Abstract
We attempted to regenerate bone in a significant osseous defect with various stem cells from deciduous teeth, extracted from puppies, and grafted them into a parent canine mandible as an allograft, parent dental pulp, and bone marrow by tissue engineering and regenerative medicine technology using platelet-rich plasma as an autologous scaffold and signal molecules. Initially, teeth were extracted from a child and parent hybrid canine mandible region and bone marrow (canine mesenchymal stem cells; cMSCs), and parent teeth (canine dental pulp stem cells; cDPSCs), and stem cells were extracted from deciduous teeth (puppy deciduous teeth stem cells; pDTSCs). After 4 weeks, bone defects were prepared on both sides of the mandible with a trephine bar. Graft materials were implanted into these defects: 1) control (defect only), 2) platelet-rich plasma (PRP), 3) cMSCs/PRP, 4) cDPSCs/PRP, and 5) pDTSCs/PRP to investigate the effect of stem cells. The newly formed bones were evaluated by histology and histomorphometric analysis in the defects at 2, 4, and 8 weeks. According to histological observations, the cMSCs/PRP, cDPSCs/PRP, and pDTSCs/PRP groups had well-formed mature bone and neovascularization compared with the control (defect only) and PRP groups at 4 and 8 weeks, respectively, and the mineralized tissues in cMSCs/PRP, cDPSCs/PRP, and pDTSCs/PRP specimens were positive for osteocalcin at 8 weeks. Histometrically, newly formed bone areas were 19.0 ± 2.9% (control), 19.7 ± 6.0% (PRP), 52.8 ± 3.5% (cMSCs/PRP), 61.6 ± 1.3% (cDPSCs/PRP), and 54.7 ± 2.2% (pDTSCs/PRP) at 8 weeks. There were significant differences between cMSCs, cDPSCs, pDTSCs/PRP, and control and PRP groups. These results demonstrate that stem cells from deciduous teeth, dental pulp, and bone marrow with PRP have the ability to form bone, and bone formation with DTSCs might have the potential to generate a graft between a child and parent. This preclinical study could pave the way for stem cell therapy in orthopedics and oral maxillofacial reconstruction for clinical application.
Keywords
Introduction
Bone defects often occur from tumor resection, congenital malformation, trauma, osteoporotic fractures, surgery, or periodontitis, and autogenous bone grafting has been the gold standard approach to treatment. However, severe invasiveness with attendant donor site morbidity and complications is not the only issue, as there is also a limited supply of autogenous bone. On the other hand, tissue engineering and regenerative medicine (TERM) using stem cells, which are of benefit from the viewpoint of reduced hospital stay, morbidity, and immune reactions, is a tool in a new clinical platform for a whole spectrum of intractable diseases. The TERM concept is to regenerate tissues using three elements—stem cells, scaffolds, and signal molecules—and it is thought that the role of stem cells is most important.
Recently, embryonic stem cells (ES cells), induced pluripotent stem cells (iPS cells), and somatic stem cells have been reported; however, ES and iPS cells have many issues to overcome for clinical use because of ethical and safety problems, immunorejection, and tumorigenesis. Somatic stem cells, especially mesenchymal stem cells (MSCs) isolated from various tissues, including bone marrow, adipose tissue, skin, umbilical cord, and placenta (7,17,26,28), have been used in clinical applications. We have used bone marrow-derived MSCs (BMMSCs), instead of autogenous bone grafting, in clinical trials for effective treatment of osseous defects, and favorable results have been obtained (22–24), but bone marrow aspiration is an invasive and painful procedure for the donor. Besides, the number, proliferation, and differentiation potential of MSCs decline with increasing age (10).
Previous studies have reported that several populations of stem cells have been isolated from dental tissues including the pulp of both exfoliated and adult teeth, periodontal ligament, and dental follicle. Dental pulp stem cells (DPSCs) and stem cells from human exfoliated deciduous teeth (SHED) have generic mesenchymal stem cell-like properties such as self-renewal and multilineage differentiation (5,13,21). DPSCs and SHED were demonstrated to have the ability to generate not only dental tissue such as dentine/pulp-like complexes but also bone tissue. Since SHED exhibit higher proliferation rates (1,5,15) and can be obtained with ease compared to BMMSCs, they might become an attractive source of autologous stem cells for bone regeneration.
