Abstract
Facioscapulohumeral muscular dystrophy (FSHD) is the third most frequent inherited myopathy. We previously demonstrated that mesoangioblasts can be efficiently isolated from FSHD muscles, although their differentiation ability into skeletal muscle was variably impaired. This correlates with overall disease severity and degree of histopathologic abnormalities, since mesoangioblasts from morphologically normal muscles did not show any myogenic differentiation block. The aim of our present study was to verify whether mesoangioblasts from differentially affected FSHD muscles reproduce in vivo the same differentiation ability shown in vitro by studying their capability to form new muscle fibers during muscle regeneration of experimentally damaged muscles. We show that a diverse ability of FSHD mesoangioblasts to engraft and differentiate into skeletal muscle of SCID mice is strictly related to the characteristics of the muscle of origin, closely replicating in vivo what was previously observed in vitro. Moreover, we demonstrate that mesoangioblasts obtained from severely affected muscles scarcely integrate into muscle fibers, remaining mainly localized in the connective tissue. This suggests a defective migration in response to chemoattractants released by damaged fibers, as indicated by cell migration assays in response to HMGB1 and very low levels of RAGE expression, along with a decreased ability to fuse or to appropriately trigger the myogenic program. Our study indicates that FSHD mesoangioblasts from unaffected muscles can be used as selective treatment to halt muscle degeneration in severely affected muscles, and suggests that pharmacological and molecular interventions aimed to ameliorate homing and engraftment of transplanted autologous mesoangioblasts may open the way to cell therapy for FSHD patients, without requiring immunosuppression or genetic correction in vitro.
Keywords
Introduction
Facioscapulohumeral muscular dystrophy (FSHD, OMIM #158900) is the third most frequent form of inherited muscle disease, following Duchenne muscular dystrophy (DMD) and myotonic dystrophy. In FSHD a high variability is observed with regard to age at onset, severity and pattern of muscle involvement, both between and within families, and the rate of disease progression appears unpredictable in a single affected individual (11,16). FSHD is a dominant disease linked to contractions of a long stretch of 3.3 kb KpnI repeat units (D4Z4) on the long arm of chromosome 4 (region 4q35). D4Z4 contains a transcriptional silencer whose deletion leads to inappropriate upregulation of one or more candidate genes whose overexpression specifically affects skeletal muscle (23). Among these, the double homeobox chromosome 4 (DUX4) overlaps the last repeat unit and the adjacent sequence. Recently, the presence of specific single nucleotide polymorphisms (SNPs) in the chromosomal region distal to the last D4Z4 repeat has been demonstrated. This produces a canonical polyadenylation signal for transcripts derived from DUX4 that improves its stability. These observations suggest that a toxic gain of function ascribable to the stabilized distal DUX4 transcript might play a pivotal role in FSHD pathogenesis (8).
Despite the identification of the genetic defect associated with FSHD, the molecular and cellular pathologic effects of this deletion remain largely unknown. This makes, at the moment, impractical to envisage a specific gene therapy to correct the deleterious consequence on skeletal muscle and improve muscle function.
In FSHD patients it is quite common to observe the coexistence of affected and apparently normal muscles. Myogenic autologous stem cells, expanded in vitro from apparently normal muscles and capable to engraft into damaged muscles, enhancing their regenerative activity, could be used for therapeutic applications, repairing, at least in part, the degenerating muscles (9).
Previous studies conducted in animal models have shown that transplanted myoblasts from phenotypically unaffected FSHD muscles participate to localized muscle regeneration (25). However, because of poor migratory capacity of these muscle progenitors that are unable to cross vessel walls, myoblast-based cell therapy protocols require multiple intramuscular injections and may target only individual muscles, posing technical challenges to develop new strategies to improve the graft success (2,3).
In our previous study, we examined primary muscle cells derived from FSHD muscles and showed that all FSHD myoblast cultures displayed several abnormalities including decreased proliferation and clonogenic abilities and reduced life span. By contrast, the differentiation ability of FSHD myoblasts was preserved (15). The studies on FSHD myoblasts are relevant in view of the possible alternative use of myogenic stem cells localized in the perivascular niche of muscle tissue, called mesoangioblasts, which are able to restore muscle morphology and function in animal models of muscular dystrophy when injected intra-arterially (20,21). This feature makes mesoangioblasts an ideal candidate for a muscle reconstitution therapy via cell transplantation. Indeed, apparently unaffected FSHD muscles are a valuable source of mesoangioblasts that can be isolated and extensively grown in culture and induced to differentiate into myotubes. Mesoangioblasts may be easily isolated also from affected muscles although their myogenic differentiation ability is variably but significantly impaired, whereas proliferation rate and life span do not differ from normal controls (15).
In the present study we show that human FSHD mesoangioblasts, obtained from differently affected muscles and transplanted into the muscle of severe combined immune deficient (SCID) mice, have a variable ability to integrate and differentiate into skeletal muscle in vivo with an efficiency that is strictly related to the characteristics of the muscle of origin. Since mesoangioblasts offer the great advantage of possible systemic delivery, our findings may open the way to future studies utilizing cells from unaffected muscles of FSHD patients for a selective treatment aimed to ameliorate the function of severely affected muscles.
