Abstract
The therapeutic potential of mesenchymal stem cells (MSCs) and their conditioned medium (MSC-CM) has been extensively studied. MSCs can repair tissue, reduce local inflammation, and modulate the immune response. Persistent renal tubular interstitial inflammation results in fibrosis and leads to chronic kidney disease (CKD). Unilateral ureteral obstruction (UUO) is a very well-accepted renal fibrosis model. In this study, we evaluated factors influenced by the administration of MSCs or MSC-CM in the UUO model. MSCs extracted from rat bone marrow were cultivated in vitro and characterized by flow cytometry and cellular differentiation. Eight groups of female rats were used in experiments (n = 7, each), including Sham, UUO, UUO + MSC (obstruction + MSC), and UUO + CM (obstruction + MSC-CM) for 7 days of obstruction and Sham, UUO, UUO + MSC, and UUO + CM for 14 days of obstruction. The MSCs or MSC-CM was administered via the abdominal vena cava after total ligation of the left ureter. After 7 or 14 days, rats were euthanized, and serum and obstructed kidney samples were collected. MSCs or MSC-CM decreased the expression of molecules, such as Collai, α-SMA, and TNF-α. We also observed reductions in the levels of caspase 3, α-SMA, and PCNA in treated animals by immunohistochemistry. Our results suggest that the intravenous administration of MSCs or MSC-CM improves fibrosis progression and factors involved in apoptosis, inflammation, cell proliferation, and epithelial–mesenchymal transition in Wistar rats subjected to UUO, indicating a potential tool for preventing CKD.
Keywords
Introduction
Chronic kidney disease (CKD) affects individuals worldwide (8,32) and is now recognized as a major public health problem. Interstitial fibrosis is regarded as one of the main pathways for CKD progression, which culminates in end-stage renal disease. Despite the great deal of effort invested in identifying therapies for CKD, the number of patients requiring dialysis and kidney replacement continues to increase (11). Because most renal disorders (whether glomerular or interstitial, congenital or acquired) can lead to renal fibrosis, there is great interest in identifying the underlying factors and contributors of this process to prevent or reverse these changes. In contrast to adults, in whom diabetes and hypertension are the major etiologies of renal failure, congenital urinary tract obstruction is the most important identifiable cause of renal failure in infants and children (30).
Unilateral ureteral obstruction (UUO) is a well-established model of renal fibrosis. In rabbits, UUO causes increases in interstitial collagens I, III, and IV, fibronectin, and heparin sulfate proteoglycan as well as proliferation of renal interstitial fibroblasts and transformation into myofibroblasts (24,27). Ureteral obstruction is followed within a few days by hydronephrosis, interstitial inflammatory infiltration, and tubular cell death attributable to apoptosis and necrosis (10). Thus, UUO model is suitable for studying the pathological and molecular changes occurring in tubulointerstitial damage since it resembles the deterioration of renal function in human chronic kidney disease (23). Animal models of UUO have been expanded and refined to elucidate the pathogenesis of obstructive nephropathy, as well as the mechanisms responsible for progressive renal fibrosis (18,26).
The administration of mesenchymal stem cells (MSCs) offers protection in several experimental models of AKI (16). Positive effects of MSC treatment on the loss of renal function in early stage CKD models were already demonstrated. In these studies, however, the incorporation and transdifferentiation of injected MSCs were rare events, suggesting that MSCs primarily have a supportive function, which is most likely by secreting growth factors and cytokines (9,15). Such a paracrine mode of action has the therapeutic potential for cell-free treatment strategies using MSC-secreted factors. Importantly, if the administration of MSC-derived secreted factors can reduce CKD progression, this may have great clinical relevance as therapy and could overcome the problems associated with (allogenic) MSC administration, such as immune incompatibility, MSC maldifferentiation (3,19), and tumorigenesis (12,17). Thus far, few studies have been carried out on MSC-secreted factor effects. To the best of our knowledge, the effect of MSC-secreted factors has not yet been investigated in the model of UUO.