MSCs have been reported to have a potent immunosuppressive effect that inhibits the T cells, B cells, and NK cells and dendritic cell function and be useful for the treatment of mesenchymal disorders. In a previous study, they demonstrated that allogeneic BMMSCs offer feasible posttransplantation therapy for osteogenesis imperfect without clinical symptoms, indicating the autoimmune response against the marrow microenvironment (6). On the other hand, DPSCs have been reported to have higher prevention ability of T-cell alloreactivity and immunosuppressive activity compared to BMMSCs (16). Because of their low immunogenic potential, MSCs from dental pulp could be used as a potential allogenic therapeutic tool for regenerative medicine, particularly for bone regeneration.
In this study, we utilized the canine mandible as a large-animal model, in which mandibular defects were established for tissue engineering and therapies (14,24,25), to examine the potential of using stem cells, autologous MSCs, DPSCs, and allologous deciduous tooth stem cells (DTSCs).
Materials and Methods
Canine Animal Models
All protocols undertaken in this study were performed in accordance with protocols approved by the Institutional Animal Care Committee and the university committee (permission number 19373). After a period of housing, hybrid dogs were operated on under general anesthesia. The first molar, premolars, and the second and third premolars in the mandible region were extracted. Bone defects on both sides of the mandible were prepared with a trephine bar with a diameter and depth of 10 mm (25). The defects were implanted with graft materials as follows: 1) control (defect only), 2) platelet-rich plasma (PRP), 3) canine mesenchymal stem cells/PRP (cMSCs/PRP), 4) canine dental pulp stem cells/PRP (cDPSCs/PRP), and 5) puppy deciduous tooth stem cells/PRP (pDTSCs/PRP), and investigated for osteogenesis. We created defects and implanted four materials at random sites (Fig. 1).
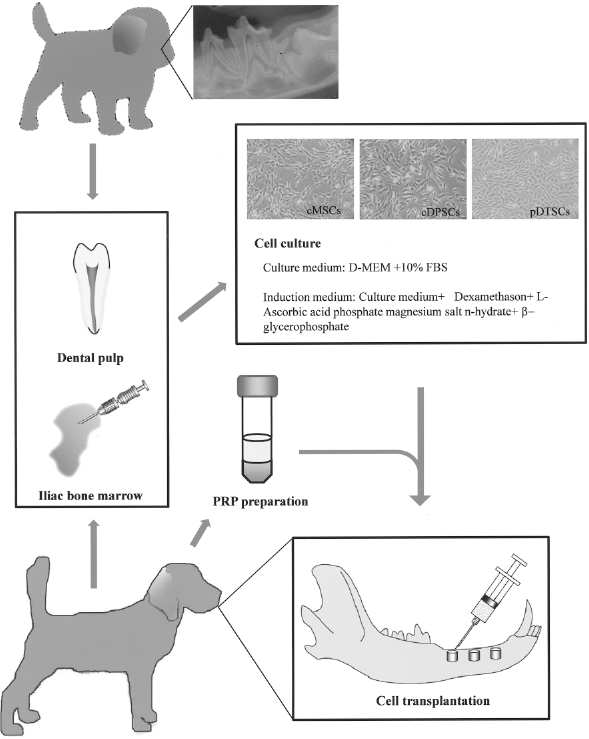
Schema of experimental protocol. Magnification of cell morphology (cMSCs, cDPSCs, and pDTSCs) (×40).