Materials and Methods
Patients
Muscle biopsies were performed after written informed consent at the Neuroscience Department of Catholic University. We used fresh and fresh-frozen muscles obtained from seven patients with genetically confirmed diagnosis of FSHD (age 30–67 years, mean 45 ± 16.5) (Table 1), one dermatomyositis (DM, age 33 years), and one normal control (CT, age 51 years). In our FSHD patients the size of the pathogenic EcoRI fragment ranged between 20 and 33 kb, indicating the presence of at least 3 KpnI repeats. No patients had infantile onset or carried very short fragments of 10–13 kb associated with the most severe phenotypes. Muscle biopsy was performed on deltoid (FSHD1 and FSHD2) or quadriceps (FSHD3, FSHD4, FSHD5, FSHD6, and FSHD7) muscles. In each patient the overall clinical severity was assessed using the clinical severity scale (CSS) (19), assigning a score ≤2 when the disease involved only facial and scapular muscles and a score of 3 (mildly affected), 3.5 (moderately affected), and 4 to 5 (severely affected) when pelvic and lower limb muscles were involved. Magnetic resonance imaging (MRI) study was performed using a 1.5 Tesla scanner. Axial images were obtained from shoulder girdle muscles and from psoas to distal foot muscles, along with coronal slices of the lumbar and pelvic muscles. To assess whether the biopsied muscle was affected and to evaluate the severity of its involvement, we used clinical (manual evaluation of muscle strength and functional tests), muscle imaging (areas of fatty fibrous substitution on T1-W images, focal areas of signal hyperintensity on T2-STIR sequences suggesting presence of inflammatory infiltrates or edema), and histopathologic criteria (increased variability of fibers size, muscle fibers with internal nuclei, presence of degenerating and/or necrotic fibers, and inflammatory infiltrates) (15). This research was approved by the Ethical Committee of our institution.
Patients, Clinical Severity, and Muscle Characterization
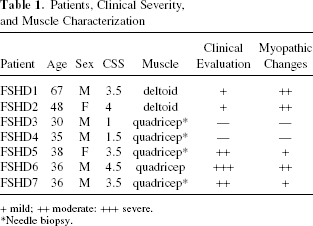
+ mild; ++ moderate: +++ severe.
Needle biopsy.
Cell and Organ Cultures
Tissue fragments of approximately 2 mm3 including intramuscular vessels and surrounding mesenchymal tissue were plated and explants were cultured in Megacell-DMEM medium (Sigma, St. Louis, MO) with 5% serum, and a cocktail of growth factors for a period of time ranging between 10 and 21 days. Tissues were digested with 0.1% trypsin and mechanically dissociated with 20-gauge needle. Cell suspension was plated in complete medium on gelatin-coated dishes. From the first passage on, cells were characterized by a triangular, adherent refractive shape and by a floating/loosely adherent round component. Since most of the cells were either poorly adherent or floating, for three passages we collected them by gently pipetting, thus eliminating most of the fibroblast and myoblast contamination (6,14). Cells were maintained in culture for several passages. At every passage, cells were counted and viability was determined by trypan blue exclusion. For growth curves, exponentially growing cells were seeded at appropriate concentrations to prevent confluence during the duration of the experiment. Viable cells were counted after 24, 48, and 72 h to determine the amount of cell proliferation.
Characterization of Human Mesoangioblasts From FSHD by Fluorescence Activated Cell Sorting (FACS)
Phenotypic characterization of all mesoangioblasts cultures was performed by examining the expression of the surface markers previously identified for human mesoangioblasts (6,14). Cells (5 × 104) were incubated with fluoroscein isothiocyanate (FITC)-, phycoerythrin (PE)-, or phycoerythrin covalently linked to Cy5 (PC5)-conjugated monoclonal antibodies (mAbs) against AC133/1 (clone CD133/1), CD34, CD56 (Miltenyi Biotec, Auburn, CA), vascular endothelial growth factor receptor II (VEGF-RII; KDR, R&D Systems, Minneapolis, MN), CD45, CD49b, CD44 (R3-242; all from Becton Dickinson, San Jose, CA), CD13 (Beckman, Fullerton, CA), anti-human fibroblasts (Gene Tex, Inc., Irvine, CA). Unlabeled samples and samples incubated with irrelevant isotype-matched fluorochrome-conjugated mAbs served as controls for background fluorescence. Cells were analyzed using Coulter® Epics XL flow cytometer (Beckman Coulter Instrumentation Laboratory, Brea, CA).
Skeletal Muscle Differentiation of FSHD Mesoangioblasts
Human mesoangioblasts differentiate down the skeletal muscle pathway under a variety of conditions, with the highest efficiency when exposed to medium conditioned by normal human myoblasts (14). Mesoangioblasts were cultured in conditioned medium for 4 days and then exposed for 7 days to differentiation medium containing 1% bovine serum albumin (BSA) (without serum). Fusion index was expressed as number of myonuclei/number of total nuclei, visualized by Hoechst 33258 staining (Molecular Probes Inc., Eugene, OR).
Intramuscular Transplantation of FSHD Mesoangioblasts Into SCID Mice
All the experimental activities involving vertebrate animals were approved by our Institutional Review Board and conducted in the Animal Care Facilities of the Catholic University strictly meeting the animal care regulations (D. L.116 issued on January 27, 1992).