We hypothesized that MSCs and MSC-CM could have a therapeutic role on CKD in this model. To support this hypothesis, we administered MSCs or MSC-CM in a UUO model in rats. The ability of the MSCs or MSC-CM to reduce fibrosis, inflammation, apoptosis, and epithelial–mesenchymal transition (EMT) was assessed.
Materials and Methods
Ethics Statement
The study was approved by the Research Ethics Committee of Federal University of São Paulo, Brazil, with animal care performed strictly according to established institutional guidelines. All surgery was performed under xylazine–ketamine anesthesia (Rhobifarma, Hortolândia, Brazil), and all efforts were made to minimize suffering.
MSC Culture
Bone marrow was collected from male Wistar rats (Cedeme, São Paulo, Brazil) by flushing the femurs and cultured with DMEM low glucose (Sigma-Aldrich, St. Louis MO, USA) with 20% fetal bovine serum (FBS; Invitrogen, Paisley, Scotland). Third- and fourth-passage cells, or their medium, were used for transplantation. CD45, CD11b, CD34, CD90, CD73, and CD44 (1:250; BD Pharmingen, San Jose, CA, USA) were evaluated by flow cytometry and used as markers of MSCs (25). Multilineage differentiation of MSCs under adipogenic and osteogenic differentiation conditions was also analyzed (data not shown).
Preparation of MSC-CM
Eighty percent confluent MSC plates were washed with PBS buffer (Gibco, Rockland, MD, USA) and then fed with serum-free DMEM low glucose (Sigma-Aldrich,) for 24 h. Supernatants from each plate were then collected, pooled, centrifuged at 1,200 rpm for 5 min and filtered in a 0.22-μm filter (Merck KGaA, Darmstadt, Germany) to eliminate any cellular debris. For each animal, we used 500 μl of conditioned medium generated by approximately 5 × 106 cells.
UUO and Injection of MSCs or MSC-CM
Adult female Wistar rats weighing 250 to 300 g were anesthetized with xylazine (Anasedan-Vetbrands, Cascavel, Brazil) and ketamine (Dopalen-Vetbrands, Cascavel, Brazil) solution. For UUO creation, a low midline abdominal incision was made. After the ureter was mobilized and isolated with minimal dissection, it was ligated with two 6-0 silk sutures (Bioline, Anápolis, Brazil) at the ureterovesical junction. Sham-treated animals underwent an identical surgical procedure without ureteral ligation. Soon after, animals received 1 × 106 MSCs resuspended in PBS buffer (Gibco) or MSC-CM (500 μl) via the cava vein. At days 7 and 14 postsurgery, the animals were euthanized. The experimental animals were separated into eight groups (n=7, each), including Sham, UUO, UUO+MSC, and UUO+CM for 7 days and Sham, UUO, UUO+MSC, and UUO+CM for 14 days. The left kidneys were extracted and washed in saline solution. One portion of each kidney was fixed in 10% formaldehyde (Labsynth, Diadema, Brazil) for histological examinations and immunohistochemical detection, while another portion was stored at liquid nitrogen for later real-time PCR.
RNA Isolation, Reverse Transcriptase, and Quantitative Real-Time PCR
Total RNA from removed kidneys was extracted using a TRIzol technique (Life Technologies, Carlsbad, CA, USA) according to the manufacturer's instructions. Reverse transcription was performed using a high-capacity cDNA for real-time PCR kit (Life Technologies). The sequences of the primers were as follows: β-actin, 5'-cctctatgccaacacagtgc-3’ (forward) and 5'-acatctgctggaaggtggac-3’ (reverse); tumor necrosis factor-α (TNF-α), 5'-actcccagaaaagcaagcaa-3’ (forward) and 5'-cgagcaggaatgagaagagg-3’ (reverse); collagen 1-α-1 (Col1a1), 5'-tggccaagaagacatccctgaagt-3’ (forward) and 5'-acatcaggtttccacgtctcacca-3’ (reverse); and α-smooth muscle actin (α-SMA), 5'-tattgtgctggactctggagatgg-3’ (forward) and 5'-agtagtcacgaaggaatagccacg-3’ (reverse). Primers were synthetized by Prodimol (Belo Horizonte, Brazil). Real-time analysis was performed on ABI Prism 7900 (Applied Biosystems, Foster City, CA, USA) using the Sybr Green PCR Master Mixes for real-time PCR (Life Technologies). β-actin was used as the endogenous control. The relative expressions were determined by a standard curve.