Isolation and Cultivation of MSCs, DPSCs, and DTSCs
The cMSCs were isolated from the parent dog's iliac crest marrow aspirate (10 ml), dental pulp tissues were obtained from extracted puppy deciduous teeth and parent permanent teeth (Fig. 1), and cMSCs, cDPSCs, and pDTSCs were isolated and cultured as previously described (5,9,13,21). Briefly, the cMSCs were incubated at 37°C in a humidified atmosphere containing 95% air and 5% CO2 and replated at densities of 3.1 × 103 cells/cm2 in control medium; Dulbecco's modified Eagle's medium (DMEM; Gibco, Rockville, MD) containing 20% mesenchymal cell growth supplement (Lonza® Inc., Walkersville, MD) and antibiotics (100 U/ml penicillin, 100 μg/ml streptomycin, and 0.25 μg/ml amphotericin B; Gibco). The pulp was gently removed and digested in a solution of 3 mg/ml collagenase type I and 4 mg/ml dispase for 1 h at 37°C. After filtration using 70-mm cell strainers (Falcon, BD Labware, Franklin Lakes, NJ), cells were cultured in the same medium at 37°C containing 95% air and 5% CO2. After primary culture, the cells were subcultured at about 1 × 104 cells/cm2. Three supplements for inducing osteogenesis, dexamethasone (Dex), sodium β-glycerophosphate (β-GP), and L-ascorbic acid 2-phosphate (AsAP) were purchased from Sigma Chemical Co. (St. Louis, MO). The cells were incubated at 37°C in a humidified atmosphere containing 95% air and 5% CO2. In culture, cMSCs, cDPSCs, and pDTSCs were trypsinized and used for implanting.
PRP, PRP Gel Preparation, and Injection of cMSCs/PRP, cDPSCs/PRP, pDTSCs/PRP Admixture
PRP and the relevant condition were prepared as previously described (24). Briefly, approximately 50 ml whole blood was drawn from the canine into centrifuge tubes containing 10 ml culture medium with 250 U/ml preservative-free heparin. The blood was first centrifuged in a standard laboratory centrifuge, Himac CT (Hitachi Koki; Hitachi city, Japan). Subsequently, yellow plasma (containing buffy coat, which contained platelets and leukocytes) was taken up into a neutral monovette with a long cannula. Second, centrifugation was performed to combine the platelets into a single pellet and the plasma supernatant, which is platelet-poor plasma (PPP) and contains relatively few cells, was removed. The resulting pellet of platelets, buffy coat/plasma fraction (PRP), was resuspended in the residual 5 ml plasma and used in platelet gel. PRP was stored for about 1 day at room temperature in a conventional shaker until use. Bovine thrombin in powder form (5,000 units) was dissolved in 5 ml of 10% calcium chloride in a separate sterile cup. Next, 3.5 ml PRP, these cells (1.0 × 107 cells/ml), and 0.5 ml air were aspirated into a 5-ml syringe, and in a second 2.5-ml syringe 500 μl thrombin/calcium chloride mixture was aspirated, and the cells were resuspended directly in PRP. The two syringes were connected with a “T” connector and the plungers of the syringes were pushed and pulled alternatively, allowing air bubble to transverse the two syringes. The contents assumed a gel-like consistency as the thrombin affected the polymerization of fibrin to produce an insoluble gel. The gel was injected into the bone defect field using a 5-ml syringe.
Histological and Histomorphometric Analysis
Each implantation site was excised with a trephine bar of 2 mm diameter at 2, 4, and 8 weeks after implantation, and sections for each experimental group were assessed by histological and histomorphometrical methods as previously (25). The specimens were fixed in 10% buffered formalin, decalcified (K-CX; Falma Co., Tokyo), and stained with hematoxylin and eosin. These specimens were examined under a light microscope and analyzed by a pathologist, blinded to the identity of each specimen, who determined the presence or absence of bone formation.
Histomorphometrical findings were analyzed with a microcomputer for image analysis. Each image of the specimens at the implantation site excised with a trephine bar of 2 mm diameter was copied on color reversal film, digitized as a 256 × 256 array of 8-bit density values, and transferred to a microcomputer for analysis (NIH Image, version 1.61; National Institutes of Health) (20). The augmented area was defined as the area enclosed within the mandible bone excised with a trephine bar of 10-mm diameter. The volume of newly formed bone in the augmented area was quantified using this computer-based image analysis system. This was calculated as the representative area percent of bone present by deducting normal bone areas from measurement areas in the section based on 2-mm biopsies.