To determine whether human FSHD mesoangioblasts obtained from muscles with a different degree of impairment recapitulated in vivo the behavior displayed in vitro, we selected four representative patients, in particular FSHD1 and FSHD2 whose biopsies presented significant myopathic changes, and FSHD3 and FSHD4 whose muscle biopsies did not show any histopathologic abnormality. Cells from FSHD patients were transplanted into the tibialis anterior (TA) of four SCID mice (females, aged 8 weeks) (Charles River, Wilmington, MA) and their ability to participate in muscle regeneration was evaluated in parallel. Two control mice were transplanted with mesoangioblasts from one DM patient and one normal control. The experiment was carried out in duplicate for each set of cells, and was performed two times. To avoid the experimental variability due to individual animals, we transplanted cells from “affected” and “unaffected” muscles on the posterior limbs (left and right, respectively) of the same mouse. Mesoangioblasts (1 × 106), all at passage 12, were transplanted by multiple injections performed into each TA (right and left) of six mice (two per group) (in two experiments, total mice number n = 12). Mice were anesthetized with an intraperitoneal injection of 10 ml/kg saline solution containing ketamine (5 mg/ml) and xylazine (1 mg/ml). To induce muscle regeneration before each injection, focal muscle damage was induced by liquid nitrogen. In detail, after opening the fascia, TA muscles of each mouse were exposed to a severe cryodamage immediately before cell implantation. The injury consisted of two cycles of freezing–thawing by direct application for 5 s of a 1.5 × 2 mm metal probe, precooled with liquid nitrogen, on the muscle surface at midway between the tendons. Freezing–thawing of the muscle was repeated two times to induce more severe muscle damage. Immediately after thawing, each muscle was injected with four aliquots of 25 × 104 cells each, both on the proximal and distal half of the cryodamaged muscle. In each treated animal, the extent of the induced muscle damage showing regenerative changes was assessed by morphological evaluation of sections serial to those used for lamin A/C and spectrin immunohistochemistry stained by H&E, alkaline phosphatase, and developmental myosin heavy chain immunofluorescence.
Analysis and Quantification of Cell Implantation In Vivo
Mice were sacrificed 2 months after the transplant. The entire TA muscles were excised and snap-frozen into liquid nitrogen-cooled isopentane. Cryostat sections were taken throughout the whole muscle, from tendon to tendon, in order to analyze human cells distribution. The presence of human cells in the host muscle was evaluated by immunolabeling of the human proteins lamin A/C and spectrin on 8-μm-thick serial sections using species-specific antibodies. In all experimental samples, transverse sections taken at 80-μm intervals along a longitudinal dimension of about 800 μm between both ends of the TA muscle were initially analyzed and all contained human cells. Within this muscle segment containing implanted cells, four representative sections were selected at regular intervals to count the number of human nuclei either located outside or inside muscle fibers. For each selected section nuclei were counted in three different microscopic fields at 40× magnification. Efficiency of muscle regeneration was calculated as previously described (24) as the ratio between the number of human nuclei within spectrin-positive muscle fibers and the total number of human nuclei (NIF = number of human nuclei inside fibers/total number of human nuclei).
Immunochemistry
This was performed on unfixed 8-μm-thick cryostat sections. For immunocytochemistry, cells were cultured on gelatin-coated optical quality plastic m-Dishes (ibidi Integrated Biodiagnostics, Munich, Germany) and fixed in 4% paraformaldehyde for 15 min. The following primary antibodies were used: monoclonal anti-skeletal myosin fast (1:100, MY-32, Sigma), monoclonal anti-human specific lamin A/C (1:200) and monoclonal anti-spectrin (1:50) (Novocastra, Newcastle Upon Tyne, UK), monoclonal anti-myosin heavy chain developmental type (1:20, MHCd, Novocastra), polyclonal anti-high mobility group box 1 protein (HMGB1, 1:100, Becton Dickinson). Detection of immunocomplexes was performed using the appropriate secondary Alexa Fluor 488 or 594 conjugated antibody (Invitrogen, Carlsbad, CA). Hoechst 33258 staining (Molecular Probes Inc., Eugene, OR.) was used to visualize cell nuclei. Samples were analyzed using a TCS SP5 laser scanning confocal microscope (Leica Microsystems, Wetzlar, Germany).
Chemotaxis Assay
Cell migration assays were performed using transwell permeable supports with 24-mm well (Corning, Lowell, MA). Polycarbonate membranes (8-μm pores) were coated with 1% type A porcine skin gelatin (Sigma). Either serum-free MegaCell-DMEM medium (Sigma) (negative control), medium containing 5% serum (positive control), or medium containing 100 ng/ml HMGB1 (HMG Biotech, Milan, Italy) were placed into the lower chamber. Then 1 × 105 mesoangioblasts/well, resuspended in 2.3 ml medium, were placed in the upper chamber and incubated overnight at 37°C in 5% CO2. After incubation nonmigrating cells remaining on the upper surface of the filters were mechanically detached, and cells that had migrated to the lower surface were stained with May Grunwald-Giemsa and counted in 20 randomly selected microscopic fields per membrane. Assays were performed in triplicate and the experiment was repeated twice.
Western Blot Analysis
Tissues were homogenized and protein extracts were obtained according to standard methods. Cells were harvested and homogenized in lysis buffer. Thirty micrograms were of protein was separated by SDS-PAGE and blotted onto nitrocellulose membranes (Schleicher& Schuell, Relliehausen, Germany). Blots were incubated with one of the following antibodies: monoclonal anti-skeletal myosin fast (1:500, MY-32, Sigma), monoclonal anti-myogenin (1:100, FD-5, Santa Cruz Biotechnology, Santa Cruz, CA), polyclonal anti-HMGB1 (1:500, Becton Dickinson Biosciences Pharmingen), monoclonal anti-receptor for advanced glycation end products (RAGE, 1:500, Chemicon, Temecula, CA), polyclonal anti-pIκBα (1:1000, Cell Signaling Technology, Beverly, MA), monoclonal anti-β-actin (1:2500, Sigma). After incubation with the appropriate HRP-conjugated secondary antibody, immunocomplexes were visualized by enhanced chemiluminescence (Amersham Healthcare, Piscataway, NJ) and quantified by densitometry.
