzyme-linked immunosorbent assays for IL-8 and CXCL1 in CM-MSC prepared from MSCs treated with SB203580 (SB) or DMSO. (C) Quantitative RT-PCR for IL-8 and CXCL1 mRNA levels of MSCs treated with SB203580 (SB) or DMSO. ∗p < 0.05; ∗∗p < 0.01 versus DMSO.
IL-8 stimulates brain or aortic endothelial cell migration (32) and is also involved in CM-MSC-induced angiogenesis by endothelial cells (15). However, neither IL-8 nor CXCL1 has been demonstrated to stimulate epithelial cell migration and wound healing. We therefore examined the stimulatory effect of IL-8 and CXCL1 on cell migration in A549 epithelial cells. Interestingly, IL-8 (Fig. 7A) and CXCL1 (Fig. 7B) both stimulated the migration of A549 in a dose-dependent manner, and blocking of IL-8 or CXCL1 activity by antibodies also inhibited the stimulatory effect of CM-MSC on A549 migration (Fig. 7C). To examine if IL-8 and CXCL1 also stimulated wound healing, wound closure between wounds treated with preconditioned medium with or without IL-8 or CXCL1 was compared. The addition of IL-8 or CXCL1 enhanced wound closure (Fig. 7D) increased the healing index (Fig. 7E) and the CD31+ capillary density (Fig. 7F) compared to wounds treated with preconditioned medium alone. These data indicate IL-8 and CXCL1 both stimulate epithelial cell migration and enhance wound closure. Thus, our data suggest MSCs promote wound healing through releasing a repertoire of paracrine factors via constitutive activation of p38 MAPK.

Interleukin-8 (IL-8) and CXCL1 stimulate epithelial cell migration and wound closure. (A) A549 epithelial cells were seeded at 10,000 cells/cm2 and cultured for 48 h. A scratch was introduced and cells were then cultured in medium (0.1% fetal bovine serum; FBS) containing indicated concentration of IL-8 (A) and CXCL1 (B), and digital pictures were taken after scratching. Cell migration was quantitatively assessed 24 h after the introduction of a scratch in monolayer cells using an image analysis program. IL-8 and CXCL1 increased cell motility compared with the cells treated with control preconditioned medium (CTR). (C) A549 epithelial cells were seeded at 10,000 cells/cm2 and cultured for 48 h. A scratch was introduced and cells were cultured in conditioned medium derived from human mesenchymal stem cells (CM-MSC; 0.1% FBS) after pretreatment with 3 μg/ml of anti-IL-8 and anti-CXCL1 for 30 min, and cell migration was quantitatively assessed 24 h after the introduction of a scratch in monolayer cells using an image analysis program. Anti-IL-8 or anti-CXCL1 inhibited cell motility compared with the cells treated with control CM-MSC (CTR). Values are the ratio of CTR (mean ± SD). ∗p < 0.05, ∗∗xlmp0.01 versus CTR. (D) Each wound received 100 μl of preconditioned medium (α-minimal essential medium; MEM) with 10 ng/ml of IL-8 (MEM + IL-8), 1 ng/ml CXCL1 (MEM + CXCL1), or medium only (MEM). The percentage of wound closure was measured from day 0 to day 14. ∗p < 0.05, ∗∗p < 0.01 for IL-8 versus MEM; +p < 0.05, ++p < 0.01 for CXCL1 versus MEM. (E) Healing index and capillary density of wounds at day 14 of treatment. ∗p < 0.05 versus MEM. (F) Immunohistochemistry for CD31 in sections of wounds at day 14 of treatment. Scale bars: 50 μm.
Discussion
Previous studies have been consistent in demonstrating the effects of transplanted MSCs on promoting wound healing; however, discrepancies existed in exploring the underlying mechanisms. MSCs were found to mediate the effects by differentiating into skin (25), vessel (12), or neural cells (16), by secreting paracrine factors (17) or by combined differentiation and paracrine effects (37). The paracrine effects of MSCs on wound healing were further elucidated by previous study (3) and ours, showing CM-MSC when applied to a mouse model of excisional wound repair accelerated wound healing compared to administering a preconditioned medium. Moreover, several factors such as cysteine-rich protein 61 (Cyr61) and pigment epithelium-derived factor (PEDF) in CM-MSC have been identified to promote angiogenesis or attract fibroblast (10,34). In the current study, we first identified IL-6, which is abundant in CM-MSC, as a factor with an important role in CM-MSC-promoted wound healing by enhancing the motility of cells such as epithelial cells. Also, antibody against IL-6 abrogated the effects of CM-MSC on promoting wound healing and enhancing cell migration. Therefore, promotion of wound healing by MSCs identified by previous reports (17,37) was partly attributable to the paracrine effect, where factors such as IL-6 play an important role.