Histological Analysis
The kidneys were collected and fixed in 10% formaldehyde (Labsynth). The tissues were subsequently embedded in paraffin, sectioned, and stained with Sirius red (Easypath, São Paulo, Brazil). A pathologist and one of the investigators observed the kidneys. The data were analyzed for fibrosis, and the changes were quantified using ImageJ™ software (NIH, Bethesda, MD, USA).
Immunohistochemical Evaluations
Immunohistochemical staining of α-SMA, (1:50; Dako, Carpinteria, CA, USA), PCNA (1:500; Dako), and caspase 3 (1:50; Millipore, Billerica, MA, USA) was performed using LSAB™ Kit (Dako) according to the manufacturer's protocol. In the control experiments, the primary antibodies were omitted and replaced by antibody diluent only.
The slides were counterstained with hematoxylin (Easypath). The changes were quantified using CorelDraw™ suite (Corel, Ottawa, Canada) and UTHSCSA Image Tool™ software (University of Texas, San Antonio, TX, USA).
MSC Tracking
To assess the homing of MSCs to renal tissue, fluorescent dye tracking was performed. Since MSCs were from bone marrow male rats we used anti-Y-chromosome antibody to detect those cells in female rats. Briefly, kidneys were frozen in liquid nitrogen and maintained at −80°C. Tissue sections were treated with 2% bovine serum albumin and incubated with the primary goat polyclonal anti-Y-chromosome antibody (1:200; Santa Cruz Biotechnology, Santa Cruz, CA, USA) overnight at 20°C. After washing with PBS, the sections were incubated with secondary FITC-conjugated rabbit anti-goat IgG antibody (1:2,000; Invitrogen) for 1 h at room temperature, and slides were mounted with fluorescent mounting medium (DakoCytomation, Carpinteria, CA, USA). Immunofluorescence staining was visualized and recorded with an Floview 1000 confocal microscope (Olympus® Bx50; Olympus, Tokyo, Japan).
Statistical Analysis
The results are expressed as the means ± standard deviation (SD). For all studies, one-way analysis of variance (ANOVA) was used followed by the Newman–Keuls test. The statistical significance level was defined as p < 0.05.
Results
TNF-α
To examine whether MSCs and MSC-CM would affect the modulation of inflammation after UUO, the proinflammatory cytokine TNF-α mRNA expression was analyzed by real-time PCR at days 7 and 14. At day 7, TNF-α mRNA expression in the UUO + MSC and UUO + CM were lower when compared with the UUO group (Fig. 1A). At day 14, we did not find any traces of the TNF-α mRNA amplification product.
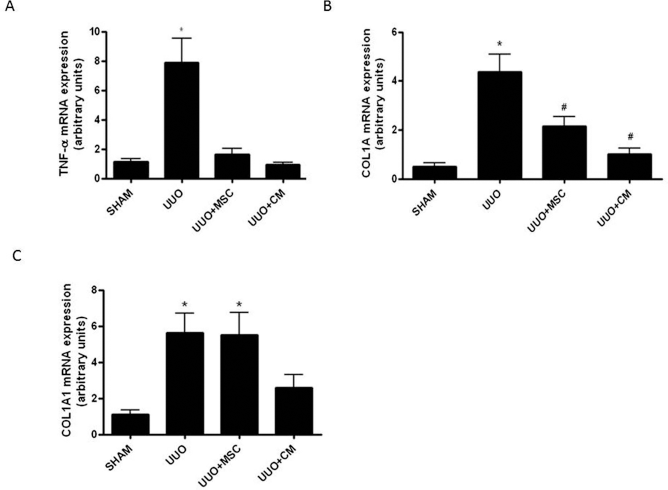
Real-Time PCR for TNF-α and Col1a1 mRNA expression. (A) TNF-α gene expression after 7 days of UUO. *Versus all groups, p < 0.05. (B) Col1a1 gene expression after 7 days of UUO. *Versus all groups, #versus UUO, p < 0.05. (C) Col1a1 gene expression after 14 days of UUO. *Versus SHAM, p < 0.05.