Reverse Transcriptase-Polymerase Chain Reaction (RT-PCR)
Total RNA was extracted from all the samples using Trizol (Invitrogen), according to the manufacturer's instructions. Total RNA (2 μg) was used as template for first-strand cDNA synthesis using Superscript II and random hexamers as primers (Invitrogen). The cDNAs were amplified for RAGE and β-actin using the following primers: RAGE, forward 5′-CACACTGCAGTCG GAGCTAA-3′ and reverse 5′-GCTACTGCTCCACCT TCTGG-3′; β-actin, forward 5′-TACATGGCTGGGGT GTTGAA-3′ and reverse 5′-AAGAGAGGCATCCTC ACCCT-3′. PCR conditions for RAGE were: 30 cycles with annealing temperature of 55°C and 1.5 mM MgCl2. Primers for RAGE and β-actin amplified a fragment of 191 and 217 bp, respectively. PCR conditions for each set of primers yielded amplification in the linear range. The PCR products were electrophoresed on 2% agarose gel.
Statistical Analysis
Data are reported as means ± SD. Statistical analysis was performed in all experiments by ANOVA for repeated measures and comparisons between groups were assessed by Student's t-test. Adjustment for multiple comparisons was made, when necessary, by the use of the Bonferroni correction method; values of p ≤ 0.05 were considered statistically significant.
Results
Patients
In this study we included seven FSHD patients different in terms of severity of clinical involvement and size of the EcoRI fragment. Two patients received a biopsy on the deltoid muscle that is frequently spared or only mildly affected in FSHD. Indeed, in these patients clinical evaluation showed a mild to moderate hypotrophy of this muscle that was associated with moderate myopathic changes on histopathology (FSHD1 and FSHD2). Out of the five patients who underwent a biopsy on quadriceps, in two patients (FSHD3, FSHD4) this muscle was unaffected on clinical and MRI examination (not shown) and showed no histopathologic abnormalities. In the other three cases (FSHD5, FSHD6, FSHD7), the quadriceps resulted unequivocally affected by both clinical and MRI evaluation, with different degrees of histopathologic involvement (Table 1).
Mesoangioblast Isolation From FSHD Muscle Biopsies
Mesoangioblasts were isolated as previously described (6,14). From the first passage on, cells were characterized by the typical morphology of human mesoangioblasts (6,14), with triangular adherent and floating/loosely adherent rounded cells. Since most of the cells were poorly adherent, they were passed three times by gentle pipetting, thus reducing fibroblast and myoblast contaminations. Cells were kept in culture up to 22 population doublings, when large flat cells undergoing senescence started to appear. There was no difference in life span, number, and morphology of cells isolated from affected or apparently normal muscles (Fig. 1A, left column). Notably, the proliferation rate of all FSHD mesoangioblasts was comparable and independent from the severity of the disease and patients' age (Fig. 1B). Doubling time from all FSHD cultures was 30.13 ± 3.35 h and did not differ significantly from what previously observed (15). There were no differences in the amount and phenotypic characteristics of cells isolated from fresh or fresh-frozen muscles. The phenotypic characterization was performed by analyzing expression of the surface markers recently identified for human mesoangioblasts (6,14). Cells from all patients, analyzed at various passages, were homogeneously strongly positive for CD44 (96.6 ± 3.2%) and CD13 (96.77 ± 3.02%), positive for CD49b (82.86 ± 6.6%), and negative for CD34, CD133, and CD45 (not shown). In particular, only a small fraction of all FSHD cultures was positive for CD56 and human fibroblast surface antigen (5.2 ± 2.8% and 1.48 ± 0.28%, respectively), indicating a negligible contamination of our cultures by myoblasts and fibroblasts. This was independent from the muscle of origin.

(A) Left column. Proliferating facioscapulohumeral muscular dystrophy (FSHD) mesoangioblasts. Phase contrast. No differences in terms of morphology were observed between cells obtained from affected and unaffected FSHD muscles and normal control. Scale bar: 10 μm. Right column. Skeletal muscle differentiation of FSHD mesoangioblasts and control mesoangioblasts. Immunofluorescence for myosin (green, nuclei are labeled in blue by Hoechst). Cells obtained from affected muscles (FSHD1 and FSHD2) generated only spare myosin-positive myotubes (fusion index 0.1–0.15). On the contrary, mesoangioblasts from unaffected muscles (FSHD3 and FSHD4) efficiently fused into multinucleated and fully differentiated myotubes (fusion index 0.6–0.7), with no significant difference from normal control mesoangioblasts. Scale bar: 100 μm. Each experiment was performed in duplicate at least two times for each patient. (B) Cell growth was assessed after 24, 48, and 72 h. Results are expressed as absolute counts. Bars represent the mean ± SD of triplicate samples of one representative experiment out of three.
In addition, all FSHD mesoangioblasts were positive for ALP (95 ± 4.21%), a marker identifying human adult mesoangioblasts as the in vitro progeny of pericytes (6,14).
FSHD Mesoangioblast Differentiation Into Skeletal Muscle
Human mesoangioblasts differentiate into skeletal muscle under a variety of conditions, with the highest efficiency when exposed to human normal myoblast-conditioned medium (14). As recently described by our group, despite their normal characteristics, FSHD mesoangioblasts differentiate into skeletal muscle to a different extent, in clear correlation with the overall disease severity and degree of involvement of the muscle of origin (as evaluated by clinical, MRI, and histopathological examination). This differentiation defect did not correlate with patients' age since, in our hands, mesoangioblasts from aged controls, DM, and polymyositis fully differentiate into multinucleated myotubes with high efficiency (14,15). However, a possible effect of age on mesoangioblasts differentiation that is peculiar of FSHD cannot be completely excluded and remains to be further investigated. Mesoangioblasts obtained from the morphologically abnormal muscles of the more severely affected patients (FSHD1, FSHD2, and FSHD6, with moderate/severe myopathic changes) were able to fuse only into spare myosin-positive myotubes (fusion index 0.14 ± 0.13). Moreover, these myotubes were short and most of them contained no more than two nuclei. Mesoangioblasts from FSHD5 and FSHD7, presenting mild myopathic changes, gave rise to a higher percentage of cells able to fuse into myosin-positive thin and mainly binucleated myotubes (fusion index 0.28 ± 0.06).