Immunofluorescence and Immunohistochemical Analysis
Eight weeks after implantation, specimens were fixed in 10% buffered formalin, decalcified (K-CX; Falma Co.), and used for immunofluorescence and immunohistochemical analysis. The sections were incubated for 32 min at 40°C with primary mouse anti-bovine osteocalcin monoclonal antibody (Takara Bio. Inc., Shiga, Japan) diluted 1:200 in PBS. Sections were then washed three times in PBS, and incubated overnight at 4°C with goat anti-mouse IgG fluorescein-conjugated secondary antibody (Chemicon International, Inc., Billerica, MA, USA) diluted 1:200 in PBS for immunofluorescence staining. Finally, the sections were washed in PBS and mounted with Vectashield with DAPI (Vector Laboratories Inc., Burlingame, CA, USA). On the other hand, the sections were incubated for 8 min at 40°C with rabbit anti-mouse IgG biotin-conjugated secondary antibody using a Ventana NexES IHC (Ventana, Tucson, AZ, USA) automated slide staining system and an iView DAB Universal kit (Ventana) according to the manufacturer's instructions for immunohistochemical staining.
Statistical Analysis
Group means and SDs were calculated for each measured parameter. Differences in newly formed bone between the control, PRP, cMSCs/PRP, cDPSCs/PRP, and pDTSCs/PRP groups were analyzed using the Tukey-Kramer test following one-way analysis of variance (ANOVA). A value of p < 0.05 or p < 0.001 indicated statistical significance.
Results
Macro Findings and Histological Evaluation of the Implants (PRP, cMSCs/PRP, cDPSCs/PRP, and pDTSCs/PRP) Compared to the Control In Vivo
The experimental design of 10-mm-long defects created in an established canine mandible model (15,25,26) was implanted with PRP, cMSCs/PRP, cDPSCs/PRP, and pDTSCs /PRP (Figs. 1 and 2). Macroscopic findings showed that bone regeneration using cMSCs/PRP, cDPSCs/PRP, and pDTSCs/PRP was to a natural marginal bone level, but regeneration using PRP and the control (defect only) was not complete (Fig. 2). In the pDTSCs/PRP group, no symptoms of pDTSCs rejection were observed in the parent recipient mandible (Fig. 2).

Macroscopic observations for bone regeneration. (A) The canine mandible healed after extraction. (B) Experimental design in the mandible prepared with a trephine bar of 10-mm diameter. (C) Representative implanted materials in bone defects. L1, defect only; L2, cMSCs/PRP; L3, pDTSCs/PRP. (D) New bone regeneration with cMSCs/PRP, pDTSCs/PRP, and control groups at 2 weeks. L1, defect only; L2, cMSCs/PRP; L3, pDTSCs/PRP. (E) New bone regeneration with cMSCs/PRP, pDTSCs/PRP, and control groups at 4 weeks. L1, defect only; L2, cMSCs/PRP; L3, pDTSCs/PRP. (F) New bone regeneration with cMSCs/PRP, pDTSCs/PRP, and control groups at 8 weeks. L1, defect only; L2, cMSCs/PRP; L3, pDTSCs/PRP. Using cMSCs/PRP and pDTSCs/PRP, bone was regenerated to a natural level, but regeneration in the control (defect only) was not complete.
Bone regeneration and implant resorption were also monitored by histological evaluation at 2, 4, and 8 weeks. Osteogenesis spread slowly through the defect base in PRP and controls. In contrast, defects filled with cMSCs/PRP, cDPSCs/PRP, and pDTSCs/PRP implants were found to show good bone formation, suggesting that bone formation occurred at about the same rate as implant resorption. Implanted and nonimplanted control regions were collected at 2, 4, and 8 weeks, and processed and decalcified for histology.
In PRP and controls, there were few bone formation and the defects were filld by fibrous tissue evan at 2, 4, and 8 weeks (Figs. 3A–F, 4A–F). On the other hand, defects filled with cMSC/PRP, cDPSCs/PRP, and pDTSCs/PRP resulted in new bone formation with active osteocytes even after 4 weeks, and matured bone formation with a well lamellar bone and abundant marrow cavity were seen at 8 weeks (Figs. 3G–O, 4G–O), but the formation by pDTSCs/PRP was slightly weak compared with cMSCs/PRP and cDPSCs/PRP (Figs. 3, 4).