Col1a1
To confirm whether the MSCs and MSC-CM can modulate the expression of collagen after UUO, the expression of Col1a1 mRNA was evaluated by real-time PCR at days 7 and 14. At day 7, we observed a transcription reduction of Col1a1 in the UUO+MSC and UUO+CM groups compared to the UUO group (Fig. 1B). However, at 14 days, the reduction only remained in the UUO+CM group compared to the UUO and UUO+MSC groups (Fig. 1C).
Sirius Red Staining of Renal Tissues
As can be observed, there was an increase in the level of fibrosis during the experiment (Fig. 2A, B). At day 7, the UUO+MSC and UUO+CM groups showed a lower fibrosis area compared to the UUO group (Fig. 2A). Surprisingly, at day 14, only the UUO + CM group continued to show a smaller fibrosis area compared to the UUO and UUO+MSC groups (Fig. 2B).
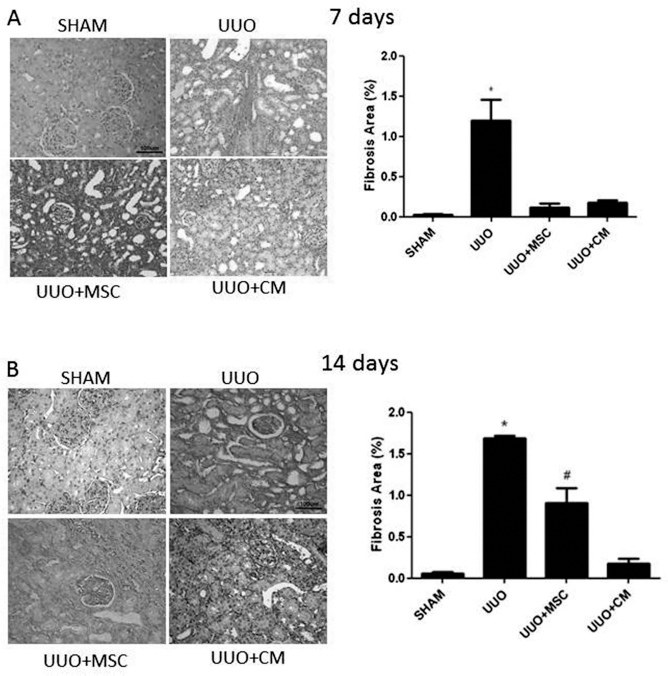
Sirius red staining (collagen I and III). (A) Representative photographs of renal tissue staining with Sirius red after 7 days of UUO and respective quantification. *Versus all groups, p < 0.05. Original magnification: 200x. Scale bar: 100 μm. (B) Representative photographs of renal tissue staining with Sirius red after 14 days of UUO and respective quantification. *Versus all groups, #versus UUO + MSC versus UUO + CM, p < 0.05. Original magnification: 200x. Scale bar: 100 μm.
Gene and Protein Expression of α-SMA in Renal Tissues
At days 7 and 14, increased expression of the α-SMA gene and protein was observed in the UUO groups. Remarkably, at day 7, there was reduction in both the gene and protein expression of α-SMA in the UUO+MSC and UUO + CM groups compared to the UUO group (Fig. 3A, B). At day 14, however, the gene expression of α-SMA in the UUO+MSC group was similar to the UUO group pattern, while the UUO + CM had even lower gene expression (Fig. 3A). When we observed the protein expression at the 14th day, both the UUO+MSC and UUO+CM groups continued to exhibit a reduced level of α-SMA (Fig. 3C).