On the contrary, mesoangioblasts from the apparently spared quadriceps of FSHD3 and FSHD4 differentiated into multinucleated myosin-positive myotubes (fusion index 0.7 ± 0.08), with no significant difference from normal control mesoangioblasts (Fig. 1A, right column, Table 1).
Of note, myoblasts isolated from the same FSHD biopsies were able to fully differentiate into multinucleated myotubes, regardless of impairment of the muscle of origin.
Intramuscular Transplantation of FSHD Mesoangioblasts Into SCID Mice
To determine whether human FSHD mesoangioblasts obtained from muscles with a different degree of impairment also keep their different myogenic potency in vivo, we transplanted them into the TA of SCID mice and evaluated their ability to participate to muscle regeneration. No significant differences between treated animals were observed regarding the extent of linear and transversal area of damaged muscle where regeneration was induced, as assessed by morphological evaluation of sections stained by H&E, alkaline phosphatase, and developmental myosin heavy chain immunofluorescence (not shown) that were serial to those used for immunohistochemistry.
Transplantation success was assessed by histological identification of human antigens within murine muscle tissue. In particular, we analyzed the nuclear envelope marker lamin A/C, to track the fate of human cells, and spectrin that shows a true fusion event between donor cells and recipient fibers. Comparable numbers of human lamin A/C-positive cells were found in all transplanted muscles, spread in a longitudinal dimension of approximately 800 ± 80 μm and transversally distributed throughout the entire muscle belly on a given section (Table 2). However, cells from affected FSHD muscles were consistently located mainly in the endomysial and perymisial connective tissue rather than integrated within regenerating fibers. This may reflect a defect in local migration of injected cells in response to chemoattractants released by damaged fibers or may be the result of a more general impairment of homing, engraftment, and differentiation. FSHD mesoangioblasts from apparently normal muscles (FSHD3 and FSHD4) were able to integrate into skeletal muscle in vivo, participating with high efficiency (p ≤ 0.05) to muscle regeneration. In fact, a high number of human nuclei were found under the basal lamina of muscle fibers expressing human spectrin (Fig. 2A, B). On the contrary, although a comparable number of human transplanted cells were detectable, mesoangioblasts obtained from affected muscles (FSHD1 and FSHD2) did not succeed in integrating in muscle fibers, as shown by a large number of human cells mainly localized in the muscle interstitium, with only few nuclei located beneath the basal lamina of spectrin-positive fibers. Furthermore, the number of spectrin-positive fibers was significantly higher in muscles transplanted with mesoangioblasts from unaffected muscles compared to mesoangioblasts from affected ones (p ≤ 0.05) (Fig. 2A–C).
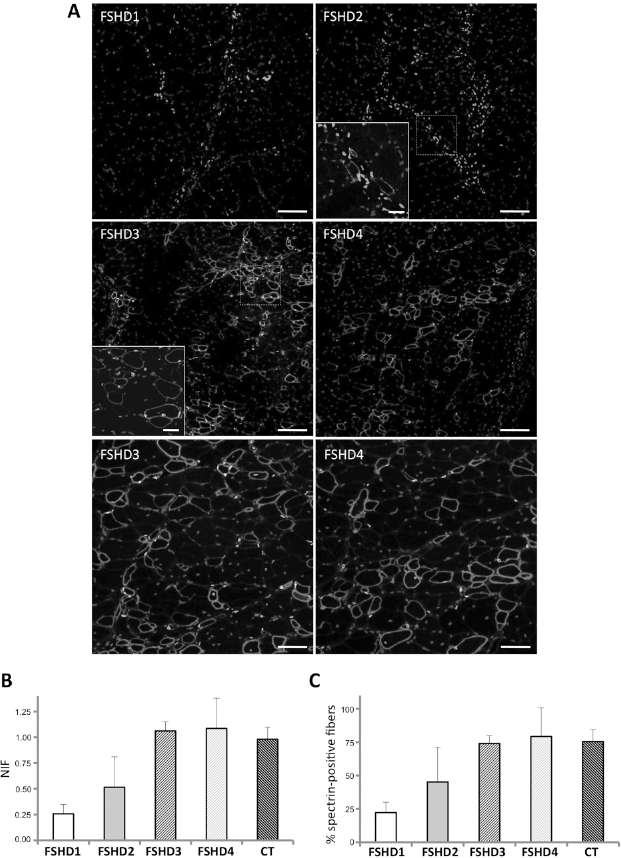
In vivo myogenicity of FSHD mesoangioblasts was tested by intramuscular transplantation in severe combined immune deficient (SCID) mice. (A) After transplants, the two TAs of the same mouse injected with FSHD mesoangioblasts from affected and apparently normal muscles showed a different ability of these stem cells to integrate and participate to muscle regeneration. In particular, in the tibialis anterior (TA) muscle transplanted with cells from affected muscles (FSHD1 and FSHD2) a large number of human nuclei (lamin A/C positive labeled in green) were localized mainly in the connective tissue, with only few human nuclei located beneath the basal lamina of spare spectrin-positive fibers (labeled in red, inset). In the muscle transplanted with mesoangioblasts from apparently normal muscles (FSHD3 and FSHD4) numerous fibers express human spectrin and contain human lamin A/C-positive myonuclei. The inset shows one spectrin-positive muscle fiber containing three human nuclei. The lower photomicrographs shows at higher magnification that the vast majority of transplanted human cells from apparently normal muscles (FSHD3 and FSHD4) have originated muscle fibers expressing human spectrin and that only rare human nuclei are located in the endomysium outside muscle fibers. Hoechst staining was used to identify both human and murine nuclei (blue). Scale bars: 100 μm in the higher four panels, 25 μm in the insets, 50 μm in the lower panels. (B) By quantitative evaluation the efficiency in muscle regeneration was significantly higher in muscles transplanted with FSHD3 and FSHD4 mesoangioblasts compared to those receiving FSHD1 and FSHD2 cells (p ≤ 0.05). NIF: number of human nuclei inside fibers/total number of human nuclei. (C) Quantitative evaluation of spectrin-positive fibers, in relation to the total number of human nuclei, showed a significant difference between muscles transplanted with FSHD3 and FSHD4 mesoangioblasts compared to those receiving FSHD1 and FSHD2 cells (p ≤ 0.05).