Although the secretion of IL-6 by MSCs (15) and the involvement of IL-6 in the immunoregulatory effects of MSCs (8) have been demonstrated, this is, however, the first time IL-6 has been identified as one of the therapeutic factors involved in the promotion of wound healing by CM-MSC or MSCs. IL-6 has been proposed as a proinflammatory cytokine involved in a lot of inflammatory diseases such as rheumatoid arthritis (1) or as an anti-inflammatory cytokine involved in the inhibition of T-cell proliferation by MSCs in diseases such as graft-versus-host disease (8). IL-6, a pleiotropic cytokine, is involved in many physiological and pathological conditions; however, whether IL-6 plays a beneficial or adverse role may be cell type and context specific.
We have further identified the p38 MAPK pathway as constitutively activated in MSCs and responsible for the secretory pathway of IL-6. The p38 MAPK pathway was activated upon exposure to stresses, such as heat shock and oxidative stress (27). MSCs or stromal cells, residing in various tissues, are inert to various injury-associated stresses, such as ischemia and hypoxia (15), and therefore may contribute to future repair and regeneration after injuries. However, the underlying mechanisms or signaling pathways involved in the inertia of MSCs upon exposure to stresses are unclear. The constitutive activation of the p38 MAPK pathway in MSCs under serum-free conditions may help explain why MSCs can survive these stresses. Further, MSCs under hypoxic conditions secretes more IL-6 than under normoxic conditions (15). IL-6 also inhibits apoptosis in many cells by activating pathways such as PI3K/Akt and Stat3 (4,19). Thus, the secretion of IL-6 by the constitutive activation of the p38 MAPK pathway in MSCs under serum depletion or hypoxic conditions may act as one of the endogenous responses to various stresses, endowing tissues or organs with the ability to resist stresses. The exploration of the roles of MSCs in overcoming the disasters encountered by life-threatening crisis may help develop new methods for treating diseases such as septicemia, acute respiratory distress, and multiple organ failure.
In addition to IL-6, we have further identified IL-8 and CXCL1 in CM-MSC, secreted by MSCs via p38 MAPK activation, are important for enhancing wound healing. Especially, the role of CXCL1 in enhancing wound healing and epithelial migration has not been explored before. We have also identified p38 MAPK as the key signaling pathways in secreting a repertoire of factors, including IL-6, IL-8, and CXCL1, which were involved in CM-MSC-induced wound healing. However, we did not observe an additional effect of the combination of these factors on stimulating epithelial cell migration, suggesting these factors did not have synergic effects in enhancing wound healing. In addition to the cytokines or proteins identified in the current study, several other proteins such as VEGF, MCP-1, and TGF-β have also been identified in CM-MSC at a concentration of therapeutic level (15,36). Future studies are needed to determine whether these factors are also important in enhancing wound healing. MSCs or CM-MSC also ameliorates lung injury-induced fibrosis by secreting the interleukin 1 receptor antagonist (29) and enhances angiogenesis by secreting angiogenic factors (15). Therefore, the paracrine factors of MSCs or CM-MSC have multiple roles in promoting tissue repair or regeneration. Elevation or suppression of certain cytokines or growth factors such as IL-6 or IL-8 is associated with several pathological conditions, including tumors (26). Thus, future application of CM-MSC or purified IL-6 or IL-8 in promoting wound healing requires more information and further investigation. However, the results provided in this study strongly support the concept of using the paracrine effects of MSCs or CM-MSC for enhancing wound healing or other tissue regeneration.
Footnotes
Acknowledgments
Grants supported by Veterans General Hospital-Taipei (R92-001-7, Stem Cell Grant-supported by HealthBanks Biotech); National Science Council (95-2314-B-010-101-MY3; 97-3111-B-010-001-); and National Yang-Ming University, Ministry of Education. This work was assisted in part by the Division of Experimental Surgery of the Department of Surgery, Taipei Veterans General Hospital. shRNA constructs were obtained from the National RNAi Core Facility at the Institute of Molecular Biology/Genomic Research Center, Academia Sinica, Taipei, Taiwan.