Real-time PCR and immunohistochemistry for α-SMA. (A) mRNA expression levels of α-SMA after 7 days of UUO. *Versus all groups, p < 0.05. (B) Representative photographs of renal tissue staining with α-SMA antibody after 7 days of UUO and respective quantification. *Versus all groups, p < 0.05. Original magnification: 200x. Scale bar: 100 μm. (C) mRNA expression levels of α-SMA after 14 days of UUO. *Versus UUO, #versus SHAM, p < 0.05. (D) Representative photographs of renal tissue staining with α-SMA antibody after 14 days of UUO and respective quantification. *Versus all groups, #versus SHAM, p < 0.05. Original magnification: 400x. Scale bar: 50 μm.
PCNA Expression
There were similar increases in the cellular proliferation in the UUO group in both periods. At day 7, the UUO + MSC and CM groups showed a reduction in the cellular proliferation compared with the UUO group (Fig. 4A). Surprisingly, at day 14, the level of PCNA increased in the UUO+MSC and UUO+CM groups compared with the UUO group (Fig. 4B).

Immunohistochemistry for PCNA. (A) Representative photographs of renal tissue staining with PCNA antibody after 7 days of UUO and respective quantification. *Versus all groups, p < 0.05. Original magnification: 200x. Scale bar: 100 μm. (B) Representative photographs of renal tissue staining with PCNA antibody after 14 days of UUO and respective quantification. *Versus UUO, #versus UUO + CM, p < 0.05. Original magnification: 400x. Scale bar: 50 μm.
Activated Caspase 3 Expression
At day 7, caspase 3 expression presented no significant differences among groups (Fig. 5A). At day 14, the level of activated caspase 3 increased substantially in the UUO group. However, the UUO+MSC showed reduced levels of caspase 3 compared to the UUO and UUO + CM groups (Fig. 5B).

Immunohistochemistry for activated caspase 3. (A) Representative photographs of renal tissue staining with activated caspase 3 antibody after 7 days of UUO and respective quantification. Nonsignificant, p > 0.05. Original magnification: 200x. Scale bar: 100 μm. (B) Representative photographs of renal tissue staining with activated caspase 3 antibody after 14 days of UUO and respective quantification. *Versus UUO, #versus UUO + MSC, φversus SHAM, p < 0.05. Original magnification: 400x. Scale bar: 50 μm.
MSC Tracking
At 24 and 48 h after MSC injection, male cells (Y-chromosome) were observed in the obstructed female rat renal tissue by immunofluorescence. The Y-chromosome-positive MSCs were found mostly in the tubulointerstitial region (Fig. 6).
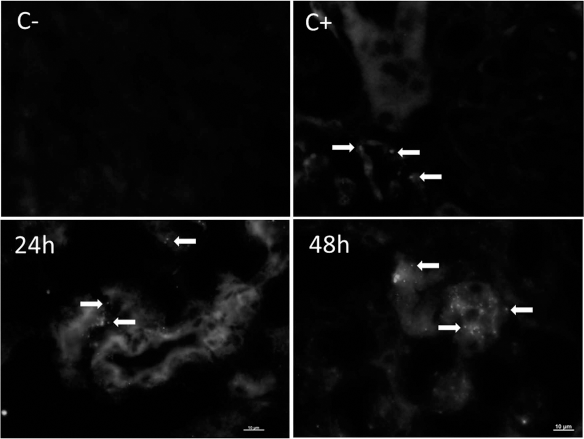
Representative photographs of renal tissue staining with chromosome Y antibody after 24 and 48 h. In negative control (C-) primary antibody was omitted. Male rats were used for positive control. White arrows indicate the presence of male MSCs in female kidney. Scale bar: 10 μm.