Human Nuclei in FSHD and Control Transplanted MusclesHuman Nuclei in FSHD and Control Transplanted Muscles

No significant difference in terms of number of human nuclei (Fig. 2B) and number of spectrin-positive fibers was observed among muscles transplanted with FSHD mesoangioblasts from unaffected muscles versus DM and normal control cells (Fig. 2C).
Chemotaxis Assay and HMGB1-RAGE Expression in FSHD Mesoangioblasts
HMGB1, a nuclear DNA-binding protein that interacts with a variety of transcription factors, is secreted in the extracellular milieu by inflammatory cells, acts as a cytokine, and can also be passively released by necrotic or damaged cells thus promoting inflammation (1,5,26). This protein is a powerful chemoattractant for muscle progenitor cells such as murine mesoangioblasts in vitro and in vivo (7,10,17,18). Because mesoangioblasts obtained from severely affected muscles remained mainly localized within perymisial-endomysial connective tissue, possibly reflecting a defect in local migration, we investigated the migration ability in vitro of all mesoangioblasts used, by comparing their transmigration across a septum of a Boyden apparatus in response to HMGB1 (7). Similar transwell assays have been already used as a reliable indicator of the mechanisms controlling cellular trafficking in vivo (13,18). A remarkable difference in migration ability in response to HMGB1 (100 ng/ml) was observed between cells from apparently normal and affected muscles (p = 0.01). Interestingly, only for cells obtained from unaffected muscle, the number of migrating mesoangioblasts in response to HMGB1 versus the corresponding control medium was significantly higher (p = 0.04) compared to those from affected muscle, indicating a reduced sensitivity to HMGB1 (Fig. 3). Since HMGB1 signaling is mainly mediated by RAGE, we investigated the expression of this receptor and found low levels of RAGE both at mRNA and protein levels in mesoangioblasts obtained from affected muscles (Fig. 4A, B). The lower expression of RAGE does not appear to simply reflect a corresponding lower expression of the ligand cytokine in the muscle of origin, since by Western blot on muscle biopsy homogenates, severely affected muscles showed a higher expression of HMGB 1 compared to apparently normal muscles (Fig. 4C). In fact, also by immunocytochemistry on frozen muscle sections, clinically and pathologically affected FSHD muscles displayed, in addition to nuclear HMGB1, increased cytoplasmic and extracellular immunoreactivity (likely due to acetylated HMGB1 for the most part) (4), that is correlated to the presence of endomysial inflammatory mononuclear infiltrates and necrotic fibers (Fig. 4D).

Migration ability was evaluated by comparing the transmigration of mesoangioblasts from affected and unaffected muscles across a septum of a Boyden apparatus in response to 100 ng/ml High mobility group box 1 protein (HMGB1). Representative May Grunwald-Giemsa staining of filters from FSHD4 and FSHD1 mesoangioblasts, after mechanical detachment of nonmigrating cells from the upper surface. Bars represent the average number of migrating cells per field of vision (FOV) ± SD from two independent experiments performed in triplicate in FSHD4 and FSHD1 cells. A significant number of mesoangioblasts from normal muscle migrated in response to HMGB1 compared to cells from affected muscle (*p = 0.01). Scale bar: 100 μm.
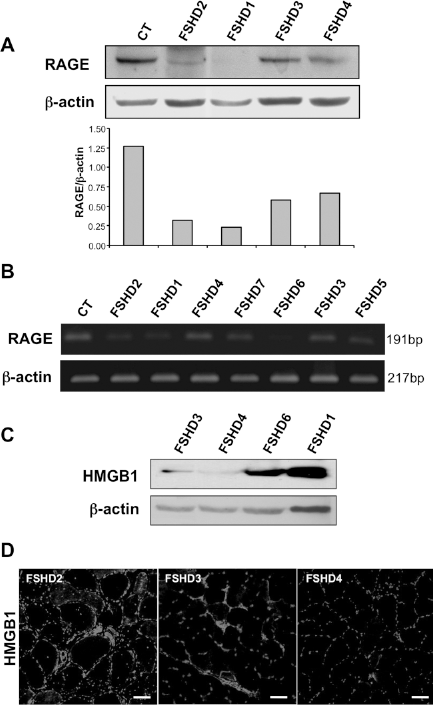
(A) Western blot analysis on proliferating FSHD mesoangioblasts using an antibody recognizing transmembrane Receptor for advanced glycation end products (RAGE) shows lower levels of this protein in cells obtained from affected muscles. A representative experiment out of three is shown. Densitometry was performed for all the samples and ratios were calculated between RAGE and β-actin values. Samples were analyzed three times with similar results. (B) mRNA expression of RAGE in all FSHD mesoangioblasts and one control. The mRNA was amplified using RT-PCR. PCR conditions yielded amplification in the linear range. (C) Western blot analysis on muscle biopsy homogenates shows higher expression of HMGB1 in severely affected muscles compared to apparently normal muscles. A representative experiment out of three is shown. (D) HMGB1 expression in muscle biopsies from patients with FSHD. HMGB1 immunohistochemistry on muscle biopsies from three representative FSHD patients is shown. Affected muscles (FSHD2) showed a higher expression of the cytokine compared to apparently normal muscles (FSHD3, FSHD4). Scale bar: 40 μm.