Histologic evaluation of control, PRP, cMSCs/PRP, cDPSCs/PRP, and pDTSCs /PRP implantations each week (lower magnification). Sections of representative implants are shown for the respective group. The sections were stained by hematoxylin and eosin. Original magnification 40× for all prints. (A) Two weeks in control group, (B) 4 weeks in control group, (C) 8 weeks in control group, (D) 2 weeks in PRP group, (E) 4 weeks in PRP group, (F) 8 weeks in PRP group, (G) 2 weeks in cMSCs/PRP group, (H) 4 weeks in cMSCs/PRP group, (I) 8 weeks in cMSCs/PRP group, (J) 2 weeks in cDPSCs/PRP group, (K) 4 weeks in cDPSCs/PRP group, (L) 8 weeks in cDPSCs/PRP group. (M) 2 weeks in pDTSCs/PRP group, (N) 4 weeks in pDTSCs/PRP group, (O) 8 weeks in pDTSCs/PRP group.
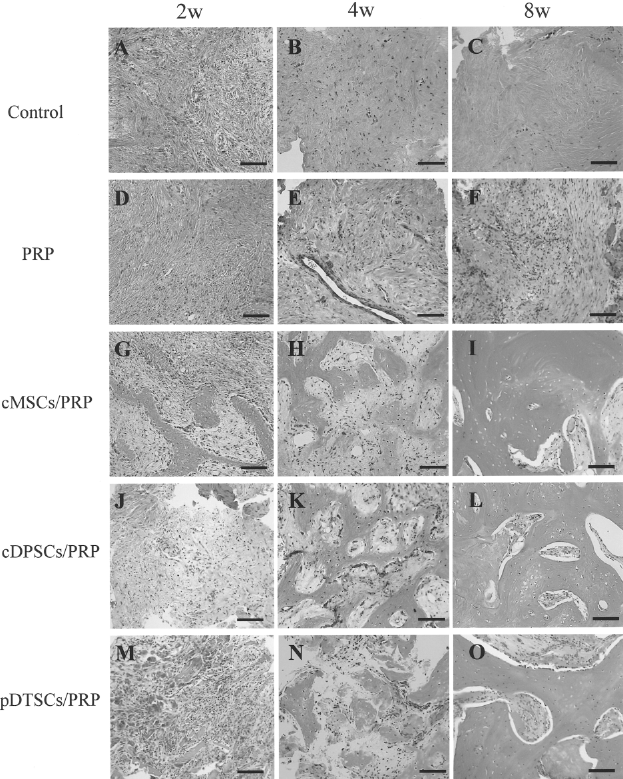
Histologic evaluation of control, PRP, cMSCs/PRP, cDPSCs/PRP, and pDTSCs/PRP implantations each week (higher magnification). Sections of representative implants are shown for the respective group. The section was stained by hematoxylin and eosin. Original magnification 200× for all prints. (A) Two weeks in control group, (B) 4 weeks in control group, (C) 8 weeks in control group, (D) 2 weeks in PRP group, (E) 4 weeks in PRP group, (F) 8 weeks in PRP group, (G) 2 weeks in cMSCs/PRP group, (H) 4 weeks in cMSCs/PRP group, (I) 8 weeks in cMSCs/PRP group, (J) 2 weeks in cDPSCs/PRP group, (K) 4 weeks in cDPSCs/PRP group, (L) 8 weeks in cDPSCs/PRP group. (M) 2 weeks in pDTSCs/PRP group, (N) 4 weeks in pDTSCs/PRP group, (O) 8 weeks in pDTSCs/PRP group.
Immunohistological analysis revealed that mineralized tissues and osteoblasts in cMSCs/PRP, cDPSCs/PRP, and pDTSCs/PRP specimens were positive for osteocalcin 8 weeks after implantation (Fig. 5). On the other hand, positive staining was not detected in control and PRP specimens (Fig. 5B, C, E, F).