Discussion
Previous studies showed beneficial effects of MSC administration in experimental models of AKI (16,15). However, the relevance of these observations in AKI and CKD is unclear, and data on MSC administration in CKD are sparse; additionally, a small number of studies have evaluated the administration of MSCs, and they have suggested prevention in the progression to CKD (5,7,9,21,22,31). Furthermore, few studies have employed MSC-CM as a therapeutic strategy (2,14,29).
In order to address this question, we carried out experiments in which UUO rats were treated with MSCs or CM. We found that both treatments could modulate collagen deposition, inflammation, EMT, cell proliferation, and apoptosis. Similar results were observed in other studies with different models. For example, Koopen et al. demonstrated that the MSC-derived CM rescues kidney function in nephrectomized rats with established CKD (29). In a model of AKI, Bi et al. demonstrated that CM from bone marrow-derived MSCs increased survival and limited renal injury (2).
There is already a study showing that arterially delivered MSCs can reduce renal fibrosis and EMT in UUO (1). However, the present study is the first, to the best of our knowledge, to evaluate the impact of MSC-CM on obstruction-induced EMT and renal fibrosis.
The tendency of the fibrotic process is to increase over time, and established fibrosis cannot be reversed. This could explain different findings concerning inflammation, EMT, and collagen deposition at 7 and 14 days. Clearly, we observed better parameters at the 7th day in comparison with the 14th day. Noteworthy, we used a single injection of MSCs or MSC-CM right after the surgery, which can be considered a preventive approach rather than a reversal treatment and may be a limitation of our study.
Recent reports suggest that microvesicles (4,13) or exosomes (6) play a central role in MSC-mediated tissue repair. In experimental myocardial infarction, the cardioprotective effects of human embryonic MSC-CM were attributed to exosomes (20,28). Other examples show that the administration of exosomes, released by mesenchymal stromal cells, promotes functional recovery and neurovascular plasticity after stroke in rats (33), and attenuated Escherichia coli endotoxin-induced acute lung injury in mice (35).
In the kidney, exosomes released by human umbilical cord MSCs protect against cisplatin-induced renal oxidative stress and apoptosis (34), and bone marrow-derived MSC exosomes repaired gentamicin-induced AKI (25).
This theory could explain why, in our study, the group treated with MSC-CM presented better results compared with the cell-treated group. Thus, we speculate that paracrine factors carried by exosomes and microvesicles generated by a pool of cells in vitro will be higher than the amounts of these generated by the few numbers of cells that can enter the injury site.
In summary, interstitial renal fibrosis is a common final pathway leading to end-stage renal failure, irrespective of the nature of the initial renal injury. Our findings provide in vivo evidence that MSCs, and especially MSC-CM, significantly attenuate tubulointerstitial damage in an experimental model of renal tubulointerstitial fibrosis after UUO. The overall effect of both treatments in this model appears to be both anti-inflammatory and antifibrotic. In view of its protective effect on different mechanisms of pathogenesis in this UUO model, MSCs and MSC-CM might be useful as an early therapeutic agent for progressive renal fibrosis.
Conclusion
Taken together, our results suggest for the first time that the IV administration of bone marrow-derived MSCs and MSC-CM improves fibrosis progression and modulates factors involved in apoptosis, inflammation, cell proliferation, and EMT in Wistar rats subjected to UUO. These results indicate a potential treatment of this pathophysiological progression and help us understand the mechanism of action of the MSCs, but further studies are necessary to explain the pathway by which MSCs or MSC-CM can modulate the responses in the kidney.
Footnotes
Acknowledgments
All authors have made substantial contributions to the concept, design, acquisition, analysis, and data interpretation. All authors have read and approved of the final manuscript. This work was supported by the CNPq (Conselho Nacional de Pesquisa), FAPESP (Fundação de Apoio à Pesquisa do Estado de São Paulo), CAPES, and FOR. The authors declare no conflicts of interest.