Nuclear factor kappa light chain enhancer of activated B cells (NF-κB) activation is required for mesoangioblasts migration in response to the tissue-damage signaling molecule HMGB1. The phosphorylation of the NF-κB inhibitor IκBα at Ser32 and Ser36 by inhibitory κβ kinase (IκKβ), leads to its polyubiquitination and proteasomal destruction. This induces the nuclear translocation of NF-κB p50/p65 and p50/c-Rel heterodimers and the transcription of target genes. Because HMGB1 induces activation of the canonical pathway of NF-κB via phosphorylation of endogenous IκBα, we examined pIκBα expression in mesoangioblasts from FSHD muscles with a different degree of impairment. Interestingly, mesoangioblasts obtained from unaffected muscles constitutively expressed higher levels of pIκBα compared to affected ones (Fig. 5A, B).
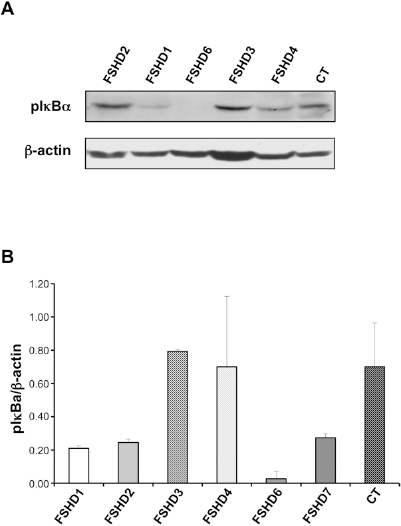
Mesoangioblasts from unaffected muscles constitutively express higher levels of pIκBα than mesoangioblasts from affected muscles. (A) A representative Western blot out of three is shown. (B) Densitometric analysis from three different Western blots performed for six FSHD samples and one control. Ratios were calculated between pIκBα and β-actin values. Bars represent the mean ± SD.
Discussion
In the present study we have analyzed human FSHD mesoangioblasts obtained from muscles with a different degree of impairment and demonstrated that their ability to integrate and differentiate into skeletal muscle in vivo is variable and strictly related to the characteristics of the muscle of origin, thus recapitulating in vivo the behavior displayed in vitro.
Molecular pathogenesis of FSHD has been unveiled for several aspects; however, mechanisms leading to progressive muscle degeneration are yet unidentified. With the goal to halt the disease progression and limit muscle damage, it is reasonable to use in vitro expanded autologous myogenic stem cells capable to engraft within damaged muscles enhancing their regenerative capacity.
The reason of the selective sparing of several muscles in FSHD in spite of the same genetic abnormality, similar to what is observed in other inherited muscle diseases such as hereditary inclusion-body myopathy or oculopharyngeal muscular dystrophy, remains elusive. However, this observation has brought up the possibility to take advantage of muscles relatively spared by the pathologic process as a source of myogenic stem cells that could be used in cell therapy protocols in FSHD patients.
Mesoangioblasts have demonstrated a significant, though partial, capability to restore damaged muscles when delivered intra-arterially (20,21). In fact, they possess the ability to colonize injured muscles by crossing the endothelium, mostly skipping the capillary filters of the body such as liver and lungs.
In a previous work we demonstrated that FSHD mesoangioblasts from apparently normal muscles can be easily isolated, extensively grown in culture, and induced to differentiate into myotubes that apparently do not display the morphological abnormalities observed in primary myoblast cultures obtained from the same muscles (15). Conversely, the myogenic differentiation ability of mesoangioblasts from affected muscles is variably but significantly impaired, with a differentiation defect correlating with the overall disease severity and the degree of histopathologic alterations of the muscle of origin. By contrast, all primary muscle satellite cells derived from unaffected and affected FSHD muscles are always able to differentiate into multinucleated myotubes, although they have impaired proliferation and clonogenic abilities and display, in some cases, abnormal morphology. This suggests that the two starting populations of myogenic stem cells are clearly distinct and may behave differently even when exposed to identical differentiating culture conditions.
The aim of our present study was to verify whether mesoangioblasts expanded from differentially affected FSHD muscles reproduce in vivo the same differentiation ability shown in vitro by investigating their capability to form new muscle fibers during muscle regeneration of experimentally damaged muscles. Although the intra-arterial delivery represents the more natural and effective way to target mesoangioblasts into damaged muscles, intramuscular transplantation avoids the introduction of additional variables that may interfere with quantification of FSHD transplanted cells, such as variable crossing of the endothelial barrier and systemic migration with loss of cells skipping the capillary filter. In order to inject simultaneously in each animal mesoangioblasts from two different patients we used intramuscular transplantation, hence eliminating most of the variability related to individual animals. This approach allowed us also to comparatively examine mesoangioblasts with a different behavior in vitro after transplantation in the same muscle microenvironment. Mesoangioblasts from apparently normal muscles were able to integrate and efficiently participate to muscle regeneration, giving rise to a significantly high number of human spectrin-positive fibers. By contrast, mesoangioblasts obtained from impaired muscles remained mainly localized in the muscle interstitium. These data confirm the differentiation defect of FSHD mesoangioblasts from affected muscles observed in vitro and demonstrate that the ability to form fully differentiated myotubes in vitro predicts their in vivo behavior. Moreover, the differentiation defect exhibited by FSHD mesoangioblasts seems an intrinsic property of these cells and is not dependent on the local environment, since FSHD mesoangioblasts always behave in the same manner (both in vivo and in vitro) in relation only to the characteristics of the muscle of origin. In addition, the fact that all the mesoangioblast sets from both apparently unaffected and affected muscles contained comparable low contaminating amounts of myoblasts and fibroblasts rules out the possibility that the results obtained simply reflect the presence of other types of cells.