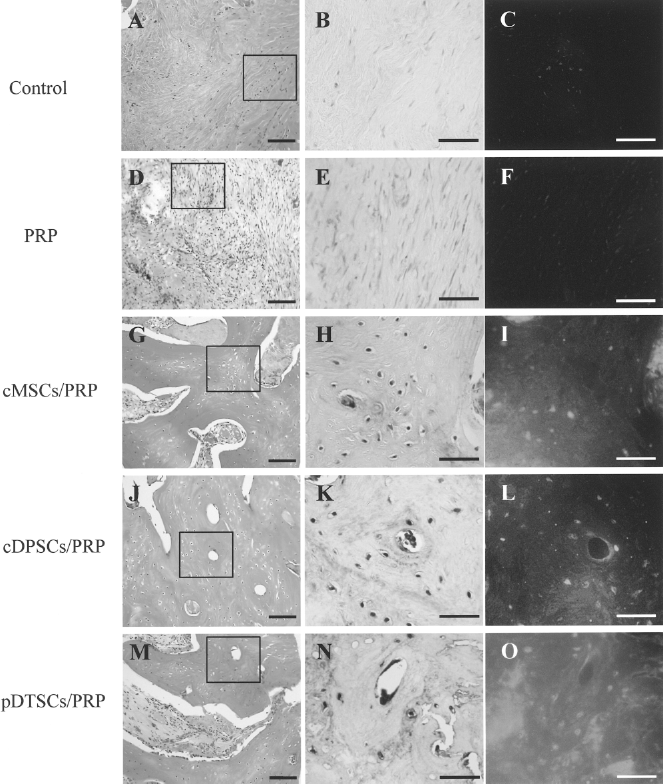
Immunofluorescence and immunohistochemical analysis of osteocalcin at 8 weeks after implantation. (A, D, G, J, M) Hematoxylin and eosin staining. (B, E, H, K, N) Immunohistochemical staining of osteocalcin. (C, F, I, L, O) Immunofluorescence staining of osteocalcin. Higher magnification of the rectangular areas in (A, D, G, J, and M) is shown in (B, C, E, F, H, I, K, L, N, and O). Scale bars: 100 μm (A, D, G, J, M); 50 μm (B, C, E, F, H, I, K, L, N, O).
Histomorphometric Analysis
The bone-regenerating ability of all implants was assessed by measuring cortical and medullary bone-regenerated areas by image analysis (Table 1). Adding PRP to the cavity did not significantly increase the cortical or medullary bone-regenerated area compared to the control. In contrast, cMSCs/PRP and cDPSCs/PRP groups showed a significant increase in the regenerated area compared with the control (p < 0.05 at 4 weeks, p < 0.001 at 8 weeks, ANOVA) or cDPSCs/PRP (p < 0.001 at 8 weeks, ANOVA), confirming the histological data; however, there was no significant difference in newly formed bone between cMSCs/PRP, cDPSCs/PRP, and pDTSCs/PRP over time (Table 1).
Histomorphology Data

Discussion
TERM by stem cells with minimal invasiveness have been attempted for clinical applications and could lead to good clinical results (22–24). Various stem cells, ES cells, iPS cells, or somatic stem cells have been identified, but the main stem cells in clinical application would be somatic stem cells. In this study, we therefore focused on stem cells, DTSCs and DPSCs, which are the most promising, and MSCs, which have been considered the standard for stem cell sources in TERM, and investigated their bone regeneration ability compared to scaffold (PRP) and defect only. The results indicated that newly formed bone areas containing these stem cells were 19.0 ± 2.9% (control), 19.7 ± 6.0% (PRP), 52.8 ± 3.5% (cMSCs/PRP), 61.6 ± 1.3% (cDPSCs/PRP), and 54.7 ± 2.2% (pDTSCs/PRP) at 8 weeks histometrically, and cMSCs/PRP, cDPSCs/PRP groups (at 4 and 8 weeks), and pDTSCs/PRP (at 8 weeks) showed a significant increase in area compared with the control and PRP, confirming the histological data (Figs. 3–5, Table 1). However, there was no significant difference in newly formed bone among cMSCs/PRP, cDPSCs/PRP, and pDTSCs/PRP (Table 1). And PRP contains various growth factors, such as platelet-derived growth factors (PDGF), transforming growth factor-β (TGF-β), and insulin-like growth factor-I (IGF-I) (12), but PRP alone did not allow osteogenesis to occur in the affected areas (Figs. 3–5, Table 1). In short, the PRP scaffold for cMSCs, cDPSCs, and pDTSCs would encourage these cells' adhesion, proliferation, and differentiation to elicit bone formation. The disappearance of the cMSCs/PRP, cDPSCs/PRP, and pDTSCs/PRP left in place induced bone tissue formation, which then self-organized according to the surrounding environment.