Yet unknown putative environmental cues, related to the specific molecular defect of the disease and likely more active in the advanced stage of myopathy, may operate in the pathological muscle milieu and determine the block of skeletal muscle differentiation of mesoangioblasts from muscles with evident pathologic abnormalities. On the contrary, mesoangioblasts from clinically and pathologically spared FSHD muscles keep a normal myogenic ability. Indeed, their regenerative capacity is comparable to that of mesoangioblasts obtained from DM muscle, which are by far more efficient than normal mesoangioblasts as they have, in our experience, the highest myogenicity (14). Since mesoangioblasts from affected muscles remained mainly localized in the connective tissue with only few human nuclei integrated within spare spectrin-positive fibers, the existence of a possible migration defect, in addition to the demonstrated differentiation impairment in culture, can be hypothesized. Several chemoattractants generated in dystrophic muscles are known to favor the homing of circulating progenitors to damaged tissues. In particular, extracellular HMGB1 and RAGE are a ligand/receptor pair sufficient for mesoangioblasts homing in vivo (18). Accordingly, here we have demonstrated a significant difference in migration ability in response to HMGB1 in vitro between cells from normal and affected FSHD muscles. Moreover, similarly to what previously described in other dystrophic muscles (18), HMGB1 expression was higher in severely affected FSHD muscles compared to apparently normal ones. Nevertheless, mesoangioblasts isolated from severely affected muscles exhibited lower levels of the HMGB1 receptor, RAGE, in comparison to mesoangioblasts from unaffected muscles. Since HMGB1 signals prevalently through RAGE, our data strongly suggest a possible dysregulation of this signaling pathway that might account for the impaired ability of these cells to respond to extracellular HMGB1 and engraft within damaged muscle. However, we cannot exclude the existence of a different unidentified chemoattractor/receptor complex that FSHD muscles might use to attract mesoangioblasts.
Our findings are in agreement with a previous study showing an activation of RAGE–NF-κB pathway induced by oxidative stress in unaffected FSHD muscles that express high levels of RAGE and hydrogen peroxide (12). An increased production of advanced glycation end products (AGEs) due to elevated reactive oxygen species (ROS) levels has been provided as a possible explanation for the high expression levels of RAGE in apparently normal FSHD muscles. Moreover, abnormal ligation of AGEs to RAGE is known to cause further ROS production and RAGE stimulation and elevated level of hydrogen peroxide may in turn induce NF-κB activation. The reason why RAGE is constitutively less expressed in cultured mesoangioblasts from affected muscles remains to be clarified.
NF-κB activation is required for mesoangioblasts migration in response to several chemoattractants released in damaged muscles, such as HMGB1. The phosphorylation of the NF-κB inhibitor IκBα leads to its polyubiquitination and proteasomal degradation and induces the nuclear translocation of NF-κB and the transcription of target genes. In the present study, we observed that even in the absence of any stimulation, mesoangioblasts from apparently normal FSHD muscles express higher levels of pIκBα reflecting NF-κB activation. The degree of NF-κB activation in mesoangioblasts from affected and apparently normal muscles may correlate with a variable receptiveness to migration stimuli present in the host regenerative skeletal muscle environment necessary for a successful engraftment.
Our data show that migration ability of FSHD mesoangioblasts in vitro may be different depending upon the biological characteristics of the muscle of origin and correlates with their myogenicity in vivo. However, whether a variable migratory ability of FSHD mesoangioblasts is indeed present in vivo and plays a significant role remains to be conclusively demonstrated. In fact, a more global impairment of engraftment of FSHD mesoangioblasts from affected muscles, beside a putative defect in local migration in response to chemoattractants released by damaged fibers, may reflect a decreased ability to fuse with existing myofibers or to appropriately trigger the myogenic differentiation program.
In conclusion, we have evaluated the ability of FSHD mesoangioblasts to engraft and differentiate in vivo in new muscle fibers and demonstrated that this capacity strongly correlates with the pathologic characteristics of the muscle of origin, exactly reproducing in vivo the distinguishing features displayed in vitro. Simple strategies for improving homing and engrafting of stem cells to damaged tissues have been described (7,22) and could be tailored also to ensure an efficient delivery of FSHD mesoangioblasts into injured target muscles. Since mesoangioblasts offer the great advantage of possible systemic delivery, our data open the way for future studies utilizing cells expanded from unaffected muscles of patients with FSHD for a selective treatment to halt muscle degeneration in severely affected muscles.
Footnotes
Acknowledgments
We thank Manuela Papacci for her precious technical assistance. This study was supported by grants from Don Carlo Gnocchi Onlus Foundation and Catholic University to R.M. and M.M. Author contributions were: conception and design, collection, and/or assembly of data, data analysis and interpretation, manuscript writing, final approval of the manuscript (R.M); collection and/or assembly of data, data analysis and interpretation, final approval of the manuscript (T.G); data analysis and interpretation, manuscript writing, final approval of the manuscript (A.B.); collection and/or assembly of data, data analysis and interpretation, final approval of the manuscript (C.G.); collection and/or assembly of data, data analysis and interpretation, final approval of the manuscript (C.S.); provision of study material or patients financial support, administrative support, final approval of the manuscript (P.A.T.); provision of study material or patients, collection and/or assembly of data, final approval of the manuscript (E.R.); conception and design, collection and/or assembly of data, data analysis and interpretation, manuscript writing, final approval of the manuscript (M.M.).