Immunohistochemical analysis of osteocalcin, which is a specific marker of osteogenesis, indicated that regenerative tissues in cMSCs/PRP, cDPSCs/PRP, and pDTSCs/PRP specimens were positive for osteocalcin and therefore identified as bone (Fig. 5). In a previous study, DSPCs were considered to possess multipotent ability and stem cell-like properties, such as self-renewal capability and multilineage differentiation, and the phenotypes were similar to those of bone marrow-derived MSCs (5,21); therefore, bone regeneration by DPSCs with TERM might resemble that by MSCs.
On the other hand, stem cells from human exfoliated deciduous teeth (SHED), DTSCs, was identified as a novel population of stem cells capable of differentiating into various cell types, such as osteoblasts, odontoblasts, adipocytes, and neural cells (4,5,13). It was suggested that SHED was able to produce woven bone and a dentin-like structure (4,11,18). DPSCs have been considered an appropriate candidate for dental tissue regeneration, a dentin/pulp-like structure, and for the treatment of general diseases (2,8); however, little is known about the differences between SHED and DPSCs. In this study, we found that DPSCs/PRP and DTSCs/PRP, in spite of an allograft, showed bone regeneration capacity similar to that of MSCs/PRP (Figs. 2–5). This might be based on a similar stem cell origin and possessing the common properties of MSCs.
Moreover, it was stated that MSCs could be used to prevent immune complications related to both hematopoietic stem cells and solid organ transplantation, and it was proposed that MSCs are suppressors of immune activity (16,19), because MSCs do not induce T-cell alloreactivity and display an immunoregulatory capacity by suppressing T-cell responses in vitro and in vivo (3,16,27). On the other hand, the immunosuppressive activity of human DPSCs was significantly higher than by bone marrow (16). Accordingly, we did not use any protocol for immunosuppression in our experimental animals as for allograft using DTSCs. The results showed that no clinical symptoms of pDTSC rejection were observed in the parent recipient canines and the parents did not reject pediatric DTSCs in this study. It is important to address whether these DTSCs trigger the immunologic system of the recipients; therefore, successful bone regeneration by allogenic DTSCs might be related to immunosuppressive activity by stem cells from dental pulp.
In conclusion, DPSCs and DTSCs from dental pulp could get a good bone formation the same as MSCs from bone marrow through natural bone formation process. Furthermore, out results indicate that DTSCs do not cause allogeneic graft rejection. Hence, promising DPSCs and DTSCs would apply as a broad potential stem cell source for bone regeneration in various bone surgeries, such as oral maxillofacial surgery, plastic surgery, craniofacial anomalies, and orthopedics.
Footnotes
Acknowledgments
The authors wish to thank Wataru Katagiri, Ryoko Yoshimi, Kazuto Okabe, Tomoyuki Kohgo, Yudai Nishino, Masato Fujio, and the members of the Department of Oral & Maxillofacial Surgery and Ms. Matsuba Kazuko of Laboratory Medicine, Nagoya University, Graduate School of Medicine for their help, encouragement, and contribution to the completion of this study. This work was partly supported by JAPAN MEDICAL MATERIALS (JMM) Corporation and Grants-in-Aid for Scientific Research (No. 20659297, 21390507, 21791848) from the Japan Society for the Promotion of Science and Akasaki Memorial Research Project by Nagoya University.
